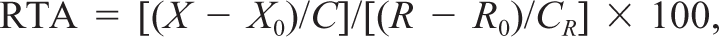Abstract
In canine mammary tumors, we examined the telomerase activity, proliferative activity by proliferative cell nuclear antigen (PCNA) immunohistochemistry, and percentage of apoptotic cells by the deoxynucleotidyl transferase-mediated dUTP-biotin nick end-labeling (TUNEL) method. The relationship between these measures and histopathologic malignancy was also investigated. PCNA index was highest in malignant tumors (adenocarcinoma: 27.0%; malignant mixed tumor: 15.7%), followed by benign tumors (adenoma: 4.4%; benign mixed tumor: 5.3%), hyperplasia (2.1%), and normal mammary gland (0.9%). In adenoma and adenocarcinoma, papillary and solid types showing higher cellularity tended to have higher PCNA indices than did cystic and tubular types. Although the TUNEL index was <1% in all cases, the relationship between this measure and histopathologic diagnosis showed the same tendency as observed in PCNA immunostaining. Telomerase activity was detectable in all adenomas, benign mixed tumors, and adenocarcinomas examined. In contrast, all normal mammary glands, hyperplasias, and malignant mixed tumors were negative for telomerase. Relative telomerase activity (RTA) of adenocarcinoma (56.5) was significantly higher than that of adenoma (27.8) and benign mixed tumor (33.9), and a significant positive correlation (P < 0.001) was noted between RTA and PCNA index. No significant correlations were noted between either PCNA or TUNEL index and clinical features such as metastasis and tumor diameter. PCNA index and telomerase activity may be useful markers for judging malignancy of canine mammary tumors.
In recent years, the life spans of companion animals such as dogs and cats have become extended. As the population of aged animals increases, the incidence of tumors has also increased. In dogs, mammary gland tumors are the most common, accounting for about 50% of all tumors in bitches. 5 Judging histopathologic malignancy and predicting prognosis are important to determine methods of treatment for canine mammary tumors. There have been a variety of studies of canine mammary tumors, and the relationship between prognosis and histopathologic type or proliferative activities of tumors has been investigated. 3 , 11 , 17 , 27 Therefore, prognosis of canine mammary tumors can be predicted with considerable reliability.
Proliferative cell nuclear antigen (PCNA) is a cofactor of DNA polymerase-δ, which is synthesized in the G1 to S phase of cell division and the expression of which is closely related to cell proliferation. 4 A correlation between the number of PCNA-positive cells and histopathologic malignancy has been reported in canine mammary tumors. 17 , 23 The deoxynucleotidyl transferase (TdT)-mediated dUTP-biotin nick end-labeling (TUNEL) method is a widely used technique for detecting DNA fragmentation or apoptotic cell nuclei in tissue sections. Combined utilization of immunohistochemistry for PCNA and the TUNEL method has enabled the quantification of proliferation activity of tumor tissue and is expected to be useful for judging malignancy and for prediction of mammary tumors in dogs and humans.
The telomere is a structure located at each end of eukaryotic chromosomes containing tandem DNA sequence repeats and is thought to be important for chromosome stability. 2 , 28 In mammalian species, the telomere repeat sequence is (TTAGGG) n . 20 The length of the telomere decreases through each cell division because of incomplete DNA replication at the chromosomal ends, and shortening of the telomere length may induce chromosomal instability, leading to cell senescence and finally death. 9 , 15 Telomerase is a ribonucleoprotein enzyme that synthesizes telomeric DNA onto chromosomal ends using an internal RNA template. 18 In humans, telomerase activity is detected in germline 14 , 26 and hematopoietic 6 cells, which constantly undergo cell division, but not in differentiated somatic cells. The activity is also detected in various immortalized cell lines and the majority of human tumors, and therefore telomerase activation is thought to be a crucial factor in immortalization of cells and tumorigenesis. 14 In human breast cancers, telomerase activity is correlated with tumor malignancy. 12 , 22 , 25 In canine mammary gland tumors, as in human breast cancers, telomerase activity is expected to be correlated with malignancy, and quantification of the activity of this enzyme might be a reliable tool for prognosis. However, there has been only one previous report 1 dealing with telomerase activity in canine tumors, and it did not include mammary tumors.
In this study, we examined the telomerase activity in canine mammary tumors of various histopathologic diagnoses to examine the relationship between enzyme activity and malignancy of the tumor. In addition, proliferative activity of tumor cells was examined using PCNA immunohistochemistry, and apoptotic index of tumor cells was determined using the TUNEL method. The relationships between these factors and telomerase activity or histopathologic type were also examined.
Materials and Methods
Canine mammary tissues
A total of 89 mammary gland tumors were obtained from bitches during surgery at the Veterinary Medical Center, The University of Tokyo, between January 1997 and March 1998. The dogs were Maltese (14), Shih-Tzu (14), Poodle (11), Yorkshire Terrier (11), and other breeds (39). The mean age of the dogs was 10.1 years (range: 6–14 years), and 9% were spayed. Normal mammary gland tissues were obtained from 3 female Beagles (1–2.5 years old) without any tumors as controls.
Histopathologic diagnosis
Mammary tissues were fixed in 10% neutral buffered formalin for 2–3 days. Paraffin-embedded sections 4 µm thick were stained with hematoxylin and eosin. According to the classification used in our laboratory, which is a modified Moulton's classification 19 (Fig. 1), all the tumor samples were divided into five histopathologic groups: hyperplasia (seven cases), adenoma (21 cases), adenocarcinoma (20 cases), benign mixed tumor (36 cases), and malignant mixed tumor (five cases). Adenomas and adenocarcinomas were further divided into cystic (six adenomas), tubular (seven adenomas, nine adenocarcinomas), papillary (five adenomas, four adenocarcinomas), and solid (three adenomas, seven adenocarcinomas) types.
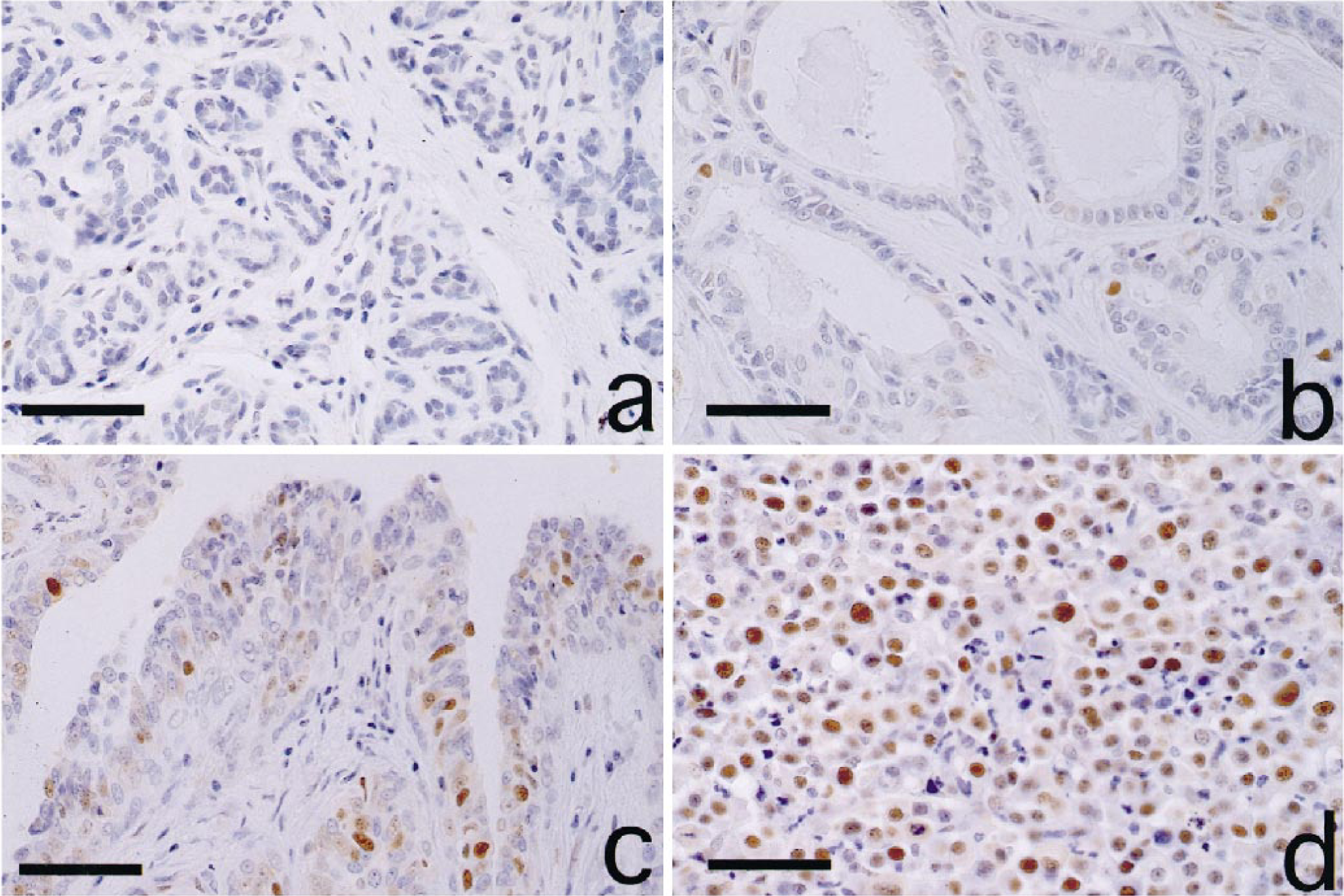
Mammary tumors; dog. Normal mammary gland (a) and tubular adenoma (b). Most cells were negative for PCNA. Papillary adenoma (c) and solid adenocarcinoma (d) showed more PCNA-positive cells. Immunohistochemistry for PCNA. Bar = 80 µm.
Immunohistochemical staining for PCNA
Immunohistochemical staining for PCNA was carried out by the avidin–biotin–peroxidase complex (ABC) method using a Vectastain Elite ABC Kit (Vector Laboratories, Burlingame, CA). Mouse anti-PCNA monoclonal antibody (PC10; Novocastra Laboratories, Newcastle upon Tyne, UK) and biotinylated goat anti-mouse IgG antibody (Kirkegaard & Perry Laboratories, Gaithersburg, MD) were used as first and second antibodies, respectively. After ABC reaction, sections were colorized in 3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma, St. Louis, MO) solution.
Detection of fragmented DNA
DNA fragmentation was examined according to a modification of the TUNEL method 8 using a commercially available apoptosis detection kit (Apop Tag™, In situ Apoptosis Detection Kit; Oncor, Gaithersburg, MD). The 3′-OH ends of multiple fragmented DNA in the sections were labeled with digoxigenin-dUTP in the presence of TdT, peroxidase-conjugated antidigoxigenin antibody was then applied, and apoptotic nuclei were visualized by peroxidase–DAB reaction.
Telomeric repeat amplification protocol (TRAP) assay
Thirty-three of 92 mammary tissues were also examined for telomerase activity. A part of the resected fresh tissue was homogenized in 200 µl of 3-[(3-cholamodopropil) dimethylammonio]-1-propane-sulfonate (CHAPS) lysis buffer supplied with the assay kit. After a 30-minute incubation on ice, the lysate was centrifuged at 12,000 × g for 20 minutes at 4 C, and the supernatant was collected. Protein concentration in each sample was measured using Coomasie Protein Assay Reagent (Pierce Chemical, Rockford, IL) and adjusted to 0.1 µg/µl. The samples were rapidly frozen and stored at −80 C until use. Telomerase activity was measured using a TRAPeze™ Telomerase Detection Kit (Oncor) according to the manufacturer's protocol. Each reaction mixture contained 2.0 µl of mammary tissue sample, 5.0 µl of 10× TRAP reaction buffer, 1.0 µl of 50× dNTPs, 1.0 µl of TS primer (5′-AAT CCG TCG AGC AGA GTT-3′), 1.0 µl of TRAP primer mixture containing a 36-bp internal standard, 2 units of Taq polymerase, and 39.6 µl of distilled water. After 30 minutes of incubation at 30 C for telomerase-mediated extension, the reaction mixture was subjected to polymerase chain reaction (PCR) for 34 cycles of 94 C for 30 seconds and 56 C for 30 seconds. As negative controls, extracts heated to 85 C for 10 minutes and CHAPS lysis buffer alone were subjected to TRAP assay. As a quantitative control, 0.1 mole of TSR8 template supplied with the kit was used. The PCR products were electrophoresed on 12.5% polyacrylamide gels (40 mA, 50 minutes), which were then stained with SYBR™ Green I (Takara Shuzo, Shiga, Japan) for 30 minutes at room temperature. DNA was observed under ultraviolet light (wavelength 254 nm), and photographs were taken with a Polaroid camera. The photographs were input into a computer (Power Macintosh 8100/100AV, Apple Japan, Tokyo, Japan) through an image scanner (IX-4015, Canon, Tokyo, Japan). Image analysis was performed using NIH image 1.56 (public domain software). The signal intensity of the TRAP band in each lane was measured, and relative telomerase activity (RTA) was calculated using the following formula:
Statistical analysis
Data were expressed as means ± SD, and statistical analysis was carried out using Student's t-test. Test for significance of the correlation coefficient was performed using the confidence limit table.
Results
Immunohistochemical staining for PCNA
Anti-PCNA antibody variably labeled nuclei of mammary cells (Fig. 1). The number of strongly labeled cells per 1,000 cells was counted on each specimen. Results are presented as averages of three counts. Table 1 shows the percentage of PCNA-positive cells (PCNA index) of each histopathologic type. The PCNA indices of malignant tumors (adenocarcinoma: 27.0%; malignant mixed tumor: 15.7%) were significantly higher than those of normal mammary gland (0.9%), hyperplasia (2.1%), and benign tumors (adenoma: 4.4%; benign mixed tumor: 5.3%) (P < 0.05). The PCNA indices of adenocarcinoma and malignant mixed tumor were six- and three-fold higher than those of benign counterparts, respectively. No significant differences were observed between normal mammary gland and hyperplasia, but benign tumors showed a significantly higher PCNA index than normal gland and hyperplasia (P < 0.05). In adenoma, PCNA indices of papillary (7.2%) and solid (6.2%) types were significantly higher than those of tubular (3.1%) and cystic (2.9%) types (P < 0.05). Although there were no significant differences among the three histopathologic types of adenocarcinoma, solid type (31.4%) tended to have higher PCNA index than the other two types (tubular: 23.8%; papillary: 26.2%).
PCNA and TUNEL indices (○ ± SD) in canine mammary tumors.
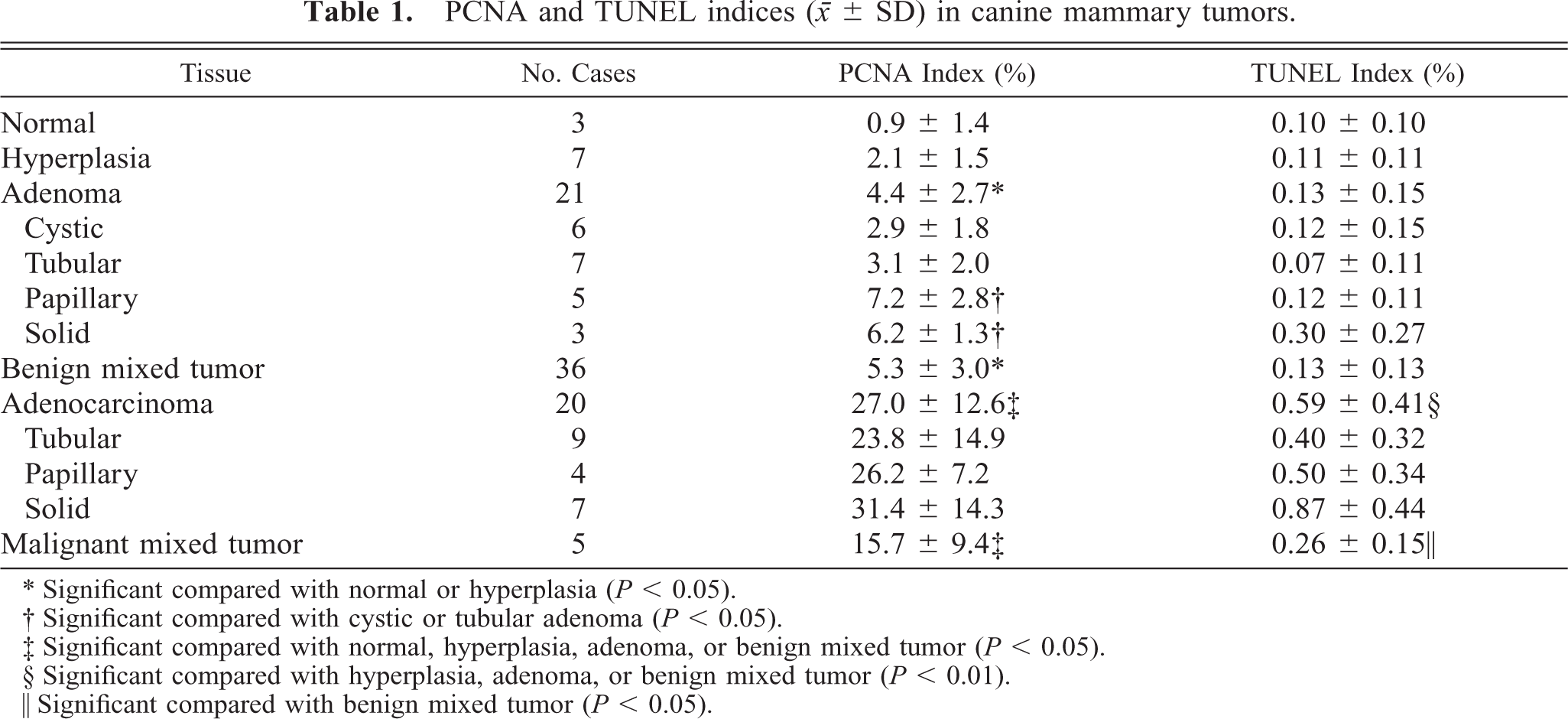
∗ Significant compared with normal or hyperplasia (P < 0.05).
† Significant compared with cystic or tubular adenoma (P < 0.05).
‡ Significant compared with normal, hyperplasia, adenoma, or benign mixed tumor (P < 0.05).
§ Significant compared with hyperplasia, adenoma, or benign mixed tumor (P < 0.01).
∥ Significant compared with benign mixed tumor (P < 0.05).
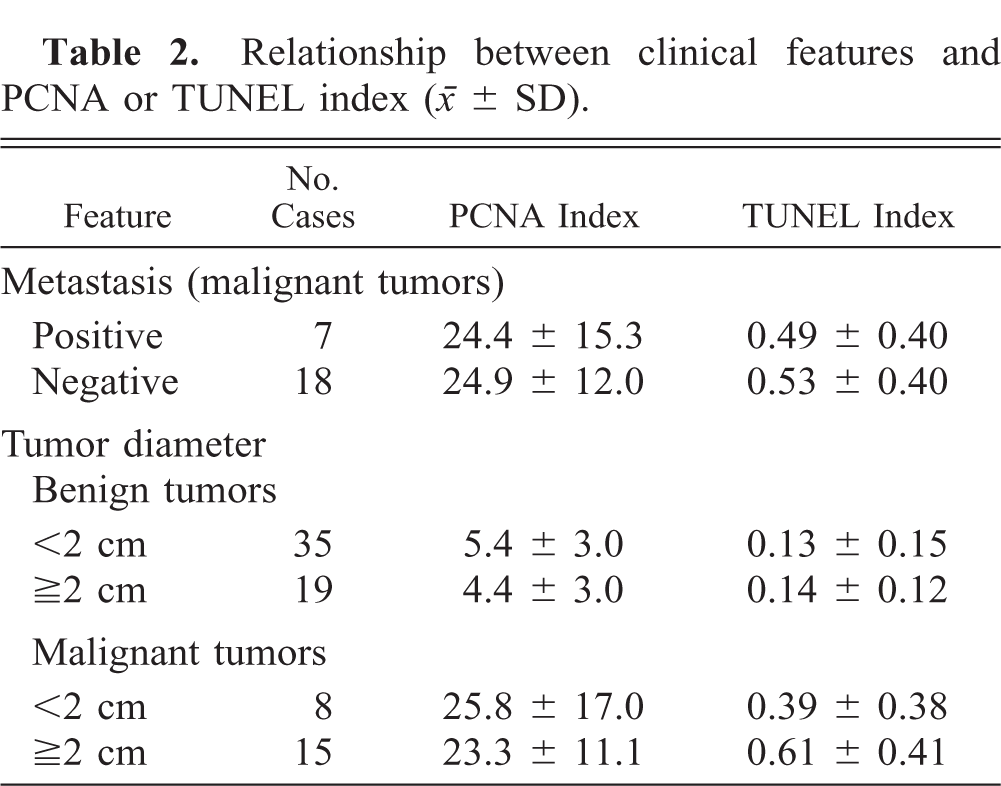
The relationships between the clinical features and PCNA index of mammary tumors are shown in Table 2. Cases without detailed clinical information were excluded. No significant correlations were observed between tumor diameter and PCNA index in benign or malignant tumors. There was also no correlation between lymph node metastasis and PCNA counts in malignant tumors.
Relationship between clinical features and PCNA or TUNEL index (○ ± SD).
TUNEL method
The percentage of TUNEL-positive cells (TUNEL index) was less than 1% in all specimens examined (Table 1, Fig. 3). The TUNEL index of adenocarcinoma was 0.59%, significantly higher than those of hyperplasia (0.11%) and benign tumors (adenoma: 0.13%; benign mixed tumor: 0.13%) (P < 0.01); it was also higher than that of normal mammary gland (0.10%), although the difference was not significant. The TUNEL index of malignant mixed tumor was 0.26%, significantly higher than that of benign mixed tumor (P < 0.05), and tended to be higher than indices of normal mammary gland, hyperplasia, and adenoma, although these differences were not significant. Solid type tumors showed the highest TUNEL indices in adenoma and adenocarcinoma, respectively. No correlations were observed between clinical features and TUNEL indices (cases without detailed clinical information were excluded) (Table 2).
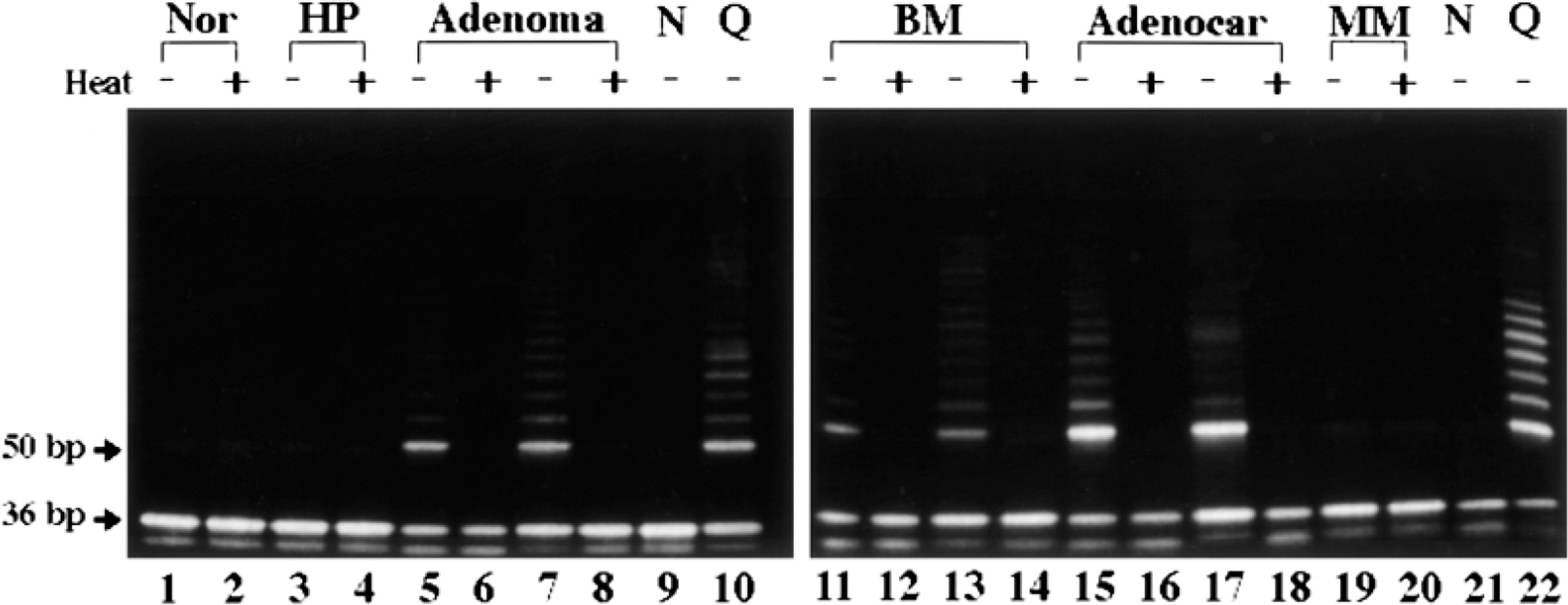
Telomerase activity in canine mammary tumors. Samples from normal mammary gland (lanes 1, 2), hyperplasia (lanes 3, 4), adenoma (lanes 5–8), benign mixed tumor (lanes 11–14), adenocarcinoma (lanes 15–18), and malignant mixed tumor (lanes 19, 20) were electrophoresed. TSR8 positive quantitation control (lanes 10, 22) and CHAPS lysis buffer-only blank (lanes 9, 21) were also included. Samples were heated (+; lanes 2, 4, 6, 8, 12, 14, 16, 18, 20) or unheated (−; lanes 1, 3, 5, 7, 11, 13, 15, 17, 19) before telomerase reaction. Telomerase activity is shown as a ladder pattern consisting of 6-bp bands starting at 50 bp. The 36-bp band in each lane is an internal control. Nor = normal mammary gland; HP = hyperplasia; BM = benign mixed tumor; Adenocar = adenocarcinoma; MM = malignant mixed tumor; N = negative control; Q = quantitative control.
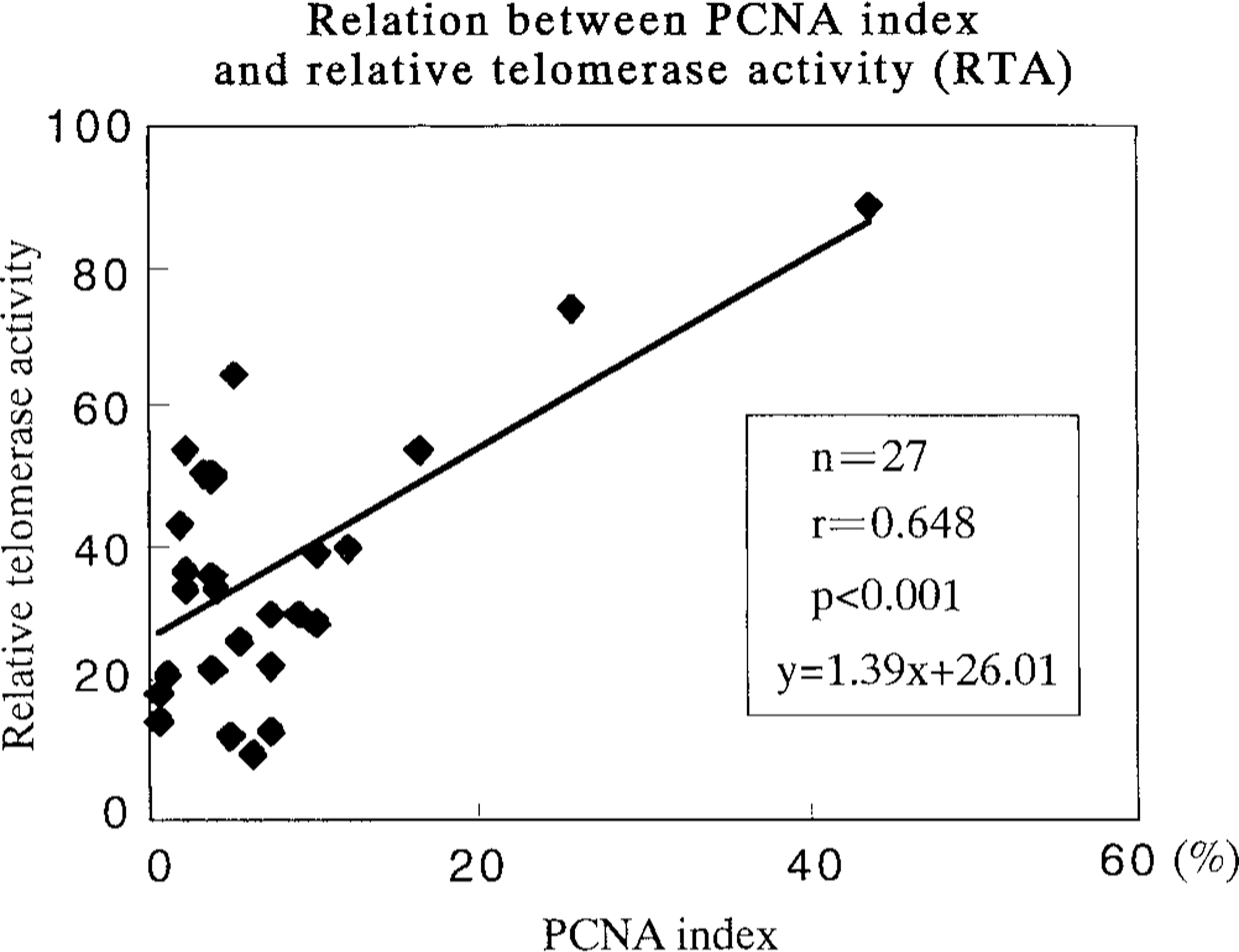
Relationship between PCNA index and relative telomerase activity (RTA). A statistically significant correlation (P < 0.001) was observed.
TRAP assay
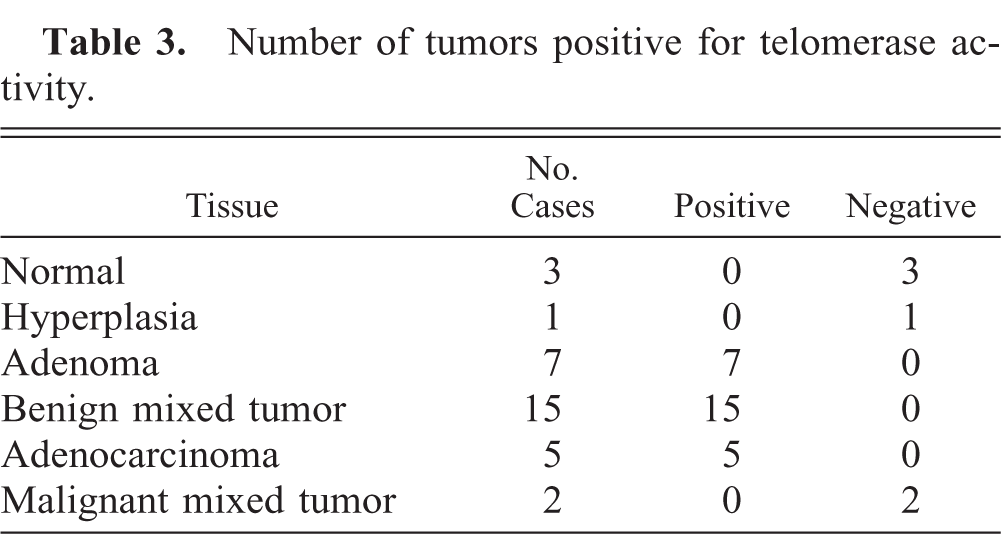
Telomerase activity was detectable in all adenomas (n = 7), benign mixed tumors (n = 15), and adenocarcinomas (n = 5), although all normal mammary glands (n = 3), hyperplasia (n = 1), and malignant mixed tumors (n = 2) were negative for this activity (Table 3, Fig. 2). Adenocarcinoma (56.5) showed higher RTA than did adenoma (27.8) and benign mixed tumor (33.9) (P < 0.05) (Table 4). Moreover, there was a significant positive relation between the PCNA indices and RTA (y = 1.39x + 26.01, r = 0.648, P < 0.001) (Fig. 3). No correlations were observed between RTA and clinical features (tumor size or metastasis) of tumors (cases without detailed clinical information and/or RTA were excluded) (Table 5).
Number of tumors positive for telomerase activity.
Relative telomerase activities (RTA) (○ ± SD) of benign and malignant mammary tumors.

∗ Significant compared with adenoma or benign mixed tumor (P < 0.05).
Relationship between clinical features and relative telomerase activities (RTA) (○ ± SD).
Discussion
In the present study, malignant tumors showed significantly higher PCNA index than the normal mammary gland, hyperplasia, or benign tumors. This finding was consistent with the results of previous studies of canine mammary tumors. 17 , 23 Therefore, immunohistochemical labeling for PCNA may be a reliable procedure for judging malignancy of canine mammary tumors. Papillary and solid types showed higher PCNA indices than did those with cystic and tubular histologic patterns in both benign and malignant tumors, suggesting that neoplastic cells with higher proliferation activity tend to form a papillary or solid pattern with higher cellular density. However, some previous reports stated that both PCNA index 23 and the nucleolar organizer region count (another indicator of cell proliferation) 3 in canine papillary adenocarcinoma were as low as those in benign tumors and hyperplasia. To resolve this conflict, further investigations are needed.
In the present study, no relationship was noted between PCNA index and clinical features (tumor metastasis or tumor size). Similar results in which the index was not correlated with lymph node metastasis, cancer stage, or tumor size have been reported in cases of human breast cancer. 10 , 21 Therefore, it seems difficult to judge the possibility of metastasis by PCNA staining in both human and canine mammary tumors. However, solid carcinoma, which showed a higher PCNA index in the present study, was reported to be characterized by shorter survival time than were other types of mammary carcinoma in dogs. 19 In addition, human breast cancers with higher PCNA indices have significantly higher recurrence rate and shorter survival time than do those with low indices. 21 , 24 The results of the present study clearly indicated that PCNA index is closely correlated with histopathologic malignancy in canine mammary tumors. Further studies of the relationships between PCNA index and clinical features such as metastasis, recurrence, and survival rate are needed.
Malignant tumors with a higher PCNA index possessed a higher TUNEL index. TUNEL-positive cells were more frequently detected in tumors with higher cellularity (papillary and solid types) in both adenomas and adenocarcinomas. In cases of human breast cancer, the high TUNEL index is associated with less tubule formation and high mitotic rate. 16 The deficiency of nutrition and/or oxygen supply due to high density of tumor cells in malignant or solid tumors may induce cell death. More apoptotic cells are seen in malignant tumors with rapid proliferation than in benign lesions, and in solid than in tubular tumors. However, in these canine mammary tumors, the absolute number of TUNEL-positive cells was so low that apoptosis might not be involved in the regulation of tumor growth.
In the present study, no telomerase activity was detectable in canine normal mammary gland or hyperplasia, as in human nonneoplastic breast tissue. 25 In some studies, telomerase activity was not detected in the majority of human benign breast tumors 12 , 22 , 25 but was positive in 70–90% of malignant cases. 7 , 12 , 22 In this study, however, both benign and malignant tumors (except for two malignant mixed tumor cases) showed telomerase activity, and RTA was significantly higher in malignant than in benign tumors. The two cases of malignant mixed tumor were negative for telomerase activity, probably because of marked cartilage or bone metaplasia and few malignant epithelial cells.
Some reports on human breast cancers indicated that metastatic or larger tumors showed higher detection rate of telomerase activity 12 and higher levels of activity, 13 but others showed no correlation. 7 , 22 , 25 In the present canine study, RTA was not related to tumor size or metastasis. Therefore, telomerase activity may be a good marker for judging histopathologic malignancy in canine mammary tumors but may be less reliable for predicting metastasis, recurrence, or survival period.
PCNA index showed a significant positive correlation with RTA. Telomerase activity of a human breast carcinoma cell line has been reported to be regulated in a cell cycle-dependent manner, increasing in the S phase and decreasing in the G2/M phase. 29 The same phenomenon might occur in canine mammary tumor cells. In the present study, both PCNA index and RTA of canine mammary tumors reflected histopathologic malignancy but not clinical features. Consequently, both PCNA expression and telomerase activity may occur along a common pathway during proliferation phase. Telomerase activity of tumor cells decreases after chemotherapy in humans. 13 If telomerase activity can be inhibited, telomere length in tumor cells may be shortened at each cell division, finally killing the cells. Telomerase inhibitors may be effective antitumor drugs in humans and dogs. Further studies of the telomerase of canine mammary tumors and human breast cancers are needed.