Abstract
Canine hepatozoonosis, caused by Hepatozoon americanum, is an emerging tick-borne disease of dogs in North America. In addition to the skeletal and cardiac myositis that are prominent features of the disease, there is disseminated periosteal bone proliferation in most dogs that manifest clinical disease. Each of six experimentally infected animals (four dogs and two coyotes) and seven of eight naturally infected dogs had gross or histopathologic osteoproliferative lesions. Experimental animals were 6–9 months of age when exposed. Naturally infected dogs were 8 months to 11 years old when subjected to necropsy. Lesions occurred primarily on the diaphysis of the more proximal long bones of the limbs; however, flat and irregular bones were frequently involved. Lesions involving metacarpals, metatarsals, and digits were infrequent. The earliest observed periosteal lesions were in an experimentally infected dog 32 days after exposure to sporulated oocysts of H. americanum. There were hypertrophy and hyperplasia of osteoprogenitor cells, and osteoblasts appeared in the cellular zone of the periosteum. Spicules of woven bone oriented perpendicularly to bone cortex followed. Later yet, periosteal new bone was remodeled and tended to become oriented parallel to the cortical bone. Horizontally oriented zones of remodeled, condensed bone sometimes occurred in multiple layers on the original cortex, forming “pseudocortices.” The osseous lesions of American canine hepatozoonosis, with few variations, are remarkably similar to those of hypertrophic osteopathy in domestic dogs and other mammalian species, including humans.
Canine hepatozoonosis was first described in India in 1906; the causative organism was classified as Leukocytozoon canis. 9 Later, the parasite was transferred to the genus Hepatozoon. 21 Canine hepatozoonosis is an emerging tick-borne disease of dogs in the USA, first confirmed in dogs from Texas 3 and later reported in Louisiana, 7 Alabama and Georgia, 11 , 19 and Oklahoma. 15 Salient clinical features of American canine hepatozoonosis (ACH) include fever, weakness, myalgia, bone pain, mature neutrophilic leukocytosis, wasting, poor response to treatment, and periosteal new bone proliferation. 2 , 11 , 19 The disease in North America is considerably more aggressive, progressive, and more often fatal than hepatozoonosis in the Old World. The causative agent of ACH was originally considered to be a particularly virulent strain of Hepatozoon canis, the etiologic agent of canine hepatozoonosis in the Old World. 3 , 4 However, differences in the clinical and pathologic features of disease in the USA and the Old World led to suspicion that the causative agent of canine hepatozoonosis in North America might be different. In 1997, because of these differences, the tissue location of merogony, and unique features in the morphology of gamonts, the parasite was designated a new species, Hepatozoon americanum. 20 This report focuses on gross and histopathologic features of the osteoproliferative lesions as observed in natural and experimental infections with H. americanum in domestic dogs and coyotes. We shall define the quality, location, and distribution of the osseous lesions of ACH and compare those with other syndromes that are characterized by widely distributed, nonerosive, osteoproliferative lesions of the periosteum.
Materials and Methods
Observations reported here were obtained by examination of four dogs and two coyote pups experimentally infected in the course of transmission studies 10 , 13 and eight naturally infected dogs that were included in a study of the tissue stages of H. americanum. 17 The experimentally infected dogs were aged 6–9 months at exposure and were infected by ingestion of experimentally infected ticks or sporulated oocysts harvested from such ticks as previously described. 13 The coyote pups were approximately 10 weeks old at oral exposure to sporulated oocysts. 10 Experimentally infected dogs and coyotes were monitored daily. Each developed clinical signs typical of the disease. Dogs were subjected to euthanasia and necropsy 4.5, 5, 10, and 26 weeks after exposure, 16 coyotes were euthanatized and necropsied 14 and 17.5 weeks after exposure. 10 Naturally infected dogs were in a clinically advanced state of disease when submitted for euthanasia and necropsy; neither age at exposure nor duration of infection was known. 17
Gross necropsy included examination of many soft tissues and bone and their preservation for histopathologic examination. The remaining skeleton was prepared for more comprehensive gross examination by submergence in water to which detergent had been added, then heated in an autoclave at 15 psi for 30–60 minutes. This procedure facilitated removal of soft tissue to expose underlying bone. Processed bones were then dried and visually examined. Formalin-fixed bone was decalcified and prepared for histopathologic examination of hematoxylin and eosin–stained sections. Sections were examined by routine and polarized light microscopy.
Description of lesions is based on a composite of observations in both dogs and coyotes with experimentally induced infections and dogs with naturally occurring disease.
Results
At necropsy the periskeletal soft tissue and periosteum of the lower legs were often perceptibly thickened and edematous. Occasionally the periosteum and other body tissues were mildly petechiate. On cut section of fresh or fixed segments of bone, there were eccentrically or circumferentially arranged plaques of spongy bone of varying thickness deposited on the original cortex. Lesions observed in the processed skeletal preparations ranged from shallow, broad, slightly elevated plaques of rather porous-appearing bone to more abrupt, thicker, and irregular plaques, to marked, more or less concentrically thickened regions of bone (Fig. 1). On transverse section, the underlying original cortex was usually covered by spongy bone and, often in cases of longer duration, with a thin layer of more compact bone at the outer margin of the bone organ (Fig. 2). Evidence of endosteal new bone deposition was not observed.
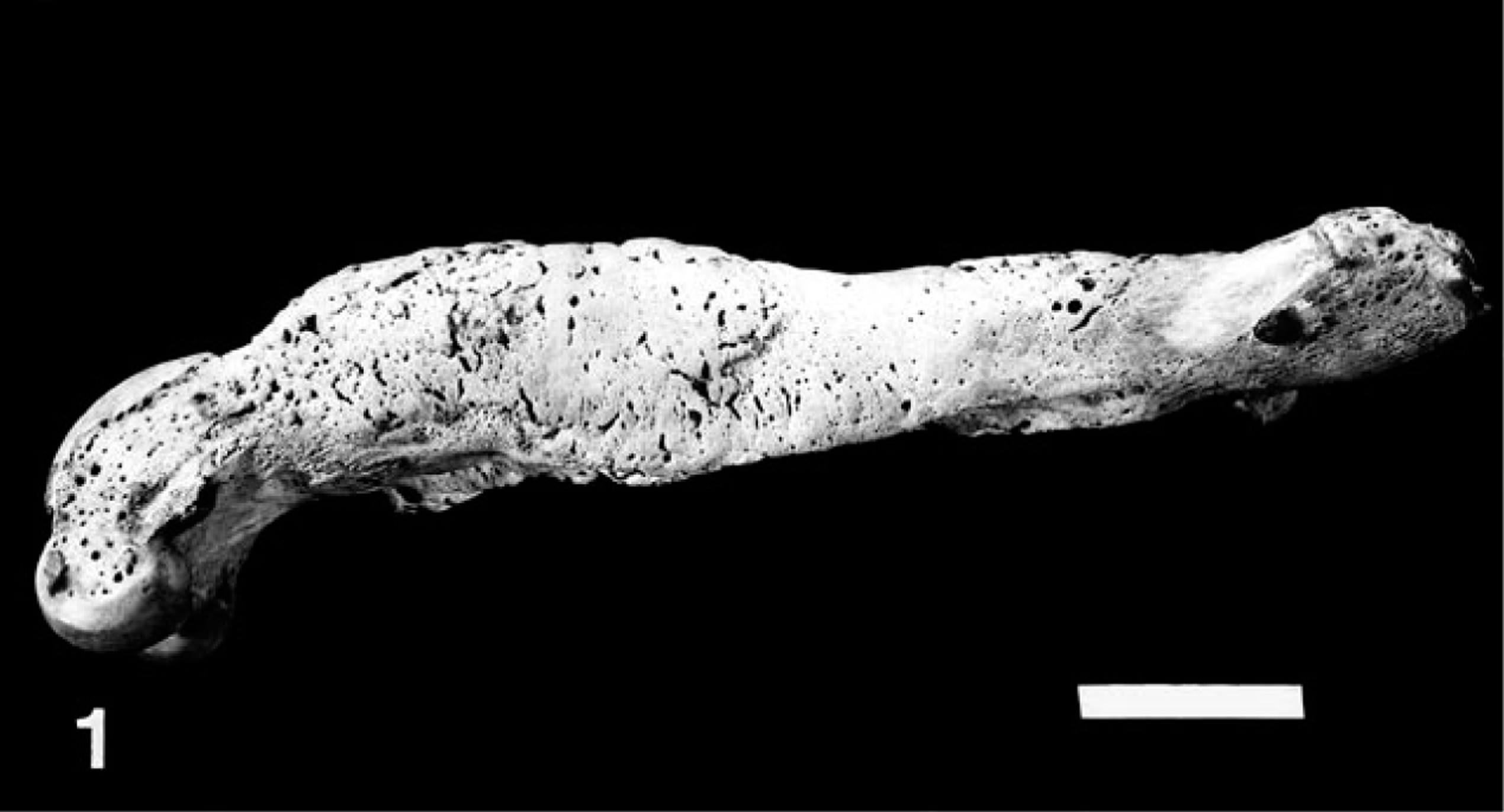
Lateral view of femur 123 days after experimental exposure to sporulated oocysts of H. americanum; young coyote. The diaphysis is diffusely but irregularly expanded by the addition of periosteal new bone. The proximal and distal metaphyses are not affected. Bar = 2 cm.
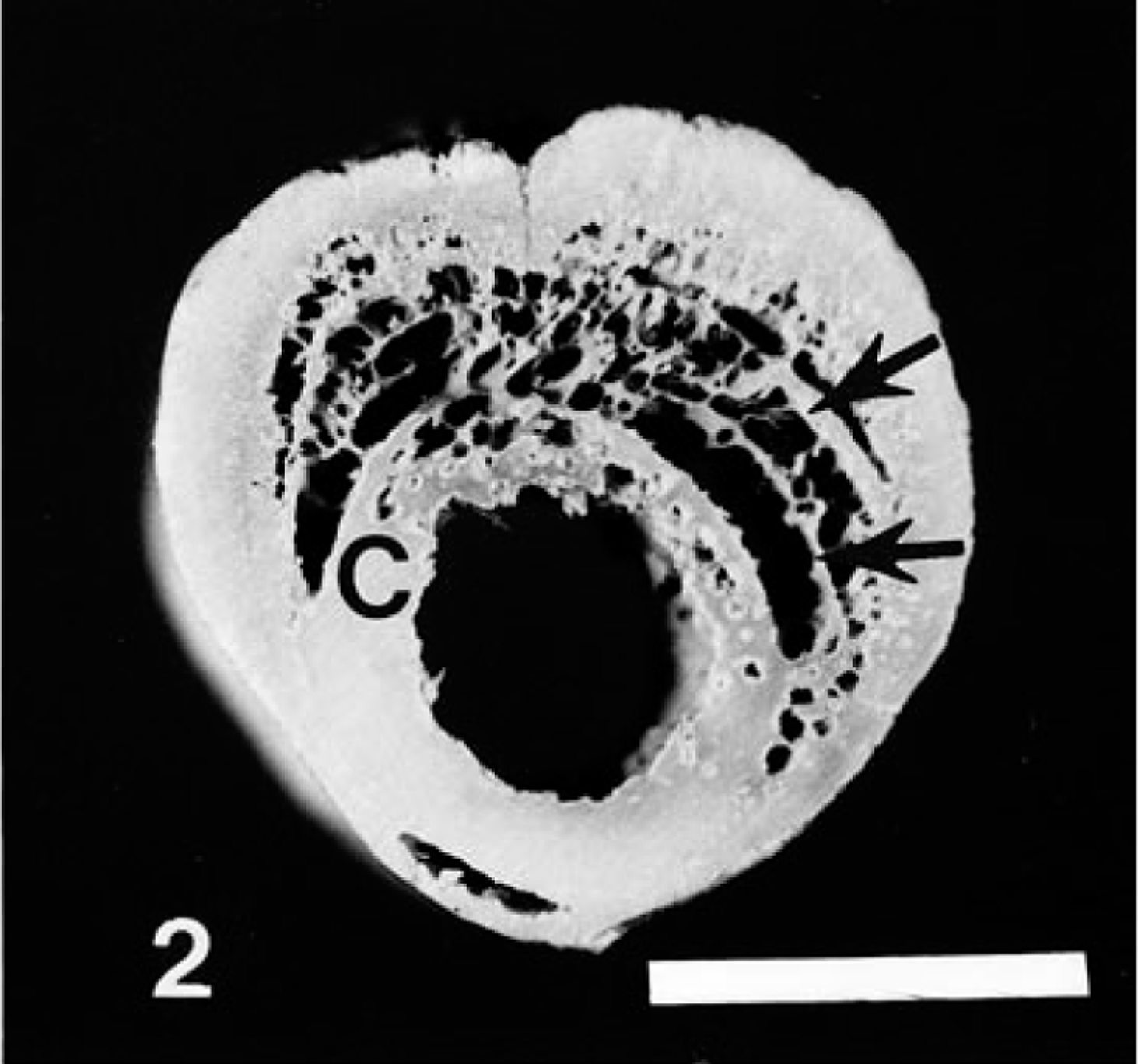
Cross-section of mid-humeral diaphysis 123 days after exposure to oocyts of H. americanum; young coyote. There is marked periosteal hyperostosis eccentrically distributed on the original cortex (C). Note pseudocortices (arrows) arranged parallel to original cortex. A portion of the original cortex is porotic. Bar = 1 cm.
Periosteoproliferative lesions were most common in the more proximal long bones of the limbs. Lesions were generally most severe on the femur and humerus, more pronounced in the distal third of the diaphysis but prominent in the proximal diaphysis as well. The metaphyses, though generally spared, occasionally had relatively mild lesions. The caudal and lateral surface of the proximal two thirds of the tibial and fibular diaphyses were commonly affected; the distal diaphyses and the metaphyses were often free of lesions. Involvement of the radius and ulna was common and most severe on the caudal and lateral surfaces of the proximal diaphysis; the distal 10–20% of the diaphyses was usually without lesions. Metacarpals and metatarsals only occasionally had mild proliferative lesions, usually involving their proximal third. Phalanges appeared to be spared. Scapulas were often diseased; periosteal bone proliferation occurred most commonly and most severely on the scapular neck and the proximal portion of the scapular spine; lesions also were frequent on the supra- and infraspinous fossas. The medial surface was less frequently affected. The prominently affected surfaces of the os coxae included the lateral surfaces of the tuber coxae, the dorsolateral area of the body of the ilium, and the dorsal surface of the ischium. The axial skeleton was generally less frequently and less severely involved than the appendicular skeleton. However, when widely distributed osteoproliferation was severe, bones of the skull and vertebral column were prominently affected. When osteoproliferation was less extensive, it tended to be present on the rostral margin of the bony orbits, the rostrolateral maxilla and mandible, and the lateral surface of the zygomatic and mandibular bones. Vertebral lesions were usually on lateral surfaces of the vertebral arches and dorsal spinous processes, less frequently on transverse and articular processes, and uncommonly on vertebral bodies. Ribs were usually without lesions.
Each of the eight naturally infected dogs had the multifocal disseminated parasitic myositis that is characteristic of the disease; 3 , 5 , 11 none of the eight had either neoplastic or inflammatory lesions in the thorax or bladder neoplasms. One dog, an 11-year-old mixed breed, neutered male, had a malignant pheochromocytoma of the left adrenal that occupied the left dorsal abdomen and infiltrated adjacent hypaxial and epaxial musculature and a single vertebral body.
Histopathology
The structure of periosteum of normal dogs includes an outer layer of mildly cellular, densely collagenous tissue whose cell nuclei are very slender and spindle shaped. Between this layer and cortical bone is a more densely cellular zone whose nuclei are somewhat more plump and whose cytoplasm slightly more voluminous (Fig. 3).

Longitudinal section of cortex and periosteum of humerus; normal dog. Note periosseous soft tissue (A), densely collagenous outer zone of periosteum (B), less densely collagenous, more densely cellular inner zone (C), and underlying cortical bone (D). HE. Bar = 50 µm.
The earliest recognizable changes in periosteal structure in this series of animals with ACH were swelling and hypertrophy of osteoprogenitor cells in the deeper layer of periosteum. Spindle cells became plump and clearly transformed to stellate morphology; they developed active, vesicular nuclei and prominent nucleoli and accumulated abundant, slightly basophilic cytoplasm that often contained a prominent Golgi zone. Such cells became aligned in single or multiple layers and underwent hyperplasia, creating a thick and densely cellular zone on the cortical surface (Fig. 4). Osteoid was deposited, osteoblasts became entrapped, and spicules of woven bone continued to be formed resulting in a thick layer of new, woven, periosteal bone. More recently formed spicules were covered by a layer of osteoblasts and were oriented perpendicularly to the cortical surface (Fig. 5). The connective tissue surrounding new bone was loosely arranged.
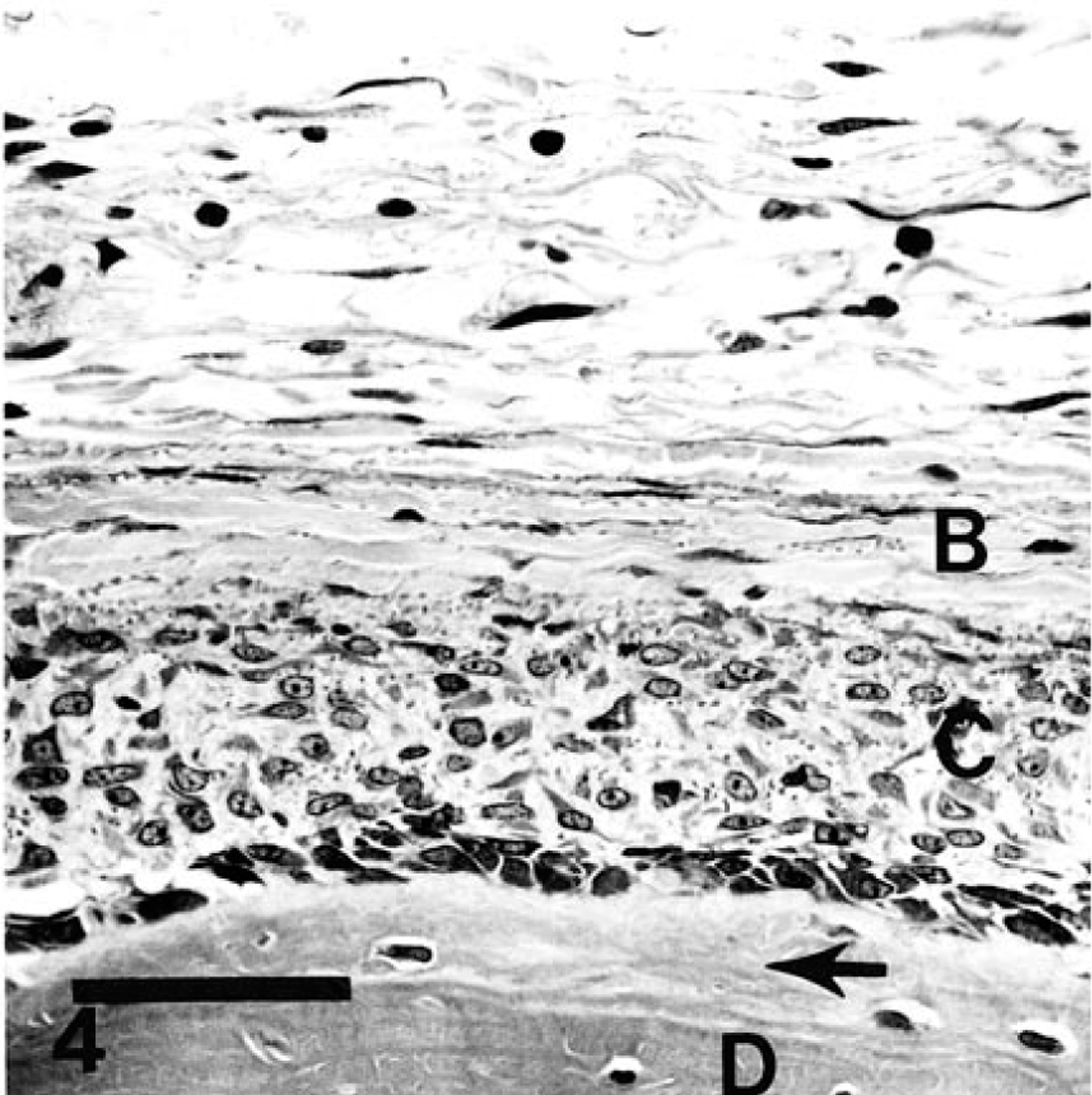
Humerus 5 weeks after oral exposure to sporulated oocysts of H. americanum; dog No. 3152. Early stage of osteoproliferation is characterized by a thin layer of new bone (arrow) on cortical surface (D), a layer of osteoblasts, a zone of hypertrophied osteoprogenitor cells (C), and an outer layer of edematous, densely fibrous periosteum (B). HE. Bar = 50 µm.
In cases of longer duration there was considerable osteoclastic activity and remodeling of woven bone apparent first adjacent, to the original cortex. As remodeling progressed, the original vertical orientation of spicules tended to change, becoming parallel to the original cortex (Fig. 5). Occasionally, presumably as a result of alternating periods of proliferation and quiescence, the development of laminae of remodeled, more dense bone resulted in the formation of “pseudocortices” 6 (Fig. 2). Spaces between spicules adjacent to the cortex occasionally contained marrow. The junction of the original cortex and remodeled new bone was sometimes difficult to distinguish. The presence of cement lines (parallel to the original cortical surface) and the smaller osteocytic lacunae and more consistently parallel orientation aided in identification of cortical bone.
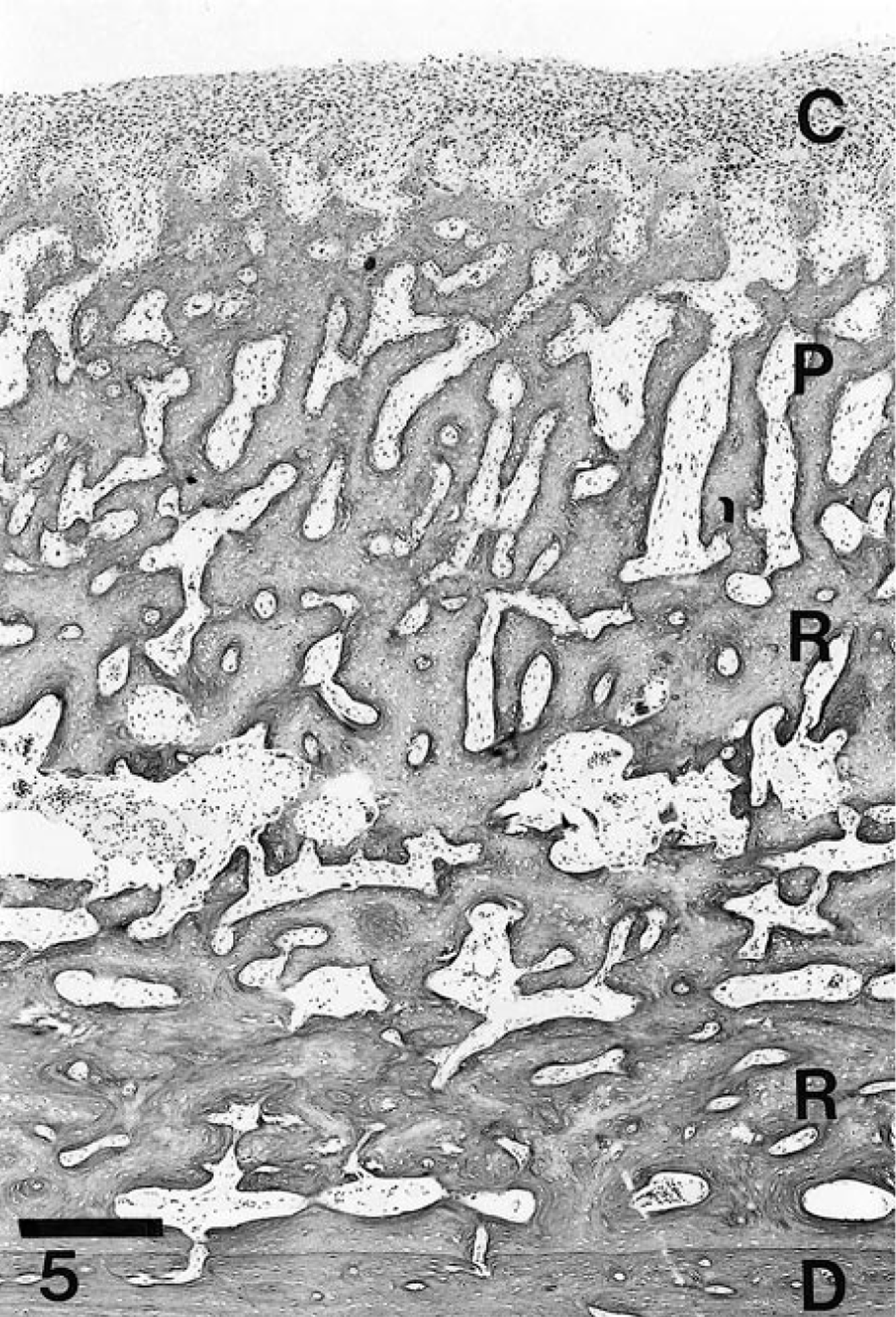
Section of femur; young coyote. Note outer, highly cellular, actively osteogenic zone of periosteum (C), perpendicularly oriented spicules of new bone (P), deep zone of partially remodeled bone (R) oriented somewhat parallel to the underlying cortical surface (D). HE. Bar = 50 µm.
Early in the osteoproliferative process, endothelial hypertrophy was obvious in vessels adjacent to and within the periosteum; vessels were dilated and prominently present but not clearly more numerous than in normal bones. In some cases severe edema was present in the periosteum and adjacent connective tissue and skeletal muscle. Muscle atrophy and eventually fibrosis often accompanied the edema. Clear evidence of inflammation within or adjacent to the periosteum was not apparent. Evidence of the periosteal lesion was observed histologically in one dog as early as 5 weeks after experimental exposure to H. americanum. In most instances there were neither parasites nor inflammation in skeletal muscle adjacent to bone lesions.
Discussion
The osseous lesions that characterize ACH fundamentally consisted of disseminated, symmetric, periosteal new bone formation that most frequently and most severely involved the diaphysis of long bones and to a lesser extent flat and irregular bones. Osseous lesions consistently developed in experimentally infected young dogs and coyotes and are present in most dogs with natural disease. 2 , 11 Seven of eight diseased dogs whose prepared skeleton was examined had lesions. Histologically recognizable bone lesions developed within 5 weeks after experimental exposure and progressed rapidly through stages of morphologic development that closely resembled those of hypertrophic osteopathy (HO). It has been suggested that the periosteal new bone is a local response to hepatozoon-induced inflammatory lesions in adjacent muscle. 5 We cannot confirm such a relationship. The disseminated and symmetric distribution of osteoproliferation is more logically attributable to periosteal stimulation by humoral factors than to local ones.
There are a limited number of diseases of humans and other mammals that include, among their pathologic effects, acquired regional or generalized bone proliferation 1 , 14 , even fewer are accompanied by generalized or disseminated periosteal new bone proliferation. Those canine diseases in the latter group include, in addition to ACH, hypertrophic osteopathy, craniomandibular osteopathy, canine panosteitis, and hypertrophic osteodystrophy. 14 With the exception of HO, lesions associated with each of these syndromes can be readily differentiated from those of ACH based on a combination of radiographic findings, distribution on the skeleton, and specific location within or on the bone organ. Differences between the bone lesions of HO and ACH, on the other hand, are not as clearly defined.
HO (hypertrophic osteoarthropathy) is a syndrome that occurs in various mammals, including humans and is characterized by regional or disseminated areas of periosteal osteophytosis. 18 Two forms of HO occur in humans—a primary, genetically dependent syndrome (familial infantile cortical hyperostosis) and secondary HO, in which development of the osseous changes is preceded by primary lesions in the thorax or occasionally other loci. 1 In human beings, where joints are frequently involved, the syndrome is usually termed hypertrophic osteoarthropathy and is characterized by 1) overgrowth of ends of digits, 2) painful arthritis, and 3) periosteal new bone formation. 8 Most or all reported cases in various other animals are of the secondary type and generally are without articular involvement or digital clubbing.
There are remarkable similarities in the gross and microscopic morphology of the osseous lesions of ACH and those of canine and human HO. 6 , 14 Each is characterized by disseminated periosteal new bone proliferation primarily involving the diaphyses of long bones. The periosteal reaction in each occurs without cortical destruction. The orientation of spicules of new bone is often clearly perpendicular to the cortex in HO, less clearly and less permanently, but nevertheless similarly oriented in ACH. In each, new bone is subject to remodeling with alteration of the perpendicular orientation of trabeculae and the formation of pseudocortices whose orientation is parallel to cortex; in each there are initial or early endothelial hypertrophy, congestion, and edema of the periosteum and adjacent soft tissue. In HO in human beings there are mild to moderate degrees of inflammation in the periosteum; 6 such changes are absent or minimal in canine HO and ACH. Other differences include the greater rapidity of onset and progression of the bony changes in ACH and the lack of an association with primary lesions in visceral organs. Whereas bony proliferation tends to occur first in metacarpals and metatarsals in canine HO, with ACH proliferation occurs initially in more proximal bones of the limbs; distal bones are usually spared.
The pathogenesis of the bone lesions of neither HO nor ACH is known. It is believed that an initial mechanism for osteoproliferation involves an increase in blood flow to and fluid retention in the limbs. These changes are followed by proliferation of vascular connective tissue and periosteum and subsequently bone deposition. 12 Hypotheses as to the cause of these events include a hormonal theory based on increased levels of circulating estrogen and growth hormone in human patients. An hypoxic theory seems negated by the existence of normal blood gas values. An intrapulmonic vascular hypothesis suggests that substances responsible for HO are humoral and, in the absence of pulmonic vascular shunts, are normally catabolized in the lung. Overproduction of humoral substances by diseased tissues may overwhelm the capacity of the lungs to inactivate the posited humoral factors. A neurologic hypothesis proposes that afferent impulses from a focus of pleural or pulmonary disease travels via intercostal or vagal nerves to initiate reflex vasodilation in the limbs. An additional hypothesis proposes that inappropriate stimulation of an extrarenal volume receptor system leads to fluid retention and limb vessel dilation followed by osteoproliferation. 1 , 12
At present, there is no unified concept of the pathogenesis of HO. It is likely there are a very limited number of mechanisms for the induction of generalized periosteal proliferation, thus increasing the probability that insight into experimentally induced ACH with its attendant generalized periosteal proliferation syndrome is relevant to the pathogenesis of other periosteal proliferative diseases. The bone lesions of ACH and HO have many features in common. It is likely that studies of the pathogenesis of the canine disease would provide fundamental information about HO in the dog and humans.
