Abstract
An adult male wild-caught true red-tail boa (Boa constrictor constrictor), imported from Surinam, was presented for anorexia, extreme lethargy, and coelomic swelling in the cranial third of the body, in the anatomic location of the thymus. The snake died a few minutes after blood sampling via cardiocentesis. Hematology revealed anemia and extreme leukocytosis (820 × 103/ml) characterized by a predominance (95%) of lymphocytes. Necropsy revealed enlargement of most of the visceral organs. Histology confirmed lymphoblastic lymphoma with a leukemic blood profile and diffuse infiltration of some of the heart, thymus, bone marrow, kidney, spleen, lung, and liver. Several large intracytoplasmic eosinophilic inclusion bodies surrounded by narrow clear “halos” were identified within gastric mucosal cells, proximal and distal convoluted tubule epithelial cells, and splenic cells. The final diagnosis was lymphoblast lymphoma with a leukemic blood profile and concurrent inclusion body disease.
Keywords
A 2.2-kg wild-caught, adult male red-tail boa (Boa constrictor constrictor) was presented to one of the authors (LS) because of anorexia, rapid weight loss, extreme lethargy, and swollen coelom in the cranial third of the body. This snake, for sale at an exotic pet shop in a suburb of Paris, had not been dewormed since it was imported from Surinam 6 months before. General husbandry was reviewed with an employee of the shop and appeared to be appropriate. Physical examination revealed emaciation, poor body condition, and lethargy. Palpation of the coelomic cavity revealed a firm mass in the region of the trachea, near the heart base, cardiomegaly, and hepatomegaly. Cardiac auscultation using a Doppler flow probe a of 8 MHz showed a normal heart rate (30 beats per min at 30°C, close to 27 beats per min when using the formula of the prediction of heartbeat frequency = 33.4 [Wtk −0.25]). 21
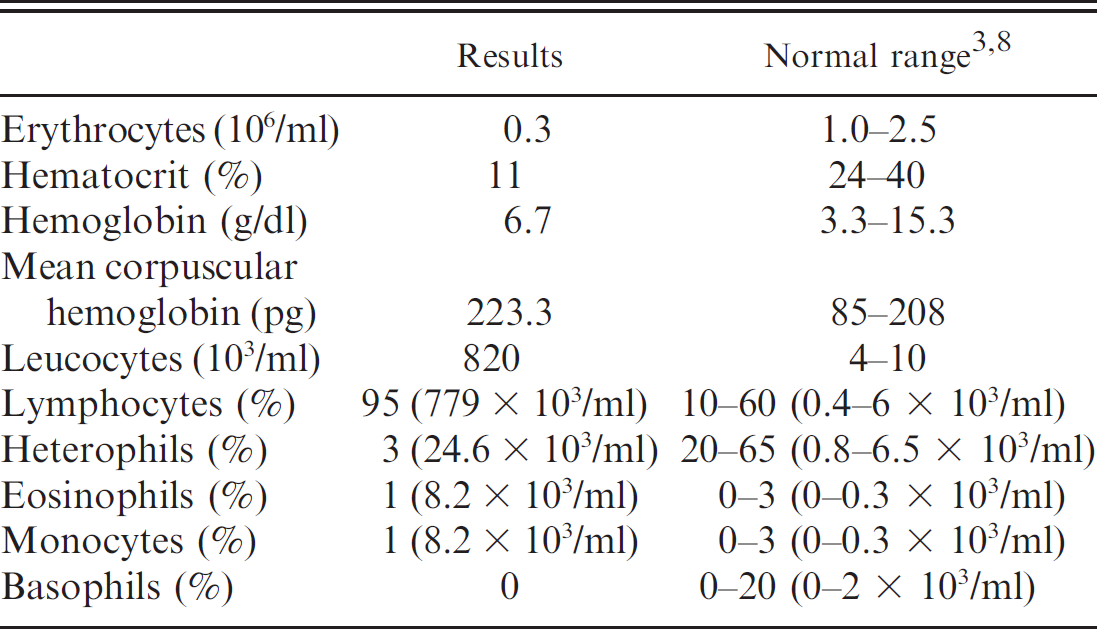
Whole blood was collected by cardiocentesis in an evacuated tube containing ethylenediamine tetra-acetic acid. The results of the complete blood count and differential white blood cell count are presented in Table 1 with appropriate reference intervals. The hemogram showed severe anemia (red blood cell count: 0.3 × 106/ml; hemoglobin concentration: 6.7 g/dl; and packed cell volume: 11%) with extreme leukocytosis (white blood cell count: 820 × 103/ml) and lymphocytosis (779 × 103/ml). 3 Examination of a stained blood film revealed the presence of numerous lymphoblastic cells at the expense of the other expected cellular populations. Biochemical test results on heparinized plasma are listed in Table 2 with corresponding reference intervals. Biochemical test results indicated hyperuricemia, uremia, and increased aspartate amino-transferase activity. 3 Unfortunately, the boa died a few min after cardiocentesis. For that reason, other ancillary tests were not performed.
Necropsy findings included enlargement and congestion of most of the visceral organs such as heart, liver and gall bladder, spleen, kidneys, pancreas, and major blood vessels. A 30-mm × 25-mm, firm, diffuse pink mass was located close to the trachea and was consistent with thymus (Fig. 1).
Sections of the mass, heart, liver, lung, kidneys, stomach, small intestine, colon, thymus, trachea, thyroid, spleen, bone marrow, and testis were collected for histopathologic evaluation. These tissues were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 3–5 μm, mounted on glass slides, and stained with Harris hematoxylin and eosin, and safranin.
The thymic mass adjacent to the trachea was composed almost entirely of neoplastic lymphoblasts and had considerable necrosis within deeper portions of the neoplasm (Fig. 2). The fine connective tissue capsule of the thymus appeared to be bulging outward and displaced towards the trachea. The thymus was predominately composed of small round lymphoid cells with large nuclei and scant, finely granular to amorphous, pale-blue cytoplasm. Individual nuclei contained 1 or more prominent nucleoli. Mitoses were infrequent. Large pale-staining macrophages with phagocytosed cellular debris were scattered throughout the thymus. Lesser numbers of pink-staining, round-to-elongated epithelioid cells with finely granular cytoplasm and densely clumped nuclear chromatin were scattered throughout the much more numerous small lymphoid cells. Thymic (Hassall's) corpuscles were infrequently identified. The small blood vessels that penetrated the thymic mass contained numerous immature lymphoid cells consistent with the lymphoblasts observed in the stained blood smears. Cell-specific immunoperoxidase-tagged monoclonal antibody staining was not performed to identify the neoplastic cell lineage.
Results of a complete blood count in a red-tail boa (Boa constrictor constrictor) with lymphoblastic lymphoma.
Blood chemistry evaluation.

Except for total aspermia, the testis was unremarkable. The myocardium (Fig. 3), spleen, lung, kidney, small intestine, colon, thyroid, bone marrow, and liver had discrete to diffuse infiltrates of lymphoblasts characterized by large, often vesicular nuclei that contained 1 or 2 prominent nucleoli. Mitotic figures were identified in most high-power fields. Of interest, large, eosinophilic inclusion bodies composed of amorphous material, and often surrounded by a narrow clear “halo,” were identified as specific cell populations. These included gastric mucosal cells, particularly those lining the gastric pits (Fig. 4); renal epithelial cells, lining both the proximal and distal convoluted tubules; and occasional splenic cells. By light microscopy, these intracytoplasmic, eosinophilic, viral-type inclusions were indistinguishable from those observed in the inclusion body disease (IBD) of boids, snakes belonging to the Boidae family. These inclusions usually consist of a 68-kd protein band unique to inclusion-positive material and represented an intracytoplasmic accumulation of an IBD virus–associated protein. 27 Without ancillary electron microscopic investigations, it was impossible to further characterize these viral-type inclusions.
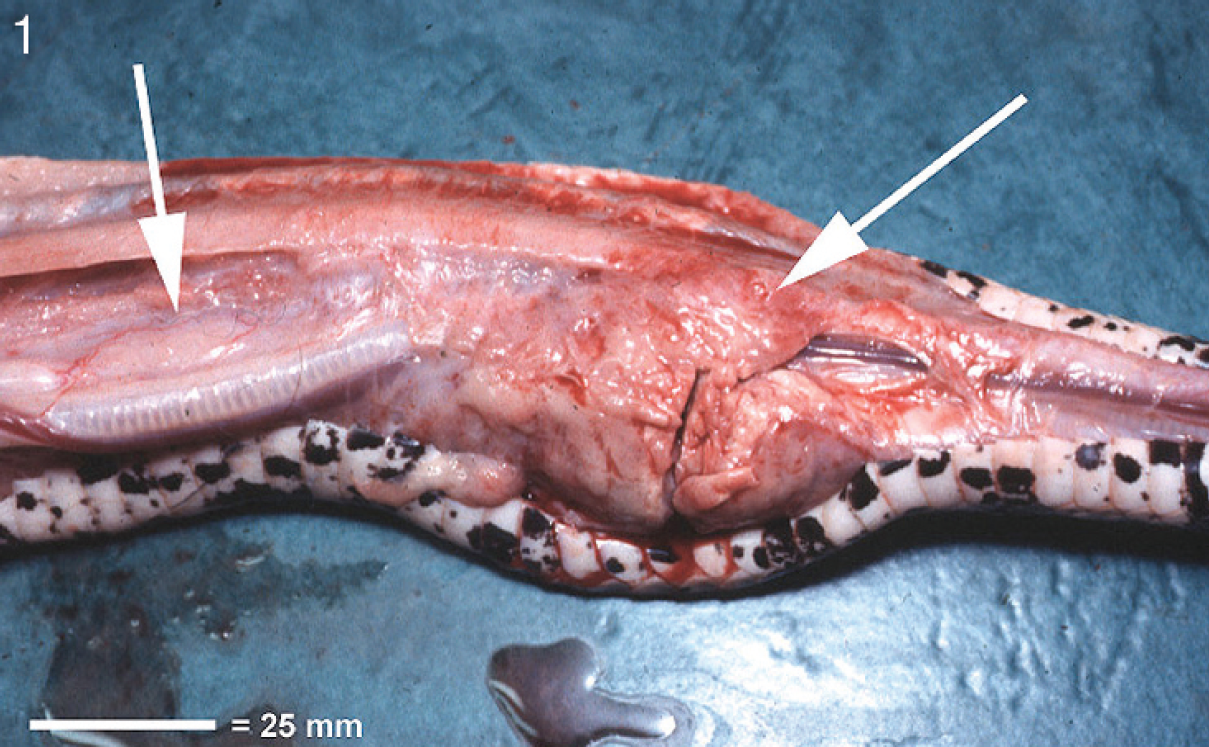
Photograph of necropsy. Note the mass of pale tissue located alongside the trachea (right arrow). This tissue extended caudal to the level of the thymus (left arrow). Bar = 25 mm.

Photomicrograph of thymic mass and trachea. The trachea occupies the upper half of this image; the thymus is seen immediately beneath it. Hematoxylin and eosin. Bar = 40 μm.

Myocardial infiltrate. Note the population of blastic round cells infiltrating throughout the myocardium. Hematoxylin and eosin. Bar = 20 μm.

Gastric mucosa. Note the presence of amorphous, magenta-staining viral-type intracytoplasmic inclusion bodies (arrows). Some of them are surrounded by clear “halos” that are present within the gastric lining, mucin-secreting, and zymogen-secreting epithelial cells. Hematoxylin and eosin. Bar = 20 μm.
Based on the histologic and hematologic findings, the definitive diagnosis was lymphoblastic lymphoma with a leukemic blood profile and concurrent IBD. Further characterization of the neoplastic cells by lineage was not possible; however, the histologic and hematologic findings suggest that the cells are of lymphoid origin. The histologic details of the thymus distinguish it from other lymphoid infiltrates inasmuch as thymic lymphocytes, large macrophages, and thymic epithelioid cells were prominently featured. The last 2 cell types, so characteristic of the thymus, were lacking in other parenchymatous organ infiltrates. Additionally, intracytoplasmic inclusion bodies were not observed in cells comprising the thymic mass. The snake's primary lymphoblastic neoplasm is not believed to be thymic in origin; however, the precise demonstration of B- or T-cell lineage would require immunohistochemical staining.
Several different malignant hematopoietic neoplasms including various types of leukemia, malignant lymphoma (lymphosarcoma) with or without a leukemic blood profile, thymoma, plasma cell neoplasia, and reticulum cell sarcoma (histiocytic sarcoma) have been described in various species of reptiles. 2,4–22,25,26,28 The precise etiology of these tumors is unknown in reptiles but may involve genetic, parasitic, environmental, and viral factors. 20 However, a viral etiology of neoplasia is poorly documented in reptiles. 13,15,19,22,25,26
Boid IBD is a multisystemic viral infection caused by a 90- to 120-nm retrovirus affecting boid snakes world-wide. 1,23,24 It is probably the most important viral disease known in captive boas and pythons to date as it has been associated with high morbidity and mortality rates. Viral infection can follow an acute or chronic clinical course, and its severity is known to be significantly worse in pythons than in boas. Adult boas may be inapparent carriers and serve as reservoirs for retroviral infection. 1 No clinical signs are pathognomonic for IBD, but affected snakes usually exhibit neurologic, digestive, or respiratory signs. 23,24 In the present report, the concomitant finding of IBD-like inclusions in a snake displaying evidence of lymphoblastic lymphoma and leukemia is noteworthy.
Acknowledgements. The authors thank Martine Mialot, Marie Lagadic, and Mercedes Estrada from the Laboratoire IDEXX Alfort, Alfortville, France.
Footnotes
a.
Vet Dopp®, Heska Corp., Fort Collins, CO.
