Abstract
Marijuana toxicosis is typically seen by companion animal veterinarians. However, with increased marijuana availability, there is a greater potential for toxicosis in other species. Herein we describe a case of suspected marijuana toxicosis in a female and a male American Mammoth donkey, aged 8 y and 20 y, respectively, fed cannabis buds. Both cases were presented because of depression and lethargy. However, the jenny had ataxia, mild colic, tachycardia, tachypnea, and decreased tongue tone. Plasma samples from the jenny on presentation and 3 d following hospitalization were submitted to the Kansas State Veterinary Diagnostic Laboratory to be screened for cannabinoids using high-pressure liquid chromatography coupled with tandem mass spectroscopy (HPLC-MS/MS). A single serum sample from the jack was taken on presentation and submitted to the Animal Health Diagnostic Center at Cornell University for Δ9-tetrahydrocannabinol (THC) and cannabidiol analysis using HPLC-MS/MS. THC was detected in all samples. Clinical signs were noted 24–36 h after ingestion, which included mild-to-moderate neurologic deficits, mild colic, tachycardia, tachypnea, and decreased tongue tone. Both donkeys recovered uneventfully within 24 h of peak effects. Utilizing a cannabinoid screening assay in collaboration with a veterinary diagnostic laboratory may be useful when an equine practitioner suspects marijuana toxicosis in a patient.
Marijuana toxicosis in companion animals is more commonly seen by veterinarians in the United States given the growing number of states legalizing recreational marijuana use by humans.2,14,16 However, to our knowledge, marijuana toxicosis in equids has not been reported in the scientific literature since 1951. 3 The main psychoactive component of cannabis (Cannabis sativa) is Δ9-tetrahydrocannabinol (THC). There are 2 primary G protein–coupled cannabinoid receptors, namely CB1 and CB2, on which THC can exert an effect.1,2,15,16 By inhibiting cyclic AMP and stimulating protein kinases, CB1 receptors modulate voltage-activated calcium ion channels and potassium channels, resulting in inhibition of neurotransmitter release.1,5,10 Some of these effects include modulating immune and inflammatory responses as well as affecting appetite, thermoregulation, mood, and movement.1,5,15,13 The CB1 receptor is abundantly expressed in the CNS and is involved in the body’s immune response, whereas CB2 receptors are more widely expressed in the periphery and have both analgesic and anti-inflammatory effects.13,15,16
Extrapolating inter-species metabolism of THC can be difficult because of the differing subsets of cytochrome P450 enzyme systems in the liver.9,13,18 The most abundant primary metabolite produced by humans is 11-hydroxy-Δ9-tetrahydrocannabinol (11-OH-THC), and this has more neurotoxic potential than THC itself because it can cross the blood-brain barrier.9,10,16 Pharmacokinetic work in humans indicates that THC-based products have variable oral bioavailability, but THC and cannabidiol (CBD) as individual compounds have low oral availability intrinsically (~6%) because they are highly lipophilic. Peak effects occur ~1 h after ingestion, and there is significant enterohepatic recirculation with these compounds, hence they are eliminated slowly from the body. 12 The terminal half-life reported for oral exposure in humans is ~22 h. 12 Lethal toxicity of marijuana is rare in veterinary species. In dogs, an oral dose of marijuana plant material at 3 g/kg did not result in death. 6 A report from the 1950s described acute death in horses and mules after ingestion of a large amount of Cannabis indica. 3 However, given the increased availability of cannabinoid-containing materials, further work is needed to characterize the pharmacokinetics and pharmacodynamics of THC across more veterinary species.2,14,16
Case 1
An 8-y-old, 449 kg (989 lb), American Mammoth Jackstock donkey jenny was presented to the primary care veterinarian because of severe lethargy and inappetence after ingesting 3–5 g of mature, fresh cannabis buds. This would equate to a maximal possible dose of 11.1 mg/kg of cannabis. The cannabis was legally grown on the property for human consumption and hand-fed to this jenny. The species of cannabis was not documented. The jenny was treated with flunixin meglumine (1.1 mg/kg PO) by the owner prior to veterinary examination. No vaccination history was available. The herd manager stated that clinical signs of slight lethargy were first noted 24–36 h after consumption. On initial presentation, the jenny had a dull, sedate attitude with moderate lethargy. Mucous membranes were pink and moist with a capillary refill time of <2 s. She was normothermic (37.8°C [RI: 36.0–38.0]; 100.1°F [96.8–100.4°F]), mildly tachycardic (heart rate 56 bpm, normal 32–44 bpm), tachypneic (respiratory rate 30 breaths/min, normal 13–25 breaths/min), and had a normal peripheral blood lactate of 0.7 mmol/L. 7 A 0.13-mg/kg dose of N-butylscopolammonium bromide was administered intravenously for treatment of abdominal pain. Rectal palpation revealed a large, gas-distended viscus to the left, consistent with a large colon displacement. Sedation (4 mg detomidine and 4 mg butorphanol IV) was needed to pass a nasogastric tube, which induced more profound ataxia than anticipated from sedation alone. Nasogastric intubation yielded no net reflux, and 5 L of water and 1 L of mineral oil were administered through the nasogastric tube. At this time, decreased tongue tone was also noted. Marked lethargy and sedation-induced severe ataxia prompted transport to a referral hospital 44 h after the noted onset of clinical signs. After a 3-h trailer ride, evaluation at the referral hospital revealed the jenny was markedly depressed and remained ataxic with significantly decreased tongue tone. Mild tachycardia (48 bpm) and tachypnea (28 breaths/min) persisted, her mucous membranes were pink and moist with a capillary refill time of <2 s, and she became mildly hypothermic (35.9°C [96.6°F]). Additional tests included rectal palpation, abdominal ultrasound, abdominocentesis, fecal floatation, and skull radiographs; all of which were unremarkable. Complete blood count (CBC, Element HT5; Heska), serum electrolytes (Element DC5X; Heska), and serum chemistry and lactate (Element DC5X) were also performed, and no clinically significant abnormalities were detected (Suppl. Table 1). The jenny was placed in a stall, and signs of disorientation, head pressing, and generalized depression persisted. A small amount of grain and hay were offered, and she was able to slowly masticate and swallow the food bolus.
The following morning, the jenny was more alert, with marked decrease of ataxia and cessation of head pressing. Tongue tone also returned to normal with more efficient mastication. The jenny remained hospitalized for 2 more days until her mentation appeared normal, and she was discharged with recommendations for continued monitoring. Plasma from the day of presentation and from day 3 in the hospital were sent to the Kansas State Veterinary Diagnostic Laboratory (KSVDL; Manhattan, KS, USA) on ice and frozen until analysis.
Case 2
A 20-y-old, 398 kg, American Mammoth Jackstock donkey jack, pastured with the jenny described in case 1, was simultaneously hand-fed ~5 g of fresh, mature cannabis, and was presented with a dull attitude and lethargy ~72 h later. This would equate to consuming ~12.6 mg/kg cannabis. Normal manure was passed just prior to veterinary evaluation. Physical examination revealed a quiet, sedate attitude, normal vital parameters, and unremarkable cardiopulmonary and gastrointestinal auscultation. The jack’s mucous membranes appeared pink and moist with a normal capillary refill time of <2 s. Tongue tone was also within normal limits, and the digital pulses were normal on palpation. Nasogastric intubation yielded no net reflux, and 6 L of water and 1 L of mineral oil were administered through the nasogastric tube. Flunixin meglumine (1.1 mg/kg IV) was administered following examination. The jack was monitored on the farm by his owner after receiving veterinary care and was reported to have returned to his normal attitude and alertness within 5 h of evaluation. Given the history of cannabis consumption and concurrent presentation of lethargy and colic in his pasture-mate, serum was submitted for THC and CBD quantification. No other equids on the farm had a history of cannabis consumption.
Plasma cannabinoid concentrations were quantified at the KSVDL using a novel high-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) method. 11 For all standards and quality control samples (Cerilliant and Cayman Chemical), cannabinoid reference standards were added to the serum and internal standard. After a protein precipitation step followed by centrifugation, the supernatant underwent solid-phase extraction and then was injected into an ultraperformance liquid chromatography system (H-class Acquity; Waters) and a triple quadrupole mass spectrometer (TQ-S; Waters) using both a positive and negative ionization mode. The response (analyte over internal standard) was plotted against the concentration (Suppl. Figs. 1–3). Linear regression with a weighing factor of 1/x was the best fit selected for the calibration of cannabinoids, and the coefficient of correlation was ≥0.99. The regression equation generated by this calibration curve was then used to calculate the concentration of THC in the jenny’s plasma. This method was validated in accordance with the U.S. FDA guidelines for the Bioanalytical Method Validation Guidance for Industry, with precision ≤20% and accuracy of 80–120%. 17
A novel HPLC-MS/MS detection was developed at the Animal Health Diagnostic Center at Cornell University (AHDC; Ithaca, NY, USA) for the determination and quantitation of THC and CBD in canine plasma using a calibration curve of 1–80 ng/mL. For standards and quality control samples, THC and CBD reference standards were added to the serum and internal standard. After a protein precipitation step followed by centrifugation, the supernatant underwent solid-phase extraction prior to evaporation and reconstitution. HPLC was performed (1260 Infinity; Agilent), which was coupled to an LC-MS/MS system (QTRAP 5500 system; Sciex). An 8-min isocratic elution was used, and compounds were analyzed in a positive ionization mode. Method validation was performed in-house in accordance with the FDA Bioanalytical Method Validation Guidance for Industry, 2018. 17 A linear regression with a 1/x weighting factor produced a best-fit curve, and the correlation coefficient was 0.9996 (Suppl. Fig. 4). The regression equation generated by this calibration curve was then used to calculate the serum concentration of THC in the jack.
In the jenny plasma samples, THC was 0.17 µmol/L (53.3 ng/mL) on presentation and 0.01 µmol/L (3.5 ng/mL) on day 3 of hospitalization. 11-OH-THC was 0.06 µmol/L (21.3 ng/mL) in the initial plasma sample and 0.003 µmol/L (1.0 ng/mL) in the subsequent sample. THC-acid (11-nor-9-Δ9-tetrahydrocannabinol), the secondary metabolite of THC in humans, was detected only in the initial plasma sample at 0.11 µmol/L (40.2 ng/mL; Table 1). No other metabolite in the cannabinoid profile was detected. THC was detected in the jack’s serum at 0.11 µmol/L (35.0 ng/mL; Table 1); CBD was not detected.
Plasma cannabinoid concentrations in the donkey jenny during hospitalization and serum cannabinoid concentrations in the jack during hospitalization.
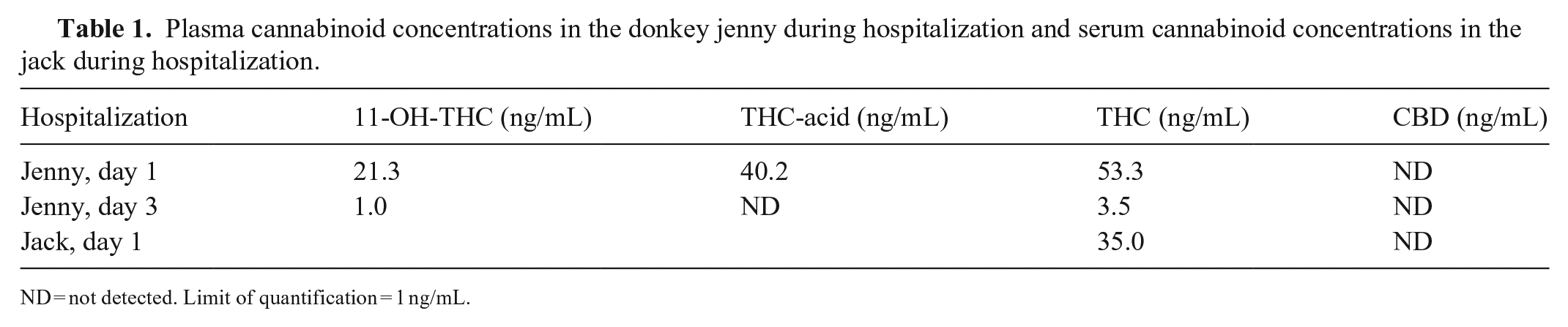
ND = not detected. Limit of quantification = 1 ng/mL.
The jack and jenny had been fed mature cannabis buds, which is the portion of the marijuana plant that contains the highest concentration of THC. 2 The concentration of THC in plant material can be as high as 20% depending on variables such as the strain of cannabis, type of soil, light exposure, and fertilization.6,16 The jenny’s clinical signs included ataxia, tachycardia, tachypnea, slow mastication, decreased tongue tone, disorientation, and head pressing. The jack’s clinical signs were milder and included lethargy and light sedation. Additionally, the jack’s mentation was normal 5 h after presentation; the jenny did not appear mentally appropriate until ~24 h following care. The amount of marijuana that these donkeys ingested is an approximation, thus it is possible that the jack received a lower dose of cannabis than the jenny or that there were other unknown factors that contributed to the difference in clinical signs and their severity. The jenny had a 1.5× higher concentration of THC in plasma compared to the jack’s serum (Table 1). THC and its metabolites are highly plasma protein–bound (~90%), whereas ~10% is distributed to red blood cells. Similar recoveries were found for cannabinoids in serum and plasma after protein precipitation and solid-phase extraction steps using gas chromatography–mass spectrometry for analysis. 8
Human or canine oral exposure to marijuana can be in the form of the raw plant material or in the form of a baked product (brownies, cookies, etc.) or other edibles. 10 The bioavailability of plant material itself is lower in comparison because conversion of tetrahydrocannabinolic acid (THCA) to its neutral counterpart THC must occur in order to have psychoactive effects. 4 During the drying of cannabis, THCA is converted to THC. 19 It was surprising that THCA was not detected in the jenny’s plasma following exposure to the fresh, undried plant material (Table 1). With oral dosing of cannabis in cattle, THCA and cannabidiol acid (CBDA) were both detected in serum. 11 It is theorized that the rumen microbial flora could decarboxylate CBDA to CBD and result in a possible therapeutic benefit to the animal. 11
Typical clinical signs of marijuana intoxication in dogs include incoordination, ataxia, urine dribbling, hyperesthesia, and vomiting.2,6,10,14,16 Similar to the donkeys, most dogs have a short recovery time of <24 h with supportive care, unless severely affected. 16 Ingestion of the plant product has been associated with nausea and vomiting in dogs as a result of direct irritation of the gastric mucosa. 16 Equids cannot vomit, but the irritation associated with the plant material could explain the mild colic signs described. Decreased tongue tone was witnessed in the jenny but is not reported as a clinical sign in dogs. This could be related to the pronounced muscle relaxant effect of THC on the central CB1 receptors, as seen in humans, or it could have been a side effect from the injectable sedative given to the jenny prior to passing a nasogastric tube. 5 Although tachycardia was described in the jenny, in a study of 213 cases of marijuana toxicosis in dogs, only 5% of dogs had tachycardia. 10 However, THC can inhibit vagal tone causing a decreased release of acetylcholine, which could explain the tachycardia. More information is needed to determine the dose of THC that produces clinical signs of toxicosis in donkeys, but equine practitioners can utilize diagnostic laboratories to assist with the diagnosis of marijuana toxicosis if suspicious clinical signs are present.
Possible treatment options for suspected marijuana toxicosis in equids may include gastric lavage, administration of oral activated charcoal, and laxatives. These adjunctive therapies are targeted at decreasing gastric absorption and facilitating excretion to limit the adverse clinical effects of cannabis. There is no scientific evidence to support the benefit of these therapies for marijuana toxicosis in equine patients. However, activated charcoal and gastric lavage are effective means of supportive treatment for marijuana toxicosis in canine patients.2,6,10
Marijuana toxicosis in donkeys has not been reported previously, to our knowledge. Equine veterinarians should be cognizant of the clinical signs of marijuana toxicosis in equids in the event owners are unaware of, or reluctant to share, information related to exposure. Veterinary diagnostic laboratories that offer cannabinoid testing to confirm the presence of THC in serum and plasma may prove useful to equine practitioners who are trying to confirm a suspicion of marijuana exposure in their patients. Both the jack and jenny had detectable concentrations of THC in serum and plasma using HPLC-MS/MS, which likely explains the clinical signs described.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211064269 – Supplemental material for Marijuana toxicosis in 2 donkeys
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211064269 for Marijuana toxicosis in 2 donkeys by Alyson H. Fitzgerald, Geraldine Magnin, Ellen Pace, Karyn Bischoff, Toby Pinn-Woodcock, Ron Vin, Michael Myhre, Emily Comstock, Steve Ensley and Johann F. Coetzee in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
Internal funding was provided by the Kansas State Veterinary Diagnostic Laboratory.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
