Abstract
Hypertrophic osteopathy is a hyperostotic syndrome of the appendicular skeleton that is most commonly associated with intrathoracic neoplasia or inflammation. The condition is rarely associated with intra-abdominal lesions. The majority of cases have occurred in dogs and human beings, with fewer cases reported in cats, horses, and other species. A 15-year-old male neutered Domestic Shorthair cat presented for swollen limbs and difficulty in ambulation. Radiographs and gross postmortem revealed severe periosteal hyperostosis of the diaphysis and metaphysis of all 4 limbs, including the humerus, radius, ulna, carpi, metacarpi, femur, tibia, tarsi, metatarsi, and phalanges. The axial skeleton was spared. Hyperostotic lesions were characterized microscopically by lamellar bony trabeculae separated by adipocytes and scant hematopoietic tissue. In several areas, fibrovascular connective tissue, woven bone, and islands of cartilage were also present. A 2.5 cm × 2.5 cm perirenal neoplasm compressed the left kidney and adrenal gland. This mass consisted of well-differentiated tubules of cuboidal epithelial cells and was most consistent with a renal tubular adenoma, because mitotic figures were rare, and no distant metastases were found. Thoracic pathology was absent. Hyperostosis was consistent with hypertrophic osteopathy secondary to the renal adenoma. The pathogenesis of hypertrophic osteopathy is uncertain, but predominant theories point to increased peripheral circulation and angiogenesis as a key initiating event. Recent literature highlights the potential role of vascular endothelial growth factor and platelet-derived growth factor in the human condition. The mechanism by which this renal adenoma caused hypertrophic osteopathy is unknown.
A 15-year-old male neutered Domestic Shorthair cat, which weighed 3.3 kg, was presented to the referring veterinarian for swelling of all 4 limbs and difficulty in ambulation. The limbs were thickened and hard on palpation. Radiographs revealed diffuse hyperostosis of the appendicular skeleton (Fig. 1). The owners elected euthanasia, and the animal was presented for necropsy. Gross examination of the long bones revealed moderate to severe periosteal hyperostosis (Fig. 2A), with preservation of the cortical bone. The humerus, radius, ulna, carpi, metacarpi, femur, tibia, tarsi, metatarsi, and phalanges were bilaterally affected. The reaction was fairly symmetric throughout and most severe at both elbow joints (Fig. 1A inset). The axial skeleton was not affected.
On microscopic examination, bony trabeculae of lamellar bone radiated from the surface of intact cortical bone to displace the periosteum to the margins (Fig. 2B). Trabeculae were separated by adipocytes and scant hematopoietic tissue. At the superficial margins of the bony expansion, lamellar bone transitioned to woven bone and scattered subperiosteal islands of hyaline cartilage (Fig. 2B inset). This immature bone was more frequently separated by fibrovascular connective tissue.
Hyperostosis of the long bones was suggestive of hypertrophic osteopathy. This condition is most commonly associated with an intrathoracic neoplasm or inflammatory process. Although there was no significant thoracic pathology in this cat, a neoplasm was present in the abdominal cavity (Fig. 3A). The 2.5 cm in diameter neoplasm was present at the cranial pole of the left kidney. The white-to-tan neoplasm was firm and well circumscribed, and compressed the adjacent kidney and adrenal gland (Fig. 3B). On microscopic examination, the neoplasm consisted of uniform tubules of cuboidal epithelial cells (Fig. 3C). There was minimal cellular atypia and little mitotic activity. The neoplastic tubules were limited by a thin, connective tissue capsule that separated the neoplasm from the adrenal gland and renal parenchyma. The cells were labeled strongly by pan-cytokeratin immunohistochemistry and were not labeled by vimentin, which confirmed their epithelial origin. The neoplastic cells were negative for the neuroendocrine marker chromogranin A, ruling out adrenal medullary origin. Melan A is used to diagnosis adrenal cortical neoplasia in human beings 8 and dogs, 23 and it has been shown to label normal adrenal cortex in cats. 24 In the cat in the present study, the normal adrenal cortex adjacent to the neoplasm labeled strongly with melan A. The neoplastic cells were negative, ruling out an adrenal cortical adenoma. Based on the findings, the neoplasm was diagnosed as a renal adenoma. Although some may argue that the size of the neoplasm may warrant a diagnosis of renal carcinoma, the lack of distant metastasis, tissue invasion, or cellular atypia attest to its benign nature. In general, renal neoplasia is uncommon in cats. Renal adenomas appear to be particularly rare. In a series of 19 feline primary renal tumors, excluding lymphoma, only 1 of 19 was diagnosed as a renal adenoma. 13
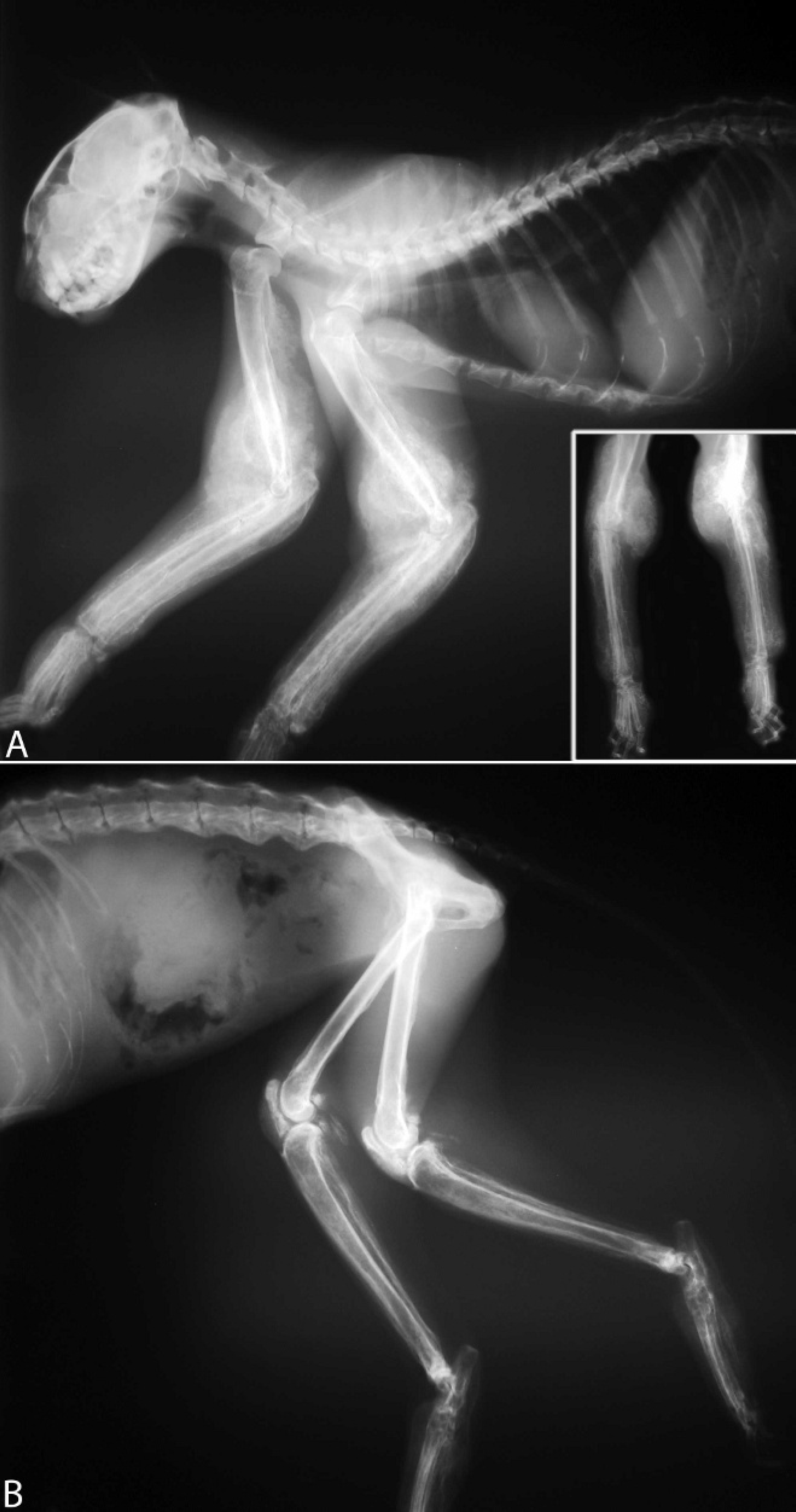
Appendicular hyperostosis in a cat. Radiographs reveal varying degrees of bony tissue radiating from the metaphysis and diaphysis of the long bones of the front (
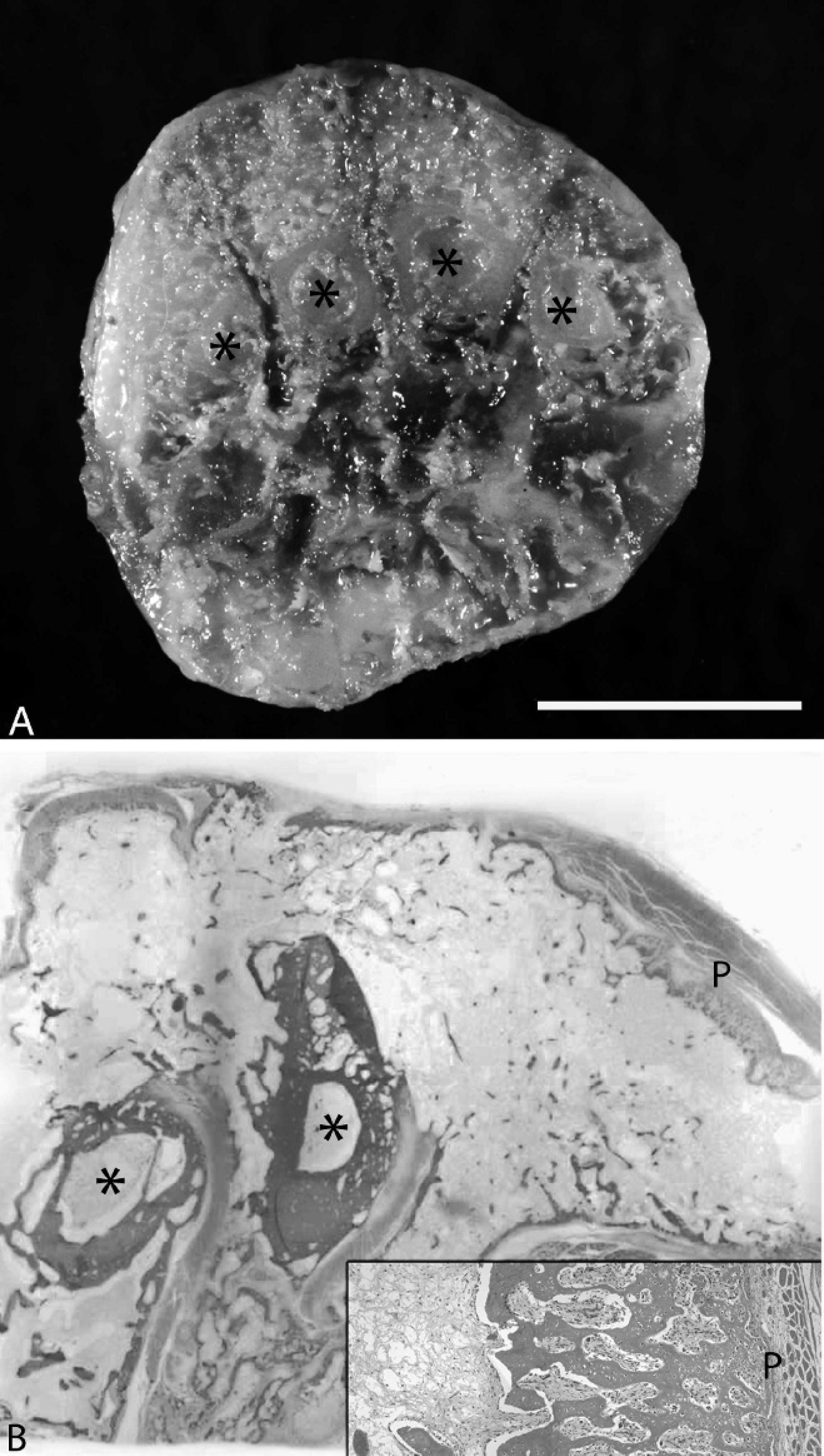
Cross section of the metacarpus.
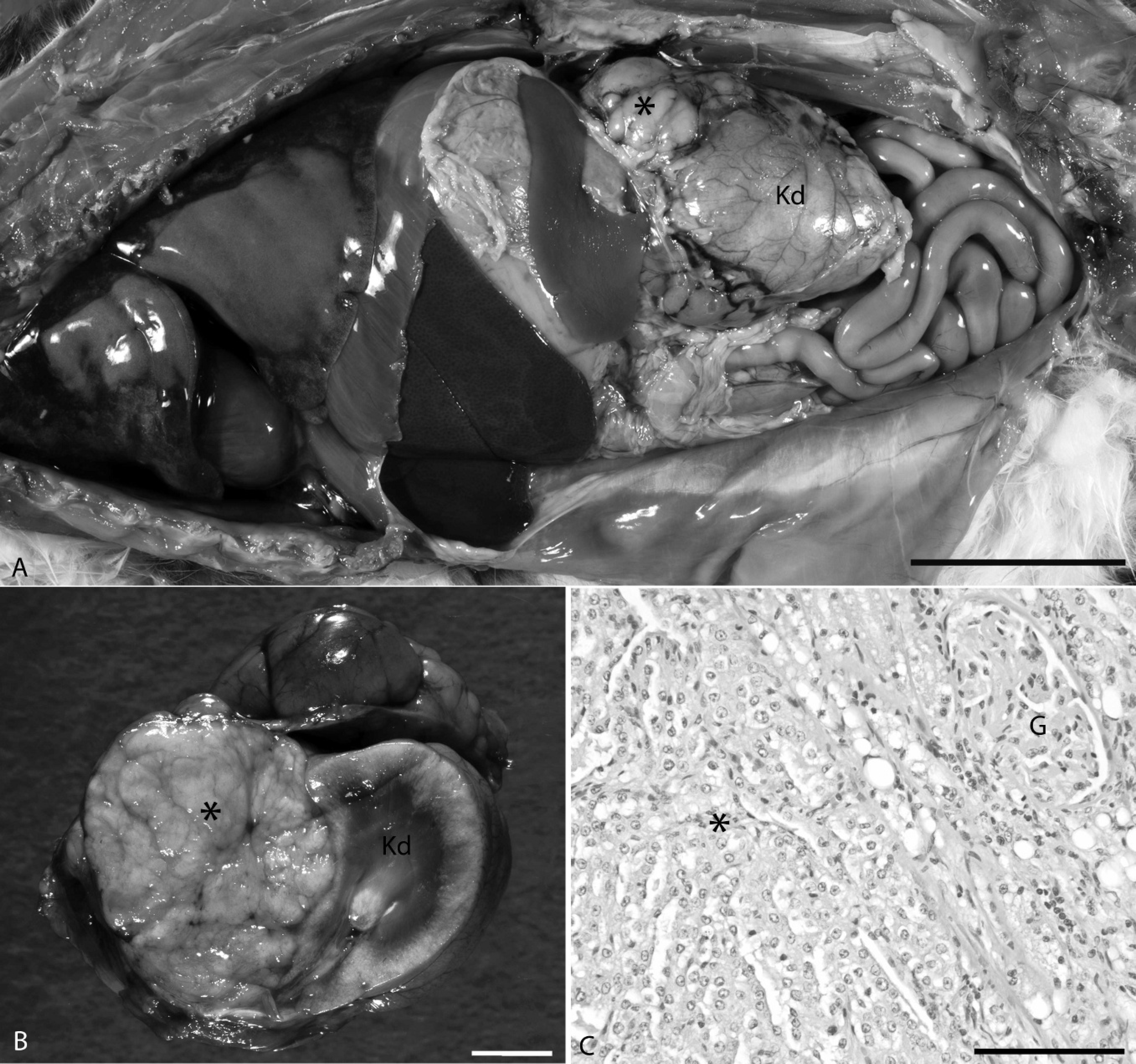
Renal adenoma.
Hypertrophic osteopathy is a syndrome of periosteal hyperostosis of the appendicular skeleton, usually occurring secondary to an underlying primary disease process. The pathogenesis of hypertrophic osteopathy is poorly defined. It is believed that increased blood flow and overgrowth of vascular connective tissue in the extremities precedes the development of fibrochondroid metaplasia and eventual hyperostosis. 2 In addition to neoplasia, hypertrophic osteopathy is also associated with a variety of inflammatory and infectious processes. The present case adds to the few reports of hypertrophic osteopathy associated with an abdominal neoplasm, rather than the more common correlation with thoracic pathology. 2
In veterinary medicine, hypertrophic osteopathy is most prevalent in dogs. The majority of these cases are associated with thoracic disease. Rare cases of hypertrophic osteopathy associated with intrathoracic disease have been reported in numerous other species, including cats, 9 horses, 17 cattle, 22 alpacas, 4 deer, 16 chimpanzees, 20 tigers, 27 mink, 28 and elk. 6 Even fewer reports describe hypertrophic osteopathy in the absence of thoracic disease. The syndrome has been described in dogs associated with rhabdomyosarcoma of the urinary bladder 3,11 and renal pelvis transitional cell carcinoma. 10 The syndrome has been diagnosed in horses with ovarian tumors 12,15,21 and a pituitary adenoma. 26 In cats, a previous study reported hypertrophic osteopathy secondary to an adrenocortical carcinoma. 2 There are also cases of feline idiopathic hypertrophic osteopathy described in the literature. 5,7
The mechanism by which such diverse lesions lead to this common end point is subject to debate. Original hypotheses included neurovascular reflexes, pulmonary vascular shunts, or tumor-derived humoral mediators. 2,5 Current literature of the analogous human condition, hypertrophic osteoarthropathy, provides evidence for the central roles of vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF). 1 It is believed that vascular shunting may allow megakaryocytes and platelet clumps to bypass the small capillary beds of the lungs and lodge in distal vascular beds, where they induce the expression and release of VEGF and PDGF in the peripheral limbs. 1 Other proposed mechanisms include inflammatory cytokine-induced production of excessive growth factors and aberrant production of growth factors by neoplastic cells. 1,14,19 Regardless of the mechanism, it is postulated that the increased concentrations of VEGF and/or PDGF in peripheral vascular beds stimulates local fibrovascular proliferation, edema, and eventual ossification. 18 Clinical support for this hypothesis in human beings comes from increases in circulating serum and plasma levels of these factors in patients with lung cancer and who develop hypertrophic osteoarthropathy relative to patients with lung cancer and without hypertrophic osteoarthropathy. 25 The potential roles of VEGF or PDGF were not investigated in the cat in the current study.
The prognosis for hypertrophic osteopathy is dependent on the underlying cause. If the initiating insult can be identified and removed, then hyperostotic lesions will usually resolve spontaneously. This was the case in a cat after surgical removal of an associated adrenocortical carcinoma. 2 A better understanding of the pathogenesis of this disorder would provide valuable insight into potential therapies that could alleviate this secondary complication in patients with incurable primary conditions or idiopathic hypertrophic osteopathy. The current case highlights the diversity of conditions that can lead to hypertrophic osteopathy and the challenges of developing a unifying mechanism that accounts for such diversity.
Acknowledgements. The authors appreciate the contribution of Dr. Steven Hubbard of Bright Veterinary Clinic, Lawrenceburg, IN. The photographic assistance of Dr. Margaret Miller of the Purdue University Animal Disease Diagnostic Laboratory and Department of Comparative Pathobiology is also greatly appreciated. Dr. Johnson was supported by a fellowship from Pfizer, organized by the American College of Veterinary Pathologists and Society of Toxicologic Pathology Coalition for Veterinary Pathology Fellows.
