Abstract
In Europe, cystic echinococcosis is rare in horses and is mostly diagnosed at slaughter or postmortem examination. Equine cystic echinococcosis can be caused by various Echinococcus taxa, but only Echinococcus equinus (the “horse strain”) is known to produce fertile cysts. In Europe, E. equinus appears to be endemic in Great Britain, Ireland, Spain, and Italy and has sporadically been reported in Belgium and Switzerland. The present report describes the first case of a molecularly confirmed E. equinus infection in a horse foaled and raised in Germany. The 19-year-old mare was presented for examination of inappetence, emaciation, and respiratory symptoms. X-ray radiographs of the thorax showed 2 well-circumscribed tumor-like masses, each approximately 10 cm in diameter in the caudal lung field. The horse was euthanized as its condition rapidly deteriorated. Necropsy revealed 2 thick-walled hydatid cysts, each 7–8 cm in diameter in the lung. The tri-layered cyst walls consisted of an outer adventitial layer, a laminated acellular intermediate layer, and an inner germinal membrane. Grossly, the cysts contained a clear, amber liquid with hydatid sand. Light microscopy of the hydatid sand revealed free protoscoleces, intact and ruptured brood capsules, calcareous corpuscles, and debris. Samples of protoscoleces underwent molecular characterization, and the diagnosis of E. equinus was confirmed by restriction fragment length polymorphism—polymerase chain reaction and sequence analysis of the complete mitochondrial nicotinamide adenine dinucleotide dehydrogenase subunit 1 gene.
Cystic echinococcosis (CE), or hydatidosis, is an important worldwide zoonotic disease caused by larval stages (metacestodes) of tapeworm parasites of the genus Echinococcus. These cyclophyllidean cestodes have an indirect, 2-host life cycle, with carnivores (mostly wild or domestic canids) as definitive hosts and herbivorous or omnivorous mammals as intermediate hosts. Adult tapeworms inhabit the small intestine of the definitive host and release gravid cestode segments (proglottids) containing eggs into the environment via feces. Upon accidental ingestion of these eggs, intermediate hosts become infected with larvae of Echinococcus spp. The developing metacestodes are fluid-filled hydatid cysts, in which protoscoleces are produced as the next generation of tapeworms. The hydatids mainly develop in liver and lung. Their growth can cause severe illness and death in the intermediate host. Depending on the temperature and other conditions, protoscoleces inside intact hydatid cysts in organs of perished or slaughtered animals stay infectious for up to 2 weeks. Consumption of viable protoscoleces by a definitive host completes the life cycle of the parasite. 5,13,17 Different species and strains of Echinococcus show considerable variation regarding their host specificity, epidemiology, morphology, and infectivity to humans. 13,20,25 This corresponds with considerable genetic differences, and recent phylogenetic studies based on DNA sequence data led to discrimination of various Echinococcus species and strains, with ongoing changes in the taxonomic classification. 13,15,19,20,22,24,25 According to the current taxonomy, Echinococcus equinus, formerly known as the horse strain (G4) of Echinococcus granulosus, is regarded as a separate species. 13,15,20,22,24
Echinococcus equinus appears to use equids (horses, donkeys, and zebras), exclusively, as intermediate hosts. 13 Other Echinococcus species, strains, or both might also cause CE in horses, but those cysts apparently do not become fertile. 28 Canids (dogs and probably foxes) serve as definitive hosts of E. equinus. 4,10,25 In dogs, the prepatent period is reported to last approximately 70 days. 4 Unlike metacestodes of other Echinococcus species, E. equinus seems to be apathogenic to humans. 20,25,27 In horses, CE is generally a rare finding, mostly incidentally diagnosed at slaughter or postmortem examination. The hydatid cysts commonly develop in the liver, lungs, or both. 2,3,7,18 Other reported localizations include the cranium, brain, pericardium, pleura, spleen, kidneys, muscle, uterus, and ocular structures. 1,18,23 The cysts have a reported longevity of several (at least 16) years 21 and rarely cause severe clinical symptoms. 18 Cases of equine cystic hydatidosis have been reported from Europe, 6,20,22 the Middle East, South and East Africa, 13,25 North America, 9 and Southeast Asia. 3 In many of these cases, the identity of the causative Echinococcus taxon was not confirmed with suitable methods. Even in Europe, recent epidemiological data on confirmed cases of E. equinus infections is limited. In Europe, E. equinus appears to be endemic in parts of Great Britain, Ireland, Spain, and Italy. 6,20,28 Especially in regions where dogs or foxes have commonly been given access to horse offal, the local prevalence of hydatid cysts in horses has been reported to reach up to approximately 25–60%. 26,28 A few cases of equine CE have also been observed in Belgium and Switzerland, 6 whereas no confirmed case of E. equinus has been reported from Germany until the current account.
The present report describes a case of cystic hydatidosis in the lung of a horse foaled and raised in Germany. The diagnosis of CE was based on characteristic morphological criteria of cyst architecture and morphology of protoscoleces, 3,25 and the allocation of the isolate to E. equinus was done by specific restriction fragment length polymorphism—polymerase chain reaction (RFLP-PCR) and confirmed by sequence analysis of the complete mitochondrial nicotinamide adenine dinucleotide dehydrogenase subunit 1 (nad1) gene. 11,12 The horse, a 19-year-old Irish Tinker mare, was foaled and raised in Germany, kept in Bavaria from 4 years of age onward, and did not have any preliminary reported sojourns outside of Germany. In 2007, the horse was first presented to the Equine Clinic of the Ludwig-Maximilians-University Munich (Munich, Germany) for examination because of weight loss and mild respiratory symptoms. Radiographs of the thorax revealed the presence of 2 well-circumscribed, homogeneous, tumor-like masses, each approximately 10 cm in diameter in the caudal lung field, which were inaccessible for ultrasonographic examination. Two years later, in March 2009, the horse was again presented to the clinic displaying inappetence, emaciation, adynamia, and acute impairment of general condition. Clinical pulmonary exam findings were an accentuated abdominal respiration with a moderately increased breathing rate of 24 gasps/min and a tightened respiratory noise audible in the right lung. Radiographic examination of the thorax confirmed the persistence of the formerly diagnosed formation of 2 comparatively radiopaque soft masses in the lung (Fig. 1A). Because its health condition rapidly deteriorated, the horse was finally euthanized in extremis. Necropsy and subsequent histopathological examination revealed a severe, extensive, chronic myocardial fibrosis of nondistinctive etiology of both ventricular walls and the interventricular septum, which was considered to be responsible for the clinical signs. Corresponding to the radiographic findings, 2 discontiguous, encapsulated, tan, thick-walled, unilocular, globoid cysts of taut consistency, 7–8 cm in diameter, were reconfirmed in the parenchyma of the left main lobe of the lung. The rather small size of the cysts, which had not noticeably grown within a period of at least 2 years, confirmed the reportedly benign nature of the parasitosis. Both cysts displayed a wall thickness of approximately 6 mm, with many low irregular folds, slightly protruding into the cyst lumen. Each cyst contained about 30 ml of clear, amber, nonropy liquid with several small pale particles (hydatid sand; Fig. 1B).
Histopathological examination was carried out on sections of paraffin and glycol methacrylate and methyl methacrylate—embedded 8 samples of both hydatid cysts. Histologically, the cysts displayed a 3-layered wall architecture, with an outer, host-derived adventitial layer, an acellular intermediate layer, and an inner germinal membrane of parasitic origin. The outer adventitial layer consisted of a chronically inflamed fibrous granulation tissue with decent attendance of epithelioid cells, lymphocytes, plasma cells, few multinucleated giant cells, accentuated fibrosis, and scattered small foci of mineralization (Fig. 1E). The laminated and germinal layers (Fig. 1E, F) of the cyst walls formed low interlacing folds, among which brood capsules with viable protoscoleces and mineralized corpuscles were evident (Fig. 1F). Microscopic examination of native hydatid sand revealed numerous free, partially evaginated protoscoleces, intact and ruptured brood capsules, calcareous corpuscles, necrotic protoscoleces, debris, and free rostellar hooklets (Fig. 1C). Morphometric characterization of protoscoleces was performed on photographic images of wet-mount preparations of native hydatid sand with the use of a commercial morphometric image analysis system. a Invaginated protoscoleces measured approximately 185 μm (±10) × 147 (±8) μm. Their rostella consisted of 2 rows of 28–38 (average: 33 ± 3) rostellar hooks, which displayed an average length of 28 (±4) μm (Fig. 1C, D). Because these morphological attributes fit in the range of various Echinococcus strains, 25 a molecular characterization of the isolate was performed. Three protoscoleces were separated from the cyst fluid with the use of a capillary pipette, and each protoscolex was individually lysed in 10 μl of 0.02 M sodium hydroxide (NaOH) at 95°C for 10 min. 16 The lysates associated with each protoscolex were used directly as templates in the PCR. For amplification of protoscolex DNA, nested PCR assays were performed with the nad1 primer pair, reaction mixtures, and amplification conditions as described previously. 11 One microliter of the protoscolex lysates was added as template. The obtained nad1 PCR products were digested with the restriction enzyme hph1. 12 The resulting banding patterns of all 3 protoscoleces matched with the pattern of E. equinus. For confirmation, the nad1 PCR products were sequenced. b The sequences of the complete nad1 gene were compared with those previously published in the GenBank database using the BLAST system (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). The obtained nad1 sequences of all 3 protoscoleces were 100% identical to each other (GenBank GQ420652). The comparison with previously published sequences gave 99.8% identity with E. equinus 14 (GenBank AF346403). Two transitions could be observed within the 894—base pair—long nad1 sequence. One occurred at position 618 (adenine for guanine), the other at position 888 (thymine for cytosine). The positions were numbered from the start codon on the basis of the complete nad1 sequence. Both transitions have no effects on the resulting protein structure.
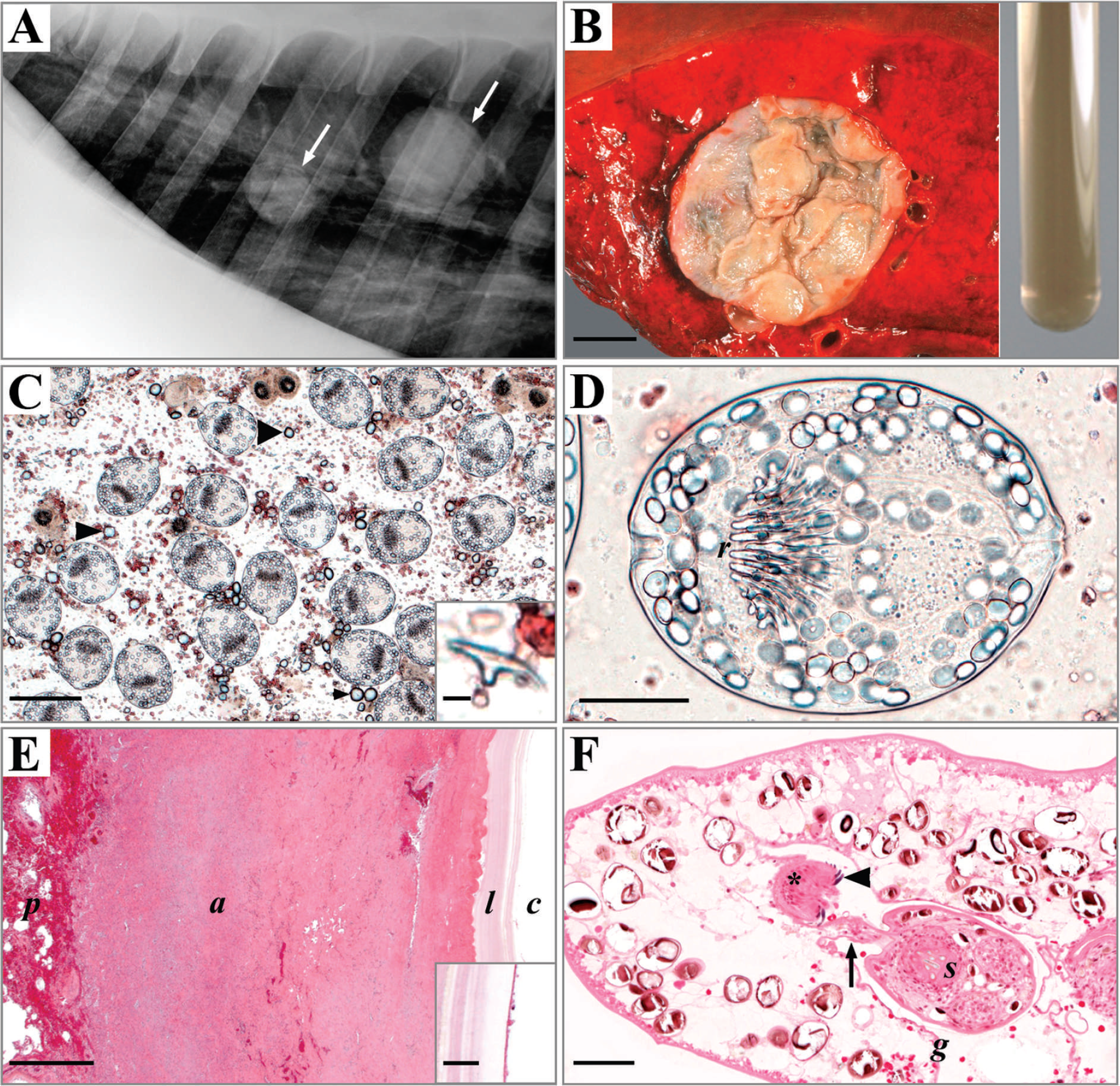
A laterolateral (right-to-left) radiographic projection of the thorax. Two spherical radiodense masses (arrows) are visible within the lung.
The present report represents the first recorded case from Germany in which the presence of E. equinus metacestodes in a horse of local origin was unambiguously ascertained by molecular species identification. In several reported cases of equine cystic hydatidosis from countries without an endemic appearance of horse-adapted Echinococcus tapeworms, the respective animals were imported from an endemic area, and infections are believed to have been established before importation. 3,9,22 In the present case, however, an autochthonous infection in Germany can be presumed. Of course, the factual path of infection of the horse remains speculative, as both origin and species of the infected definite host responsible for fecal contamination of pasture with E. equinus eggs is not known. In this regard, contact with feces of an E. equinus—infected dog imported from an endemic area outside of Germany could be regarded as a most likely source of infection. Otherwise, dogs or wild carnivores (e.g., foxes) of local origin, which might have had access to infectious horse offal, have to be taken into consideration, although the existence of a stable, autochthonous transmission cycle appears rather improbable given the conditions for offal disposal in Germany. According to German law, carcasses of perished horses, and offal from slaughters displaying signs of diseases communicable to humans or animals, must be disposed in accredited rendering plants, and stringent legal regulations direct the treatment of offal from slaughter and its utilization as pet food.
Thus, more data on the prevalence of equine cystic hydatidosis in Germany and central Europe is needed to achieve a better assessment of the epidemiological situation of this important parasitic infection. Also, additional data on the prevalence of E. equinus among its definitive hosts is required, with special regard to wild carnivores. Therefore, every suspicious case of equine cystic hydatidosis incidentally found during postmortem examination or slaughter, or, as in the present case, as an arguable differential diagnosis to radiographically displayable tumor-like lesions, should be thoroughly investigated. Next to a precise investigation of the provenance and historical background of the infected horses (and potential contact with dogs coming from endemic areas), these investigations should include routine gross and histopathological, as well as parasitological, examinations of the hydatid cysts and their contents. The morphological diagnosis of metacestode stages of Echinococcus spp. in equids should always be completed through subsequent identification of the definitive strain, species, or both by genetic sequence analysis. Recognition and broad diagnostic coverage of equine cystic hydatidosis therefore demands particular attention and cooperation of diagnosticians, pathologists, and parasitologists. The risk of a potential establishment of this parasite in central Europe requires the effective prevention of factors that support the maintenance of E. equinus in equine hosts. These factors predominantly concern the perpetuation of the life cycle of E. equinus by transmission of infective parasite stages from intermediate to definitive hosts, which can be prevented by vigilant handling of infectious cyst material and strict obedience of statutory provisions regulating adequate carcass disposal.
Acknowledgements The authors thank Heike Sperling for excellent technical assistance.
Footnotes
a.
Videoplan™ image analysis system, Zeiss-Kontron, Eching, Germany.
b.
Seqlab GmbH, Göttingen, Germany.
