Abstract
A 12-month-old American Miniature horse colt was presented to the Virginia Tech Veterinary Teaching Hospital with a 7-month history of progressive ataxia. Physical examination revealed a head intention tremor, base-wide stance, and ataxia. Necropsy findings were confined to the brain. There were bilateral areas of liquefactive necrosis and cavitation corresponding to the dorsal accessory olivary and lateral (accessory) cuneate nuclei. Cerebellar folia of the dorsal vermis were thin. Microscopically, the cerebellar cortex was characterized by patchy areas of Purkinje cell loss with associated variable thinning of the molecular and granule cell layers and astrogliosis. Dorsal accessory olivary and lateral cuneate nuclei were cavitated and had mild glial response around their periphery. Additionally, a focus of necrosis and neuropil vacuolization was found in the right putamen. These findings indicate the presence of a neurodegenerative disorder centered, but not confined to, the cerebellum and its connections in this American Miniature horse colt.
There are numerous reports in the veterinary literature of juvenile-onset degenerative conditions of the cerebellum (cerebellar abiotrophies), and these have been reported in a variety of species, including dogs, 5 , 20 cats, 2 cattle, 10 , 13 , 21 , 22 sheep 7 , 17 , 19 and horses. 4 , 6 , 16 In these diseases lesions predominate within the cerebellar cortex where Purkinje cell loss is usually the most common finding. In some cases neurodegeneration within the cerebellum and its connections may be associated with neuronal loss at other sites functionally unrelated to the cerebellum. In domestic animals, these disorders are called multiple system abiotrophies, while in human neuropathology the general term systems degenerations is used. 18 There are few reports of multiple systems abiotrophies in the veterinary literature, and most of these are in dogs. 8 , 14 , 15 Here we describe the clinical and morphologic findings in a yearling American Miniature horse that presented clinically with cerebellar signs and at necropsy was found to have neuronal loss in the cerebellar cortex, dorsal accessory olivary nuclei, lateral cuneate nuclei, and right putamen.
A 12-month-old American Miniature horse colt was presented to the Virginia Tech Veterinary Teaching Hospital with a history of progressive neurologic disease that began when the animal was weaned at 5 months of age. Reported clinical signs were loss of balance, head tremor, and falling over. There was no history of trauma or previous illness. The animal was from a closed breeding herd of American Miniature horses with a history of two similar cases in the previous 10 years, both exhibiting progressive neurologic abnormalities consistent with cerebellar disease first noticed at weaning. The parentage of all three affected horses was unknown because multiple stallions were used in an unobserved, random pasture-mating program. Physical examination revealed the colt to be alert and responsive but exhibiting an intention head tremor and a base-wide stance in the forelimbs. The pulse rate, respiratory rate, and temperature were within normal limits. A cranial nerve examination was normal except for an absent menace response with normal vision. There were proprioceptive deficits, hypermetria (grade II/IV forelimbs, grade I/IV hindlimbs), truncal sway, ataxia (grade III/IV hindlimbs, II/IV forelimbs), a tendency to fall to the side or back, and spastic hyperextension of the forelimbs when the colt was startled. There was no evidence of neuromuscular weakness. Clinical signs were consistent with a cerebellar lesion. Hematologic values were normal. Serum vitamin E concentration was 2.8 µg/ml (normal: 1.7–9.5 µg/ml). Cerebrospinal fluid (CSF) analysis was unremarkable. Western immunoblot and polymerase chain reaction tests for Sarcocystis neurona performed on CSF were negative. The colt was euthanatized. Postmortem computed tomographic scan of the brain identified no abnormalities. The brain was removed and fixed whole. Spinal cord was not examined. Serial transverse brain sections revealed thinning of the cerebellar cortex within the vermis (midline cerebellum), especially dorsally. Bilaterally symmetric regions of lacunar-like cavitation were noted within the medulla oblongata corresponding to the dorsal accessory olivary nuclei (DAON) and lateral (accessory) cuneate nuclei (LCN) 23 (Fig. 1). There were no other macroscopic findings. Histologic examination of the brain revealed patchy moderate to marked Purkinje cell loss within the cerebellar vermis that was associated with cortical thinning (Fig. 2). Immunohistochemical staining of cerebellar cortex for glial fibrillary acidic protein (GFAP) revealed a prominent increase in Bergmann astroglial process number and thickness that was spatially associated with regions of Purkinje cell loss (Fig. 3). Associated with this astrogliosis were frequent focal areas of mild Bergmann astrocyte proliferation mainly confined to the deeper parts of the molecular layer. Some Purkinje neurons had slightly shrunken and vacuolated cytoplasm suggesting degeneration. Microscopic examination of DAON and LCN confirmed the presence of large regions of cavitation containing cellular debris and low numbers of activated macrophages characterized by abundant cytoplasm (gitter cells). Parenchyma immediately surrounding regions of necrosis was characterized by a mild glial response and vacuolization (Fig. 4). There were increased numbers of astrocytes with hypertrophied processes (GFAP stain). Activated microglial cells with rod-shaped nuclei were present in low numbers. Capillaries were congested and contained swollen endothelial cells. Occasional swollen axons were present in adjoining parenchyma. There was a narrow crescenteric region of neuropil vacuolization and liquefactive necrosis that measured about 0.8 cm in greatest dimension within the right putamen. It contained low numbers of activated microglial cells with abundant cytoplasm. A similar lesion was not identified in the left putamen despite examining multiple sections from this region. Other brain regions, including the substantia nigra and pontine and cerebellar nuicroscopically unremarkable. Staining of sections of affected brain for ubiquitin using an antibody known to cross-react with equine ubiquitin 9 failed to identify increased staining of neurons or glial cells when compared with horse control brain.
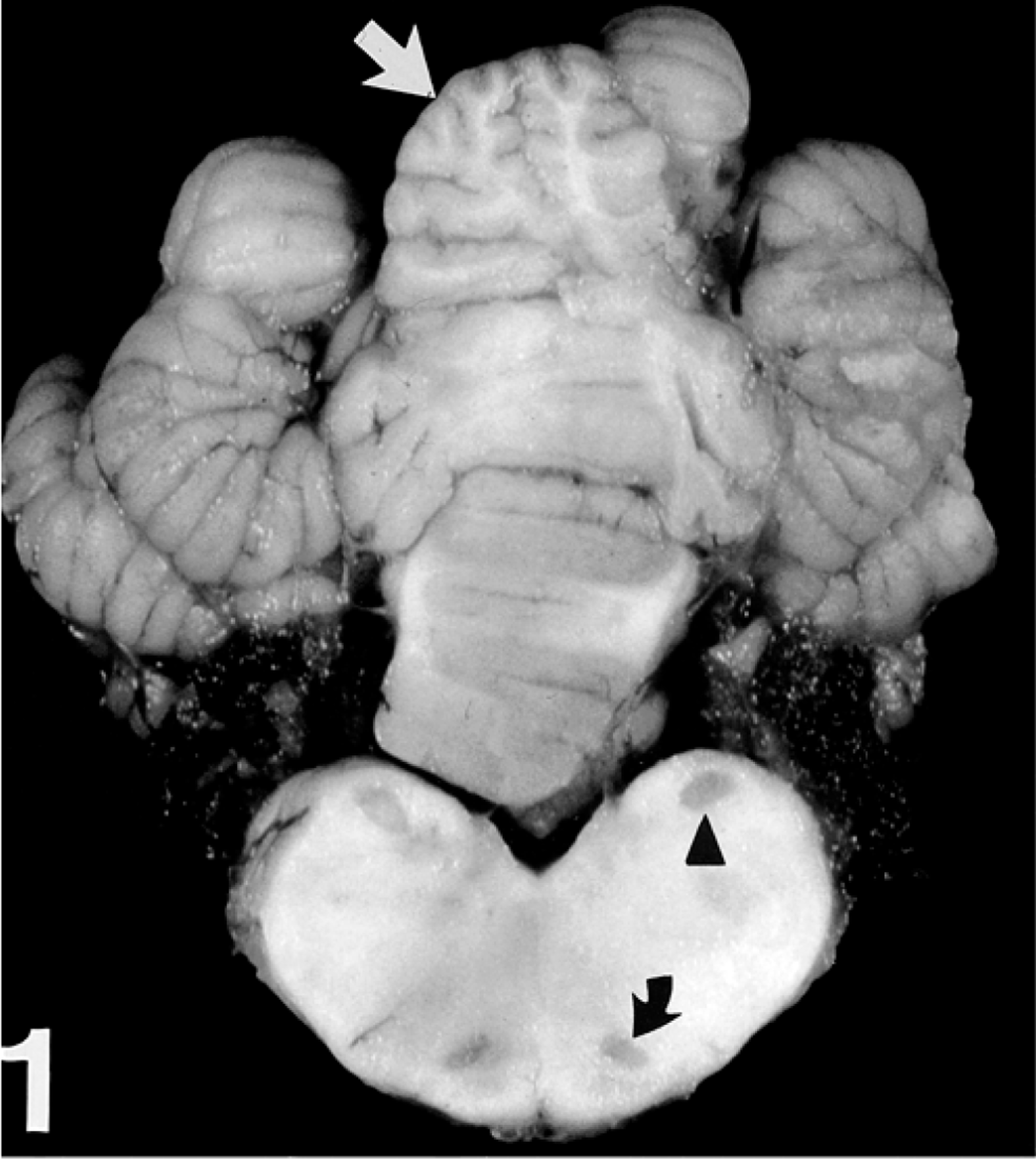
Cerebellum and medulla oblongata; horse. Bilateral foci of cavitation are present within the lateral (accessory) cuneate nuclei (arrow head) and dorsal accessory olivary nuclei (curved arrow). The dorsal cerebellum vermis shows subtle cortical thinning (straight arrow).
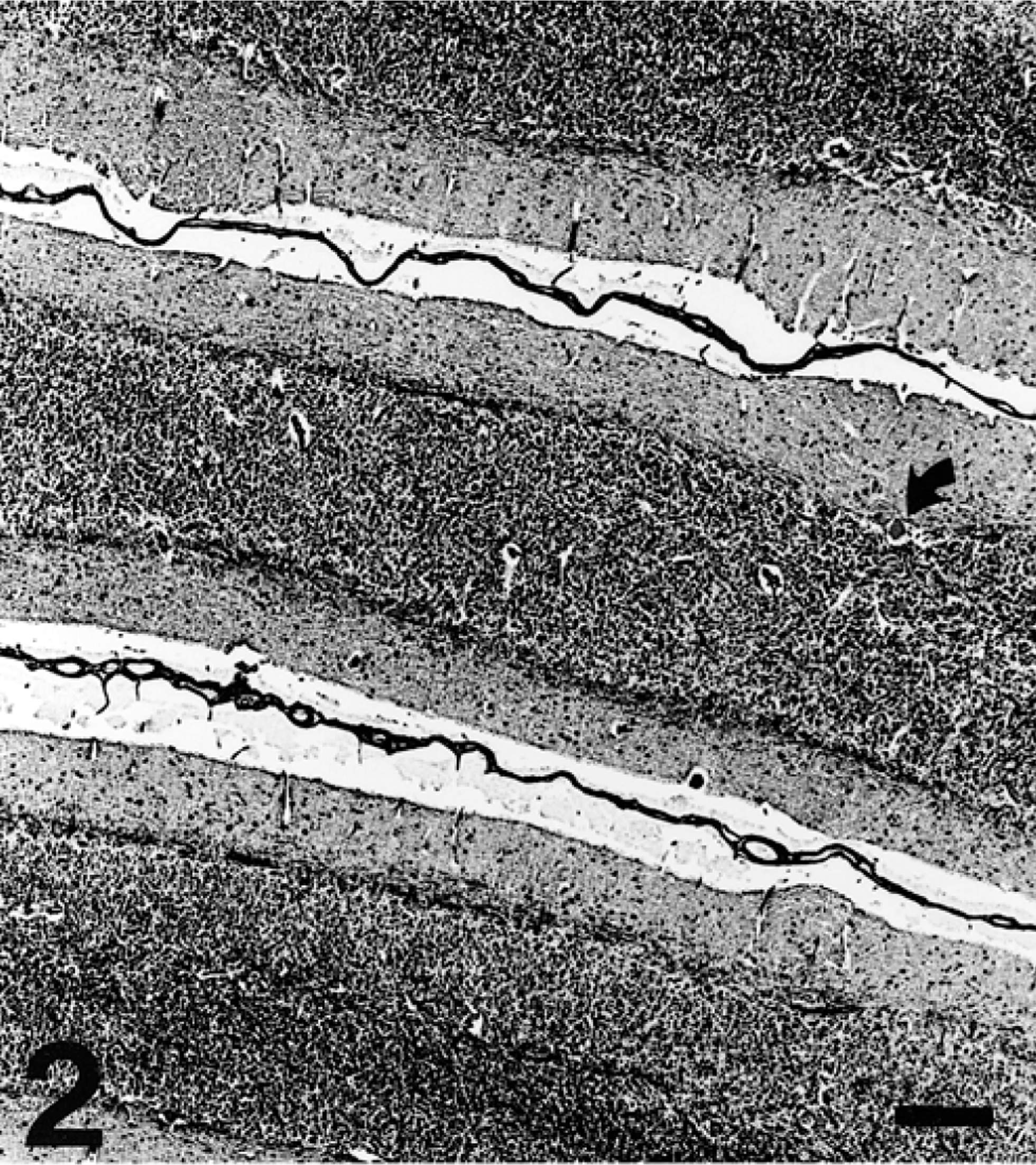
Cerebellar cortex of vermis; horse. Purkinje cells are marked by their absence; only one is noted (curved arrow). The molecular layer shows variable thickness. Bielschowsky stain. Bar = 200 µm.
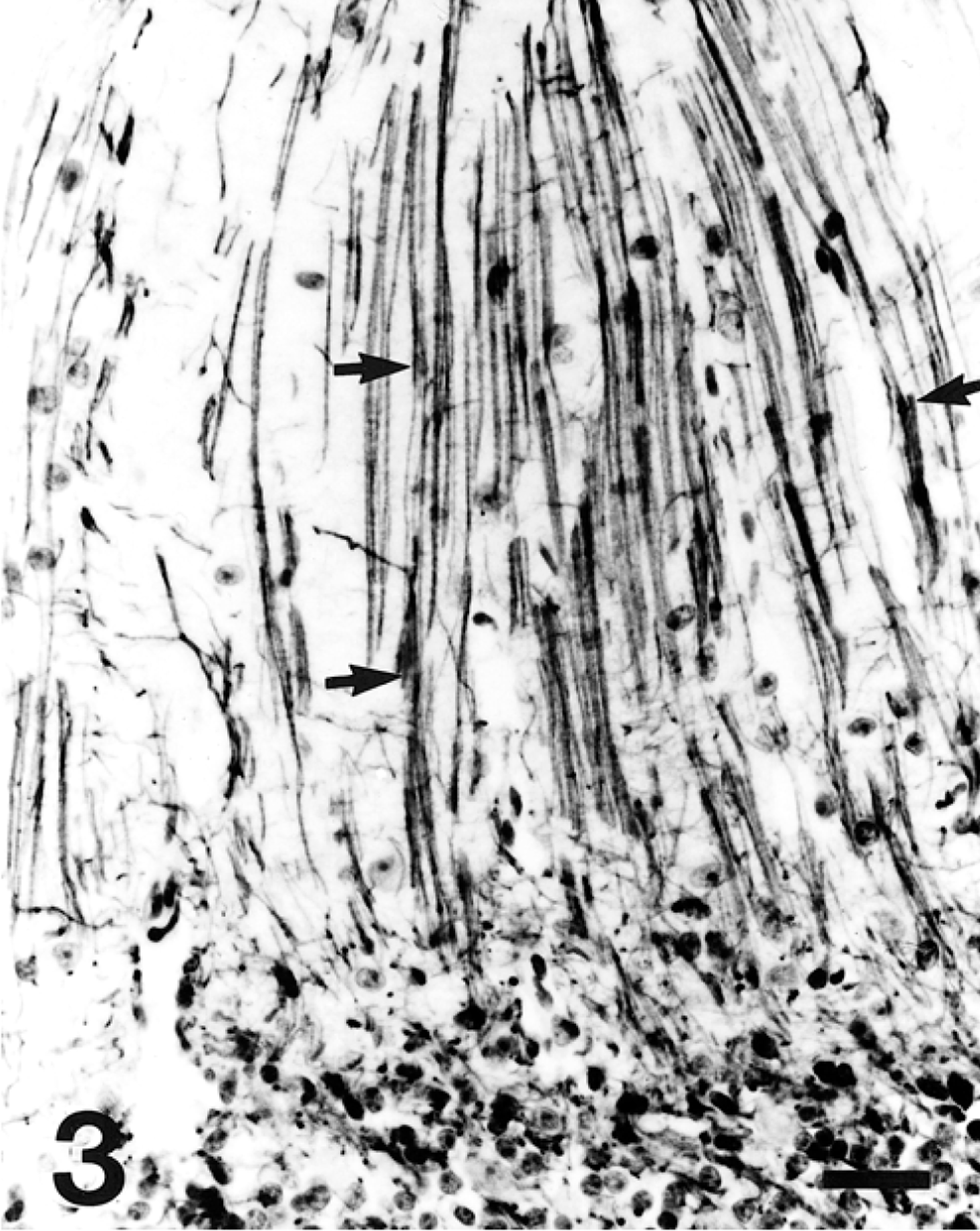
Cerebellar cortex; horse. Within a region of molecular layer Bergmann glial processes are increased in number and thickness (between straight arrows). Note the absence of Purkinje cells. Avidin-biotin complex immunoperoxidase with anti-GFAP antibodies. Bar = 50 µm.
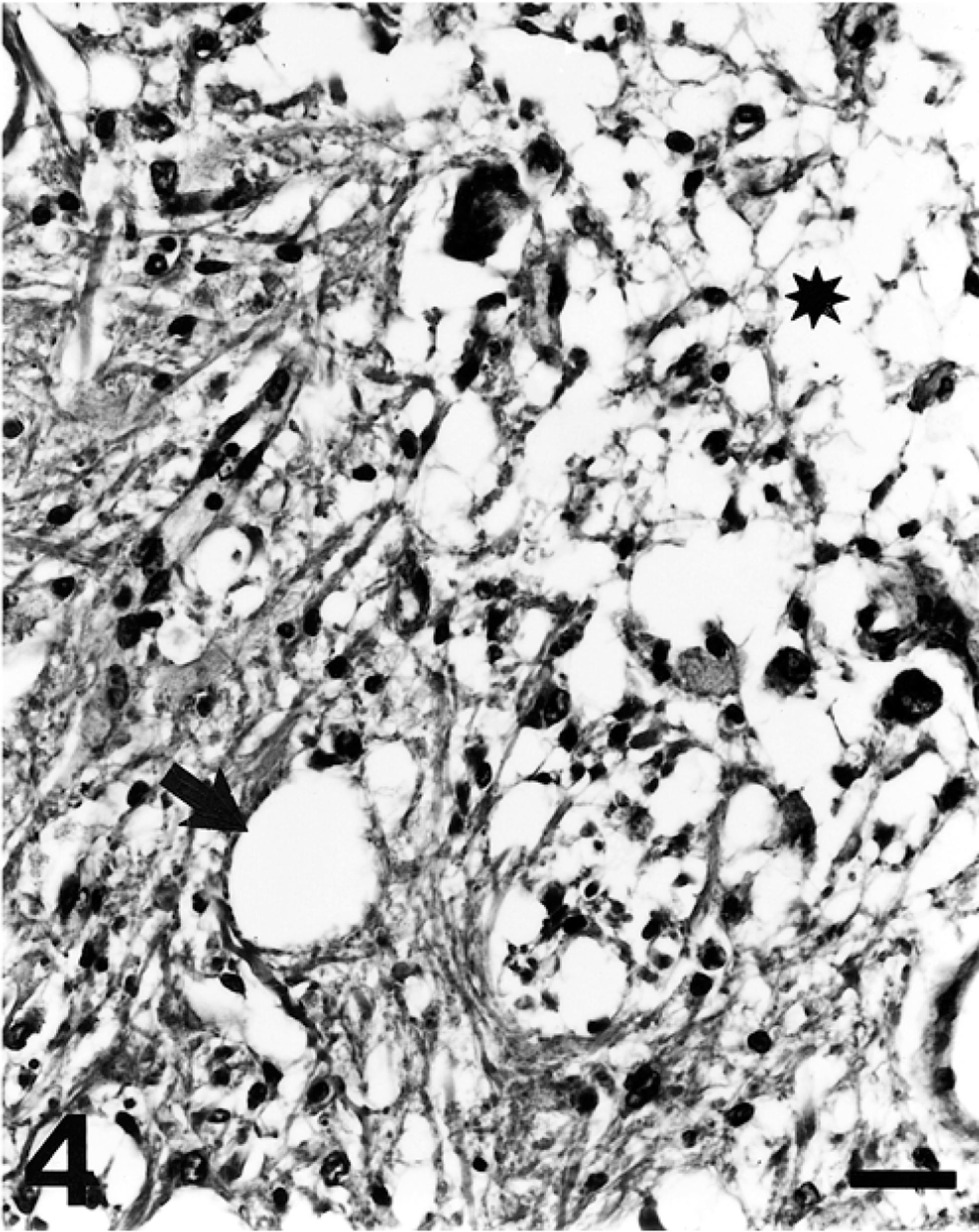
Medulla oblongata, dorsal accessory olivary nucleus; horse. There is cavitation (asterisk) and more peripheral neuropil vacuolization (arrow). HE. Bar = 50 µm.
In horses cerebellar abiotrophy has been described in Arabian 16 and part Arabian 3 foals and Swedish Gotland ponies 4 and is thought to be autosomal recessive in inheritance. The lesions identified in the brain of this American Miniature horse colt include cerebellar abiotrophy and additional features not previously described in the horse. While vacuolization of the olivary nuclei has been described in some cases of equine cerebellar abiotrophy, the presence of cavitating lesions within the medulla oblongata or putamen necrosis has not. Dorsal accessory olivary and LCN necrosis and cavitation in this case may have resulted from retrograde transsynaptic degeneration as both of these nuclei have direct afferent connections to the cerebellar cortex. 1 The LCN is part of the dorsal spinocerebellar system and is concerned with unconscious proprioception. Impulses arising from muscle spindles and Golgi tendon organs above the level of the eighth cervical spinal nerve travel in axons in the dorsal columns of white matter to reach the LCN. These axons synapse with LCN neurons that send axons to the cerebellum via the caudal cerebellar peduncle. The olivary nuclei form an important relay station in connections between the cerebral cortex and the cerebellum. Olivary neurons have axons that project to the cerebellar cortex and form a major part of the climbing fiber system with synaptic input on Purkinje and to a lesser degree other cerebellar cortical neuronal types. Olivary nuclei receive input from the cerebral cortex, basal nuclei, cerebellar nuclei, mid-brain, and spinal cord. The putamen is part of the basal nuclei, it lacks direct connections with the LCN or cerebellar cortex. 1 The failure to identify a lesion in the left putamen might be explained by chronological age of the putamen lesion. The lesion in the putamen was much more recent than the cerebellar and associated lesions: possibly a bilateral lesion would have developed at this site if the animal had survived long enough. The absence of a menace response with normal vision is explained by the involvement of the cerebellum in this reflex. 12
The possibility of inbreeding and historical information suggesting previously encountered cases in this herd of horses could be consistent with an inherited condition, most likely autosomal recessive. In humans, cerebellar ataxias can be inherited by autosomal recessive or dominant modes, and for several conditions the genetic defect has been identified. 11 Much less is known about the genes involved in similar diseases in domestic animals. Unfortunately, pedigree analysis was not possible as breeding histories were not known. Other possible causes such as neurotoxicity, nutritional deficiency, or a spontaneous dominant mutation cannot be ruled out.
