Abstract
A vestibulocerebellar disorder was observed in 2- to 4-month-old swine after consumption of broken rice contaminated with 13% Aeschynomene indica seeds. Affected animals recovered in 2–14 days after removal of the contaminated food. To reproduce the syndrome, 10 pigs were fed 13% A. indica seeds in commercial pig ration or rice. They showed clinical signs similar to those observed in the spontaneous cases, 1 hour to 6 days after ingestion. Three pigs recovered after the withdrawal of the contaminated food. The others were killed. Two pigs that were fed for 25 days with food containing 3% and 6% seeds and two control pigs did not show clinical signs. Histologic lesions were characterized by symmetric focal degeneration in the cerebellar and vestibular nuclei. The initial lesion, observed 24 hours after the first ingestion, was characterized by vacuolation of the neuropil. Subsequently, there was progressive loss of parenchyma, vascular reaction, a few small spheroids, astrocytosis, and accumulation of gitter cells. The brains from four pigs were perfused with a buffered solution of glutaraldehyde-paraformaldehyde 12–48 hours after they started to ingest the seeds. No ultrastructural lesions were observed in the cerebellar nuclei after 12 hours of seed consumption. At 24 hours, endothelial cells and pericytes were separated by empty spaces from astrocyte foot processes, suggesting perivascular edema. The astrocytes were enlarged, consistent with intracellular fluid accumulation. These results suggest that A. indica seeds are toxic, causing functional derangement of the vestibulocerebellar system, followed by alterations in the microvasculature in the cerebellar and vestibular nuclei.
An outbreak of intoxication occurred in a group of seven Landrace × Duroc Jersey pigs, weighing 20–30 kg in the municipality of Pelotas, state of Rio Grande do Sul. All animals were fed with broken rice. Clinical signs, observed 24 hours after first ingestion of the contaminated broken rice, were characterized by an uncoordinated gait, falling, and difficulties in rising, followed by lateral recumbency. Reflexes, sensorium, and limb movements were conserved. Appetite was maintained. Affected animals recovered in 2–14 days after removal of the contaminated food, and no pathologic studies were performed. The broken rice contained 13% Aeschynomene seeds. The adult plants grown from the seeds were identified as Aeschynomene indica. A complete description of this species is given by Kissman and Groth. 7 Voucher specimens were identified as Brazil, Rio Grande do Sul, Claudio Timm, No. 17598 (PEL).
A. indica L. from the Fabaceae (Leguminoseae) family is a weed abundant in irrigated rice fields in the Brazilian state of Rio Grande do Sul. This plant has black or dark brown, beanlike seeds measuring 3–5 mm (Fig. 1). Seeds may be harvested with rice, and during processing of rice they contaminate screenings composed mainly of broken rice. These screenings are often used for animal feed.

Seeds of A. indica mixed with broken rice. Bar = 10 mm.
This article reports a neurologic syndrome with symmetric focal degeneration in the cerebellar and vestibular nuclei in swine fed with rice screenings containing 13% A. indica seeds. The histologic and ultrastructural lesions in experimentally intoxicated swine are also described.
Materials and Methods
Two groups of pigs were used for the experimental reproduction of the intoxication. The first group included 11 Landrace × Large White pigs (Nos. 1–11), weighing 28–37 kg, from the University of Pelotas farm. The second group of three pigs (Nos. 12–14) weighed 10–22 kg. They were of the Brazilian Piau breed and came from a small dairy farm. After being made to fast for 24 hours, the pigs were fed broken rice ad libitum (pig No. 1) or commercial pig ration (pig Nos. 2–7 and 12–14) mixed with 13% A. indica seeds. Two other pigs were fed for 25 days with commercial pig ration containing 6% (pig No. 8) and 3% (pig No. 9) seeds, respectively. The A. indica seeds used in the experiments were separated manually from the broken rice and later mixed with the ration. Contaminated screenings were obtained from a rice-processing plant. Pig No. 1 was fed ground seeds, whereas the other pigs were fed intact seeds. One control pig (No. 10) was fed commercial ration without A. indica seeds and another (No. 11) was fed only uncontaminated broken rice. For pig Nos. 1–4 and 8–11, blood samples were obtained at the start of the experiment and every 2 days. Serum creatine kinase (CPK), aspartate aminotransferase (AST), and alanine aminotransferase (ALT) activities as well as urea and creatinine concentrations were determined using standard methods in accordance with the recommendations of the German Society of Clinical Chemistry. 10 The analyses were performed within 8 hours after collection.
Necropsy and complete histologic evaluation were performed in four experimental pigs (Nos. 2–5), which were killed by exanguination after anesthesia with sodium pentobarbitone. Samples of liver, kidney, lung, lymph nodes, spleen, thyroid, adrenal, stomach, small and large intestines, cardiac and skeletal muscles, peripheral nerves, and the whole central nervous system (CNS) were fixed in 10% neutral formalin, embedded in paraffin, and sectioned at 6 μm. The brain was sectioned in 4- to 5-mm transverse sections, and all sections were processed for histology. Transverse sections from the cervical, thoracic, and lumbar spinal cord were also studied. All sections were stained by hematoxylin and eosin (HE), and sections of the nervous system were also stained with Luxol fast blue for myelin and Sevier-Munger-Luxol fast blue and Bielschowsky stain for nerve fibers.
Four experimental pigs (Nos. 6, 7, 13, and 14) were injected intravenously with heparin, then deeply anesthetized with sodium pentobarbitone. A midventral sternal thoracotomy was performed. A cannula was inserted in the aorta through the left ventricle. The abdominal aorta and the subclavian arteries were clamped, and the right atrium was opened. A buffered saline solution was injected through the cannula for 1 minute, followed by perfusion with 4–6 liters of a fixative of 2% glutaraldehyde and 2% paraformaldehyde in phosphate buffer (pH 7.4) for 30 minutes. Small fragments from the right cerebellar nuclei were fixed in the same fixative for 2 hours, postfixed in 1% osmium tetroxide buffered in 0.4 M sodium cacodylate (pH 7.4), and embedded in Epon 812. Semithin sections were stained with methylene blue. Ultrathin sections were stained with lead citrate and uranyl acetate and examined with an EM 109 Zeiss transmission electron microscope at 80 kW. Histologic examination of the perfused pigs was performed for the left brain using the methods mentioned previously.
Results
The 10 pigs (Nos. 1–7 and 12–14) that ingested 13% A. indica seeds were affected with neurologic signs similar to those observed in the spontaneous cases. Clinical signs, the amount of seeds consumed, and the outcome of the intoxication are summarized in Table 1. Pig Nos. 1–7 ingested the food in small amounts, and clinical signs appeared in 1–6 days (Table 1). In contrast, pig Nos. 12–14 ingested the food immediately and continuously until the observation of the first clinical signs, merely 1–2 hours later (Table 1).
Experimental intoxication by A. indica seeds in swine. Seeds consumed, observation of clinical signs, and outcome of the intoxication.
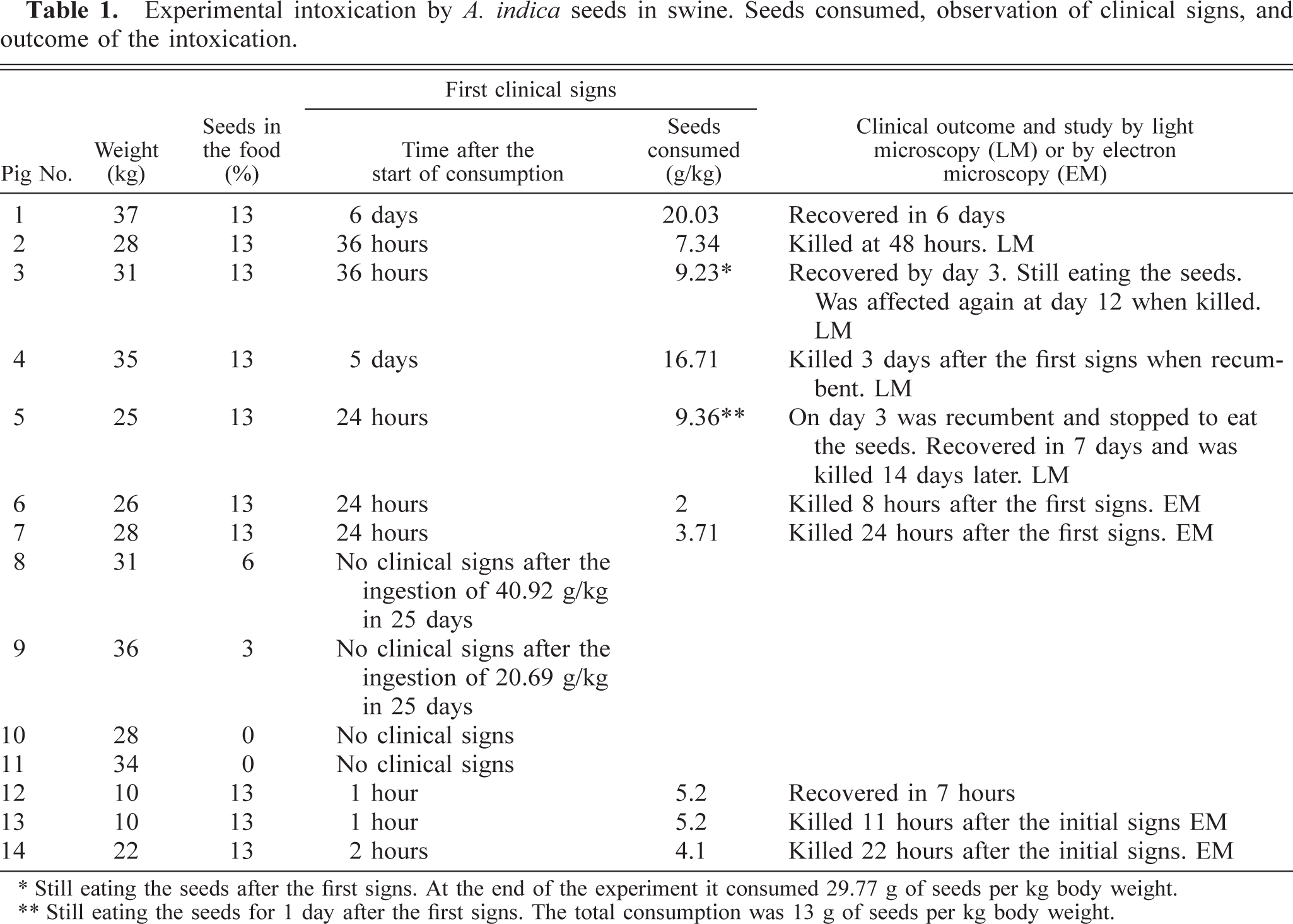
∗ Still eating the seeds after the first signs. At the end of the experiment it consumed 29.77 g of seeds per kg body weight.
∗∗ Still eating the seeds for 1 day after the first signs. The total consumption was 13 g of seeds per kg body weight.
Clinical signs in pig Nos. 1–7 were characterized by an uncoordinated gait (Fig. 2), swaying when standing, wide-based stance and falling, followed, in 24–48 hours, by sternal or lateral recumbency. Some animals, after being raised to their feet, stood for only a few minutes, with the legs widely placed and the nose touching the ground (Fig. 2). Reflexes, sensorium, and limb movements were conserved. Muscular tremors were also observed. Appetite was maintained, but the animals were unable to eat while recumbent. Serum CPK, ALT, and AST activities and urea and creatinine concentrations were within normal values.

Pig No. 1. It has severe incoordination with the nose touching the ground.
One hour after starting to eat, pig No. 12 had severe incoordination followed by lateral recumbency. Continuous masticatory movements, vomiting, and severe respiratory distress were also observed. Four hours later, this pig showed only incoordination, and it had fully recovered 7 hours after the onset of signs. Pig No. 13 developed incoordination followed by loss of equilibrium, salivation, masticatory movements, and vomiting 1 hour after starting to ingest the ration. Later, the incoordination was more accentuated, and the animal stayed in recumbency. When the first signs were observed, the animal had eaten 400 g of contaminated food (5.2 g of seeds per kg body weight), and in the following 4 hours, it had eaten another 200 g. Pig No. 14 had masticatory movements 2 hours after the start of ingestion. Six hours later, it became uncoordinated and, when excited, had masticatory movements followed by vomiting. In the following 20 hours, it started to eat the food again on three occasions, but these attempts were followed by vomiting. When the first signs were observed, the animal had eaten 700 g of contaminated food (4.1 g of seeds per kg body weight) and in the following 4 hours, another 230 g. Twenty-four hours after the initiation of clinical signs, when it was euthanatized and perfused, the pig had masticatory movements and incoordination.
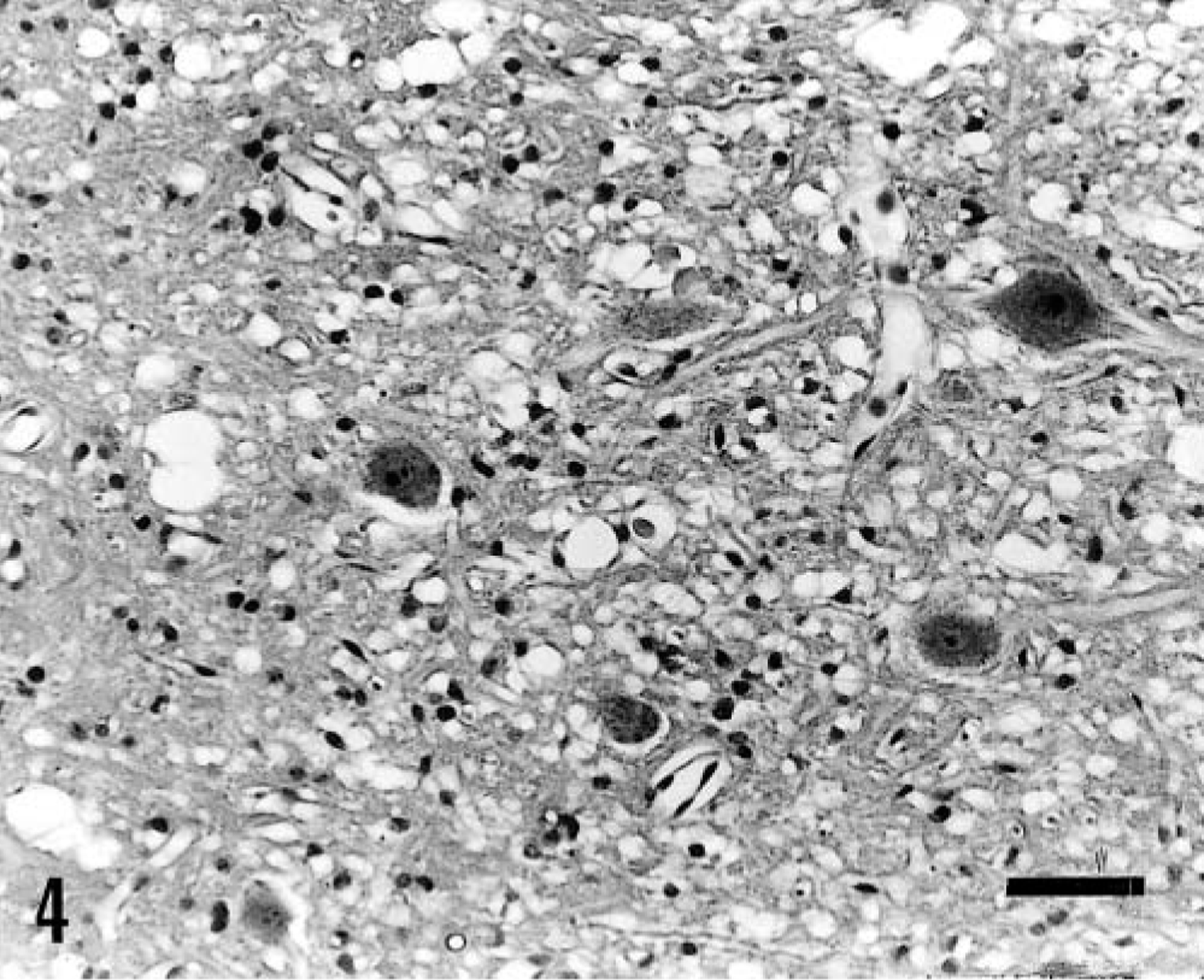
Macroscopic lesions were not observed, and no significant histologic lesions were found in pig Nos. 13 and 14, except for perivascular hemorrhages in one fastigial nucleus in pig No. 14. In pig Nos. 2, 3–7, and 14, histologic lesions were characterized by symmetric focal degeneration in the cerebellar and vestibular nuclei (Fig. 3), and to a lesser extent, in white matter adjacent to these nuclei. These lesions appeared more severe in the cerebellar than in the vestibular nuclei. In pig Nos. 2, 6, 7, and 14, lesions in the cerebellar and vestibular nuclei were characterized by diffuse vacuolation of the neuropil (status spongiosis) (Fig. 4). Perivascular hemorrhages were observed around some blood vessels. The neurons and glial cells appeared normal. In pig Nos. 3 and 4, lesions were characterized by vacuolation of the neuropil, prominent capillaries with swollen endothelium, a few small spheroids, astrocytosis, and collections of gitter cells, where neurons, glial cells, and axons had been lost (Fig. 5). In most affected areas, there was a sharp demarcation between necrotic and viable nervous tissue (Fig. 5). In some lesions, a peripheral rim of viable neurons within the cerebellar or vestibular nucleus was seen at the margin of the lesion (Fig. 5). Scattered axonal spheroids and digestion chambers sometimes containing axonal residue and macrophages were occasionally observed in the periphery of the lesion (Fig. 5). Luxol fast blue staining showed considerable phagocytosis of myelin. In pig No. 5, killed 15 days after recovery, astrocytosis was more prominent than in the previous cases; large myelin vacuoles, sometimes containing macrophages or axonal residue, were frequently observed, and blood vessels were surrounded by numerous gitter cells. In this pig, the Bielschowsky stain showed a reduction in the number of axons, but surviving axons appeared normal. Axonal density appeared to be greater in areas of the cerebellar nuclei that were unaffected.
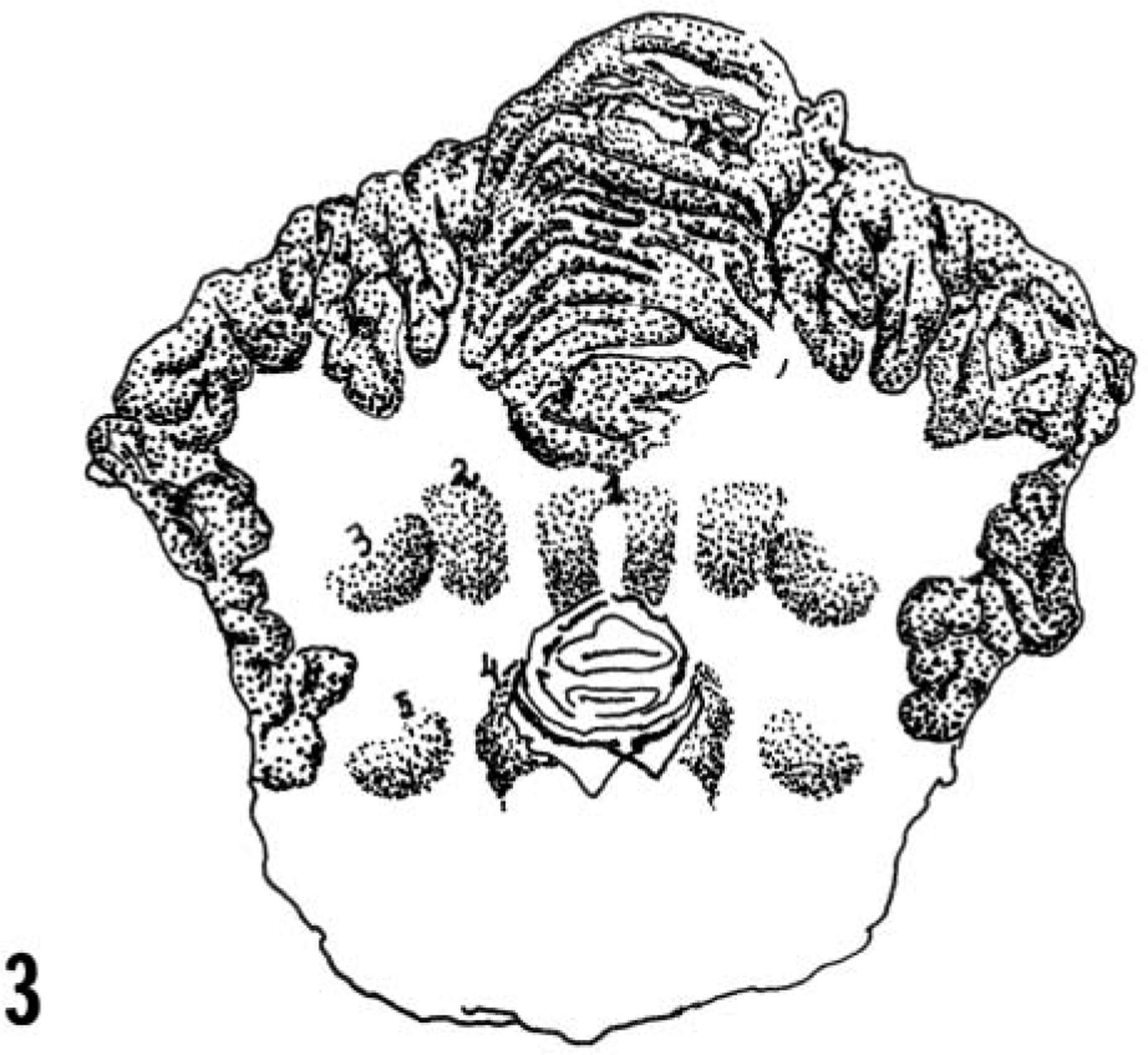
Drawing of the trasversal section of the cerebellum, cerebellar peduncles, and pons showing the fastigial cerebellar (1), interposital cerebellar (2), lateral cerebellar (3), medial vestibular (4), and lateral vestibular (5) nuclei. Modified from Yoshigawa. 13 .
Fastigial cerebellar nucleus; pig No. 6. Diffuse vacuolation (status spongiosus) is observed in the neuropil with apparently viable neurons within the nucleus. HE. Bar = 25 μm.
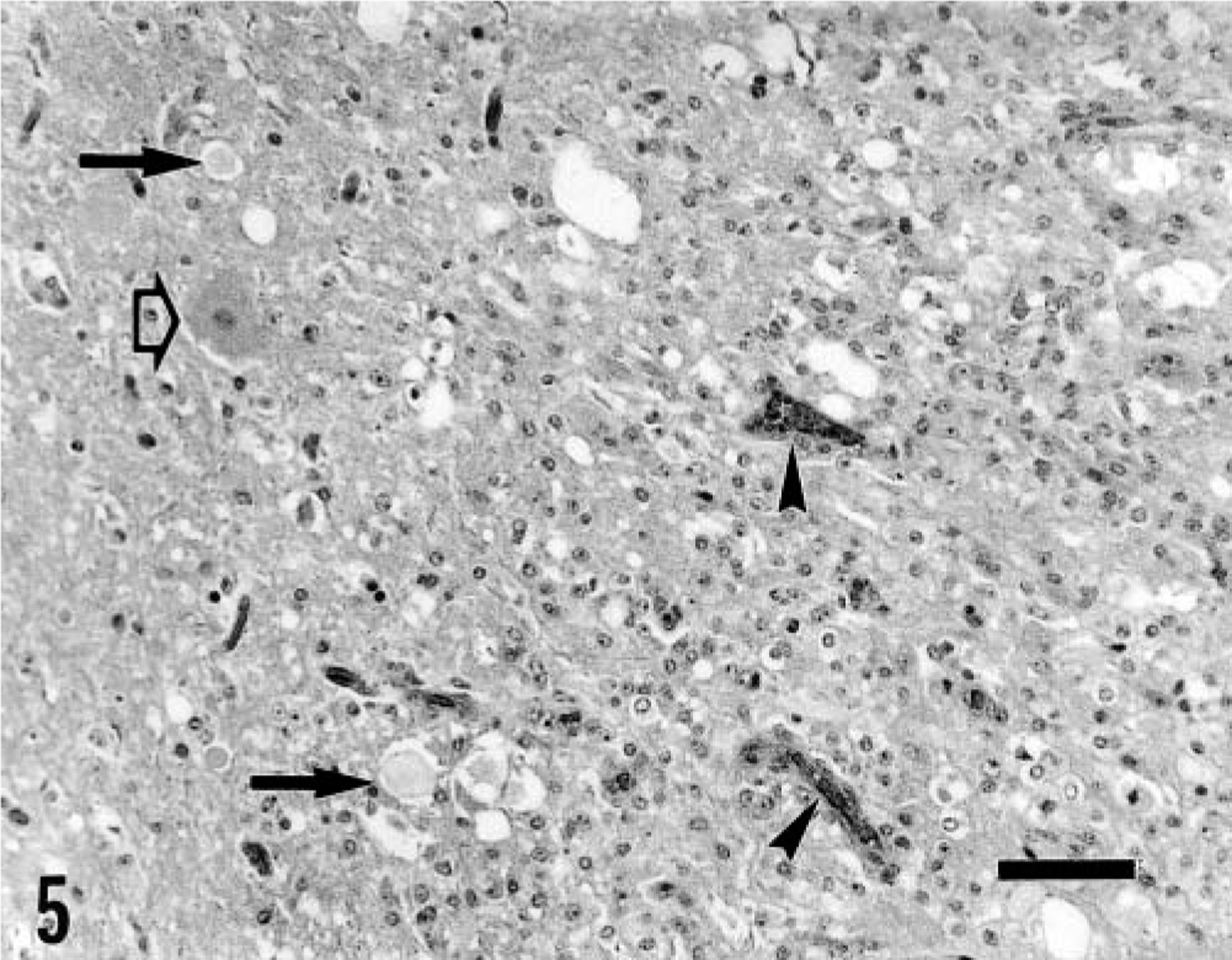
Fastigial nucleus; pig No. 4. Within the lesion (at right), the parenchyma had been lost and is substituted mainly by gitter cells and glial cells. Prominent capillaries with swollen endothelium (arrow heads), axonal spheroids (black arrows), and digestion chambers are also observed. An apparently viable neuron is present at the border of the lesion (open arrow). There is a sharp demarcation between necrotic and viable nervous tissue. Bar = 25 μm.
In pig No. 13, killed 12 hours after ingesting A. indica seeds, no significant lesions were observed by electron microscopy of the cerebellar nuclei. In pig No. 14, killed 24 hours after ingestion, the capillary endothelial cells and pericytes were separated by empty spaces from the astrocyte foot processes, suggesting perivascular edema (Fig. 6). Occasionally, astrocyte processes were separated from the perivascular microglial cells. The astrocytes were enlarged, indicating intracellular fluid accumulation (Fig. 6). In pig No. 6, the cerebellar nuclei were not identified in the small pieces of tissue collected for electron microscopy. In pig No. 7, killed 48 hours after ingestion, ultrastructural lesions were characterized by a mixture of degenerate and necrotic neurons and degenerate axons and myelin sheaths. Macrophages, lipid membranes, and axonal debris were often found inside degenerated myelin sheaths. Residual bodies, lipid droplets, and, occasionally, erythrocytes were observed in the cytoplasm of macrophages. Perivascular hemorrhages were observed very frequently. In the edematous neuropil, a few unaltered axons were found admixed with cellular debris. Unaltered terminal axons with synaptic junctions were seen in close contact with degenerate neurons.
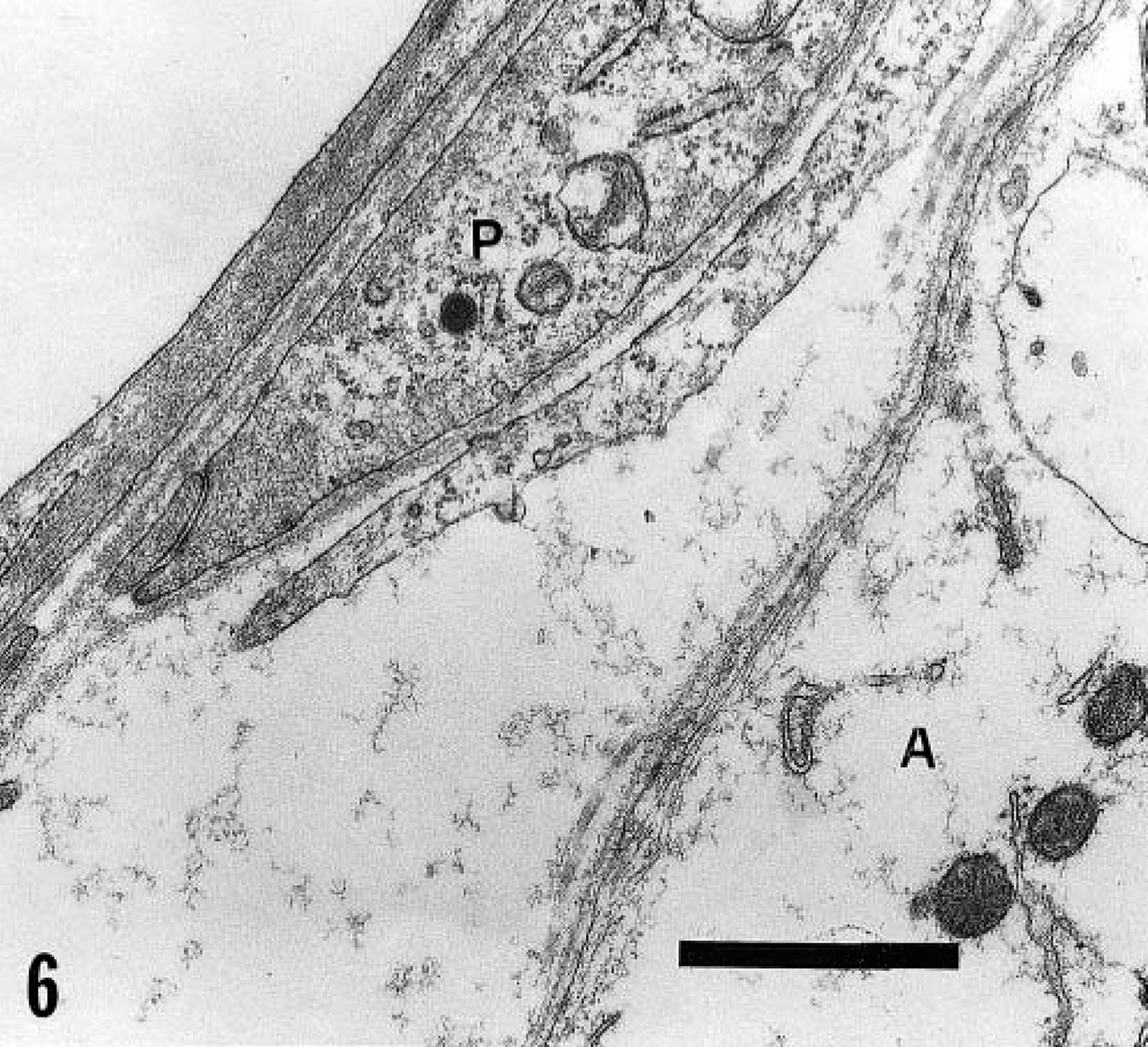
Cerebellar nucleus; pig No. 14. The capillary endothelial cells and pericytes (P) are separated by an empty space from the astrocyte foot processes (A). Bar = 1 μm.
Discussion
The genus Aeschynomene has approximately 150 species, mainly weeds, found in South America, Central America, North America, Africa, and Asia. 9 A. indica (Fabaceae) is a weed that contaminates cultures of irrigated rice in Asia, Pacific Islands, Australia, Africa, Puerto Rico, and, in Brazil, in the state of Rio Grande do Sul. 7 , 9 Until now, no species of Aeschynomene have been reported to be toxic to domestic animals or humans. Few phytochemical investigations have been reported in the genus Aeschynomene. A flavonoid, reynoutrin, and the amino acid, potassium aeshynomate, were isolated from A. indica. 8 , 11 The neoflavonoid mimosifoliol and the styrylcycloheptenone derivative mimosifoleon were isolated from A. mimosifola. 3
Experimental reproduction of the clinical signs in pigs receiving the same proportion of A. indica seeds as the spontaneous cases substantiated the suspected etiology of the disease. High concentrations (13%) in the food were necessary to produce intoxication. Pigs fed 3% and 6% seeds in the food for 25 days were not affected, suggesting that the seeds do not have a cumulative effect. No apparent differences were observed in the toxicity of the ground seeds administered to pig No. 4 compared with the toxicity of the whole seeds administered to the other pigs. The period between first ingestion and the onset of clinical signs was quite different in pig Nos. 1–7 than in pig Nos. 12–14. In the latter group, the signs were observed only 1–2 hours after ingestion, probably because food was eaten continuously until the start of clinical signs. This eating behavior was probably due to the different conditions in which the pigs were raised: pig Nos. 1–11 were raised on a farm under a confinement production system using only balanced pig diet; and pig Nos. 12–14 were raised on a farm under a production system using pastures and different kinds of food. The different behavior in the two groups of pigs could be due also to breed or body size differences.
No references could be found in the veterinary or human literature regarding degenerative lesions in the cerebellar and vestibular nuclei in association with plant intoxication. However, focal symmetric degenerative lesions in specific areas of the CNS are reported in other diseases. In horses, intoxication by Centaurea spp. causes necrosis of the substantia nigra and globus pallidus. 2 , 14 In sheep and cattle, the toxins of Clostridium perfringens type D cause symmetric focal necrosis of the internal capsule, basal ganglia, substantia nigra, and cerebellar peduncles. 5 In swine, toxicosis caused by selenium results in symmetrical spinal poliomalacia affecting the ventral gray matter of the cervical and lumbar intumescenses. 4 , 12 A number of familial neurodegenerative disorders in dogs can target specific neuronal populations such as the cerebellar and vestibular nuclei. 1
Uncoordinated gait, swaying when standing, a wide-based stance, falling, and vomiting are characteristic of a vestibulocerebellar syndrome 6 and are explained by the lesions in the cerebellar and vestibular nuclei observed in the affected pigs. Standing with the nose touching the ground and the legs widely placed after having been raised is, presumably, for better balance. Permanent sternal or lateral recumbency, not common in the cerebellar and vestibular syndromes in other species, is probably a consequence of severe difficulties in rising and standing caused by the lesions in the vestibular and cerebellar nuclei. This may also explain the absence of signs related to other areas of the brain; the pigs had normal sensorium, and the appetite, limb movements, and reflexes were maintained.
The occurrence of clinical signs only 1 hour after A. indica ingestion and the absence of histologic and ultrastructural lesions in the pig killed 12 hours after starting the ingestion suggest a functional derangement of the vestibulocerebellar system caused by an unknown toxin contained in the plant. This functional derangement is probably followed by alteration in the microvasculature, causing perivascular edema and hemorrhages, and intracellular fluid accumulation in astrocytes. These lesions seem to be responsible for vacuolation of the neuropil observed in pig Nos. 2, 7, and 14. Later on, these changes are followed by neuronal degeneration and death, myelin degeneration, accumulation of gitter cells, and loss of parenchyma as observed in pig Nos. 3–5.
The recovery of the spontaneous cases and experimental pig Nos. 1, 5, and 12 indicated the potential for reversibility of the clinical signs. This reversibility suggests that, in natural cases, withdrawal of the contaminated food will result in recovery of affected animals. Despite the reversibility of the clinical signs, the morphologic lesions are not reversible, as was observed in pig No. 5, killed 16 days after clinical recovery. In this animal, the astrocytosis and the numerous gitter cells observed are characteristic of a healing lesion of the CNS.
Footnotes
Acknowledgements
This work was financially supported by the Programa de Apoio a Núcleos de Excelência (PRONEX), Grant No. 7697102600.
