Abstract
Broiler chickens with transmissible proventriculitis have severe lymphocytic infiltration of the proventricular glands. The distribution of T cells and B cells in these infiltrates was studied histopathologically, and their identity was confirmed immunohistochemically (CD3, CD4, CD8, and B cells). To reproduce this disease, 1-day-old commercial boilers were orally gavaged with homogenized proventriculi from broilers with proventriculitis. Resulting lesions were examined at both acute (7 days postinoculation [dpi]) and chronic (14 and 21 dpi) time points. Lymphocytic infiltrates in the proventricular glands and the mucosal lamina propria were present at all time points and were most prominent and demarcated at 14 dpi. T and B lymphocytes were present during acute and chronic proventriculitis, but their distribution varied within the glands. Lymphocytic infiltrates in the proventricular glands and in the lamina propria were predominantly CD3+ T cells, and most of these were also CD8+. B cells and CD4+ T cells formed aggregates in chronic proventriculitis. Thus, both cell-mediated and humoral immune responses are induced during transmissible proventriculitis, and the cell-mediated immune response is morphologically greater.
Proventriculitis is a transmissible disease that occurs in commercial broiler chickens. It is characterized by enlargement of the proventriculus, retention of ingesta in the lumen, and weakness of the gastric isthmus. During routine evisceration at processing, affected proventriculi rupture at the isthmus and retained ingesta spills into the body cavity, which results in condemnation of affected carcasses for contamination. The disease has also been associated with impaired growth and poor feed conversion. 10,11 Microscopically, degeneration and necrosis of the proventricular glandular epithelium is accompanied by marked lymphocytic infiltration. 4,10,11,20
The etiology of proventriculitis is currently unknown. Several agents have been implicated as potential causes. Noninfectious causes include oral exposure to biogenic amines, 2 mycotoxins, 21 lack of dietary fiber, 22 and excessive copper sulfate. 5,12 Infectious agents that have been implicated include adenovirus, 15 reovirus, 14 infectious bronchitis virus (IBV), 29 infectious bursal disease virus, 4,11 and megabacterium. 24 However, none of these noninfectious or infectious agents have been found consistently. 19 Viral particles have been detected in acute lesions using electron microscopy, but isolation of a virus from affected proventriculi has been unsuccessful. 10,11 Proventriculitis has been reproduced by inoculation of proventricular homogenates produced from diseased chickens 4,11,20 and bacteria-free filtrates of these homogenates, showing that the initiating cause is transmissible and viral. 11 However, lesions are more severe after exposure to unfiltered homogenate, suggesting that proventriculitis has a complex etiology involving both viral and bacterial agents. 11
Transmissible proventriculitis is characterized by a marked lymphocytic infiltrate in the proventricular glands. 4,10,11,20 The purpose of this study was to characterize the lymphocytes composing this infiltrate in order to determine what immunologic mechanisms chickens use to respond to this infection. To accomplish this we experimentally infected commercial broiler chickens with proventricular homogenates from affected broilers and studied the proventricular lesions using histopathology, by staining for lymphocyte cell-surface markers, and by identifying the distribution of these different lymphocyte subsets.
Materials and Methods
Chickens
One-day-old unvaccinated broiler chicks were obtained from a commercial hatchery. All chicks were wing banded, weighed, separated into groups, and maintained in positive pressure Horsfal isolation units. Feed and water were provided ad libitum.
Proventricular homogenates
An infectious proventricular homogenate (+ PV) and a noninfectious proventricular homogenate (− PV) were prepared from proventriculi of 2- to 4-week-old chickens as described previously. 20 In brief, proventriculi collected from chickens with proventriculitis were washed in sterile normal saline (phosphate-buffered saline [PBS]) three times on a magnetic stirrer to remove feed and toxins. Washed proventriculi were then diluted 1 : 1 wt/vol in PBS and homogenized with a commercial blender (Waring, New Hartford, Connecticut). The same procedure was followed using proventriculi from clinically normal 14-day-old commercial broilers, obtained from a local hatchery and raised in isolators, with no gross or microscopic signs of proventriculitis. Both homogenates were frozen at −70 C and thawed immediately before use.
Experimental design
Twenty-four 1-day-old commercial broilers were separated into two groups. The first group was inoculated by oral gavage with 1 ml of − PV. The second group received 1 ml of + PV. All chickens were observed daily for development of clinical signs. At 7, 14, and 21 days postinoculation (dpi), four birds from each group were weighed and killed by cervical dislocation. The bursa, proventriculus, and thymus from each bird were weighed. Relative organ weights were obtained using the formula (organ weight/body weight)100. A portion of each organ was fixed immediately by immersion in 10% neutral buffered formalin for 24 hours for histopathology and processed using routine histologic techniques and embedded in paraffin. The proventricular samples were trimmed by taking three longitudinal sections from the proventriculus of each chicken, each of approximately 3-mm thickness, and placing them into histology cassettes flat on their side. The remaining portion of each organ was placed in cryostat embedding media (Cryo-Gel, Instrumedics, Inc., Hackensack, New Jersey) and immediately frozen in liquid nitrogen and kept at − 70 C for preparation of frozen sections for immunohistochemistry.
Histopathology
Paraffin-embedded tissues were sectioned, mounted, stained using hematoxylin and eosin, and examined, with the investigator blinded to treatment, for lesions using light microscopy. All sections were assigned a lesion severity score as described previously. 20 In brief, for all tissues, a lesion score of 1 represented no lesions. For bursal sections, 2 was defined as mild variation in follicle size, 3 as moderate variation in follicle size, and 4 as either necrosis or follicle atrophy. For thymic sections, 2 was defined as mild cortical thinning, 3 as moderate cortical thinning, and 4 as absence of cortical lymphocytes. For proventricular sections, 2 was defined as mild glandular lumenal ectasia, 3 as ectasia, necrosis of the glandular epithelium, plus lymphoid infiltrates in the interglandular interstitium, and 4 as either acute glandular necrosis or ductal metaplasia with lymphoid infiltrates. The total proventricular mural thickness was measured on the slide with a millimeter ruler at the widest point on each proventricular histologic section, and an average thickness per proventriculus per bird was obtained.
Monoclonal antibodies
Monoclonal antibodies for T lymphocytes (Southern Biotechnology Associates Inc., Birmingham, Alabama) were mouse anti-chicken CT-3 (anti-CD3), CT-4 (anti-CD4), and CT-8 (anti-CD8). HisCl antibody (Cedi Diagnostics BV, Lelystad, The Netherlands) was used for B lymphocytes.
Immunohistochemistry
Optimal conditions for immunohistochemical staining with each monoclonal antibody were determined using bursa and thymus tissues from normal chickens. All antibodies stained cell populations in positive control tissues with equal intensity. These tissues were included as controls during the staining of each group of slides. Frozen tissue blocks were sectioned at 5 µm using a cryostat; replicate sections were mounted on charged slides (Superfrost Plus, Fisher Scientific, Pittsburgh, Pennsylvania). These were fixed immediately in acetone for 10 minutes and stored at −70 C until stained. Immunostaining was accomplished using a nonbiotin peroxidase kit (DAKO Envision System, DAKO, Carpinteria, California), according to the manufacturer's recommendations. In brief, the sections on slides were placed in a moist chamber and washed for 5 minutes in 0.1 M PBS, followed by incubation for 5 minutes in peroxidase-blocking reagent (DAKO Envision System). Sections were then washed in PBS for 5 minutes and incubated with one of the monoclonal antibodies at 4 C overnight (CD3, CD4, and CD8 were used at a dilution of 1 : 100 and HisCl at a dilution of 1 : 50). After primary antibody incubation, sections were washed in PBS for 5 minutes and incubated with the secondary antibody (peroxidase-labeled polymer conjugated to goat anti-mouse immunoglobulin, DAKO Envision System) at room temperature for 45 minutes. After washing in PBS for 5 minutes, bound antibody was detected by a 5- to 10-minute incubation with 3,3′-diaminobenzidine substrate-chromagen (DAKO Envision System). Finally, sections were counterstained with hematoxylin, air dried, coverslipped, and examined using light microscopy. All sections were evaluated independently by two observers (M. J. Pantin-Jackwood, T. P. Brown) without knowledge of the treatment group or stage of infection. The predominant subset of stained lymphocytes was identified and localized in each organ examined.
Statistical analysis
The relative organ weights and lesion scores were analyzed using one-way analysis of variance and comparison of means for all pairs using the Tukey-Kramer test. Significance was set at a 0.05 level of probability.
Results
Clinical signs and macroscopic lesions
No clinical signs were observed in any of the chickens in control or experimental groups. Gross lesions were observed in all proventriculi from + PV–inoculated chickens. At 7 and 14 dpi proventriculi were enlarged with a mottled appearance and a distended gastric isthmus. At 21 dpi, the enlargement was still present but was less severe. No proventricular enlargement was present in chickens given − PV. The proventricular wall of chickens inoculated with the + PV was thickened (Table 1), with an intramural white lobular pattern. No macroscopic lesions were observed in any other organ of experimentally infected or control chickens.
Body weight gain, relative proventriculus weight, proventriculus lesion score, incidence of proventriculitis, and proventriculus wall thickness of commercial broilers orally challenged at 1 day of age with a negative proventricular homogenate (− PV) or a positive proventricular homogenate (+ PV) and necropsied at 7, 14, and 21 dpi (mean ± standard deviation).∗.
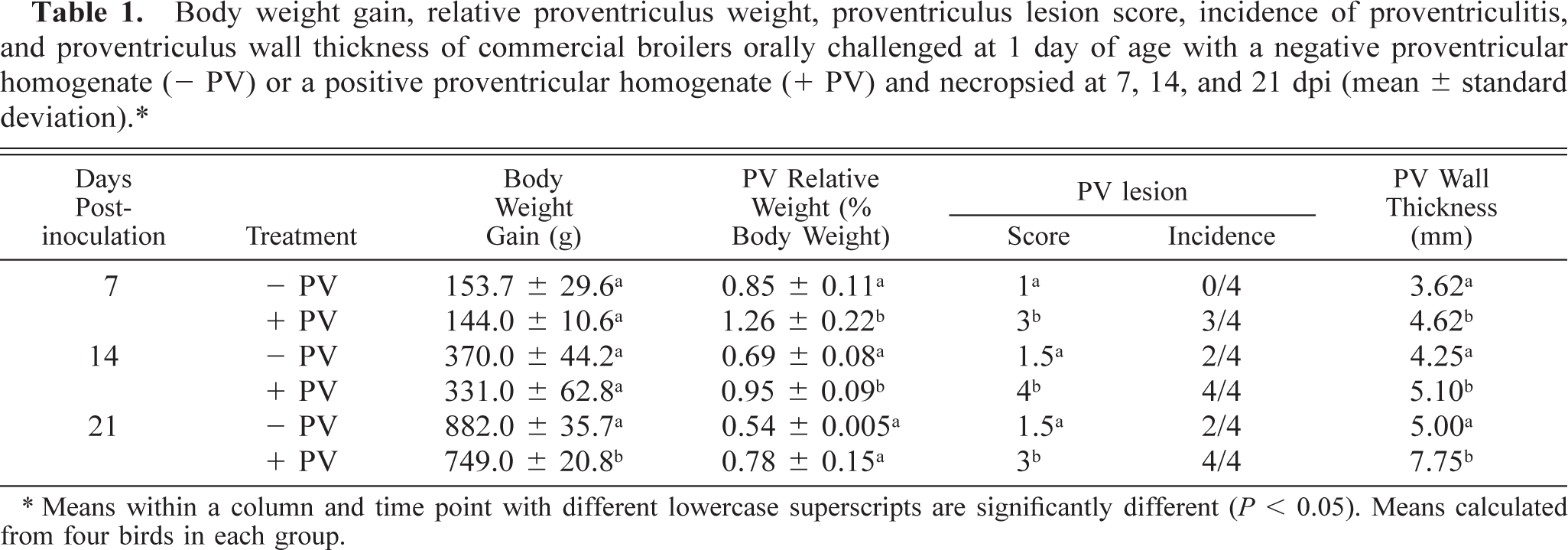
∗ Means within a column and time point with different lowercase superscripts are significantly different (P < 0.05). Means calculated from four birds in each group.
Body weight gain
At 7 and 14 dpi, chickens inoculated with + PV had no significant suppression of weight gain compared with age-matched chickens given − PV. At 21 dpi, there was suppression of body weight gain in birds that received + PV (Table 1).
Organ weights
Chickens that received + PV had increased proventricular organ/weight ratio at 7 and 14 dpi compared with those receiving − PV (Table 1). There was no difference in the bursa and thymus organ/weight ratio between birds inoculated with + PV and those given − PV (data not shown).
Microscopic lesions
No lesions were present in the proventricular glands of the − PV–treated birds at 7 dpi. Proventricular lesion scores in − PV–treated chickens were the same at 14 and 21 dpi and were significantly milder than those present in chickens given + PV (Table 1). Lesions present in the − PV–treated chickens consisted of mild lumenal ectasia of the glands. Lymphocytes were present at all time points in the lamina propria of the mucosa, especially in areas surrounding the orifices of the excretory ducts of the proventricular glands (Fig. 1). By 21 dpi, these lymphocytes formed small aggregates in the proventricular glands (Fig. 2).
Proventriculus; chicken inoculated with negative proventricular homogenate (− PV) at 7 dpi. Lymphocytic infiltration in the lamina propria of the mucosa and surrounding the orifice of the secretory duct (arrows). Oxynticopeptic cells form the glandular epithelium (insert). HE.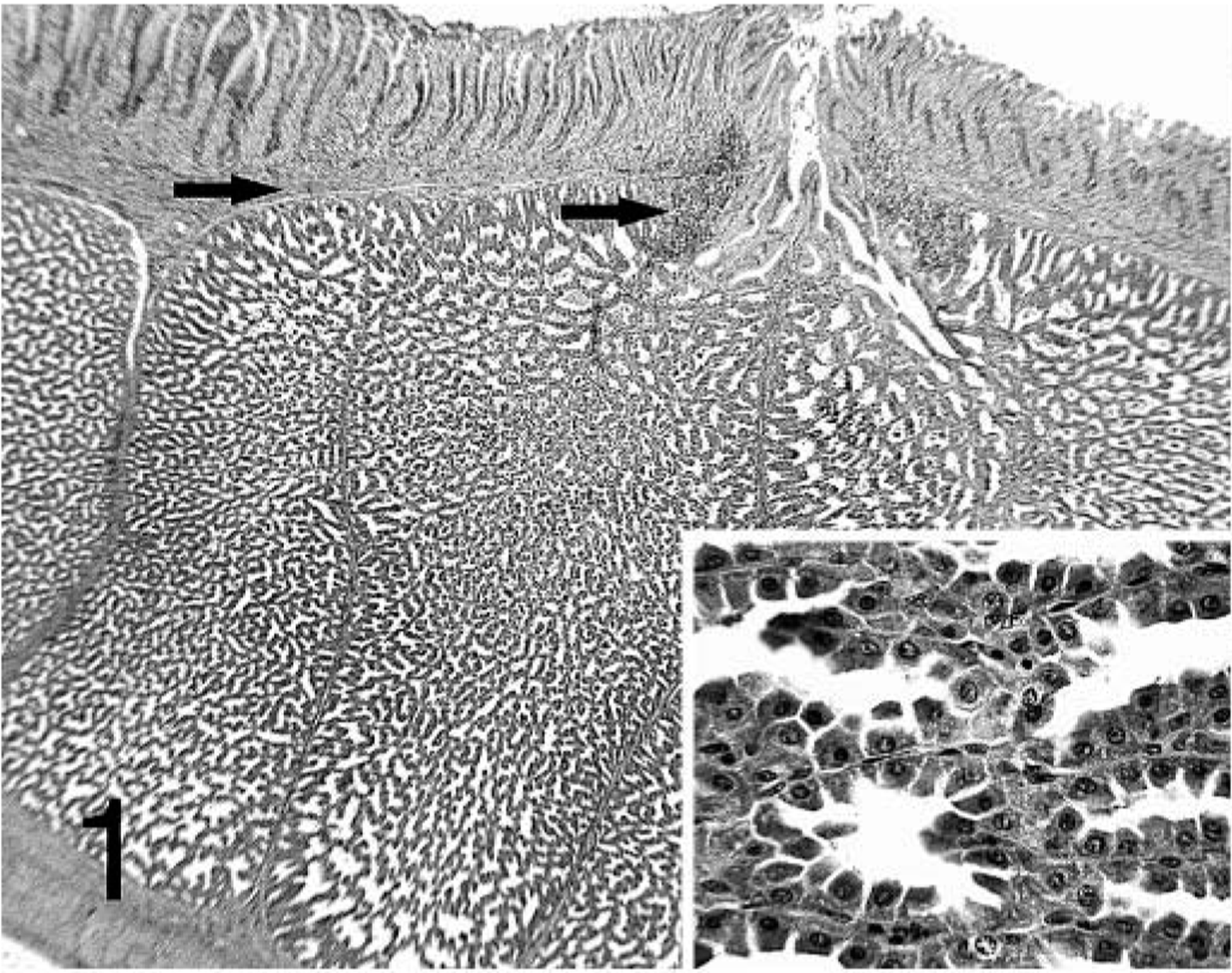
Proventriculus; chicken inoculated with negative proventricular homogenate (− PV) at 21 dpi. Small lymphocyte aggregates are present in the proventricular gland (arrows). HE.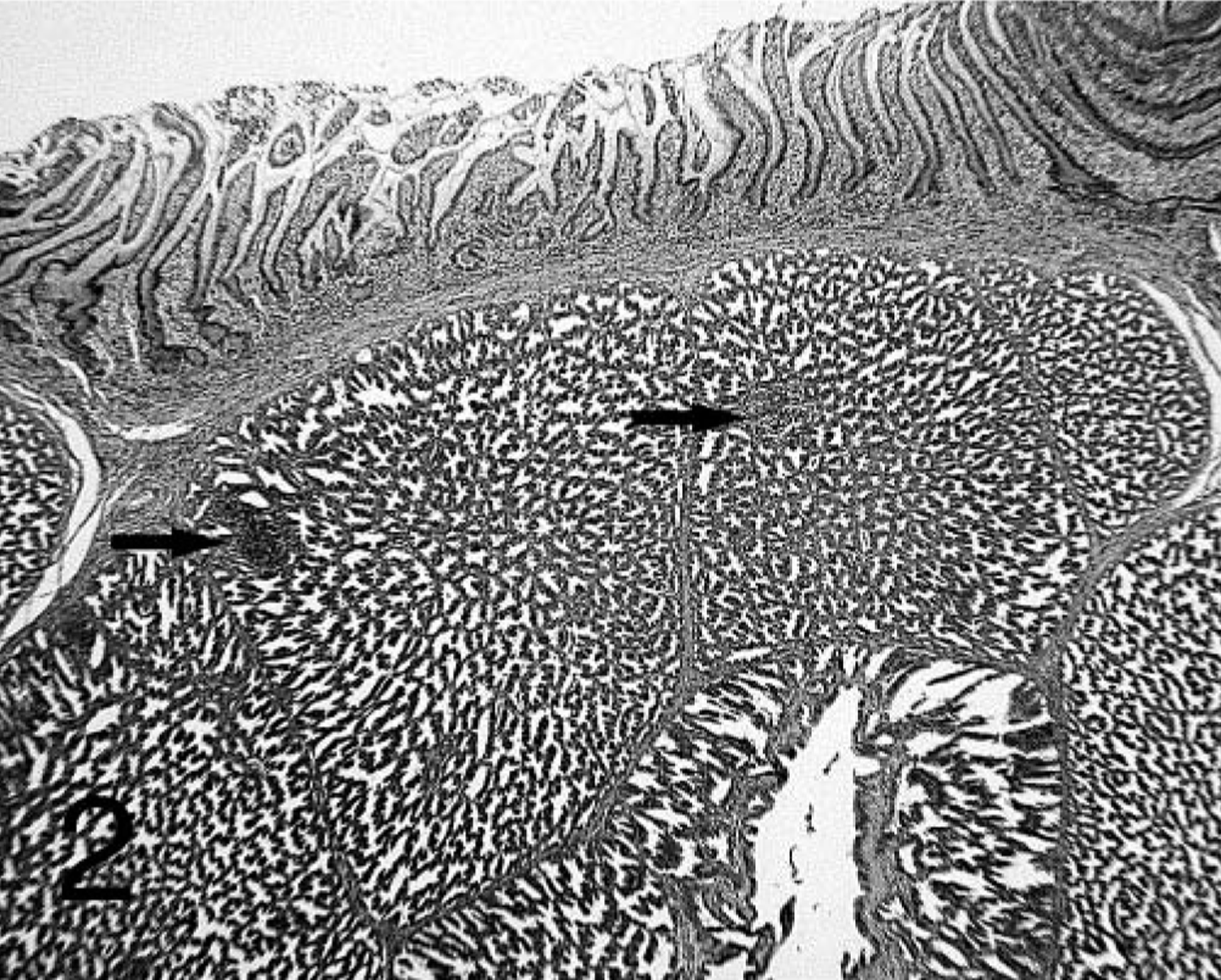
Lesions were present in the proventriculus of all chickens that received + PV. No lesions were present in the thymus and bursa from these birds (data not shown). Also, the incidence of lesions in the proventriculus was higher in chickens that received + PV compared with those receiving − PV (Table 1). Proventriculi of chickens exposed to + PV had necrosis of the glandular epithelium at 7 dpi (Fig. 3). Collecting sinuses of the glands were dilated and contained desquamated epithelium and debris. Nuclei of glandular epithelium were enlarged and pale, with marginated chromatin. At 7 and 14 dpi, lymphocytic infiltrates were present in large numbers in the lamina propria of the mucosa and also in affected glands expanding the glandular interstitium (Fig. 4). At 14 and 21 dpi, the glandular epithelium in the affected glands was replaced by hypertrophic and hyperplastic ductal epithelium (Figs. 5, 6). At 21 dpi, there was also less necrosis of glandular epithelium, but lymphocytes were still forming aggregates or germinal centers in affected glands and segments of the mucosa (Fig. 6).
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 7 dpi. Severe necrosis of the glandular epithelium, dilation of sinus with desquamated epithelium, and lymphocytic infiltration of the proventricular gland (arrow). Nuclei of glandular epithelium are enlarged and pale, with marginated chromatin (insert). HE.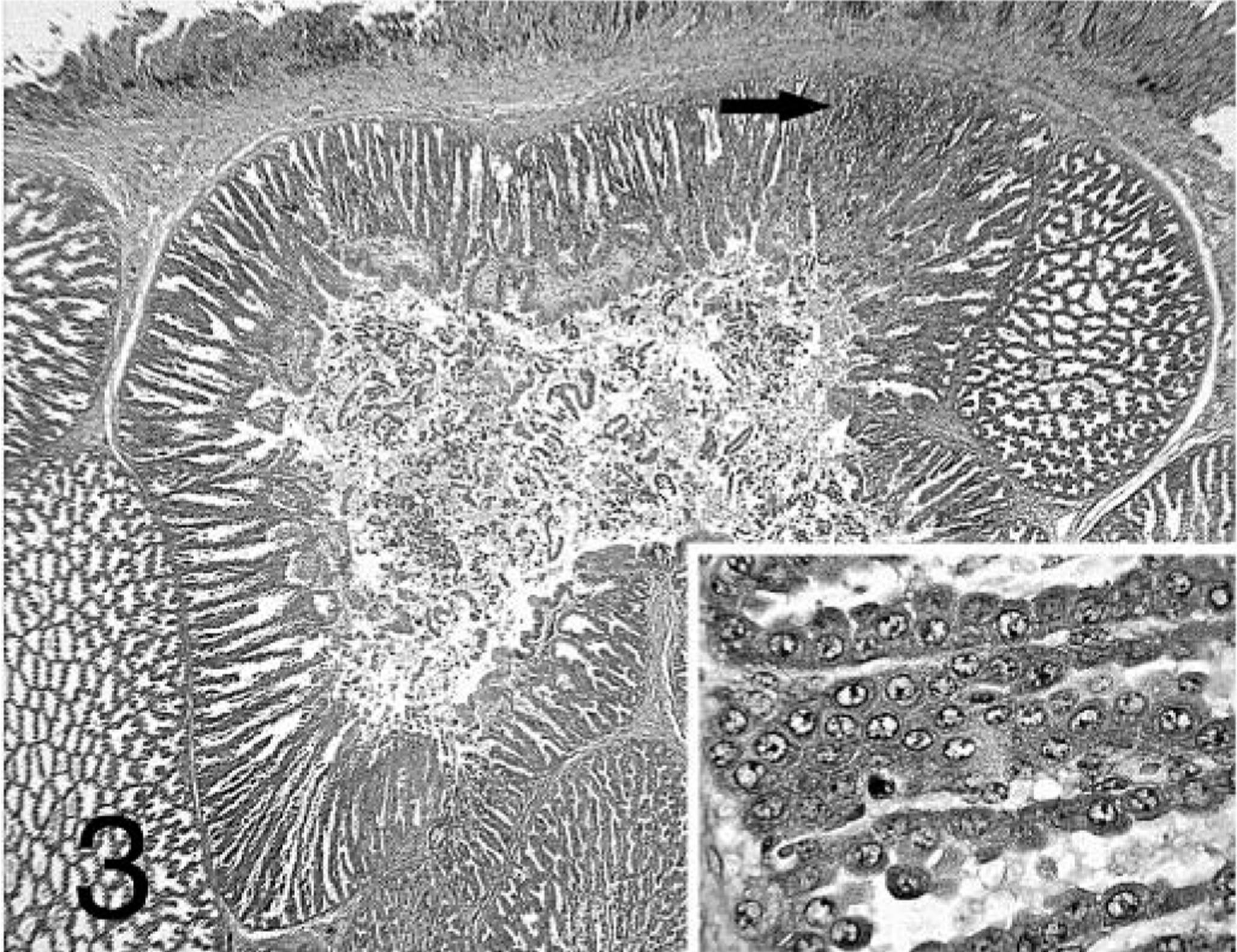
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. Sheets of lymphocytes in the proventricular gland and the lamina propria of the mucosa. HE.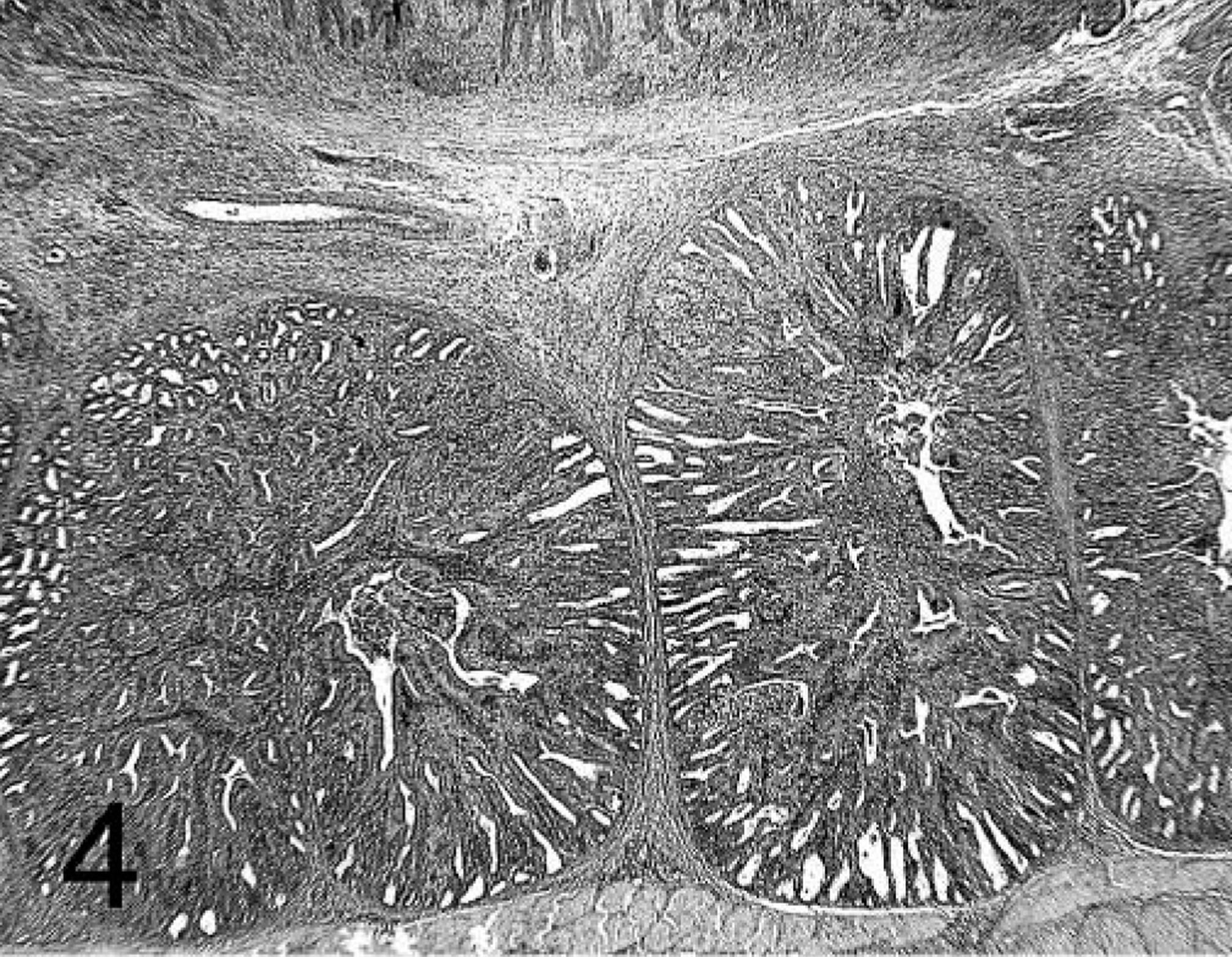
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. Diffuse lymphocytic infiltration in the proventricular gland and the lamina propria of the mucosa. Ductal epithelium replacing glandular epithelium (insert; area marked with asterisk). HE.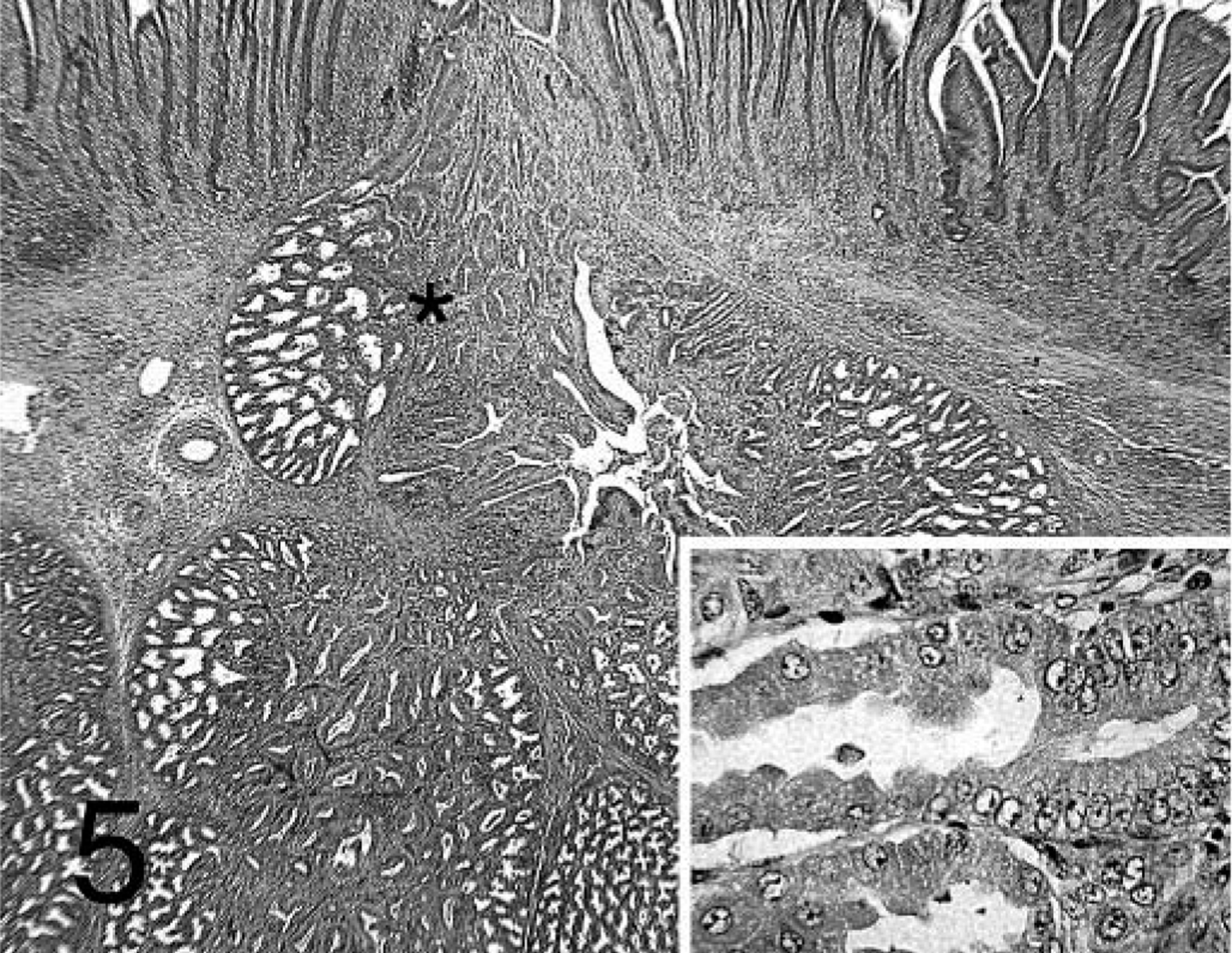
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 21 dpi. There is less glandular epithelial necrosis than at 7 and 14 dpi. Lymphocytes in affected proventricular glands, and the lamina propria of the mucosa form well-demarcated aggregates. HE.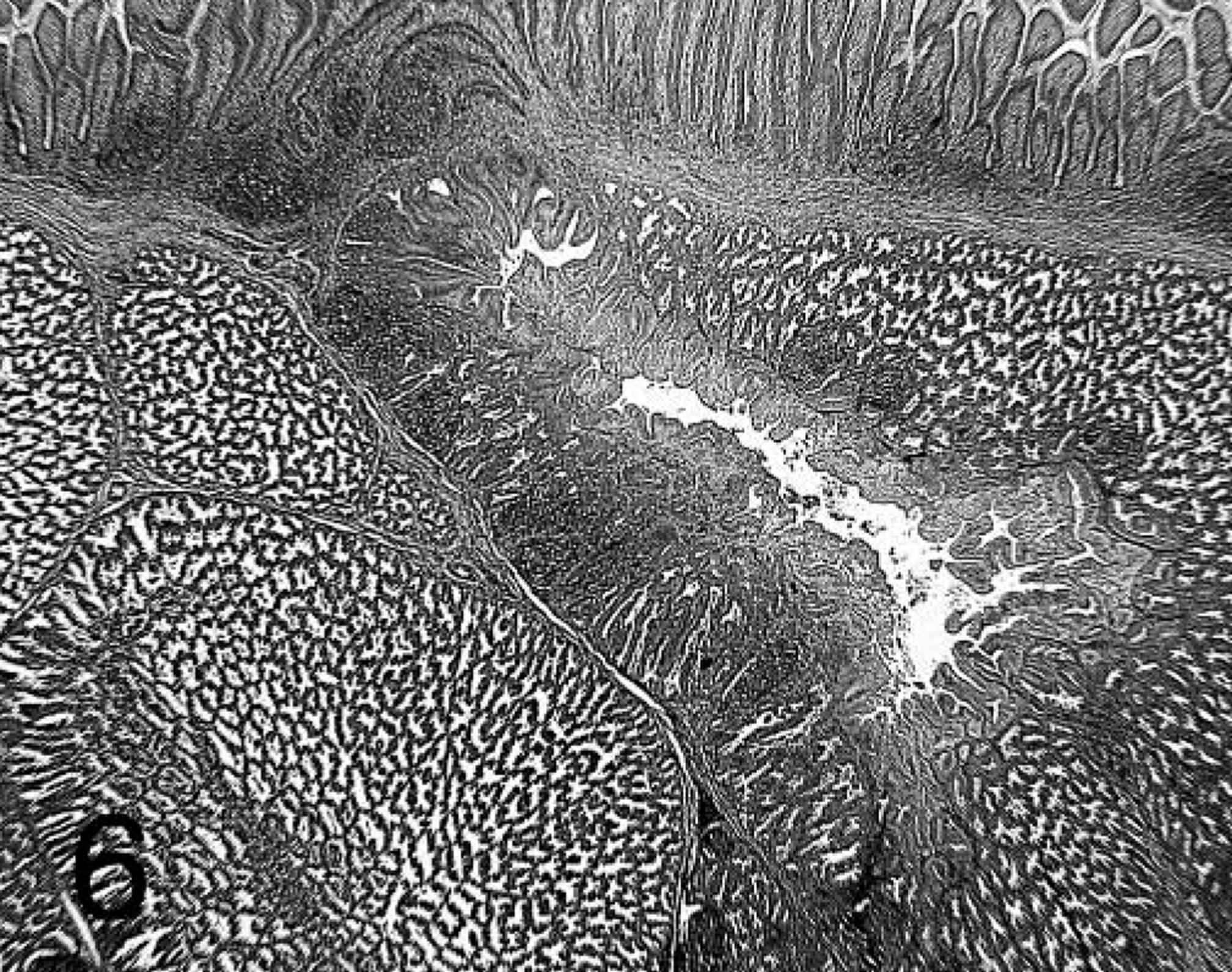
Localization of CD3+, CD4+, CD8+, and B cells
Both T and B cells were present in the lamina propria of the proventricular mucosa of chickens treated with − PV (Figs. 7, 8). Most lymphocytes in the proventricular glands were T cells, and were localized to the interstitium between the glands and intraepithelially as individual lymphocytes. Small lymphoid aggregates were present in the glands at 14 and 21 dpi and were mostly composed of B cells.
Proventriculus; chicken inoculated with negative proventricular homogenate (− PV) at 7 dpi. B-cell staining mostly found in the lamina propria of the mucosa. Immunohistochemistry staining: nonbiotin peroxidase method. Hematoxylin counterstain.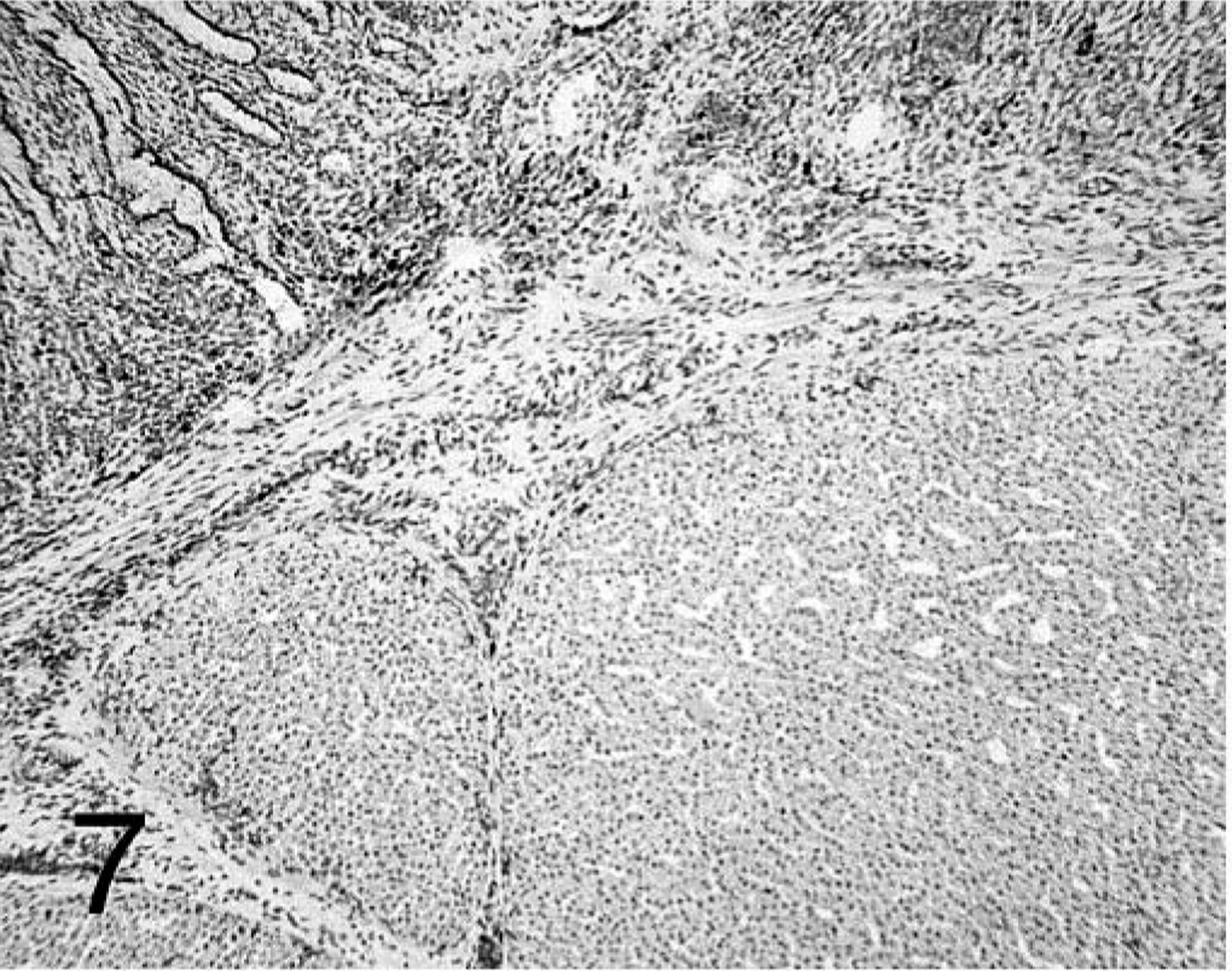
Proventriculus; chicken inoculated with negative proventricular homogenate (− PV) at 7 dpi. CD3+ T-cell staining in the lamina propria of the mucosa, interstitium between proventricular glands, and deep in the glands. Immunohistochemistry staining: nonbiotin peroxidase method. Hematoxylin counterstain.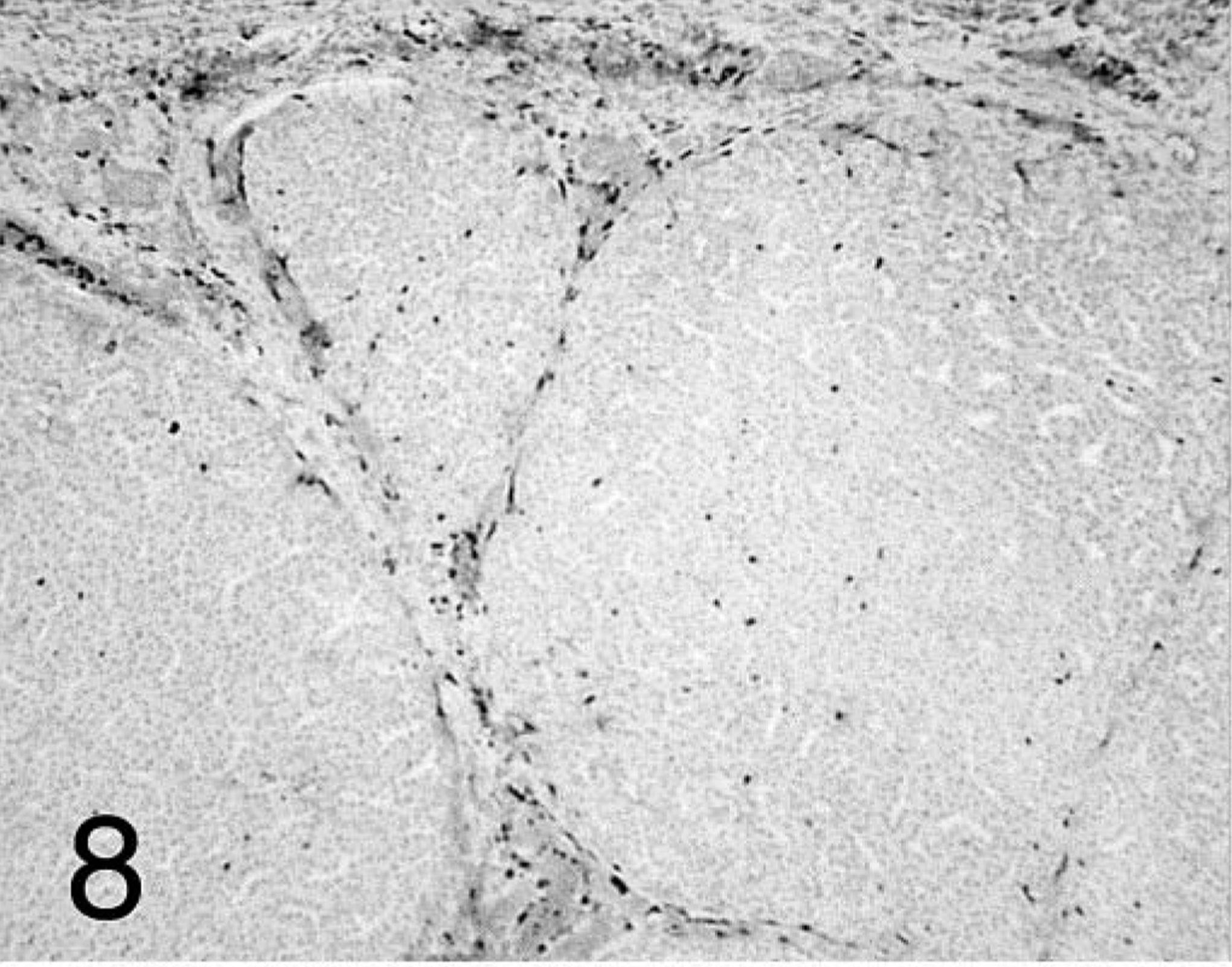
In chickens that received + PV, T cells predominated at all time points and were dispersed within the lamina propria of the mucosa and in deeper areas of proventricular glands. B cells were also present, and their distribution varied depending on the stage of the proventriculitis. Initially, B cells were localized similar to T cells but in lower numbers. As the proventriculitis progressed, B cells formed highly organized aggregates (germinal centers) in the proventricular glands and in the lamina propria of the mucosa (Fig. 9). T cells surrounded these germinal centers and infiltrated the proventricular glands and the mucosa (Fig. 10).
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. B-cell staining observed in the forming germinal centers in the proventricular glands. Immunohistochemistry staining: nonbiotin peroxidase method. Hematoxylin counterstain.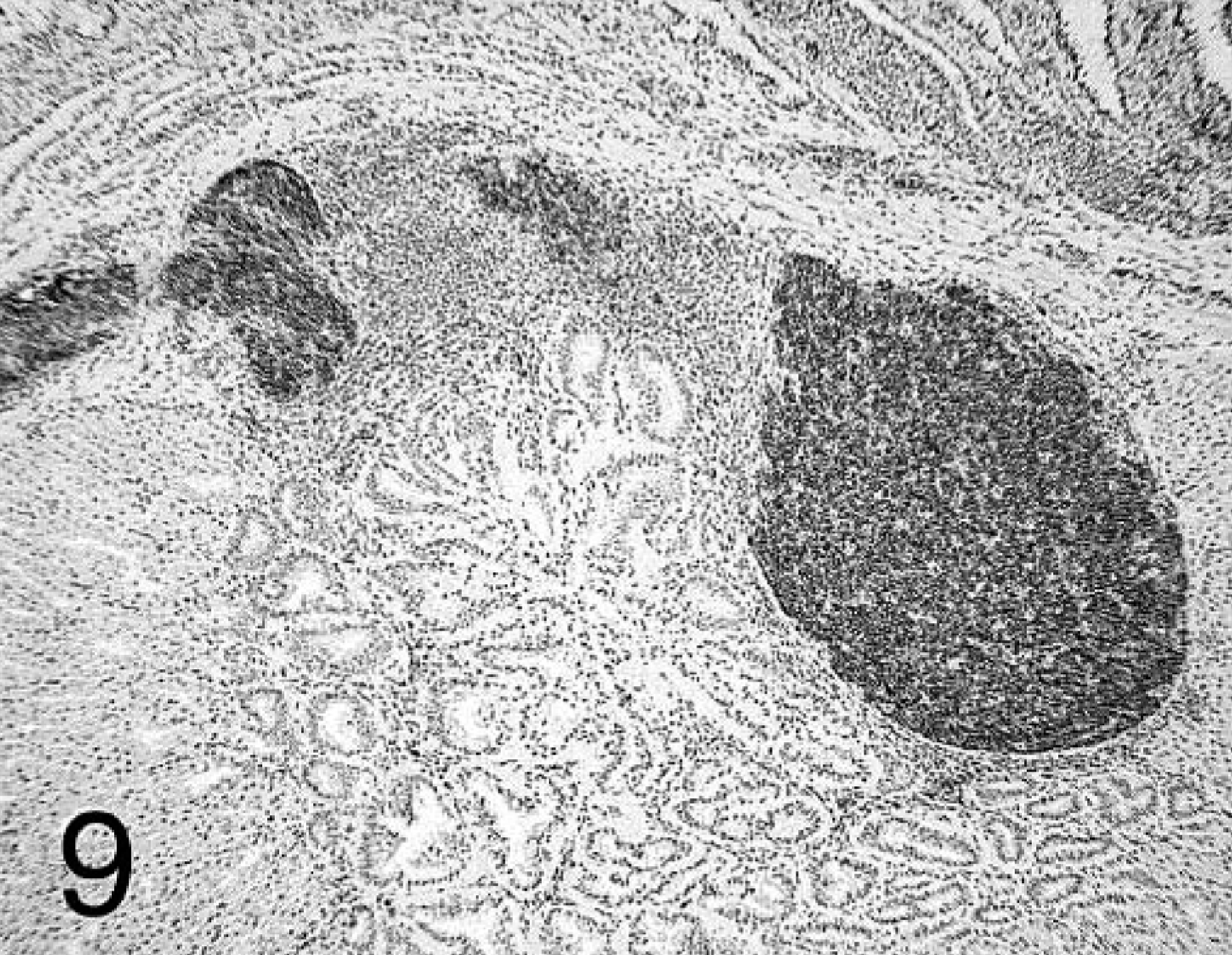
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. CD3+ T-cell staining in the lamina propria of the mucosa and deep in the proventricular glands. Immunohistochemistry staining: non-biotin peroxidase method. Hematoxylin counterstain. 25×.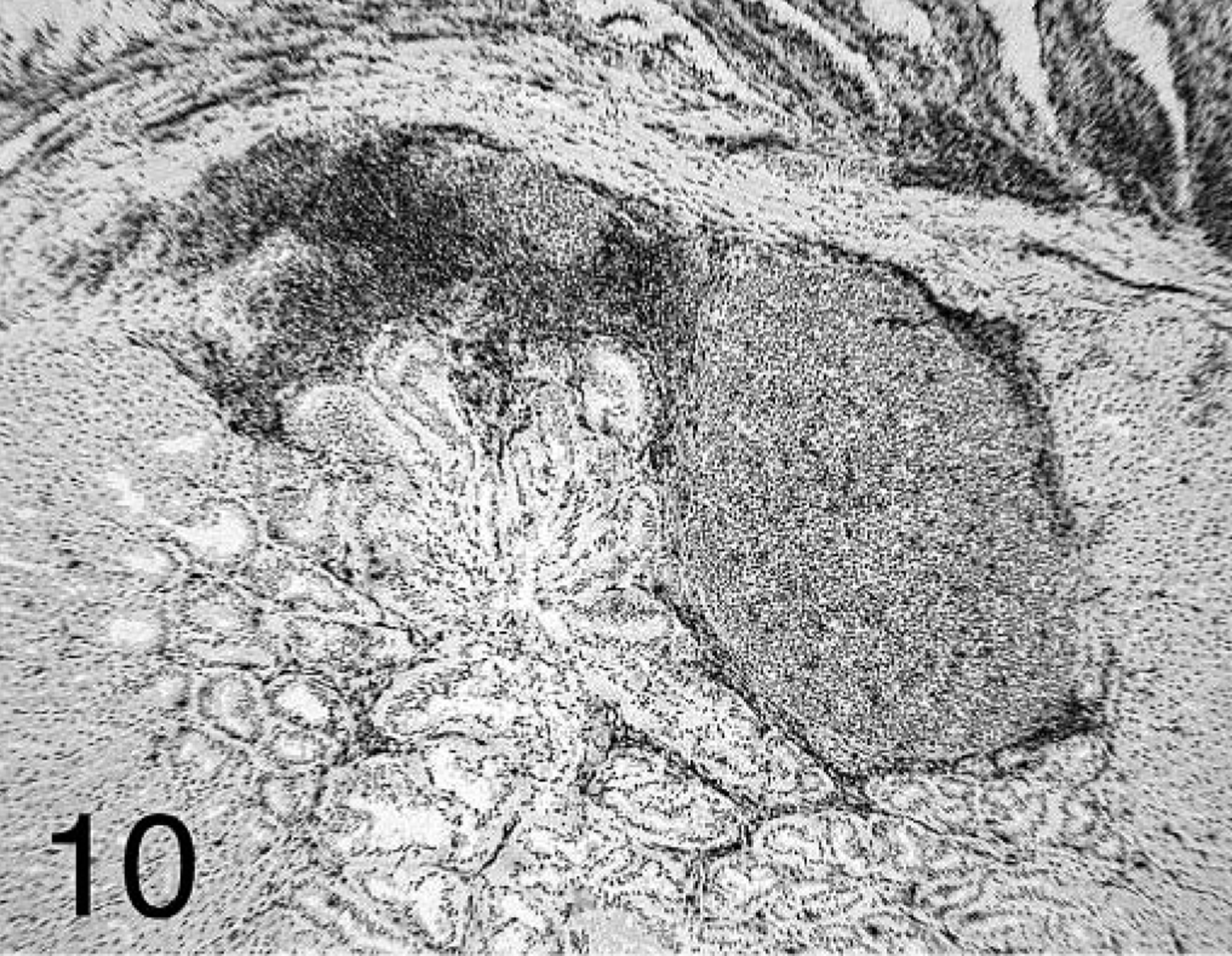
The two subsets of T lymphocytes studied (CD4+ and CD8+) were distributed differently in affected proventriculi. Both subsets were found at all time points in large quantities in the lamina propria of the mucosa, with CD4+ predominating at 7 dpi. In glands with acute necrosis of the epithelium, CD8+ lymphocytes were the predominant cells found. At 14 and 21 dpi, CD4+-positive cells were found mostly surrounding and infiltrating the B-cell germinal centers but also forming aggregates in the deep proventricular glands (Fig. 11). The CD8+ cells were more widely distributed in the mucosa and infiltrating the proventricular glands in the intraepithelial spaces (Fig. 12).
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. CD4+ T-cell staining in the lamina propria of the mucosa and the proventricular glands surrounding and in the germinal centers. Immunohistochemistry staining: nonbiotin peroxidase method. Hematoxylin counterstain.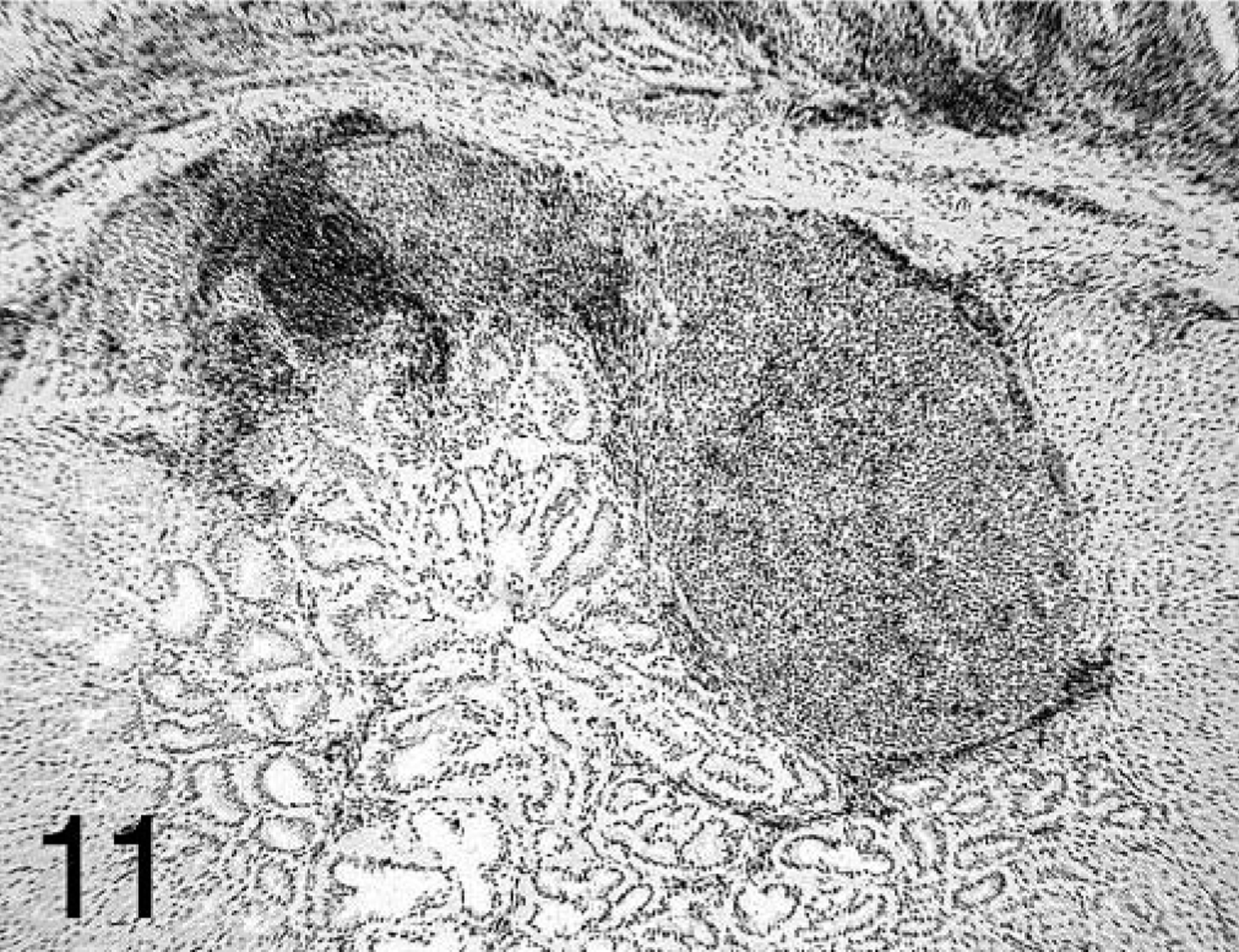
Proventriculus; chicken inoculated with positive proventricular homogenate (+ PV) at 14 dpi. CD8+ T-cell staining in the lamina propria of the mucosa and deep in the proventricular glands. Immunohistochemistry staining: non-biotin peroxidase method. Hematoxylin counterstain.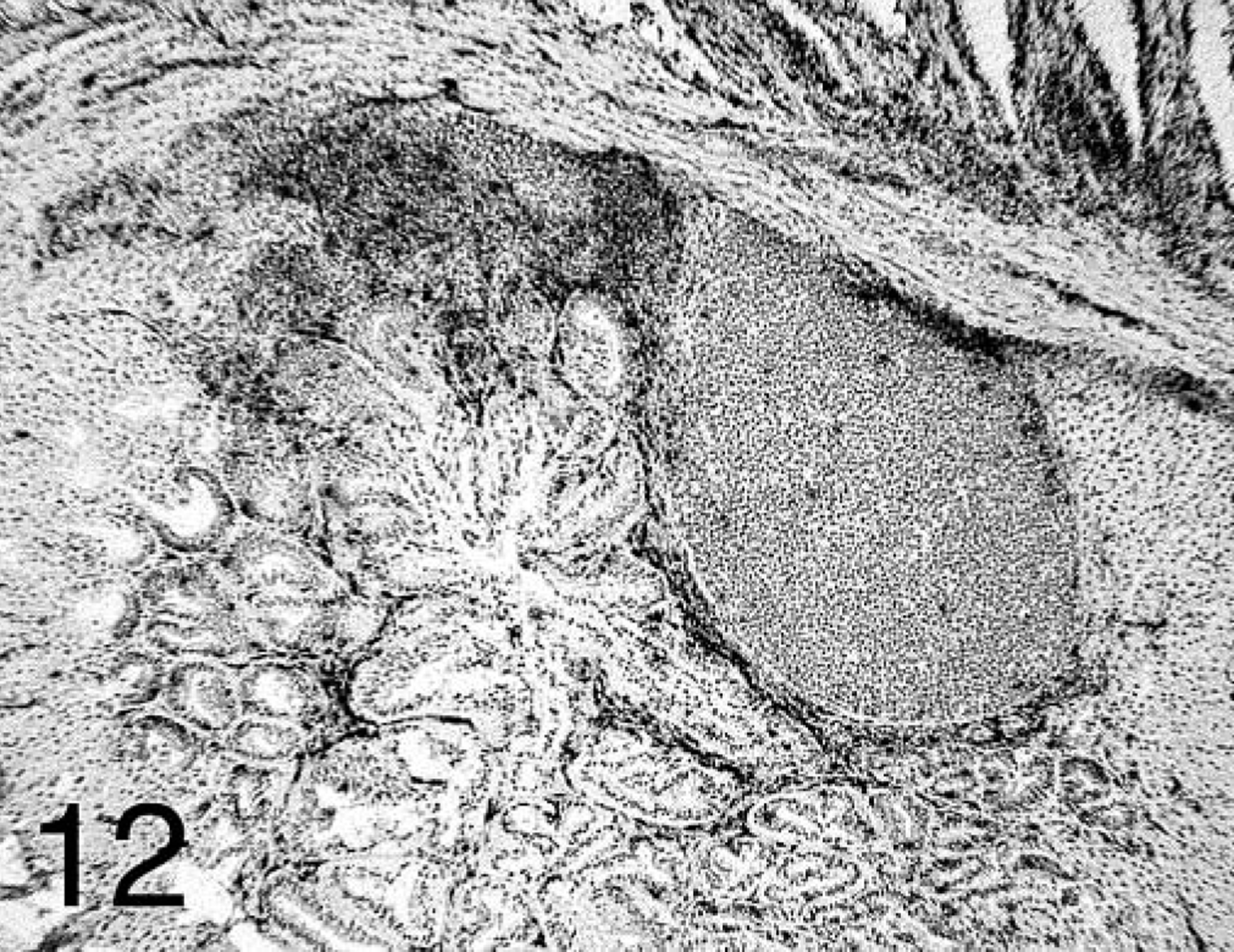
Discussion
In this study, lymphocyte subpopulation changes during proventriculitis were investigated. Proventriculitis was successfully reproduced by inoculation with a proventricular homogenate derived from proventriculi collected from broiler chickens affected with proventriculitis (+ PV). Microscopic changes in these proventriculi included necrosis of the glandular epithelium and replacement of this epithelium with ductal epithelium. This loss of glandular tissue and ductal hyperplasia may result in loss of function of the proventriculus. 10 This would explain the poor feed conversion and reduced growth rates observed in naturally affected chickens with proventriculitis and the reduced body weight observed in our experimental chickens at 21 dpi.
Severe lymphocytic infiltration was observed in all experimentally infected chickens. The distribution of these lymphocytes in the proventriculus varied. In the acute or early stages, lymphocytes were present as sheets in large numbers in the lamina propria of the mucosa and infiltration affected glands. In the later stages, the lymphocytes formed aggregates in both the lamina propria of the mucosa and deep in the proventricular glands. These chronic changes were accompanied by less necrosis and ductal metaplasia and hyperplasia. Staining of these lymphocytes showed that both B and T cells are increased in number during proventriculitis but occupied different histologic locations within the proventriculus depending on the stage of the disease.
Lymphocytes are present in the mucosa of normal chicken organs as the mucosal-associated lymphoid tissue. 3 This complex immune apparatus has developed in the chicken in response to agents entering the body through mucosal surfaces lining the respiratory, digestive, and genitourinary tracts, providing a first line of defense against these agents. Matsumoto and Hashimoto 17 described the normal distribution and developmental changes of the lymphoid tissues in the chicken proventriculus. They observed the development of lymphoid masses in the proventricular lamina propria underneath the surface epithelium and near the duct orifice, which suggested that the local mucosal immune mechanism develops primarily with a dominant participation of T lymphocytes in the early posthatching period. The development of B lymphocytes occurs after the invasion of antigens associated with food intake because of immunologic information from the prerequisite T lymphocytes. In our study, the response to a nonidentified infectious agent present in our + PV homogenate induced proliferation of the lymphoid tissue present in the proventriculus. This immune response was similar to that observed in the mucosa of other organs in response to different pathogens 3,6,8,16,18,23,25,27,28 . Intraepithelial lymphocytes could be observed in the deep proventricular gland, and Matsumoto and Hashimoto 17 identified them as γδ T lymphocytes, similar to those found in the chicken intestine. These authors could not demonstrate the presence of M cells in the proventriculus, suggesting that there are alternative routes for uptake of intraluminal antigens.
The cause of proventriculitis is not known, but it seems most likely that a virus is the primary agent involved. 4,11 T-cell–mediated immune responses to viral pathogens are well established and occur by a number of mechanisms, including induction of cytotoxic activity, recognition of target antigens in conjunction with the major histocompatibility complex, and production of lymphokines such as interferon-γ, interleukin-2, and tumor necrosis factor-β 13,26 Cells mediating these different activities can be identified by cell-surface antigens, CD4+ for helper T cells, CD8+ for cytotoxic and suppressor T cells, and CD3+ as a shared T-cell antigen. The activation of B cells and their differentiation into antibody-secreting plasma cells is triggered by antigen and requires helper T cells. 1 In our study, CD4+ T cells were the most abundant lymphocyte subset found in the lamina propria of the mucosa in the early stages of proventriculitis. These lymphocytes were later found surrounding and infiltrating what appeared to be B-cell germinal centers.
The CD8+ T cells were the most abundant lymphocytes found in the affected proventricular glands and formed sheets infiltrating the glandular epithelium. The influx of CD8+ T cells suggests cytotoxic activity associated with pathogen clearance. The CD8+ cytotoxic T lymphocyte (CTL) response has been shown to be critical for the control of primary, persistent, and reactivated virus infections. 7 Kotani et al. 13 studied the lymphocytic subsets in the trachea of chickens inoculated with IBV and concluded that the chicken's immune system may use specific CTL to eliminate IBV at the early stage of infection and in the later stage may depend on humoral immunity to control viral infection. Songserm et al. 25 also observed an increase of CD8+ cells in the intestine of chickens inoculated with malabsorption syndrome homogenate. They found that an increase of cytotoxic activity was associated with the intestinal lesions and weight gain depression. In our study, the influx of CD8+ cells in the proventriculus appeared to occur after the onset of the necrosis of the glandular epithelium. The epithelial necrosis and influx of T lymphocytes appeared to start in the area surrounding the mucosal papillae and spread to the glands that drained through these papillae. Not all glands were affected simultaneously, some glands presented lesions and others appeared normal. In a previous study, chickens immunosuppressed with cyclosporine, a T-cell suppressor, developed more severe lesions in the proventriculus and had higher incidence of proventriculitis than immunocompetent chickens, underlining the importance of cell-mediated immunity in controlling this disease. 20
In addition to CD4+ and CD8+ cells, natural killer (NK) cells may play a role in the defense against gastrointestinal pathogens. NK cells are phenotypically defined as CD8+ cells lacking T- or B-lineage–specific markers. Gobel et al. 9 demonstrated by this criterion that approximately 30% of CD8+ intestinal intraepithelial lymphocytes were NK cells. The physiologic role of the intestinal NK cells is not known. They may be the first line of defense once epithelial cells are infected, thus serving a function similar to cytotoxic T cells. 9 In our study, because we did not have a marker for NK cells, this specific subset was not analyzed and we cannot draw conclusions about the role of NK cells in proventriculitis. Most of the lymphocytes observed in the affected proventriculi that stained with the cytotoxic T-cell marker (CD8+) also stained with the pan-T-cell marker (CD3+), with only a low percentage not staining for the CD3+ marker. The presence of NK cells in the proventriculus and their role in proventriculitis need to be further investigated.
In this study, staining of B-cell immunoglobulins (IgG, IgM, and IgA) was not performed. IgM and IgA in the intestinal secretions prevent environmental antigen influx into internal body compartments, neutralizing viruses and microbial toxins and preventing their adherence and colonization of mucosal surfaces. 16 The role of these immunoglobulins, if any, is not clear for proventriculitis, and further study of their importance in this disease is required.
In conclusion, the influx of CD4+ cells during proventriculitis suggests that these cells are involved in the induction of the immune response, whereas the CD8+ cells most likely act as effector cells. The influx of B cells and the formation of highly organized germinal centers indicate that antibody-mediated mechanisms are also involved in the immune response in proventriculitis. Germinal center formation occurred during the chronic phase when the proventricular lesions were less severe.
Footnotes
Acknowledgements
We thank Gwin Kerce for technical assistance with immunohistochemistry.
