Abstract
Postmortem examination of a free-range white-tailed deer (Odocoileus virginianus) revealed severe emaciation, bilateral firm proliferation of the metatarsal diaphyses, and a large intrathoracic mass associated with the accessory lung lobe. Smaller masses were evident in the abomasum, duodenum, omentum, and the capsular surface of the liver. Microscopically, the masses were similar and were diagnosed as eosinophilic granulomas with intralesional fungal hyphae characteristic of Zygomycetes spp. Fungal hyphae were identified as Conidiobolus incongruus by 18S ribosomal RNA sequencing on fresh lung tissue. Furthermore, the proliferative lesions of the metatarsal bones along with the intrathoracic mass were compatible with hypertrophic osteopathy.
Conidiobolus species are opportunistic fungal pathogens classified in the order Entomophthorales of the class Zygomycetes. This genus is distributed worldwide and is found in soil and decaying organic material. 11 Three species, Conidiobolus coronatus, Conidiobolus lamprauges, and Conidiobolus incongruus are capable of causing illness and death in humans and animals. 11 In wild or domestic animals, C. incongruus has previously been reported to infect sheep 4,10 and red deer in Australia. 3 In both cases, the fungus was associated with nasal swelling, rhinocerebral mycosis, exophthalmia, and disseminated disease.
An approximately 2.5-year-old, male, free-range white-tailed deer was observed in Ledges State Park (Boone County, IA) to be in poor body condition and debilitated with swollen hind legs. Upon physical examination, the deer had a dull hair coat, bilaterally sunken eyes, and diffuse firm proliferations along the entire metatarsal diaphyses of both hind legs. Necropsy revealed marked emaciation with scant retroperitoneal fat and a 25 cm × 13 cm × 15 cm ovoid intrathoracic mass associated with the accessory lung lobe, which displaced adjacent lung lobes (Fig. 1). The mass was firm, contained multiple 1–4-cm cystic structures that were filled with slightly viscous yellow/orange fluid, and was adhered to the major vessels leaving the heart, pericardium, caudal lung lobes, and diaphragm. The cut surface was pale green with interspersed gritty, light brown regions and multiple cavernous areas filled with yellow to brown caseous material (Fig. 2). Exploration of the abdominal viscera revealed an approximately 5 cm × 5 cm × 5 cm large delineated mass embedded in the omentum near the abomasum. In addition, the abomasal pylorus contained a 4-cm raised, circular, ulcerated and hemorrhagic mass. A 3-cm transmural, circular, raised mass was also present on the antimesenteric side in the duodenum, approximately 4 cm from the abomasal pylorus. Furthermore, there were two 0.5-cm tan nodules in the liver.
Appropriate tissue sections were placed in 10% phosphate buffered formalin, fixed overnight, processed, and embedded in paraffin wax. Tissue blocks were sectioned at 4 μm, mounted on glass slides, and stained with hematoxylin and eosin for routine microscopic examination. Pulmonary and abomasal masses were aerobically and anaerobically cultured for bacteria and fungi; additional fresh tissue was stored at −80°C.
Microscopically, the accessory lung lobe was greatly expanded by abundant well-vascularized fibrous connective tissue that replaced normal parenchyma. Intermixed and walled off from the normal lung parenchyma were multifocal to coalescing irregular inflammatory nodules containing numerous macrophages, eosinophils, nondegenerative to degenerative neutrophils, fewer lymphocytes, and multinucleated giant cells that were frequently centered on flame figures and karyorrhectic cellular debris. Deeply basophilic to nonstaining fungal hyphae were rarely visible. The hyphae were 10–15 μm in diameter, had nonparallel walls, lacked septa, contained occasional 30-μm bulbous dilations of the wall, and exhibited infrequent nondichotomous branching. Rare 30–40-μm sporangia were also present. Similar fungal hyphae and inflammatory cells were present in the abomasal and duodenal lesions. Additional findings included multifocal ulcerations of the mucosa, transmural inflammation, and necrotic adipocytes adjacent to the serosa within the associated mesentery. Fungal hyphae were also occasionally visible within areas of collagenolysis and necrotic debris. Periodic acid–Schiff (PAS) and Gomori methenamine silver (GMS) stains of pulmonary, abomasal, and duodenal lesions revealed numerous fungal hyphae with characteristics as described above (Fig. 3).
The small discrete tan liver nodules were focal granulomas associated with the capsular surface. The granulomas were delineated from the hepatic parenchyma by small amounts of fibrous connective tissue and contained eosinophils, flame figures, and fungal hyphae similar to the other lesions. In addition, fungal hyphae and granulomatous inflammation were present at the base of the aorta within the tunic adventia and tunic media. Bilaterally, metatarsal diaphyses were thickened by woven and trabecular bone that was orientated perpendicular to cortical bone and contiguous with the periosteum. The periosteum was further expanded by mature fibrous connective tissue that extended into and between the new woven and trabecular bone.
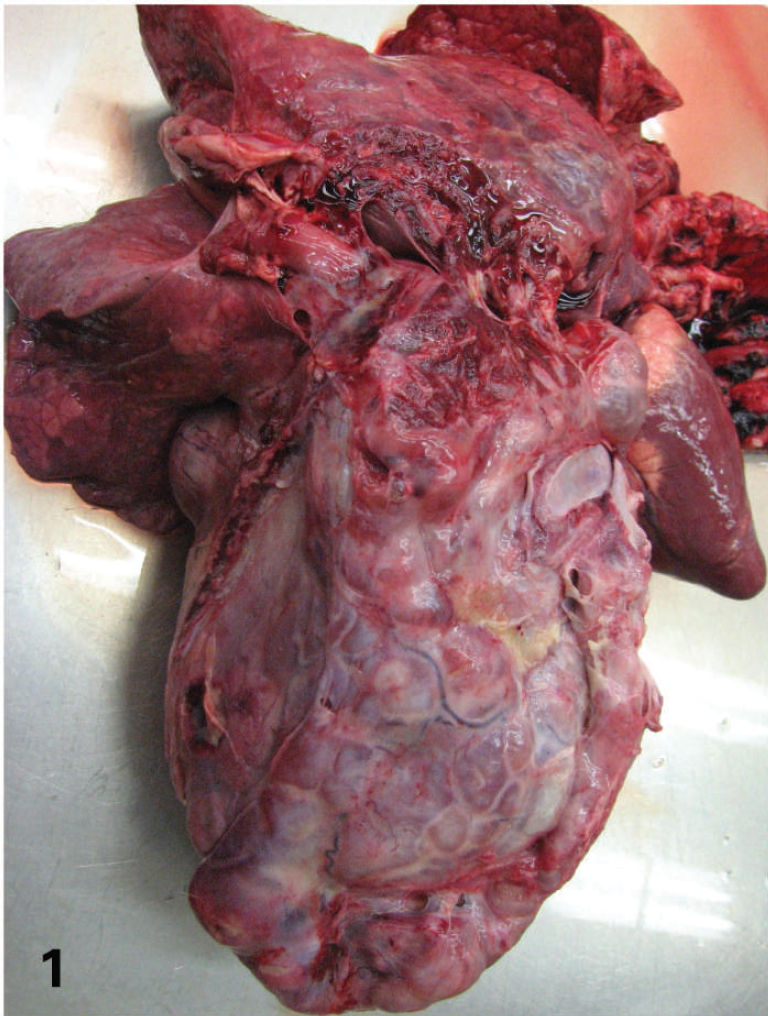
Lung, white-tailed deer. Thoracic mass (25 cm × 13 cm × 15 cm) associated with the accessory lung lobe. The mass contains multiple 1–4-cm cystic structures and is firmly adhered to the major vessels leaving the heart, pericardium, adjacent lung lobes, and diaphragm.

Lung, white-tailed deer. The cut surface of the thoracic mass is pale green with interspersed regions that are gritty and light brown with multiple cavernous areas filled with yellow to brown caseous material.

Lung, white-tailed deer. Fungal hyphae are 10–15 μm in diameter, have nonparallel walls, lacked septa, contain occasional approximately 30-μm bulbous dilations of the wall, and exhibit infrequent nondichotomous branching. Periodic acid–Schiff stain. Bar = 50 μm.

Left metatarsal, white-tailed deer. Lateral radiograph. The metatarsal periosteum is diffusely thickened by radiodense perpendicular woven bone.
No aerobic or anaerobic bacteria or fungal organisms were isolated by routine culture methods, and fresh sections of the associated lung mass were processed for fungal DNA amplification by polymerase chain reaction (PCR). Briefly, fungal genomic DNA was extracted using a commercially available extraction kit a according to the manufacturer's instructions. Primers 18SF (5′-ATTGGAGGGCAAGTCTGGTG) and 18SR (5′-CCGATCCCTAGTCGGCATAG), binding to highly conserved regions of the fungal 18S ribosomal RNA (rRNA) gene, were used for PCR amplification. 6 The PCR reaction contained 5 μl genomic DNA extract, 5 μl 10× PCR buffer II, b 3 μl MgCl 2 (25 mmol b ), 1 μl deoxynucleotide triphosphates (10 mmol c ), 2.5 U AmpliTaq Gold DNA polymerase, b 50 pmol of each primer, d and sterile double-distilled water for a final volume of 50 μl. The primers amplify a 486 base pair (bp) region of the 18S rRNA gene. The amplification was performed as described, 6 and the PCR product was analyzed on an ethidium bromide-stained 1.5% agarose gel. Amplified product was gel purified using a commercial gel extraction kit. e Both strands of the PCR product were sequenced twice using BigDye terminator chemistry. b The same primer set (18SF and 18SR) used for PCR amplification was used for sequencing. The sequence was then compared with known 18S rRNA gene sequences in GenBank using BLAST (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) to confirm the fungal species.
Double-strand sequencing of the 486-bp PCR amplicon confirmed the presence of Conidiobolus spp. in the lung sample. The sequences obtained showed 99% homology to 18S rRNA gene of C. incongruus (GenBank accession no. AF113419) and 95% homology to 18S rRNA genes of C. coronatus (GenBank accession nos. EF392543, AF113417, AF113418, AF296753, D29947) and C. firmipilleus (Gen-Bank accession no. AF368507).
To the authors' knowledge, this is the first report of a domestic or wild animal in North America in which C. incongruus was associated with disease. Previous reports of C. incongruus affecting animals were limited to Australia 3,4,10 ; although, C. incongruus has been reported as the cause of disease in an immunocompromised human patient in North America. 14 Alternatively, C. coronatus and C. lamprauges have been reported and associated with disease in domestic animals in North America. 9,13 Similar to C. incongruus, both C. coronatus and C. lamprauges cause respiratory disease and dermal lesions. 8
In the present case, the gross and microscopic lesions were similar to previous reports. In both sheep with nasal granulomas 4,10 and in the red deer 3 with pulmonary lesions, the cut surface had a green tinge with areas of necrosis. Microscopically, granulomatous inflammation with neutrophils, eosinophils, and fibroplasia predominated within the lesions; few fungal hyphae were visible. The present case differed from previous reports by the observation of collagenolysis and the absence of a Splendore-Hoeppli response. Conidiobolus spp. are known to produce collagenase, lipase, and other proteases in vitro, 11 and the production of these virulence factors may have contributed to the formation of flame figures and fat necrosis in the mesentery. Splendore-Hoeppli phenomenon in cases of Conidiobolus infection is thought to be related to host immune competence. The lack of this phenomenon has been associated with immunosuppression in humans 11 and animals. 3 The immunocompetence of the deer reported herein was not assessed.
Previous reports of C. incongruus in sheep were herd outbreaks. 4,10 In one of these outbreaks, heavy rainfall in the preceding months was believed to have contributed to abundant decaying plant material, which was associated with increased fungal survival. 4 The affected white-tailed deer in the present case was observed in Ledges State Park in the fall of 2007. The Des Moines River is the western border of Ledges State Park, and the low-lying areas of the park regularly flood during excessive rainfall. In mid-to-late August of 2007, excessive rainfall occurred in the Midwest region of the United States, and the Des Moines River flooded. This flooding may have contributed to excessive decomposition of plant material in the park where the deer was found.
Another prominent clinical feature in the white-tailed deer in the current case was the presence of thickened bilateral metatarsal bones. Figure 4 is a radiograph of the left metatarsal bone of the white-tailed deer demonstrating the proliferative lesion. Histologically, the subcutaneous tissues were mildly edematous; however, the majority of the expansion caused by new bone (woven bone) formation that was orientated perpendicular to the existing cortical bone. This lesion, in combination with the presence of an intrathoracic mass and the lack of other bone-related issues, is compatible with hypertrophic osteopathy. To date, the pathogenesis of hypertrophic osteopathy remains unknown, but many theories have been postulated. 5,7 Hypertrophic osteopathy is often associated with a neoplastic or granulomatous intrathoracic mass, although hypertrophic osteopathy has been seen and associated with other disease manifestations. 1,7 Hypertrophic osteopathy has been reported in humans, 2 domestic animals, 5 and wild animals, including a roe deer (Capreolus capreolus) with pulmonary abscesses. 12 To the authors' knowledge, this also is the first report of hypertrophic osteopathy in a white-tailed deer. Conidiobolus incongruus should be considered a potential respiratory pathogen capable of causing disseminated disease in susceptible North American white-tailed deer. Furthermore, granulomatous pulmonary disease associated with C. incongruus may result in hypertrophic osteopathy.
Footnotes
a.
Soil Master™ DNA Extraction Kit, EPICENTRE Biotechnologies, Madison, WI.
b.
Applied BioSystems, Foster City, CA.
c.
USB Corp., Cleveland, OH.
d.
Integrated DNA Technologies Inc., Coralville, IA.
e.
MinElute® Gel Extraction Kit, Qiagen Inc., Valencia, CA.
