Abstract
Eight pigs were inoculated subcutaneously with a highly virulent hog cholera virus (HCV) strain ALD. The infected pigs developed severe illness and became moribund on postinoculation day (PID) 7 or PID 10. Histologic lesions were characterized by severe generalized vasculitis, necrosis of lymphocytes, and encephalitis. HCV antigen was detected in crypt tonsilar epithelial cells, macrophages, and reticular endothelial cells of lymphoid tissues. Antigen localization corresponded well with histologic lesions. Five pigs were inoculated with less virulent HCV Kanagawa/74 strain and were euthanatized on PID 30. All five infected pigs recovered from the illness but became stunted. They also had a slight follicular depletion of lymphocytes, histiocytic hyperplasia, and hematopoiesis in the spleen. Less virulent HCV antigen was observed in the tonsils, kidneys, pancreas, adrenal glands, and lungs. Although antigen localization was less associated with histologic lesions, immunoreactivity was stronger than that in the pigs infected with the ALD strain of HCV. An almost complete loss of B lymphocytes was recognized in pigs infected with the ALD strain and was correlated with follicular necrosis in lymphoid tissues. Loss of B lymphocytes was not prominent in the pigs infected with Kanagawa/74 strain. The number of CD4+ and CD8+ T lymphocytes was significantly higher than that in the noninfected control pigs.
Hog cholera (HC) is a highly contagious disease affecting swine and can result in severe economic losses. Virulent strains cause a fatal hemorrhagic illness in susceptible pigs of all ages. Less virulent strains cause subacute, chronic, or inapparent illness. 2 12 16 These acute and/or chronic forms of infections with HC virus (HCV) have been observed under both natural and experimental conditions. 2 5 9 10 15 17 HCV affects the immune system, resulting in a generalized leukopenia. Lymphoid organs are affected by the infection. Both B-lymphocyte deficiency and T-lymphocyte activity have been observed in pigs infected with a virulent strain of HCV. 8 12 However, there is little information on immunocompetence in pigs infected with a less virulent strain of HCV. There is one report on immune reactions in 4-day-old gnotobiotic piglets infected with the less virulent Kanagawa/74 strain of HCV. 7
The present study was undertaken to compare the responses of both B lymphocytes and T lymphocytes (CD4+ and CD8+) in lymphoid tissues of pigs during the acute stage of HC caused by a highly virulent strain and during the chronic stage of HC caused by less virulent strains of HCV. The histologic distribution of HCV antigen was studied using immunohistochemistry in formalin-fixed, paraffin-embedded tissues.
Materials and Methods
Animals
Fifteen weaned pigs each weighing 25–30 kg were used in this study. They were born from nonimmune sows and were HCV susceptible. Preexposure sera were negative for antibody against HCV. The pigs were divided into three groups and were housed separately in different buildings to prevent cross-infection.
Virus
A highly virulent HCV strain, ALD, 11 and a less virulent HCV strain, Kanagawa/74 4 7 11 isolated from a pig with chronic HC, were passaged in pigs. Blood samples were collected at the peak of infection. Infected blood was maintained at −80 C. These two strains were used as inoculum for the pigs.
Experimental procedure
Before inoculation with HCV, the pigs were anesthetized with ketamine hydrochloride (30 mg/kg intramuscularly) and xylazine (2.0 mg/kg, intramuscularly). Thirteen pigs were inoculated subcutaneously with 2 ml of each viral strain. Infectivity of the viruses was 105.0 TCID50/ml of HCV (Table 1). 4 Eight pigs (Nos. 1–8) were inoculated with the ALD strain of HCV. Two separate pigs from this group were euthanatized on postinoculation days (PIDs) 3, 5, 7, or 10. Five pigs (Nos. 9–13) were inoculated subcutaneously with Kanagawa/74 strain and were euthanatized on PID 30. Euthanasia was by exsanguination after azaperone and thiopental administration. Two additional pigs (Nos. 14, 15) were inoculated with noninfected pig blood and were used as negative controls for the experiment.
Distribution of HCV antigen∗ in pigs infected with ALD or Kanagawa/74 strains of HCV.
- = negative; + = small number; ++ = moderate number; +++ = large number.
PID = postinoculation day.
Blood was collected from eight ALD pigs that were alive on PIDs 3, 5, 7, and 10 and from five Kanagawa/74 pigs on PIDs 7, 14, 21, and 28. The total number of leukocytes for each blood sample was determined using a Coulter counter. Clinical observations were recorded daily.
Immunohistopathologic examination
Tissue specimens from each pig were fixed in 10% neutral buffered formalin, embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin (HE). For the detection of T-cell marker antigens CD4 and CD8 and of B-cell marker antigen, tissue samples from fresh tonsils, spleen, and mesenteric lymph nodes were embedded in Tissue-Tek O.T.C. Compound (Sakura Finetechnical Co., Tokyo, Japan) and stored frozen in liquid nitrogen. Five-micrometer sections were cut using a cryomicrotome, air-dried, fixed in cold acetone for 5 minutes, and kept at −20 C until staining.
For the detection of HCV antigen, specimens from the spleen, tonsils, kidneys, lymph nodes, lungs, adrenal glands, pancreas, and ileum were fixed in 10% neutral buffered formalin for 7 days, processed, and embedded in paraffin. Sections were stained by the avidin–biotin complex (ABC) immunoperoxidase method (Vectastain ABC kit, Vector Laboratories, Burlingame, CA). Four-micrometer sections were cut and deparaffinized. Deparaffinized tissue and frozen sections were incubated with 1% hydrogen peroxide in methanol for 30 minutes to block endogenous peroxidase activity. For antigen reactivation, the deparaffinized sections were incubated in antigen-unmasking solution (Vector Laboratories) for 1 minute. After blocking the sections with normal horse serum for 30 minutes at room temperature, the primary antibody was incubated for 18 hours at 4 C. A commercially available anti-HCV monoclonal antibody (WA303, Central Veterinary Laboratory, New Haw, Addlestome, UK) was used at a dilution of 1:50. Commercially available CD4 and CD8 monoclonal antibodies (Veterinary Medical Research and Development, Pullman, WA) were used at dilutions of 1:50 and 1:10, respectively. Anti-swine B-cell monoclonal antibody (STH249, Dr. M. Shimizu, National Institute of Animal Health, Tsukuba, Japan) was used at a dilution of 1:50. Biotinylated horse anti-mouse secondary antibody was incubated alone for 30 minutes and with the ABC for 60 minutes, both at room temperature. Between each step, the slides were washed 3 times for 10 minutes each time in phosphate-buffered saline. The chromogen, 0.5% 3,3′-diaminobenzidine tetrahydrochloride (DAB) diluted 1:10 in 0.05 M Tris containing 0.3% hydrogen peroxide, was applied to the slides for 5 minutes. The sections were counterstained with methyl green. Tissue sections from pig Nos. 14 and 15 and serum from a nonimmunized mouse were used as controls. The mean number of CD4+ and CD8+ T lymphocytes and B lymphocytes were counted in 10 fields of 200× magnification. 7
Results
Clinical observation
Pigs given the the ALD strain of HCV developed fever (41–42 C), depression, anorexia, and leukopenia. These pigs became moribund on PID 7 or PID 10. The mean (± SD) number of leukocytes in the eight pigs examined was 16.3 ± 3.7 × 103 cells/mm3 before inoculation. Leukocytes in these pigs decreased to 5.4 ± 0.7 × 103 cells on PID 3 (n = 8 pigs), 7.9 ± 1.1 × 103 cells on PID 5 (n = 6 pigs), 3.6 ± 1.6 × 103 cells on PID 7 (n = 4 pigs), and 5.0 ± 4.1 × 103 cells on PID 10 (n = 2 pigs).
Pigs given the Kanagawa/74 strain of HCV developed anorexia, depression, fever (40–41.5 C), and slight leukopenia between PID 3 and PID 20. The average number of leukocytes in the five pigs examined was 7.1 ± 3.3 × 103 cells/mm3 on PID 7, 7.8 ± 3.3 × 103 cells on PID 14, 11.8 ± 3.2 × 103 cells on PID 21, and 18.6 ± 4.5 × 103 cells on PID 28. After PID 21, fever decreased in all pigs, there was no increase in body weight, and they all became stunted.
Macroscopic and histologic lesions
Macroscopically, multiple and severe petechial hemorrhages were observed in the kidneys and urinary bladder of the pigs given the ALD strain. Lymph nodes were swollen, edematous, and hemorrhagic. Splenic infarctions were observed in four pigs (Nos. 3–6), and thymus atrophy was observed in six pigs (Nos. 3–8). Extravasation in the larynx and epiglottis was found in three pigs (Nos. 9, 11, 12) and severe thymus atrophy was found in all five pigs (Nos. 9–13) given the Kanagawa/74 strain.
Histologic lesions in pigs given the ALD strain induced generalized hydropic degeneration and necrosis of endothelial cells lining the vascular system. Follicular depletion of lymphocytes and/or necrosis in the spleen was prominent in six pigs (Nos. 1–6). Histiocytic hyperplasia in the lymphoid tissues was found in two pigs (Nos. 7, 9), and cortical depletion of thymic lymphocytes was found in six pigs (Nos. 3–8). Encephalitis characterized by cerebral vasculopathy was found in five pigs (Nos. 3, 5–8).
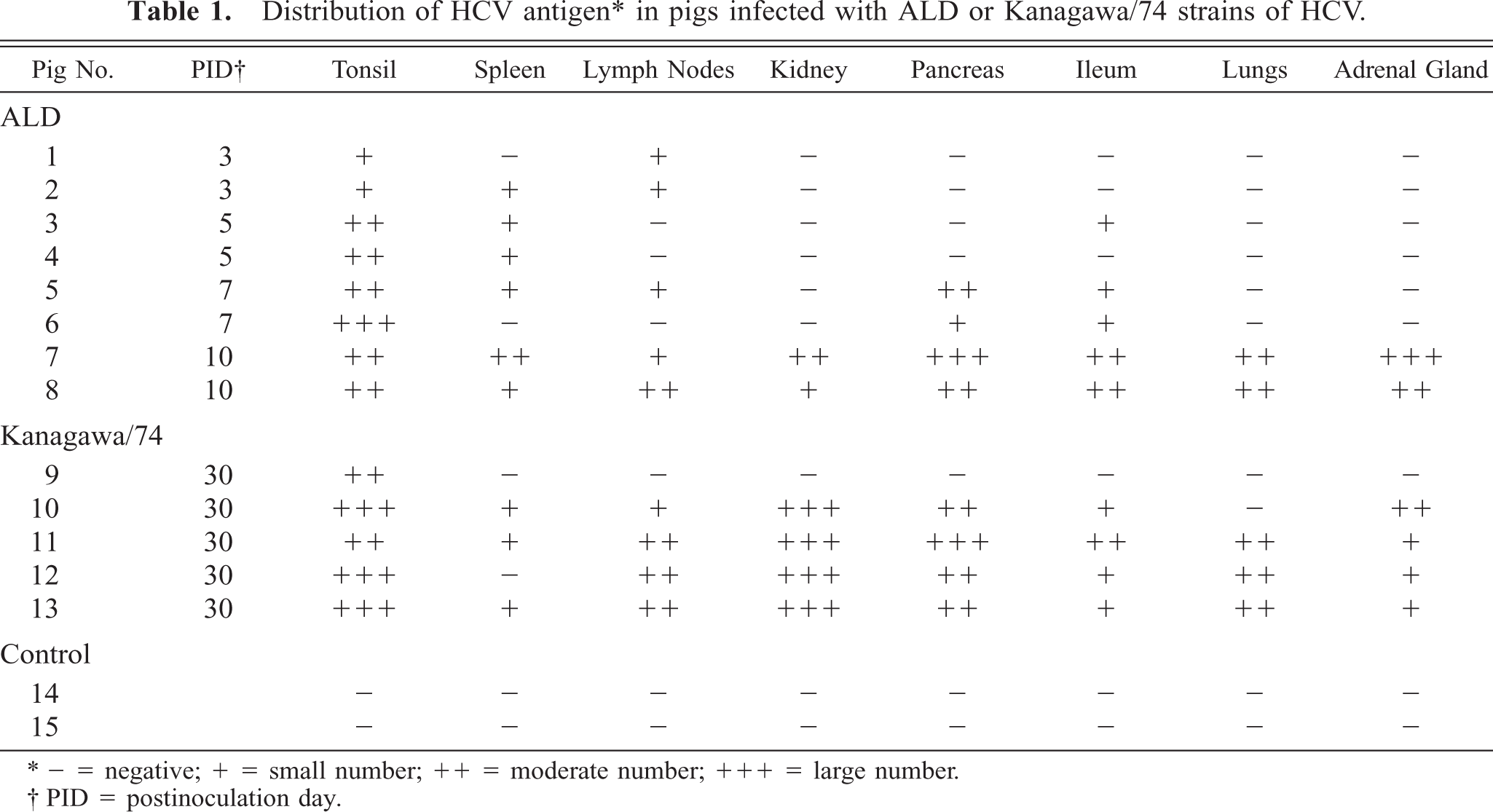
Histologic lesions in pigs given the Kanagawa/74 strain induced slight follicular depletion of lymphocytes without necrosis in three pigs (nos. 10, 11, 13), histiocytic hyperplasia in lymphoid tissues of two pigs (Nos. 9, 12), hematopoiesis in spleens of three pigs (Nos. 9, 10, 12) (Fig. 1), severe cortical depletion of thymic lymphocytes in all five pigs (Nos. 9–13), and encephalitis in one pig (No. 12). No macroscopic or histologic lesions were found in the noninfected control pigs.
Spleen; pig No. 9. Hematopoiesis in pig infected with Kanagawa/74 strain of HCV. HE. Bar = 50 μm.
Distribution of HCV antigen
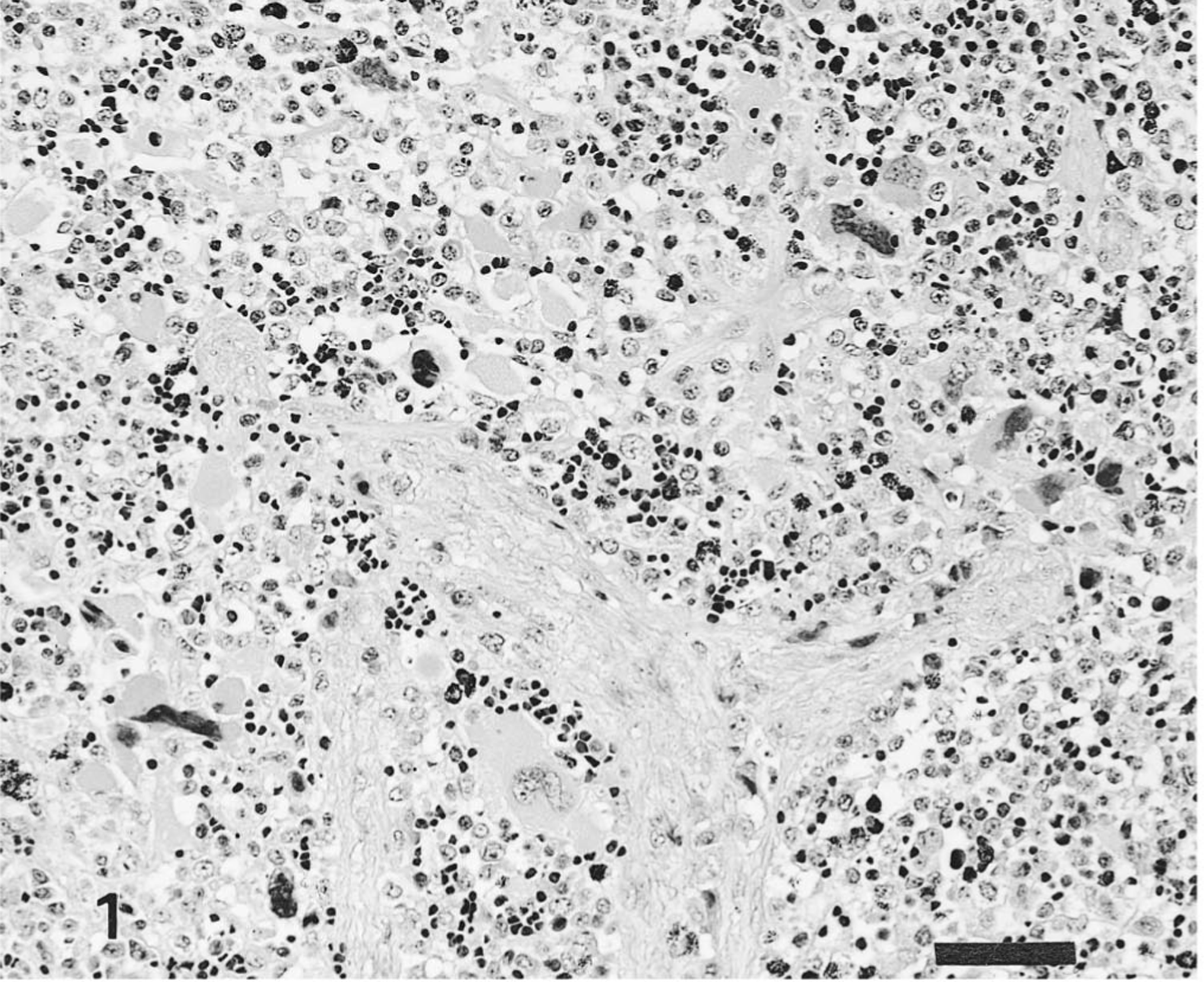
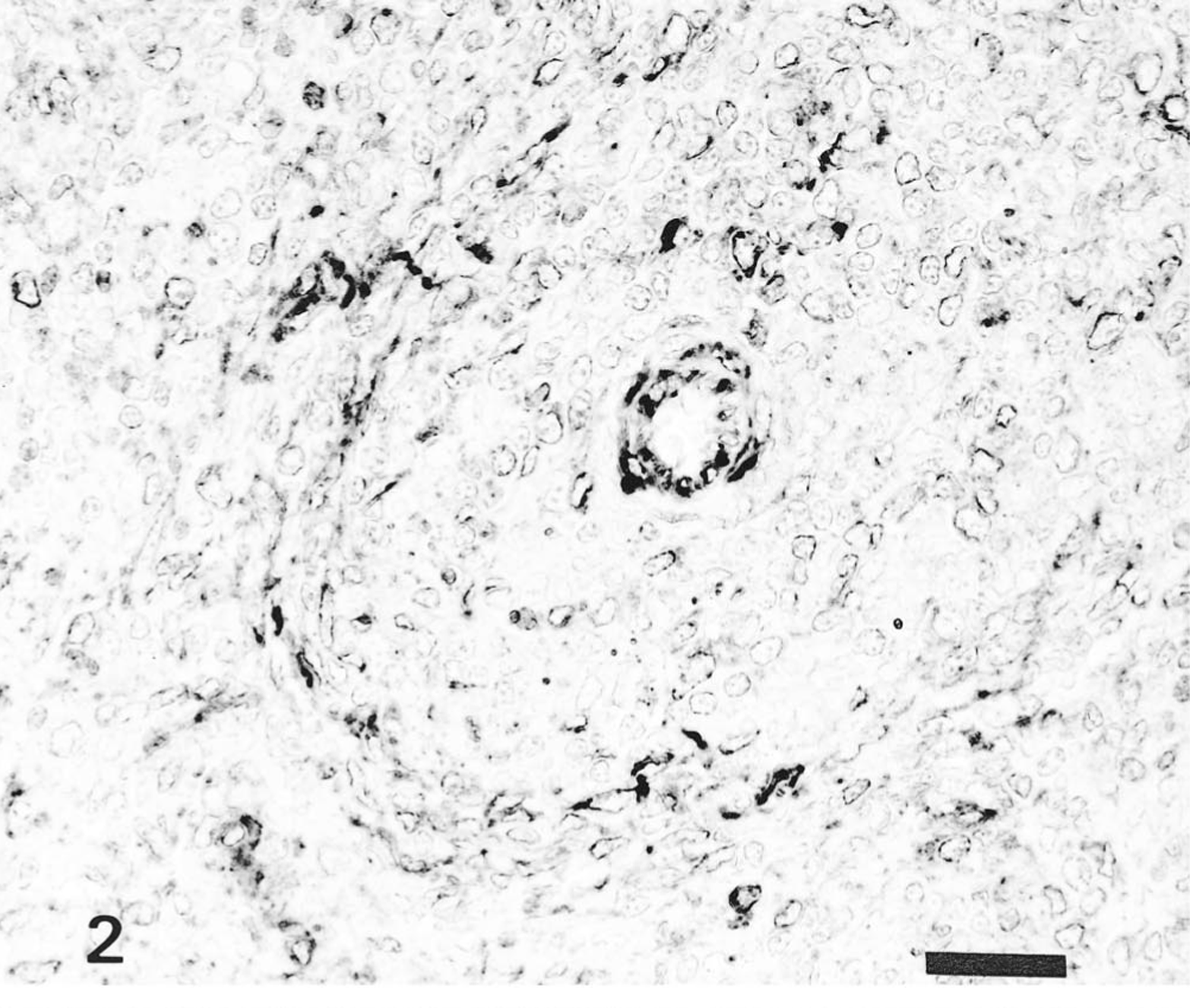
The distribution of HCV antigen in the tissues is shown in Table 1. Pigs infected with the ALD strain had HCV antigen in superficial and crypt tonsilar epithelial cells and in tonsillar endothelial cells on PID 3 and PID 7. Macrophages and endothelial cells of the spleen (Fig. 2) and lymph nodes contained antigens on PID 3 and PID 10. HCV antigen was also observed in the superficial and crypt mucosal cells of the ileum, scattered macrophages and lymphocytes of Peyer's patches (Fig. 3), pancreatic acinar epithelial cells, and pancreatic ductal epithelial cells on PID 5 or PID 7. Occasionally, HCV antigen was found in the smooth muscle cells of blood vessels distributed in the mesenterium. Specific antigen was also found in the convoluted and collecting duct epithelial cells of the kidneys, the bronchial and bronchiolar mucosal epithelial cells of the lungs, and the zona reticulata and zona fasciculata of the adrenal gland cortex on PID 10.
Spleen; pig No. 7. HCV antigen in reticular and epithelial cells of pig infected with ALD strain of HCV. Anti-HCV ABC. Bar = 25 μm.
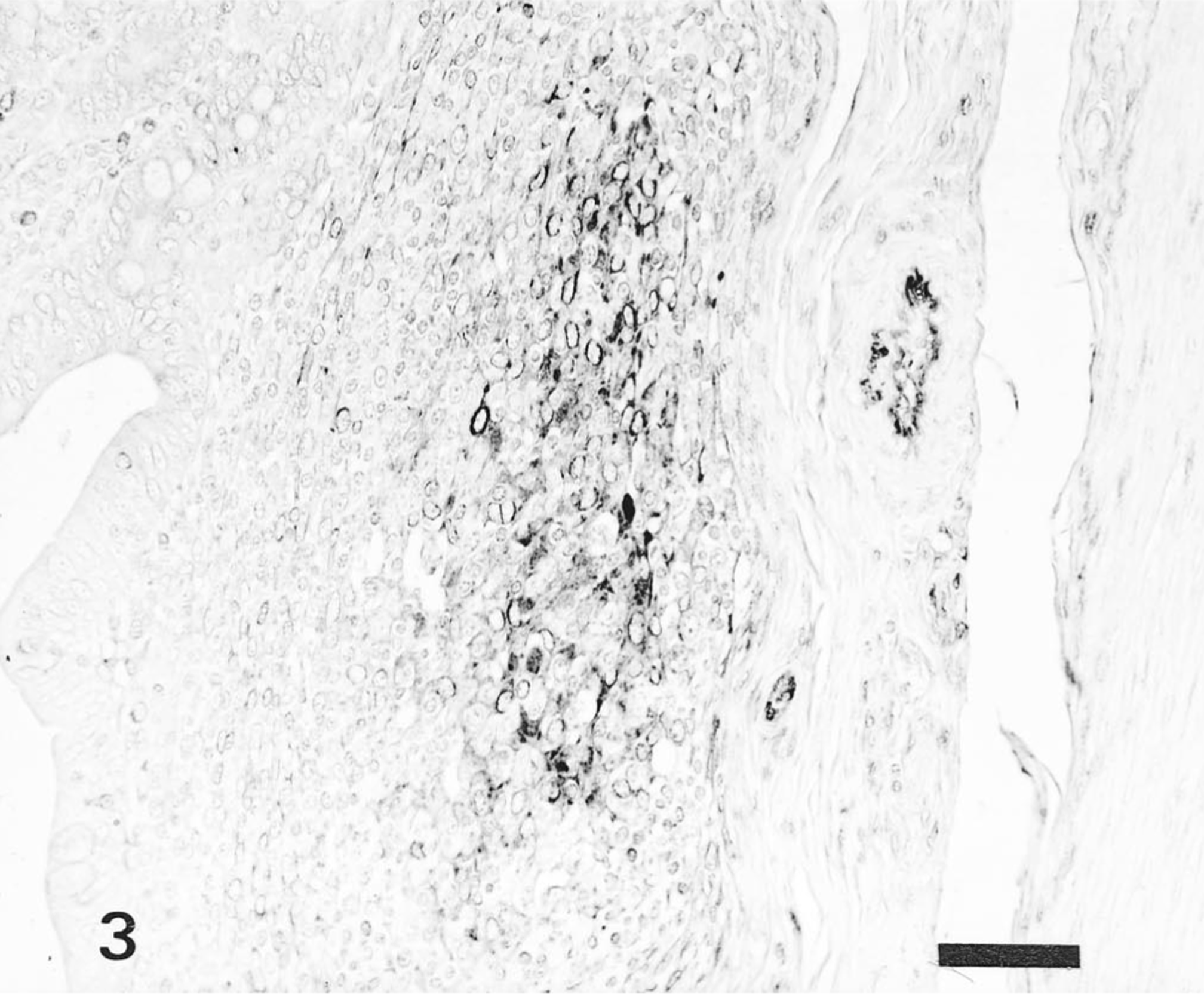
Ileum; pig No. 8. HCV antigen in the macrophages and lymphocytes of Peyer's patches of pig infected with ALD strain. Anti-HCV ABC. Bar = 50 μm.
In pigs infected with the Kanagawa/74 strain, the distribution of HCV antigen was similar to that of moribund pigs infected with the ADL strain but was not associated with histologic lesions. Immunoreactivity in certain tissues, such as the tonsils (Fig. 4), kidneys (Fig. 5), lungs, pancreas (Fig. 6), and adrenal gland, was stronger in the pigs infected with the Kanagawa/74 strain than in those infected with the ALD strain (Table 1).
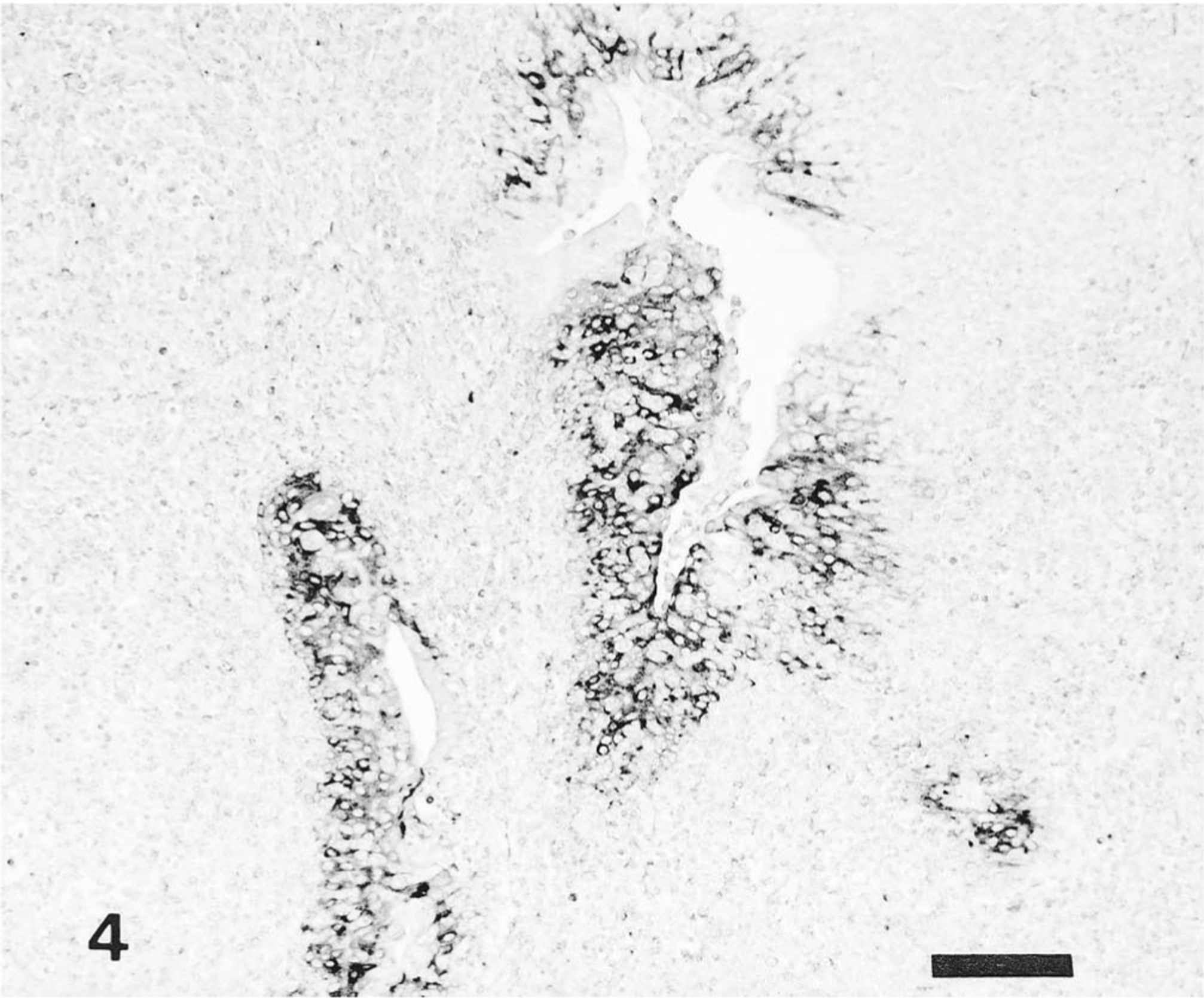
Tonsil; pig No. 10. Large amount of HCV antigen in crypt tonsilar epithelial cells of pig infected with Kanagawa/74 strain. Anti-HCV ABC. Bar = 100 μm.
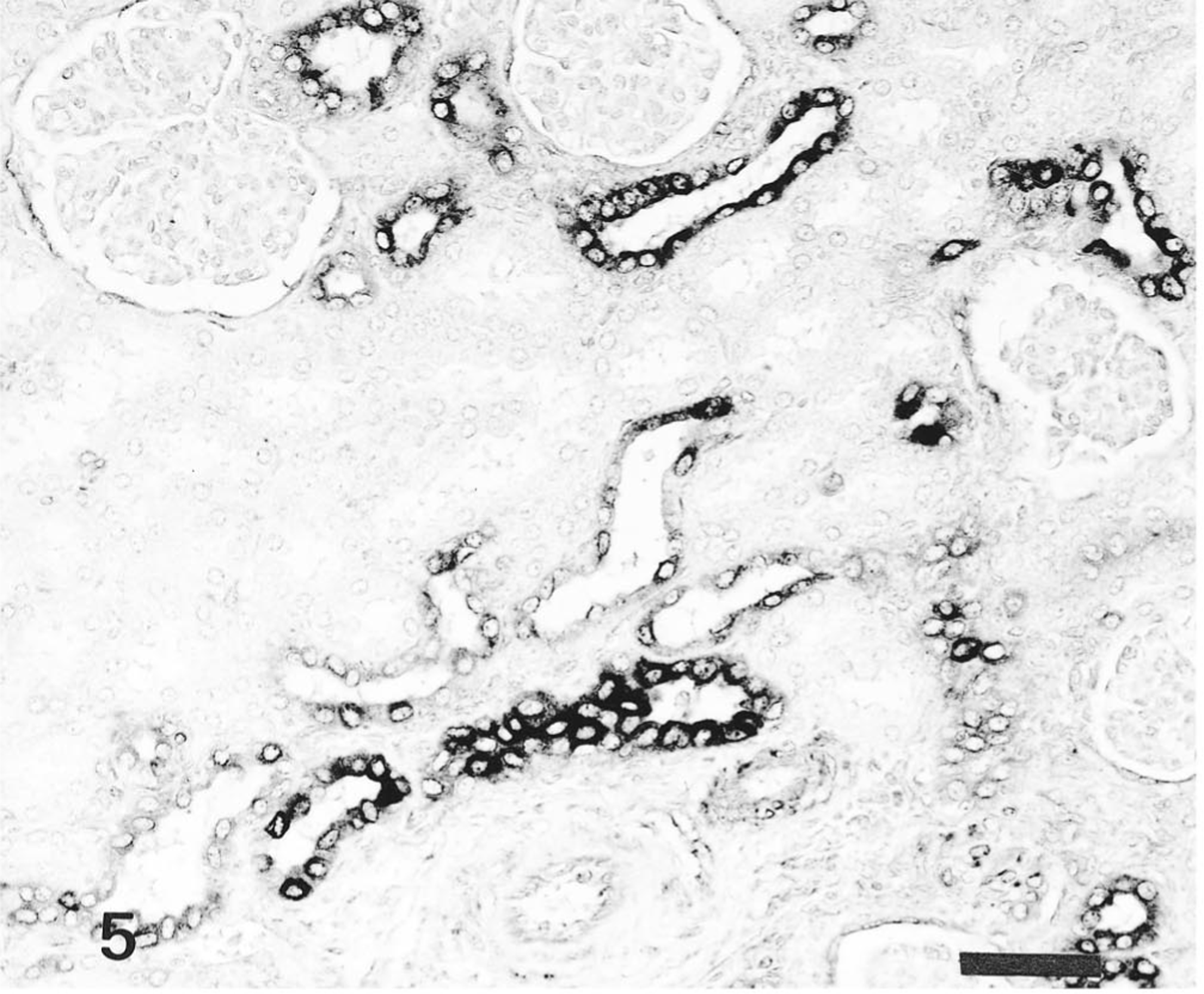
Kidney; pig No. 11. HCV antigen in convoluted and collecting duct epithelial cells of pig infected with Kanagawa/74 strain. Anti-HCV ABC. Bar = 50 μm.
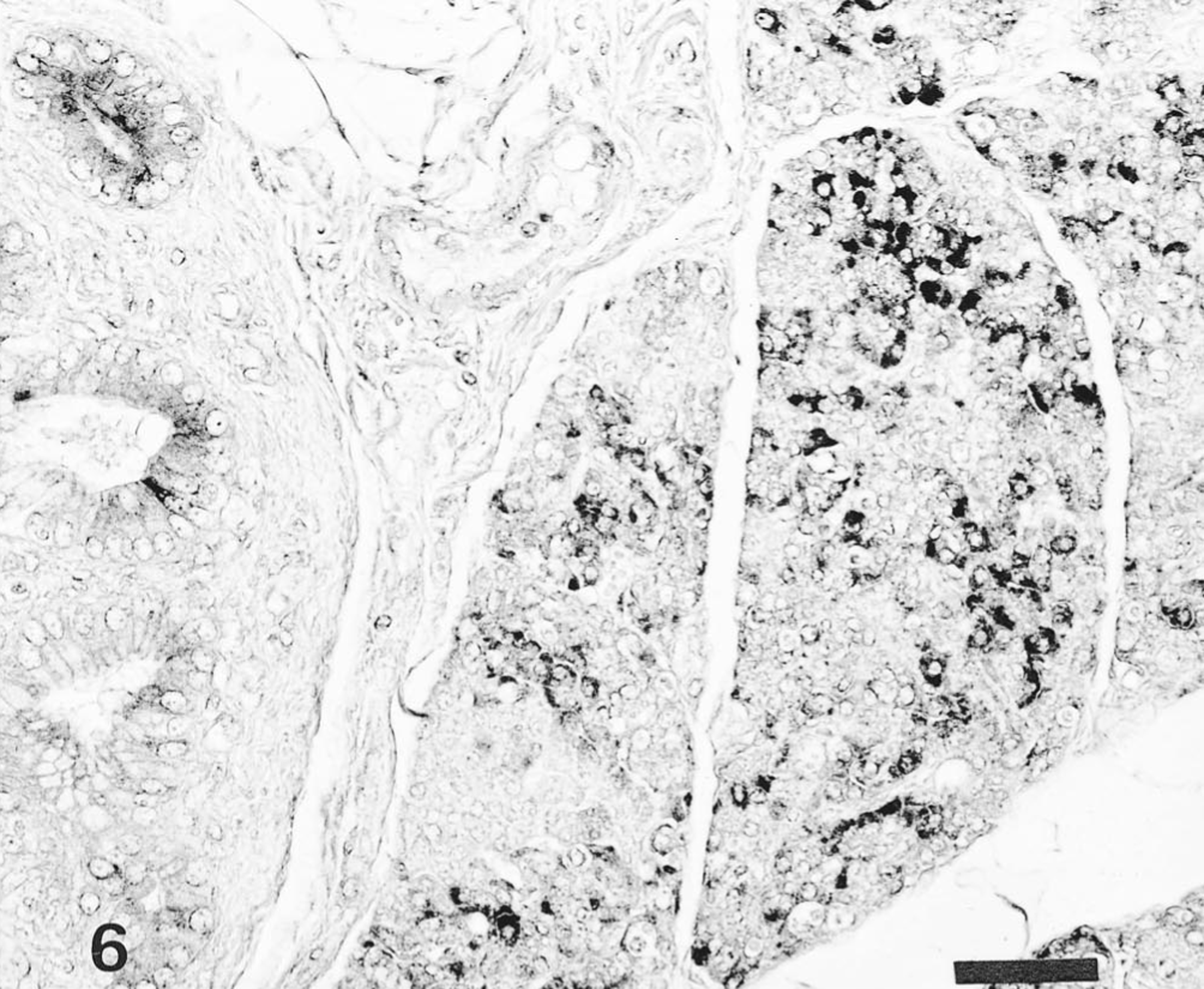
Pancreas; pig No. 11. HCV antigen in the pancreatic acinar epithelial cells and pancreatic ductal epithelial cells of pig infected with Kanagawa/74 strain. Anti-HCV ABC. Bar = 50 μm.
Distribution of T and B lymphocytes
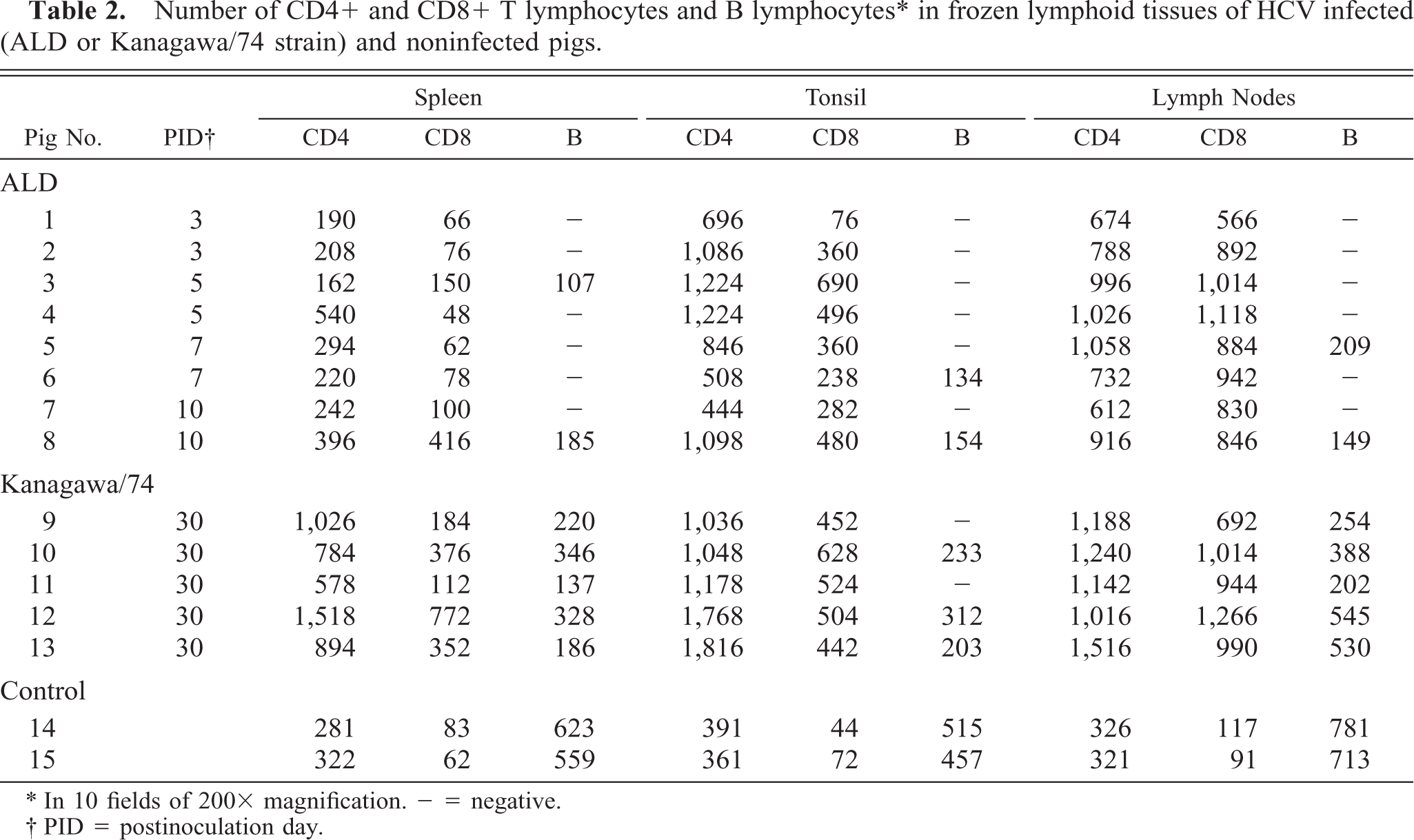
The distribution of T and B lymphocytes after HCV infection is shown in Table 2. In the noninfected control pigs, T lymphocytes (CD4+ and CD8+) were located in the periarteriolar lymphoid sheath of the spleen, interfollicular areas of the tonsils, and perifollicular or interfollicular areas of the central cortex of lymph nodes. B lymphocytes were found in the follicles of lymphoid tissues. In the spleen, the number of CD4+ and CD8+ T lymphocytes in the pigs infected with the ALD strain did not increase, but in pigs infected with the Kanagawa/74 strain, CD4+ T lymphocytes increased 3–5 times and CD8+ T lymphocytes increased 3–10 times. In tonsils and mesenteric lymph nodes, CD4+ T lymphocytes increased 2–4 times, and CD8+T lymphocytes increased 6–12 times in all pigs infected with the ALD and Kanagawa/74 strains of HCV (Figs. 7, 8).
Number of CD4+ and CD8+ T lymphocytes and B lymphocytes∗ in frozen lymphoid tissues of HCV infected (ALD or Kanagawa/74 strain) and noninfected pigs.
In 10 fields of 200X magnification. - = negative.
PID = postinoculation day.
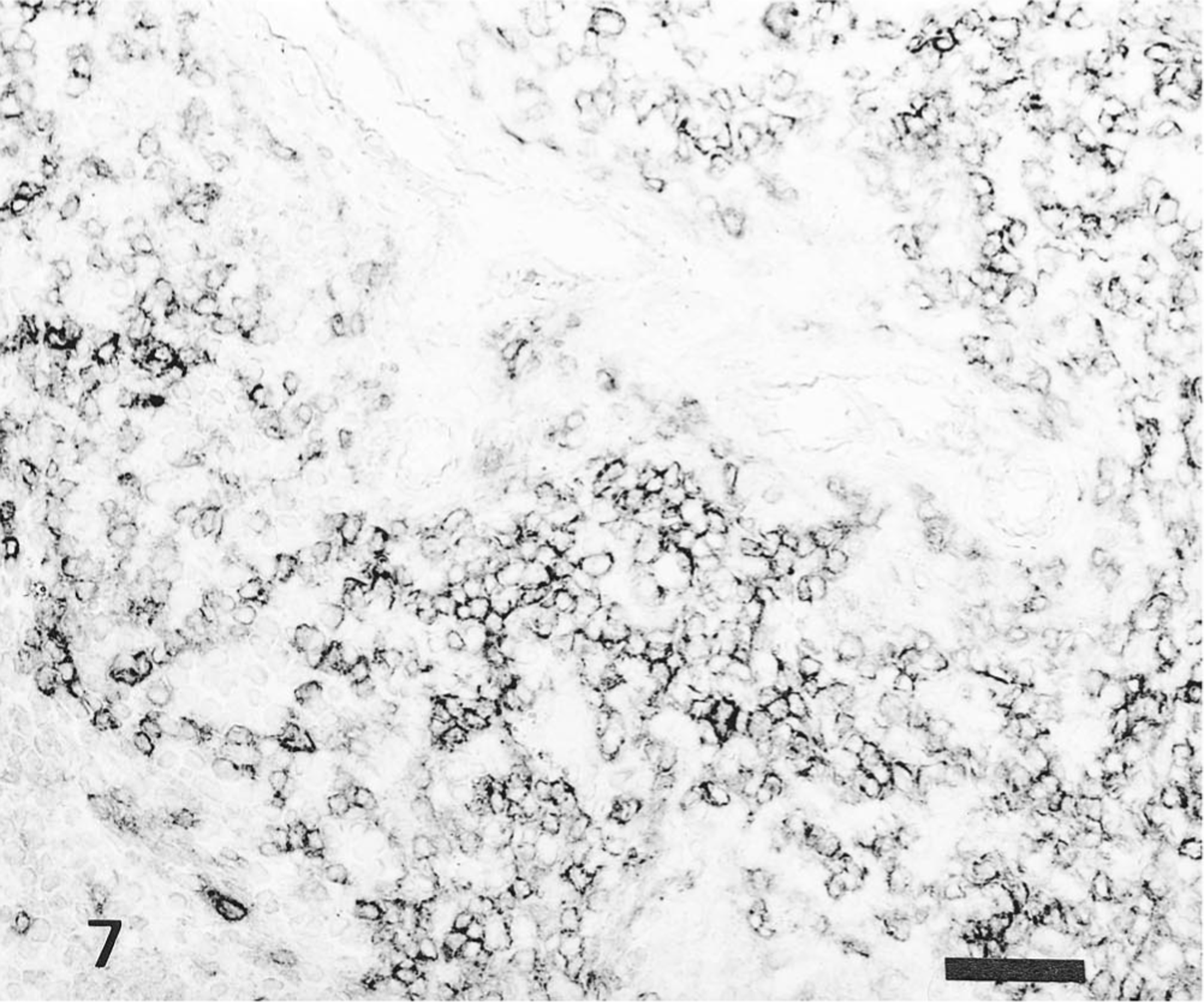
Spleen; pig No. 9. CD4+ T lymphocytes in periarteriolar lymphoid sheath of pig infected with Kanagawa/74 strain. Anti-CD4+T lymphocyte ABC. Bar = 50 μm.
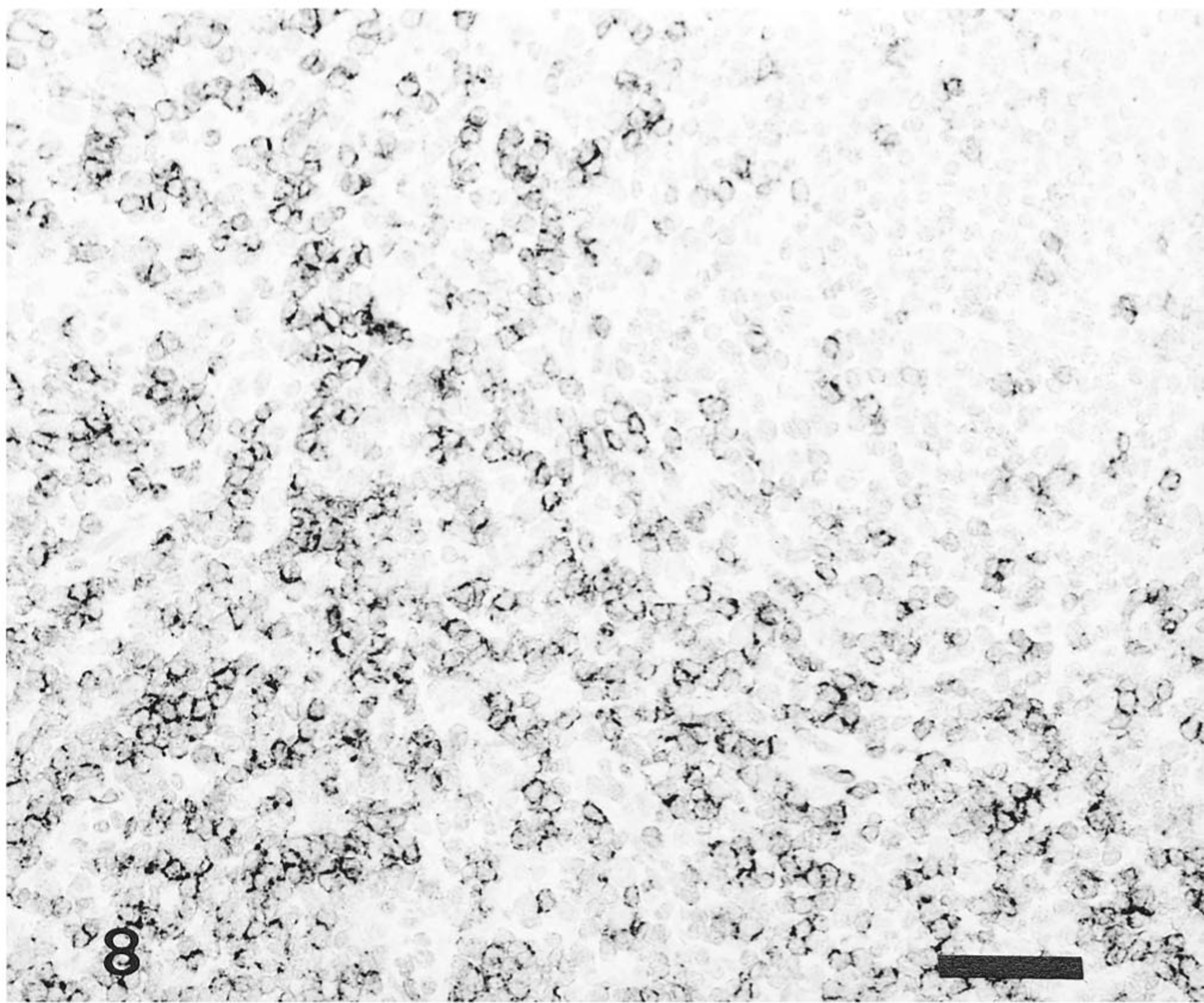
Lymph node; pig No. 12. CD8+ T lymphocytes in interfollicular areas of pig infected with Kanagawa/74 strain. Anti-CD8+T lymphocyte ABC. Bar = 50 μm.
There was an almost complete loss of B lymphocytes in the lymphoid follicle during the acute stage of infection with the ALD strain, but reduction of B lymphocytes was less severe at the chronic stage of infection with the Kanagawa/74 strain.
Discussion
Most HCV-infected animals die with widespread petechiation, thrombosis, splenic infarction, and encephalitis within 3 weeks of infection. 12 16 In the present study, pigs infected with the ALD strain of HCV developed severe illness characterized by depression, high fever (>41 C), anorexia, and leukopenia leading to fatal hemorrhagic illness. They had histologic lesions that included severe generalized vasculitis, necrosis of lymphocytes, and encephalitis. These clinicopathologic findings closely resembled those in pigs infected with highly virulent HCV strains. 9 10
Persistent infection with HCV has been observed under both natural and experimental conditions, suggesting that HCV may persist in host tissues throughout the course of an illness. Chronic HC had been divided into three clinical phases: an early acute reaction characterized by anorexia, depression, and fever; a period of general clinical improvement; and a period of relapse and death. 5 The most prominent histologic lesions caused by persistent HCV infection are atrophy of the thymus, slight depletion of lymphocytes, plasmacytosis, and extramedullary hematopoiesis. 13 17 By contrast, the gnotobiotic piglets infected with the Kanagawa/74 strain of HCV did not develop illness at the acute stage of infection but had moderate necrosis of endothelial cells, depletion of lymphocytes, hyperplasia of histiocytes, and plasmacytopoiesis. 7 In the present study, the pigs infected with the Kanagawa/74 strain all survived 30 days and had anorexia, depression, fever, and slight leukopenia during the acute phase of illness. All infected pigs recovered from the disease by PID 30 but they were stunted. The most significant histologic lesions were slight follicular depletion of lymphocytes, histiocytic hyperplasia, hematopoiesis in the spleen, and cortical depletion of thymic lymphocytes. Therefore, the characteristics of the histologic lesions at PID 30 in pigs infected with the Kanagawa/74 strain resembled those observed in cases in the second phase of HCV persistence.
The organic and chronologic distribution patterns of HCV antigen in infected tissues are well known from several immunohistologic studies using immunofluorescence techniques on frozen tissue samples. 2 10 HCV antigen, 6 and antigens of many other viruses, can be identified in formalin-fixed tissues by immunohistochemistry. Pigs acutely infected with the ALD strain had HCV antigen detectable first in crypt tonsilar epithelial cells, follicular macrophages, reticular and endothelial cells in the spleen and lymph nodes, and smooth muscle cells of the blood vessels. Localization of the antigen corresponded to the areas of histologic lesions similar to those of previous reports. 2 5 13 17 Pigs chronically infected with the Kanagawa/74 strain had HCV antigen in the tonsil, spleen, lymph nodes, pancreas, ileum, lungs, and adrenal glands. This finding resembled the distribution of moderately virulent HCV in dead pigs from PID 41 to PID 70. 2 The immunohistochemical staining patterns of the two strains of virus are strikingly similar given the difference in clinical disease caused by the two isolates. Moreover, there is a strong immunohistochemical positivity with the Kanagawa/74 strain, and areas of staining corresponded very closely in pattern and intensity with the staining seen with the ALD strain near the time of death. Therefore, the high concentration of HCV antigen within the tissue infected with the Kanagawa/74 strain might persist for longer periods in target cells until the pig relapses and dies. Widespread distribution of HCV antigen, without concomitant tissue lesions, resembled the distribution of bovine viral diarrhea virus antigen in aborted calves with weak calf syndrome. 1 3
Classical swine fever affects the immune system mainly by causing generalized leukopenia. 8 13 Lymphoid organs are also affected by infection, and deficiency of B lymphocytes has been recorded. 14 A general dysfunction of T lymphocyte activity has also been observed. 8 In the present study, an almost complete loss of B lymphocytes in the lymphoid organs was noted in the pigs infected with the ALD strain of HCV, but this loss was not so prominent in pigs infected with the Kanagawa/74 strain and euthanatized on PID 30. These results are consistent with the histopathologic lesions of follicular depletion of lymphocytes and/or necrosis in pigs infected with the ALD strain of HCV. The number of CD4+ and CD8+ T lymphocytes was significantly higher in pigs infected with both strains of HCV than in the noninfected control pigs. The B-lymphocyte deficiency was probably caused by viral destruction of the germinal centers in lymphoid tissues and was the most significant immunopathologic consequence of acute infection with the virulent strain of HCV. HCV appears to alter the immune mechanisms of the pigs, causing the B-cell reaction and increasing T lymphocytes in chronic stages.
Footnotes
Acknowledgements
We thank Mr. M. Kobayashi and Miss M. Shimada for preparing the histologic tissue sections, and Dr. Y. Ando and Mr. T. Fujisawa for preparing the photographs.
