Abstract
Hypertrophic osteopathy (HO) is a condition in which periosteal bone forms along long bone diaphyses and metaphyses. Lesions generally affect all 4 limbs, and often involve the distal portions. The pathogenesis is incompletely understood, and many, but not all, cases are associated with concurrent neoplasia or space-occupying masses. Among veterinary species, most cases are reported in dogs, cats, and horses, with sporadic cases in other domestic and non-domestic species. In cervids, this condition is often associated with fungal granulomas, typically in the lung. We report HO, with findings consistent to other veterinary species, in a farmed white-tailed deer (WTD) with bacterial pneumonia and in 3 free-ranging WTD, one of which had fungal pneumonia. Recognition of HO in WTD and potentially associated conditions can lead to improved sample collection in the field. Such information can improve characterization of the disease, complement our understanding of comparative pathology of this condition, and raise awareness among individuals or institutions working with cervids.
Hypertrophic osteopathy (HO), also known as hypertrophic pulmonary osteoarthropathy or Marie disease, is a condition of incompletely understood pathogenesis in both humans and veterinary species. 4 In HO, periosteal bone forms along the long bone diaphyses and metaphyses, especially those of the distal limbs. 4 In most veterinary species, lesions are bilateral and often involve all 4 limbs.11,20 Most reported cases involve an intrathoracic mass; however, extrathoracic masses, cardiac abnormalities, and a variety of inflammatory conditions have also been associated with this syndrome. 4 If the initiating cause of the bony lesions can be recognized and resolved, such as via surgical removal of a neoplasm, bone lesions potentially regress. 1 However, in cases of severe disease and debilitation, affected animals may be euthanized due to poor prognosis.
Four white-tailed deer (WTD) with HO were examined by several wildlife diagnostic institutions over 18 y (2004–2022) in the eastern United States. Case 1 was a wild, hunter-harvested 2.5-y-old doe with reported swelling of all 4 limbs from which samples were collected in the field (Fig. 1). A forelimb, a hindlimb, and whole, fresh brain were submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS; Athens, GA, USA) from the Kentucky Department of Fish and Wildlife Resources. No other field findings (e.g., gross lesions in other organs) were reported, and no gross or histologic lesions were observed in the brain. The metacarpal and metatarsal bones were diffusely and irregularly thickened along the metaphyses and diaphyses (up to 6 cm diameter) by expansion of hard proliferative periosteal bone without involvement of the joints.
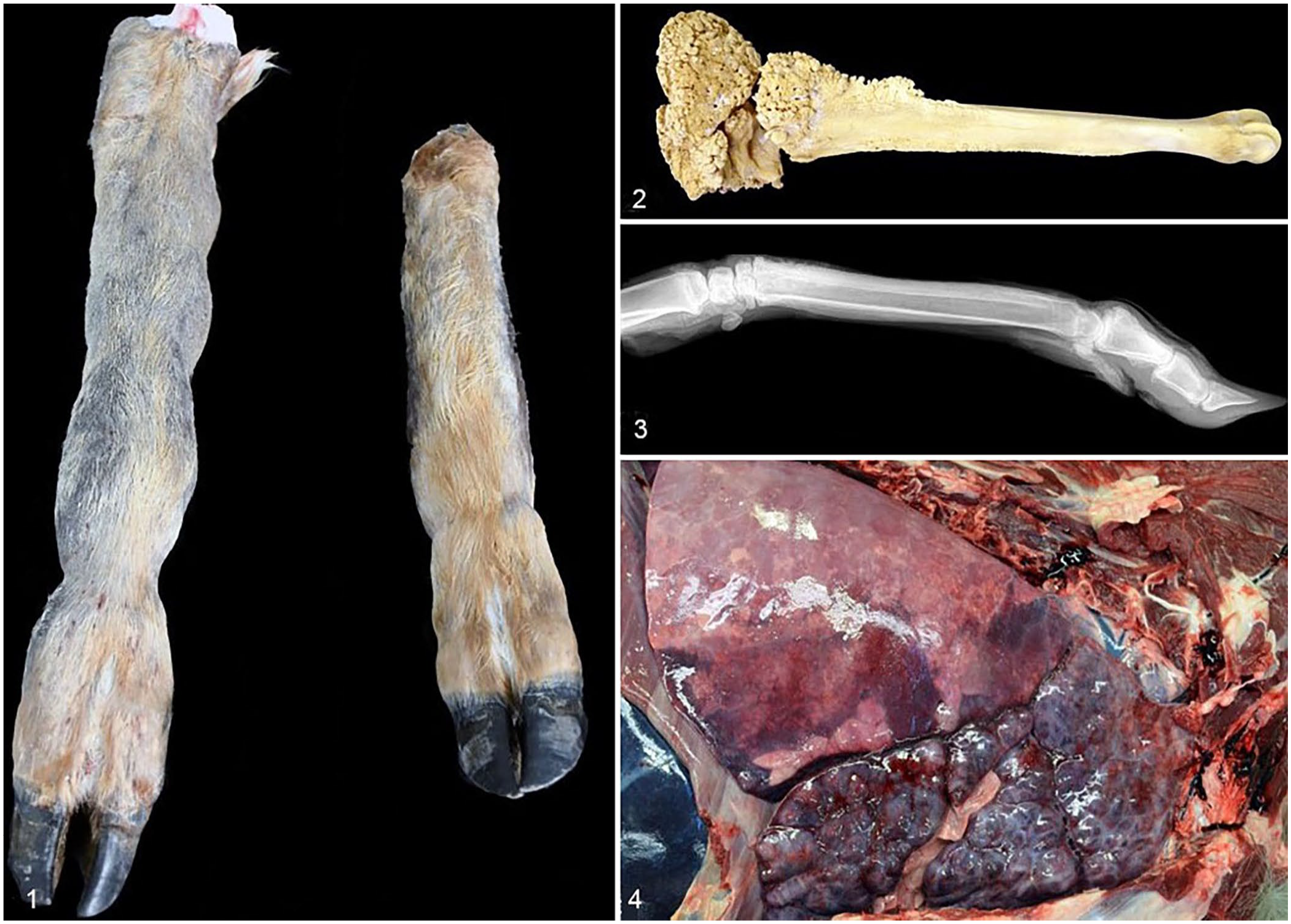
Hypertrophic osteopathy and associated pneumonia in white-tailed deer.
Case 2 was a wild, 1.5-y-old WTD doe that was euthanized and autopsied in the field. It was observed in poor nutritional condition with thickened lower limbs. Formalin-fixed sections from a large pulmonary abscess and cross-sections of the distal tibia were submitted to SCWDS from the West Virginia Division of Natural Resources. Microscopically within the lung, there was severe fibrosis and coalescing necrotic nodules containing intralesional fungal hyphae that stained with both periodic acid–Schiff reaction and Grocott methenamine silver stain (GMS). No microorganisms were evident by a Ziehl–Neelsen acid-fast stain on the lung.
Case 3 was a wild, >2.5-y-old (adult) WTD male from the Pennsylvania Game Commission that was euthanized due to poor nutritional condition and autopsied in the field. Abscesses were found in both lungs, and all 4 distal limbs were expanded by hard swellings. The distal limbs were submitted to the SCWDS. In both metatarsal and metacarpal bones, the periosteum was expanded focally and diffusely by bony proliferation of the diaphysis and metaphysis. Orthogonal radiographs of a forelimb and hindlimb were obtained at the College of Veterinary Medicine, University of Georgia (Athens, GA, USA) and showed irregular expansion of the periosteum of the diaphysis and metaphysis of both the metacarpal and metatarsal bones by rough-edged tissue of bone-like opacity.
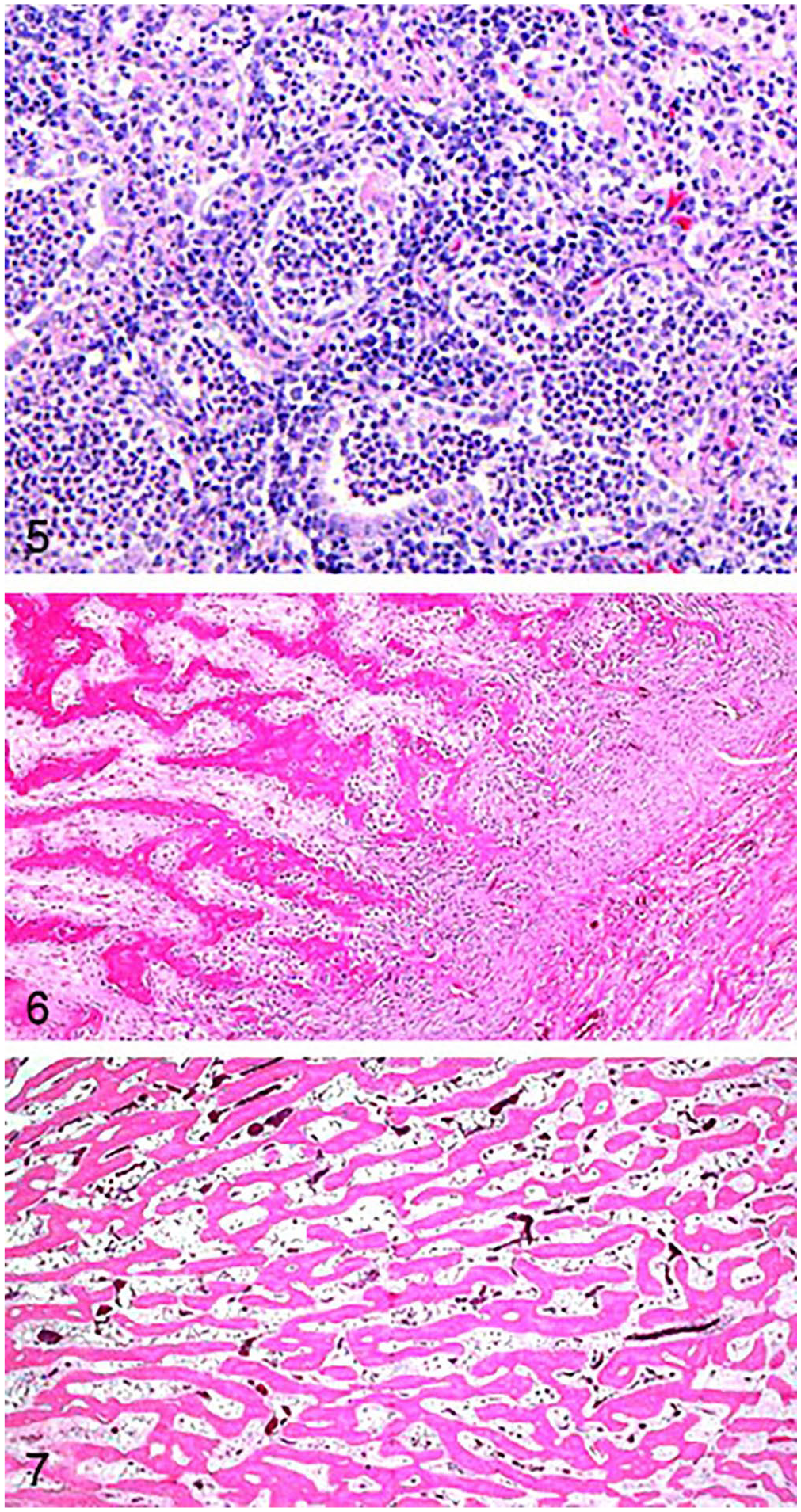
Case 4 was a captive 2-y-old WTD buck carcass that was submitted to the Pennsylvania State University–Animal Diagnostic Laboratory (ADL; University Park, PA, USA) after being found dead in its enclosure. Available case history noted the deer was in poor nutritional condition and had difficulty walking. All 4 distal limbs had hard swellings of the metaphyses (Fig. 2). Orthogonal radiographs of a forelimb taken at the VCA Metzger Animal Hospital (State College, PA, USA) had similar radiographic changes to case 3, with the phalanges additionally affected (Fig. 3). The cranioventral lung lobes had bilateral consolidation with abscesses and fibrous adhesions to the parietal pleura and the pericardium, affecting ~60% of the total lung volume (Fig. 4). Microscopically, alveoli and bronchioles were diffusely filled with neutrophils, fibrin, and edema with foci of necrosis and large colonies of bacteria (Fig. 5). Foci of organizing fibrin with type II pneumocyte hyperplasia and fibrosis entrapped bronchioles and compressed and replaced alveoli; lymphoplasmacytic pleuritis was diffuse and chronic. A GMS stain showed intra- and extracellular argyrophilic pleomorphic bacteria (coccobacillary-to-filamentous) within bronchioles. No fungal hyphae were highlighted with H&E or GMS stains. The tracheal mucosa was covered by red-brown, finely granular material, and, microscopically, was diffusely hyperplastic and formed folds with erosions. The lamina propria and submucosa were infiltrated by abundant lymphocytes and plasma cells. No significant lesions were observed in other organs. Aerobic culture of the lung yielded heavy growth of Pasteurella multocida, and anaerobic culture yielded moderate growth of Fusobacterium necrophorum. Mycoplasma spp. were not detected by genus-level PCR of a bronchial swab.
Bronchopneumonia and hypertrophic osteopathy in a captive white-tailed deer, case 4.
Microscopic lesions in bone sections from all 4 deer were similar and included marked expansion of the periosteum by thick trabeculae of periosteal new bone, which were covered by plump osteoblasts that radiated perpendicular to the cortex, with various degrees of overlying fibrosis, and hematopoietic elements filling the intertrabecular spaces (Figs. 6, 7).
In cervids, HO has been reported in 5 free-ranging and captive animals (reviewed here) and is most often associated with disseminated fungal disease. A free-ranging WTD in Iowa, USA had systemic Conidiobolus incongruus infection and was diagnosed with HO. 11 The emaciated buck had HO lesions affecting the distal hindlimbs and a large pulmonary mass, with smaller masses throughout the abdominal cavity and viscera. Similar disease was reported in 2 farmed wapiti (Cervus canadensis) in association with Aspergillus fumigatus pneumonia. 8 The yearling bulls were from Indiana, USA, and had anorexia, weight loss, and progressive lameness. Both had firm, bilateral swelling of the metatarsi, metacarpi, distal radii and ulnae, and calcanei. The dorsal aspects of the bones were affected more severely than the palmar and plantar surfaces. In both wapiti, the lungs had pyogranulomatous nodules with fibrous pleural adhesions. One of the wapiti also had granulomas in several lymph nodes, a kidney, and the myocardium, as well as concurrent ulcerative glossitis, rumenitis, and interstitial nephritis. Fungal hyphae histomorphologically compatible with Aspergillus spp. were detected within the pulmonary, cardiac, and renal granulomas, and exhibited strong immunolabeling with Aspergillus spp. antibodies. In surveillance of free-ranging Slovenian roe deer (Capreolus capreolus) from 2000 to 2017, 4 of 586 deer examined had pulmonary mycosis attributed to A. fumigatus infection. 19 Of these, HO was diagnosed in one deer, an emaciated, ~5-y-old buck. The buck had marked bilateral swelling surrounding the metacarpo- and metatarsophalangeal joints. These swellings caused decreased range of motion, and the deer had hoof overgrowth, possibly secondary to the bony lesions. The lungs contained caseous nodules with associated fibrous adhesions. Radiographic examination of the limbs revealed periosteal proliferation of the tarsal, carpal, metacarpal, and phalangeal bones. Histologically, the necrotizing and granulomatous lung lesions contained intralesional hyphae consistent with A. fumigatus (identified during aerobic culture). The last case was in an ~3-y-old, roe deer buck with a pulmonary abscess. 16 The distal phalanges, metacarpi, and metatarsi all had characteristic periosteal proliferation and hyperostosis. This buck was hunter-harvested and reportedly was not clinically affected. Thus, of the 5 reported cases, at least 4 were associated with fungal disease, with aspergillosis occurring most frequently. In 4 cases, all limbs were affected, and in the remaining case, both metatarsi were affected, consistent with descriptions in other veterinary species.
The clinical and pathologic findings in our HO cases in WTD are consistent with those reported in the literature. Our results support that this condition occurs sporadically in WTD in both captive and free-ranging populations and can be associated with chronic pulmonary disease. In one of our cases, bacterial pneumonia due to P. multocida and F. necrophorum was associated with HO, an association not reported previously in WTD. We suspect that the pulmonary abscesses and chronic inflammation contributed to the skeletal lesions, compatible with findings in other species. Our case 2 had fungal pneumonia, consistent with reports that fungal pulmonary disease can be associated with HO in cervids. Although one of our cases lacked ancillary diagnostic testing and histologic examination of the lung mass, the gross appearance of the pulmonary nodule was similar to cases of chronic fungal pneumonia in cervids seen previously at SCWDS (unpublished data).
Although the pathogenesis of HO remains incompletely understood, a variety of proposed factors are suggested. For cases with pulmonary lesions, stimulation of visceral nerves, increased peripheral blood flow, and angiogenesis are thought to induce proliferation of distal limb periosteum and associated connective tissue.4,20 Shunts bypassing pulmonary circulation have also been shown to increase peripheral vascular circulation, which may lead to periosteal thickening from lower oxygenation of blood in the distal limbs.4,13 Other suggested contributing factors noted in the human medical literature include tumor or inflammatory cytokine-induced production and release of growth factors, such as vascular endothelial growth factor and platelet-derived growth factor, in peripheral limb vasculature, and altered platelet circulation.4,20 In humans, hypertrophic osteoarthropathy is secondary in 95–97% of cases, although a primary, autosomal dominant form is also described. 21 The secondary form can be classified as generalized or localized (1 or 2 limbs affected), with visceral lesions associated with the generalized form and various vascular anomalies and hemiplegia associated with the localized form. 21 The release of growth factors is thought to stimulate vascularity, fibroblasts, and osteoblasts, in turn leading to bony changes. 21 Both vagotomy and surgical removal of the initiating pulmonary lesions can lead to regression of limb lesions, supporting the role of a neural vascular reflex.4,13
In the veterinary medical literature, most HO reports are in domestic animals, particularly dogs, cats, and horses, and patients often have concurrent intrathoracic lesions. In dogs, HO is most commonly a paraneoplastic syndrome that has been associated with a variety of neoplasms, including primary and metastatic pulmonary, renal, hepatic, mammary, bone, and urinary bladder neoplasms. Other associated conditions include bacterial endocarditis, pneumonia, pulmonary abscesses, heartworm disease, and congenital megaesophagus.2,13,14,20 In cats, associated conditions include renal, adrenocortical, and pulmonary neoplasia, and less commonly, megaesophagus, nutritional disorders, and cardiac malformations.1,6,7 In horses, HO has been associated with ovarian and gastric neoplasia, nodular pulmonary fibrosis, pulmonary abscesses and neoplasia, and intrathoracic and extrathoracic granulomatous disease.10,12,15,18
In non-domestic species, HO has been described in both free-ranging and captive animals. In free-ranging wildlife, reports include several cervid species with pulmonary masses, koalas (Phascolarctos cinereus) with pulmonary actinomycosis, and 2 raccoon dogs (Nyctereutes procyonoides) with nodular pulmonary lesions.9,17 Other reported animal groups and species affected include non-human primates, large non-domestic felids, alpacas, donkeys, cattle, mink, and sheep.3,12,17 Although many cases involve obvious thoracic masses or other suspected predisposing causes, some cases are considered idiopathic, even after extensive diagnostic evaluation.5,11
Hunter-harvested wildlife cases present an additional complication in determining possible predisposing causes, as carcasses are not as thoroughly examined as those submitted to a diagnostic laboratory, and thus gross lesions may be missed, especially if they are subtle or in organs less-often examined and/or collected. HO has similar characteristic gross, histologic, and radiographic lesions in cervids as in domestic veterinary species, and chronic pulmonary disease should be considered as an inciting cause in cases in which HO is suspected. Based on the reported cases of HO in cervids, thorough evaluation of the lungs is warranted to look for potential predisposing causes. If no masses are found in the lungs, examination of other thoracic and abdominal viscera may be indicated, and a more detailed autopsy may be needed to work-up cases without an obvious gross cause.
Footnotes
Acknowledgements
We thank SCWDS member state wildlife agencies for diagnostic case submission, including the states of Alabama, Arkansas, Florida, Georgia, Kansas, Kentucky, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. Further support was provided by SCWDS federal wildlife agency partners, including the U.S. Fish and Wildlife Service National Wildlife Refuge System and the U.S. Geological Survey Ecosystems Mission Area. We also thank the personnel of the ADL, VCA Metzger Animal Hospital, and Athens Veterinary Diagnostic Laboratory for technical support in autopsy, microbiology, histopathology, and radiology.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the sponsorship of the SCWDS by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kansas, Kentucky, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. Support from the states to SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
