Abstract
The aim of our study was to compare serum levels and protein tissue of human epidermal growth factor receptor–2 proto-oncogene (HER2) and mucin 1 (MUC1) using an antigen-capture enzyme-linked immunosorbent assay and immunohistochemistry (IHC) in canine mammary carcinomas and investigate how the 2 markers correlate with dogs with metastasis and without metastasis to a regional lymph node. Forty-eight female dogs were selected, including 14 with non-metastatic cancer, 14 with lymph node metastasis, and 20 healthy animals. Serum samples were collected from all the animals and tissues from 28 dogs with malignant mammary tumor with or without metastasis for evaluated HER2 and MUC1 expression. Tissue sample were evaluated for MUC1 and HER2 immunoexpression by IHC. The results showed measurable serum levels of MUC1 and HER2 in all groups. While serum MUC1 levels were significantly higher in animals with metastasis than the other 2 groups, no increase was observed in HER2 serum levels. The MUC1 IHC results showed that only membrane immunostaining was significantly different between the groups. Statistically, there was an association between immunostaining and the serum HER2 levels. Our results indicate that serum concentrations of HER2 and the IHC staining pattern for HER2 in primary tumor do not correlate with the presence of regional metastasis. However, increased concentrations of MUC1 in the serum of dogs with mammary cancer are associated with the presence of metastasis to regional lymph nodes. A membrane pattern of IHC staining for MUC1 in the primary tumor suggests that metastases to regional lymph node are present.
Spontaneous mammary gland tumors are the most frequent type of neoplasm in female dogs, and the prevalence of malignant lesions varies from 26% to 73%.2,4 To date, surgery is the primary and most cost-effective treatment; however, because of local recurrence and early metastases, the postsurgery overall survival rate is low. Thus, there is a great interest in veterinary medicine to better monitor tumor development and response to therapy. 2
The human epidermal growth factor receptor–2 proto-oncogene, also known as HER2 and
Another protein found in breast cancer is mucin 1 (known as MUC1 in tissue and CA15.3 in serum), which is a type 1 transmembrane glycoprotein that is aberrantly overexpressed in various cancer cells. 11 Studies have demonstrated that increased expression of this glycoprotein in human cancer cells is associated with tumor invasion, the potentiation of cellular signaling, and resistance to genotoxic anticancer reagents, thus suggesting that MUC1 expression is closely associated with tumor malignancy and a poor prognosis. 13 We have previously shown that MUC1 serum levels are detectable in female dogs with mammary cancer and that the levels tend to increase with metastasis. 2 Therefore, the objectives of our current study were to compare serum and protein tissue HER2 and MUC1 by antigen-capture enzyme-linked immunosorbent assay (ELISA) and immunohistochemistry (IHC), respectively, in dogs with mammary gland cancer and to investigate how the 2 markers correlate with the evolution of disease.
Our study was conducted in accordance with the Animal Experimentation Ethics Committee of the Federal University of Minas Gerais (UFMG; Belo Horizonte, Minas Gerais, Brazil; protocol no. 279/10). Forty-eight female dogs, randomly selected, were admitted to the Veterinary Hospital of the UFMG, and histological testing, proposed in a previous study,
3
were performed by a veterinary pathologist from the Laboratory of Comparative Pathology of UFMG. Twenty-eight female dogs were diagnosed with malignant mammary tumors (14 without metastatic disease and 14 with metastasis to regional lymph nodes), and 20 female dogs were healthy (with no evidence of mammary neoplasm during a routine checkup). To evaluate metastatic disease, all patients were subjected to pulmonary radiographic analysis and histological examination of the regional lymph nodes. One regional lymph node was processed routinely for formalin-fixed, paraffin-embedded sections from every dog, and a minimum of 2 slides were examined per lymph node. Based on the presence or absence of visible microscopic metastases, the cases were grouped into dogs with regional metastasis (
Peripheral venous blood samples were obtained from subjects during the preoperative stage and routine checkups (control group). After collection and clot retraction, the samples were centrifuged at 2415 RCF for 5 min, aliquoted to avoid freeze–thaw cycles of sample and loss of bioactive, and stored at −20°C until use. The serum tests were performed according to the protocol of each manufacturer. All assays were in duplicate, and a positive and a negative control (supplied with the kit) with a known concentration range was also measured. To assess MUC1 a (based on 2 mouse monoclonal antibodies: Ma695 as catcher antibody recognizing a sialylated carbohydrate epitope expressed on the MUC1 antigen and Ma552 as tracer antibody targeting a hydrophilic peptide of the protein core) and HER2 b (using anti-human sHER2 that detects the soluble circulating fragment of HER2, p185HER2) serum, ELISA kits were used. In all assays, positive control serum (supplied with the kit) was also measured. The reagents used were the same as the reagents used for human diagnostics.2,9,12 A microplate reader c was used for the measurements. The cutoff point to evaluate the serum test results was conducted through the receiver operating characteristic curve, and the established positive results were >0.50 U/mL and >0.62 U/mL for MUC1 and HER2, respectively.
For IHC, samples from the primary tumors samples were fixed in 10% formalin for 24–48 hr and routinely prepared. Sections immunoreacted with a polyclonal rabbit anti-human c-erbB-2 oncoprotein d (1:100, polyclonal) and a mouse monoclonal anti-MUC1 d (1:50, clone E29) and incubated overnight (16–18 hr) at 4°C. After incubation, target antigens were visualized using horseradish peroxidase e with diaminobenzidine as the chromogen. A human breast carcinoma tissue known to overexpress the receptor (+3) was used as a positive control for HER2. For MUC1, the internal positive controls were the apical surface of normal glandular epithelial cells. Negative controls were obtained by omitting the primary antibodies. MUC1 and HER-2 IHC was performed on primary tumors.
The expression of HER2 was scored by 2 observers in accordance with the Bertagnolli 1 guidelines for reporting HER2 immunostaining. In our study, specimens with scores of 0 and +1 were regarded as negative, and scores of +2 and +3 were defined as HER2 overexpression. 7 To evaluate MUC1 expression, a semiquantitative analysis was performed in accordance with a previous stuy. 5 Immunostaining of the diffuse cytoplasmic expression, plasma membranes, and nuclei was evaluated. For association analysis, specimens with ≤20% of the cells labeled were regarded as negative, and cells with ≥20% of the cell labeled were considered MUC1 positive.
Statistical analysis was performed with commercial software.
f
When possible, analysis of variance was used, followed by the Student–Newman–Keuls (SNK) and Tukey tests or Mann–Whitney test (for nonparametric analysis); correlations were investigated with the Pearson or Spearman tests. Results were considered significant when the test probability was
The age of the animals with mammary cancer ranged from 8 to 16 years (mean: 10.2 ± 2.2 years). The mean ages of the subjects with and without metastasis were not significantly different (10.3 and 11.4 years, respectively;
The serum concentration of MUC1 was significantly elevated in female dogs with metastatic mammary gland cancer to a regional lymph node as compared with the control dogs and dogs with no metastasis to a regional lymph node group (both
MUC1 and HER2 serum levels in female dogs from groups I, II, and III (mean ± standard deviation).*

Different superscript letters in the same column differ statistically with a
All tumors expressed MUC1, but the expression pattern differed among them. Figure 1C and Table 2 show that 26 out of 28 (92.8%) were positive for cytoplasmic MUC1 expression, 18 out of 28 (64.2%) were positive for membrane MUC1 expression (Fig. 1D; Table 2), and only 3 out of 28 (10.7%) were positive for nuclear MUC1 expression (Fig. 1E; Table 2). A total of 18 out of 28 (64.2%) exhibited both cytoplasmic and membrane MUC1 immunostaining (Fig. 1F; Table 2). A significant difference for membrane expression for MUC1 was observed (
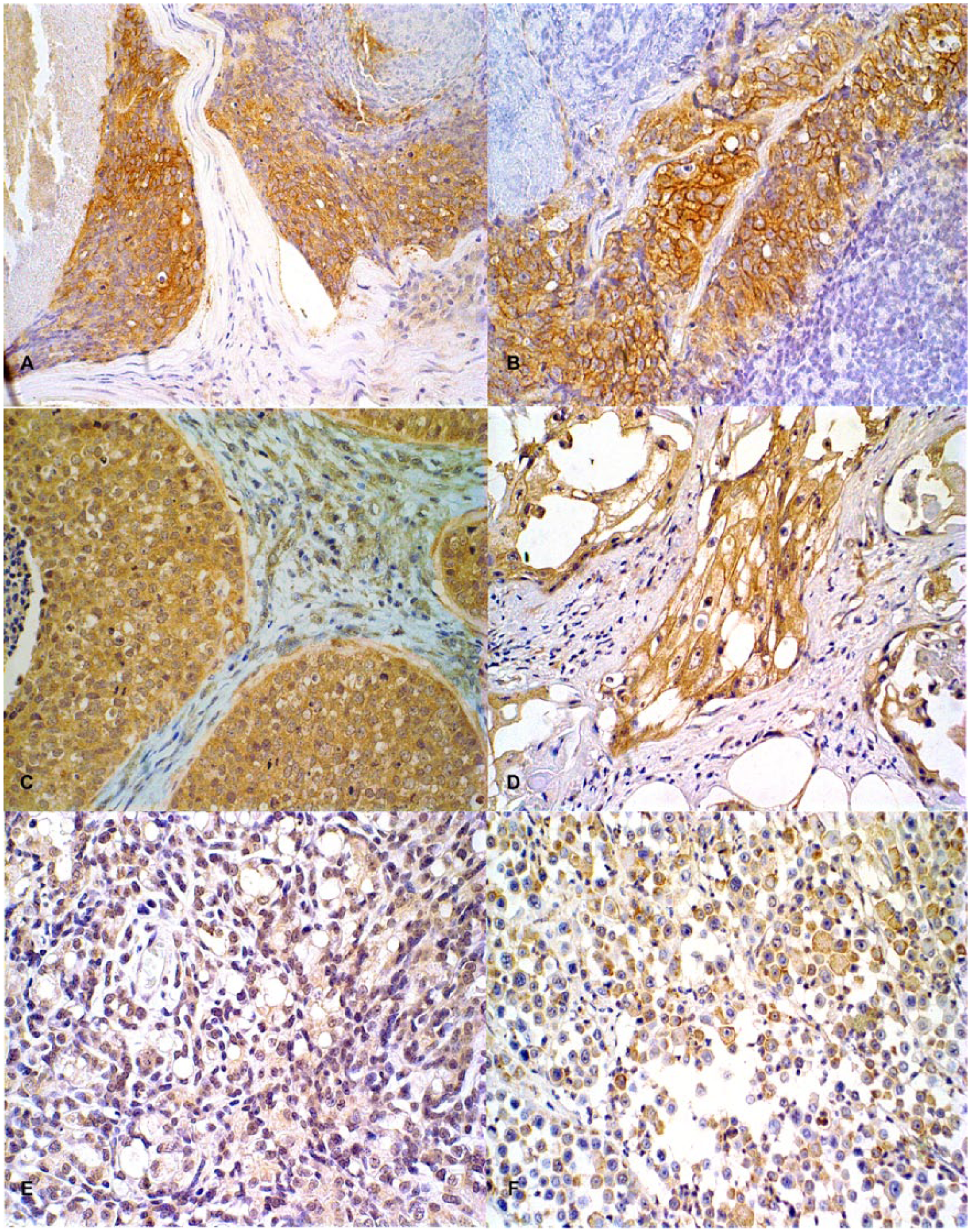
Immunohistochemical staining of canine mammary carcinoma sections.
Association of immunohistochemical (IHC) expression and MUC1 serum levels in female dogs with mammary cancer (
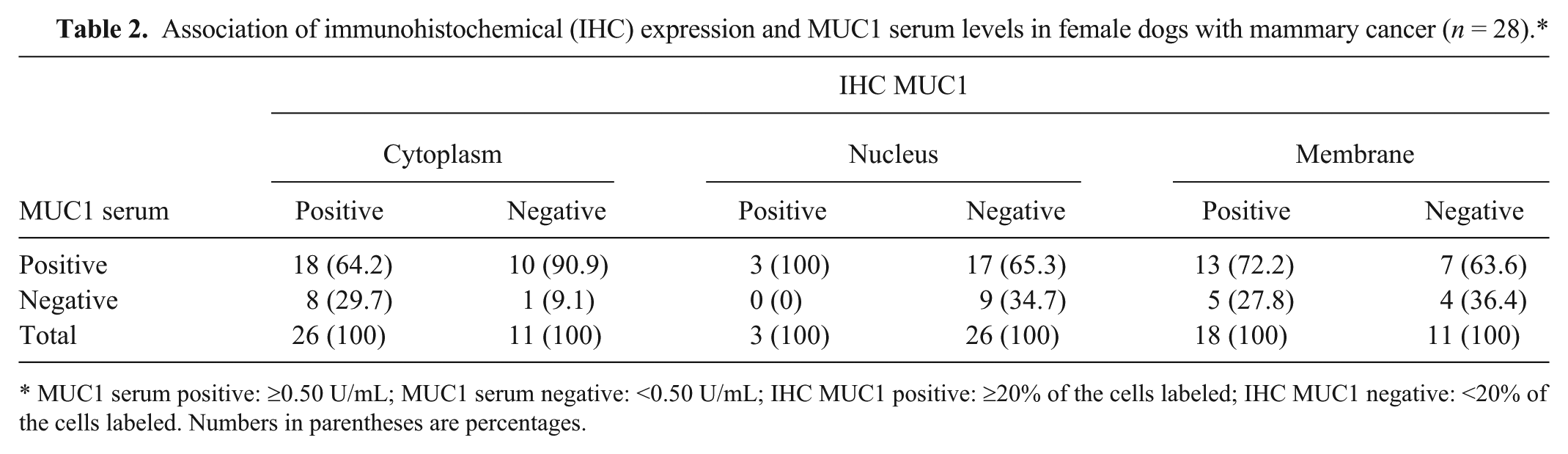
MUC1 serum positive: ≥0.50 U/mL; MUC1 serum negative: <0.50 U/mL; IHC MUC1 positive: ≥20% of the cells labeled; IHC MUC1 negative: <20% of the cells labeled. Numbers in parentheses are percentages.
Evaluation of immunohistochemical expression of HER2 and MUC1 in female dogs from group II (
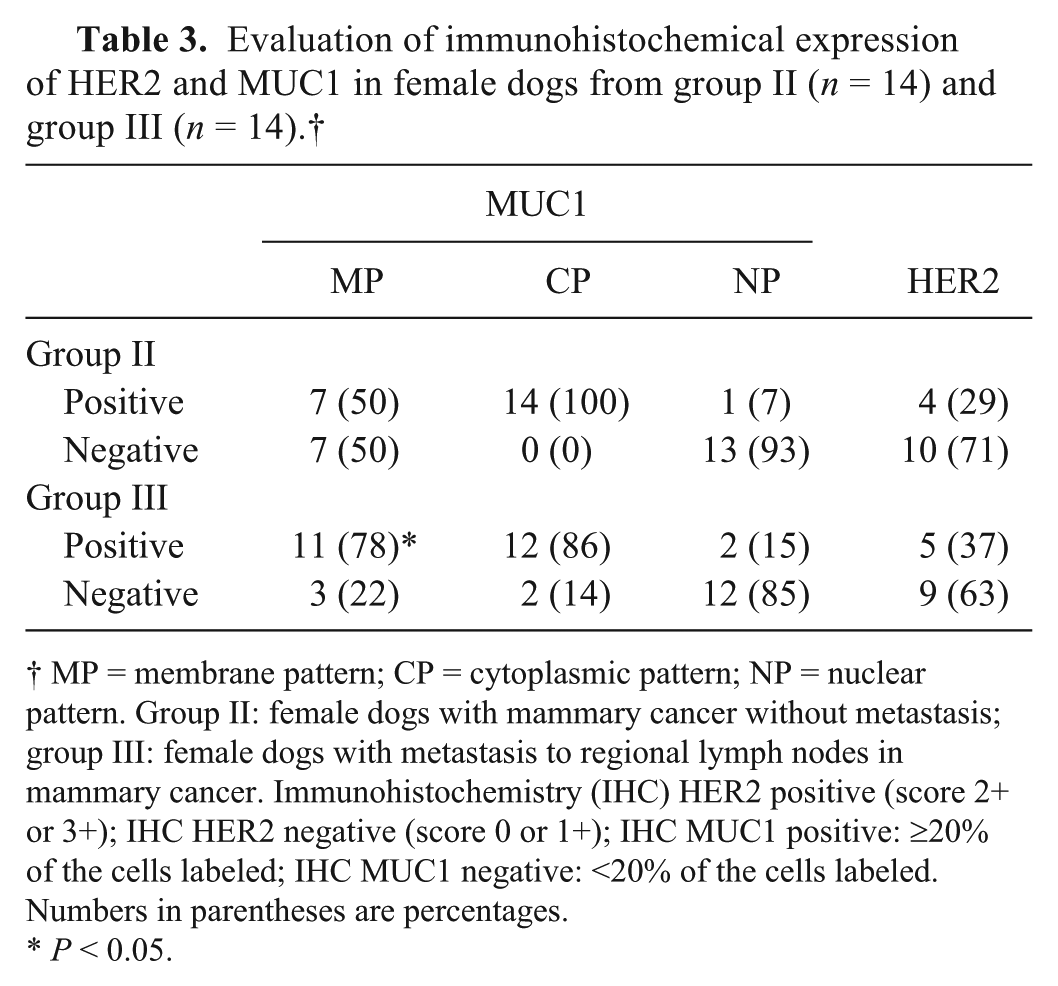
MP = membrane pattern; CP = cytoplasmic pattern; NP = nuclear pattern. Group II: female dogs with mammary cancer without metastasis; group III: female dogs with metastasis to regional lymph nodes in mammary cancer. Immunohistochemistry (IHC) HER2 positive (score 2+ or 3+); IHC HER2 negative (score 0 or 1+); IHC MUC1 positive: ≥20% of the cells labeled; IHC MUC1 negative: <20% of the cells labeled. Numbers in parentheses are percentages.
HER2 protein levels did not differ between groups (
Nineteen out of 28 dogs (67.8%) showed negative HER2 immunostaining while 9 (32.1%) were positive (Fig. 1A, 1B; Table 4). When HER2 protein expression was compared between groups, a significant difference was not observed (
Association of immunohistochemical (IHC) expression and HER2 serum levels in female dogs with mammary cancer (

HER2 serum positive: ≥0.62 U/mL; HER2 serum negative: <0.62 U/mL; IHC HER2 positive (score 2+ or 3+); IHC HER2 negative (score 0 or 1+). Numbers in parentheses are percentages.
In our study, no significant correlations were observed between HER2 protein expression and MUC1 protein expression in the cytoplasm (
Evaluation of immunohistochemical expression of HER2 and MUC1 in canine mammary neoplasms (
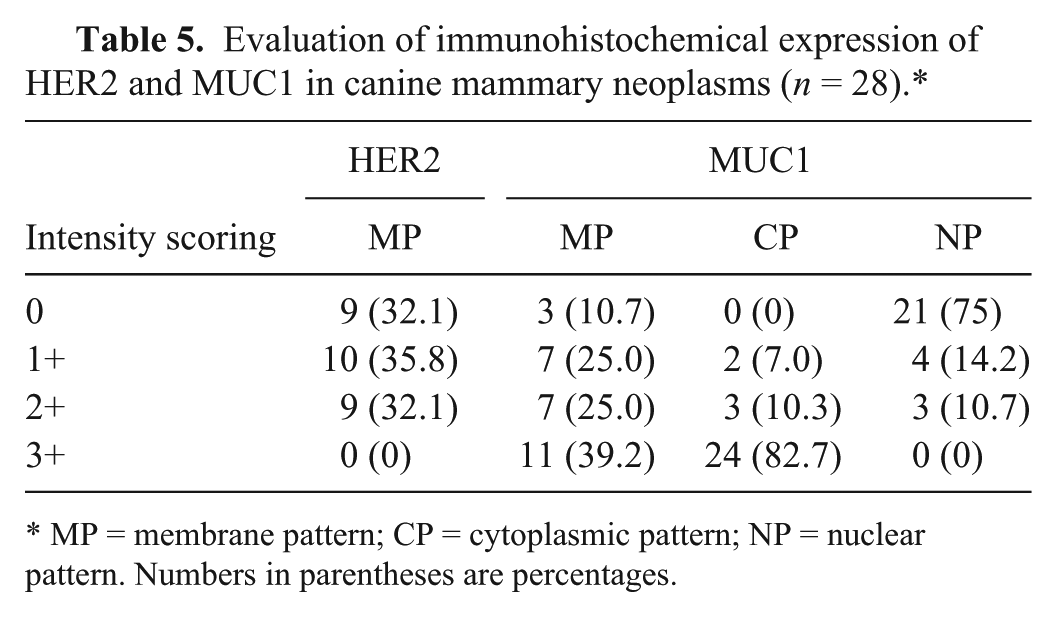
MP = membrane pattern; CP = cytoplasmic pattern; NP = nuclear pattern. Numbers in parentheses are percentages.
In our study, the possibility of using human antigens kits to determine HER2 serum concentrations in dogs was demonstrated. However, the present data shows that this protein cannot be used as a tumor marker in the serum of dogs, as the HER2 serum concentrations were not significantly different between healthy animals and those with cancer. Moreover, after measuring the HER2 serum levels in 20 normal subjects, the mean for the normal group was determined to be 0.55 ng/mL, which has not been previously determined, to the authors’ knowledge.
In the present study, HER2 protein overexpression was observed in 32.1% (9/28) of canine malignant mammary tumors. This percentage was similar to what has been reported in another study. 6 HER2 overexpression is associated with malignancy, high histological tumor grade, large tumor size, and p53 expression, suggesting a role in canine mammary gland carcinogenesis 1 and a poor prognosis. 6 On the other hand, current research shows a controversial relationship of HER2 in canine tumors. 10 Our results do not show a significant difference in HER2 protein expression between groups of dogs with or without metastasis to a regional lymph node, suggesting that there is no prognostic significance in the analysis of HER2, in isolation in the serum or in the primary epithelial mammary tumors. The association of HER2 serum and tissue expression levels was positive and significant similar to human patient–based studies. 14 This finding is interesting because an ELISA is a quantitative method, whereas IHC is a morphological, in situ, and semi-quantitative test. Moreover, the ELISA for serum HER2 is a dynamic test that can be performed at any time and can be used when primary tumor samples are unavailable, thus eliminating the need for a biopsy. The present data demonstrate a significant increase in the serum concentration of MUC1 concentrations between dogs with metastasis to a regional lymph node as compared with dogs without detectable node metastasis and control dogs. MUC1 expression was detected in the primary mammary tumors from all cases. Cytoplasmic immunostaining was predominant, which agreed with findings reported in a human study. 5 However, our study showed a significant increase in membrane immunostaining by MUC1 on primary tumors of dogs with metastasis to a regional lymph node compared with tumors of dogs without detectable node metastasis.
There was no evidence that serum concentrations of HER2 or IHC staining of HER2 in primary canine mammary cancer correlated with the presence of metastasis to regional lymph nodes. However, these results indicate that determining the serum concentration of MUC1 and examining the primary tumor with IHC for membrane distribution of MUC1 helps predict the likelihood of metastasis to a regional lymph node in dogs with a mammary tumor without pulmonary metastases determined by radiographs of the thorax.
Footnotes
Authors’ contributions
LC Campos, E Ferreira, and GD Cassali contributed to conception and design of the study. LC Campos, MR Araújo, and E Ferreira contributed to acquisition, analysis, and interpretation of data. LC Campos, JO Silva, and FS Santos contributed to acquisition and analysis of data. GE Lavalle contributed to acquisition of data. LC Campos, E Ferreira, and GD Cassali drafted the manuscript. LC Campos, MR Araújo, E Ferreira, and GD Cassali critically revised the manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
CanAg, Fujirebio Diagnostics AB, Göteborg, Sweden.
b.
eBioscence Diagnostic Inc., Vienna, Austria.
c.
SpectraMax 190 absorbance microplate reader, Molecular Devices Corp., Sunnyvale, CA.
d.
Dako North America Inc., Carpinteria, CA.
e.
Advance HRP visualization method, Dako Denmark A/S, Glostrup, Denmark.
f.
GraphPad Prism v. 5.0, GraphPad Software Inc., San Diego, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Fundação de Amparo à Pesquisa de Minas Gerais (FAPEMIG), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil.
