Abstract
To date, two isoforms of estrogen receptors (ER) have been identified, cloned, and characterized from several species, estrogen receptor-α (ERα) and estrogen receptor-β (ERβ). Although the presence of ERα has been demonstrated in normal and tumoral canine mammary tissues, the issue of ERβ expression has not been addressed in the dog. In this study, we have analyzed the expression of ERβ in formalin-fixed, paraffin-embedded tissue samples of nonaltered mammary gland, 30 malignant (six complex carcinoma, 12 simple carcinoma, three carcinosarcoma, and nine carcinoma or sarcoma in benign tumor), and five benign (one fibroadenoma, one complex papilloma, one complex adenoma, and two benign mixed tumors) mammary tumors of the dog by using a polyclonal ERβ antibody and the avidin-biotin-peroxidase complex immunohistochemical technique. Our results show that high numbers of normal ductal and acinar epithelium and approximately one third of canine mammary tumors express ERβ. This expression was higher in benign than in malignant tumors. Furthermore, expression was higher in complex and mixed histologic subtypes of malignant tumors when compared with simple subtypes.
The presence of estrogen receptors (ER) in variable proportions of benign and malignant tumors of the mammary gland of the dog has been demonstrated by both biochemical and immunohistochemical (IHC) methods. 4,5,12–14 Its presence has been associated with a favorable prognosis in malignant tumors. 12,13 After the cloning of a second ER in rats and humans, designated ERβ, 9,11 it became known that currently reported data correlated with the isoform α of ER (ERα). In humans, messenger ribonucleic acid, western blot, and IHC analyses have shown that ERβ is expressed in a wide variety of tissues including the mammary gland and breast cancer cell lines and tumors. 2,3,8,15,16 In this study, we have analyzed the expression of ERβ by immunohistochemistry in formalin-fixed, paraffin-embedded tissue samples of normal mammary gland, benign tumors, and malignant tumors of the dog.
Proliferative mammary lesions were collected from 28 female dogs aged 4–16 years (mean 9.3 years). Nonaltered mammary tissues were from glands without tumoral lesions included in regional or block mastectomy pieces. The 35 excised mammary lesions (four dogs had multiple tumors) were fixed in neutral-buffered formalin and embedded in paraffin wax. The histologic types of tumors, determined on hematoxylin and eosin–stained tissue sections, 10 were as follows: 30 malignant tumors (six complex carcinoma, 12 simple carcinoma, three carcinosarcoma, and nine carcinoma or sarcoma in benign tumor) and five benign tumors (one fibroadenoma, one complex papilloma, one complex adenoma, and two benign mixed tumors). Enlarged regional lymph nodes free of metastases were observed in seven dogs. The polyclonal rabbit anti-ERβ antibody developed against the N-terminal region of the human ERβ sequence (Upstate Biotechnology, Lake Placid, NY) diluted 1:50 was used, using the avidin–biotin–peroxidase complex Vectastain kit for polyclonals (Vector, Burlingame, CA). According to the manufacturer, the antibody recognizes ERβ and does not detect ERα. Dewaxed and rehydrated sections were subjected to high-temperature antigen retrieval by incubation with 0.01 M citrate buffer, pH 6.0, at 95 C for 25 minutes in a water bath. After cooling down at room temperature for 20 minutes, endogenous peroxidase activity was blocked with 3% hydrogen peroxide in methanol for 30 minutes at room temperature, rinsed (phosphate-buffered saline [PBS], pH 7.6, thrice for 10 minutes), treated with 10% normal goat serum in PBS, and incubated overnight at 4 C. The reaction was developed by incubation with the chromogen 3,3′-diaminobenzidine tetrahydrochloride (DakoCytomation, Glostrup, Denmark) for 15 minutes, rinsed in tap water, counterstained with Mayer's hematoxylin, dehydrated, and mounted. Normal canine tissues known to contain the antigen sought (ovary, uterus) were used as external positive controls. Tissue sections under study that were treated with normal rabbit serum, instead of the specific antibody, were run as negative controls. The number of positive cells in a given sample was determined as the percentage of positively stained cells: −, when either none or less than 5% of nuclei were stained; +, when positive nuclei represented between 5 and 19% of nuclei observed; ++, when positive nuclei represented between 20 and 59% of nuclei observed; and +++, when 60% or more of the nuclei are positive.
No staining was observed in the negative controls. In the canine ovary, ERβ expression was found in the nuclei of granulosa cells and ovarian stroma (Fig. 1a). In the canine uterus, immunoreactive products to ERβ antibody were found in the nuclei of endometrial (surface and glandular epithelial, and stromal fibroblasts) and myometrial (smooth muscle) cells (Fig. 1b). In the normal canine mammary gland, immunoreactive products to ERβ antibody were observed in the nuclei of numerous acinar and ductal glandular epithelial cells of several lobuli. The nuclei of isolated basal-myoepithelial cells were occasionally stained (Fig. 2), whereas stromal cells were unreactive. In addition to the strong nuclear staining, faint staining was found in the cytoplasm of all types of cells (Figs. 1, 2).
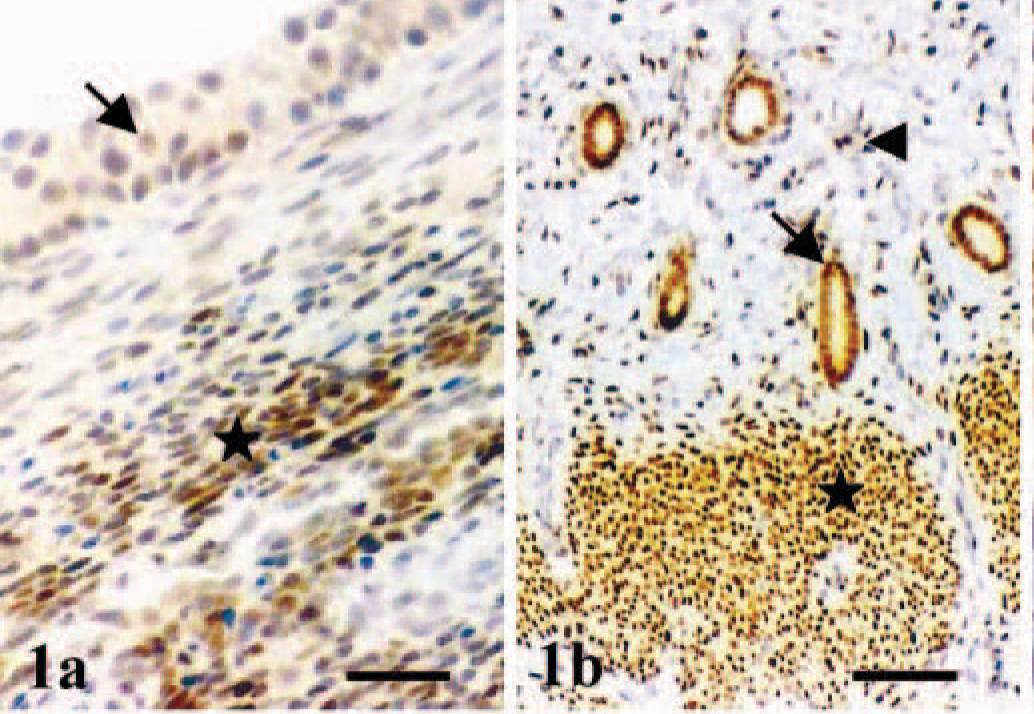
Ovary and uterus; dog.
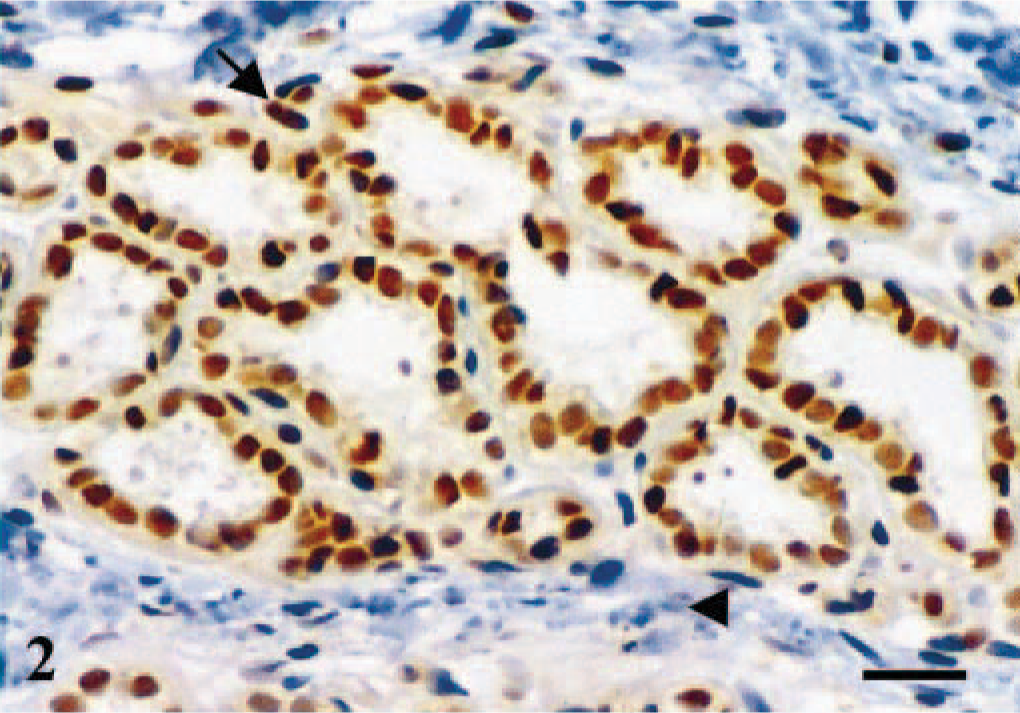
Mammary gland; dog. Normal lobule. Immunoreactive products to ERβ antibody are observed in the nuclei and, with lower intensity, in the cytoplasm of the majority of glandular epithelial cells of all acini. Both stained (arrow) and unstained (arrowhead) fusiform nuclei occupying a basal position in the acinus are also seen. Stromal cells are unreactive. ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 µm.
In the tumors, immunoreactive products to ERβ were found in the nuclei of glandular epithelial-type cells (Figs. 3–6). Additionally, a faint cytoplasmic staining was sometimes seen in the same type of cells (Figs. 3–6). Myoepithelial-type cells of complex (Figs. 3, 6) and mixed tumors (Fig. 4), chondrocytes of mixed tumors (Fig. 4), and sarcoma cells were unreactive. Eleven tumors expressed ERβ (31%) (Tables 1, 2). ERβ-positive cases included eight malignant tumors (26% of the group of malignant tumors) (Table 2) and three benign tumors (60% of the group of benign tumors) (Table 1). The number of positive cells was scored (+++) in 87% of malignant and 67% of benign tumors.
Estrogen receptor–β expression in five benign mammary tumors of the dog.

Estrogen receptor beta expression in 30 malignant mammary tumors of the dog.

∗ Carcinoma or sarcoma in benign tumor.
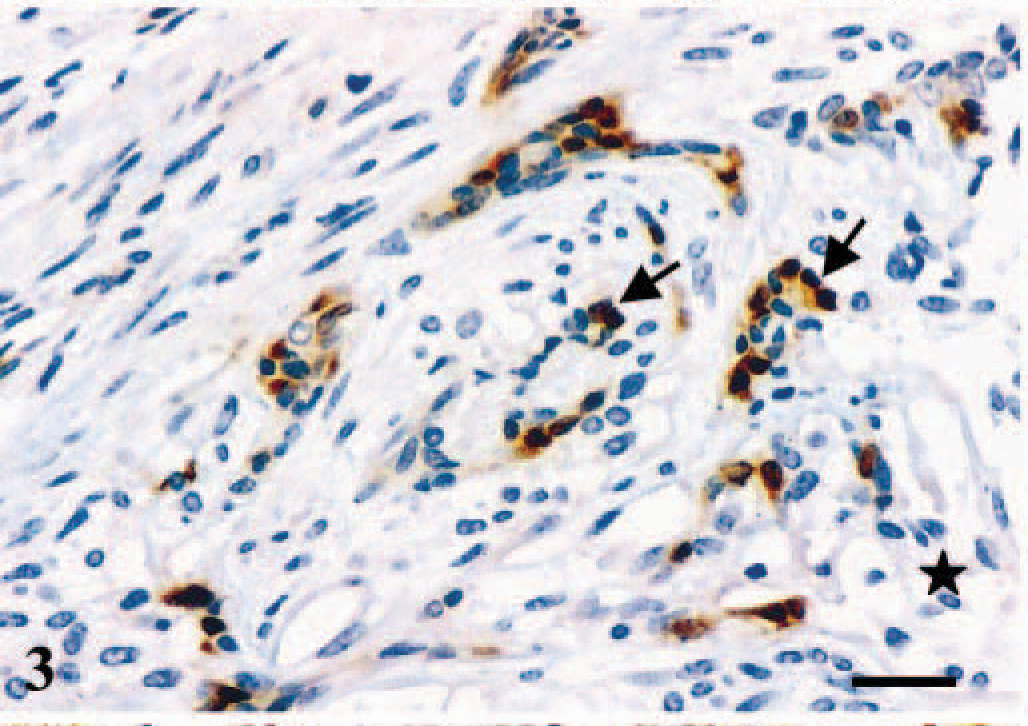
Mammary gland; dog. Complex adenoma. Immunoreactive products to ERβ antibody are observed in the nuclei and the cytoplasm of the majority of glandular epithelial-type tumor cells (arrows). The immunoreactivity is strongest in the nucleus. The myoepithelial cell component of the neoplasm is unreactive (star). ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 µm.
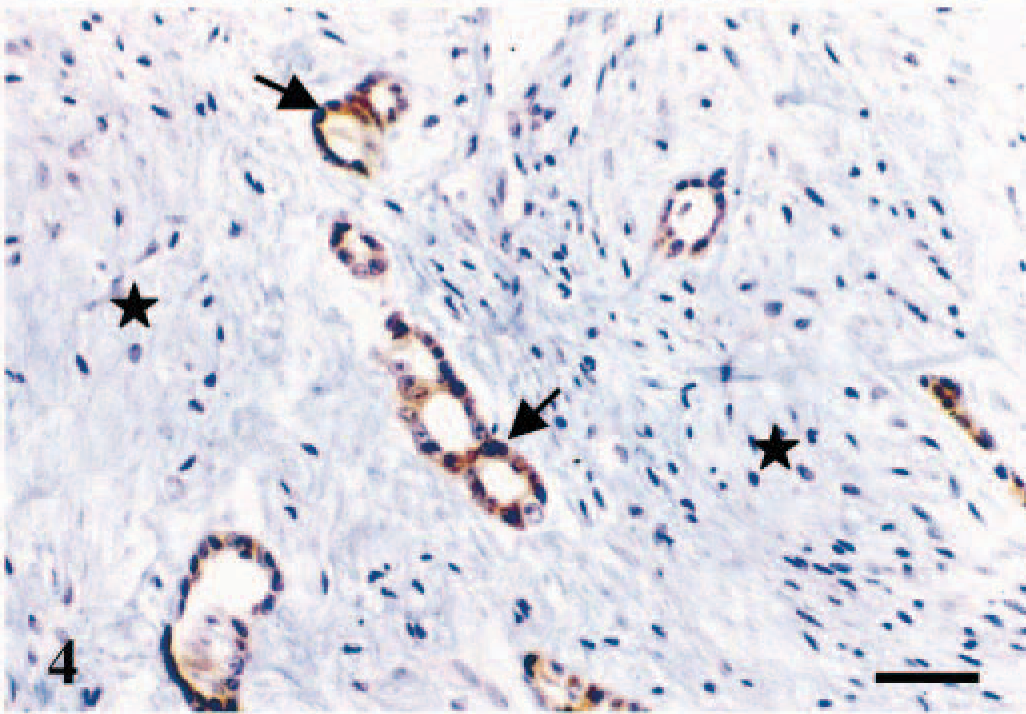
Mammary gland; dog. Benign mixed tumor. Immunoreactive products to ERβ antibody are observed in the nuclei and the cytoplasm of the majority of glandular epithelial-type tumor cells (arrows). The intensity of the reaction is similar at both levels. Stromal cells are unreactive (stars). ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 25 µm.
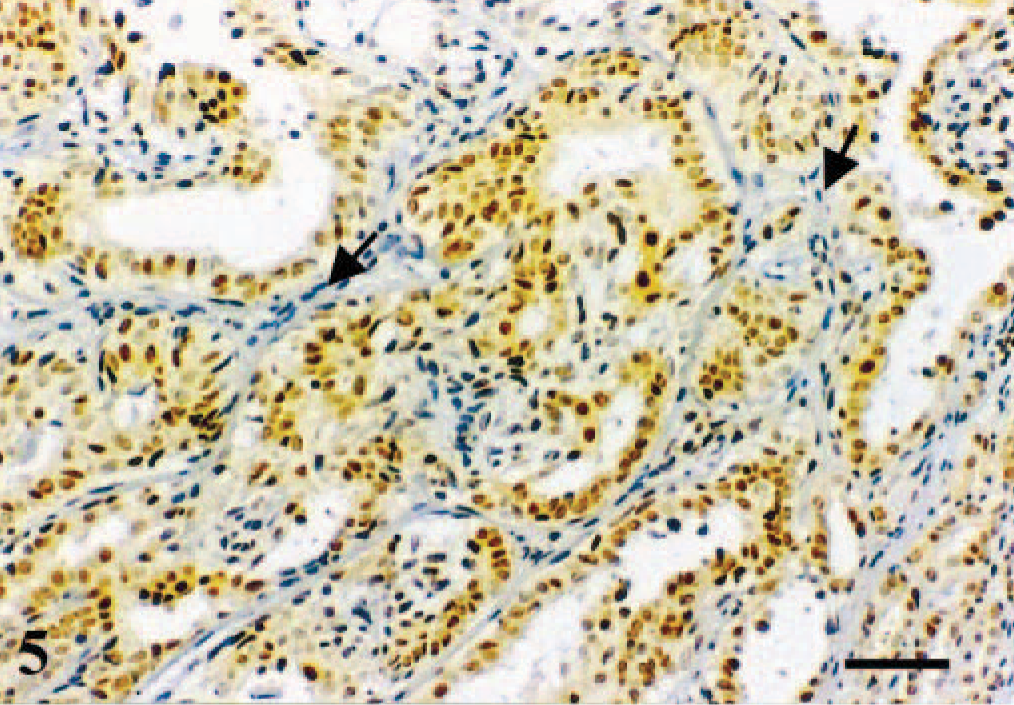
Mammary gland; dog. Simple tubular carcinoma. Immunoreactive products to ERβ antibody are observed in the nuclei of the overwhelming majority of the neoplastic epithelial cells. A fainter reaction is also observed in the cytoplasm of these cells. Stromal cells are unreactive (arrows). ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 25 µm.
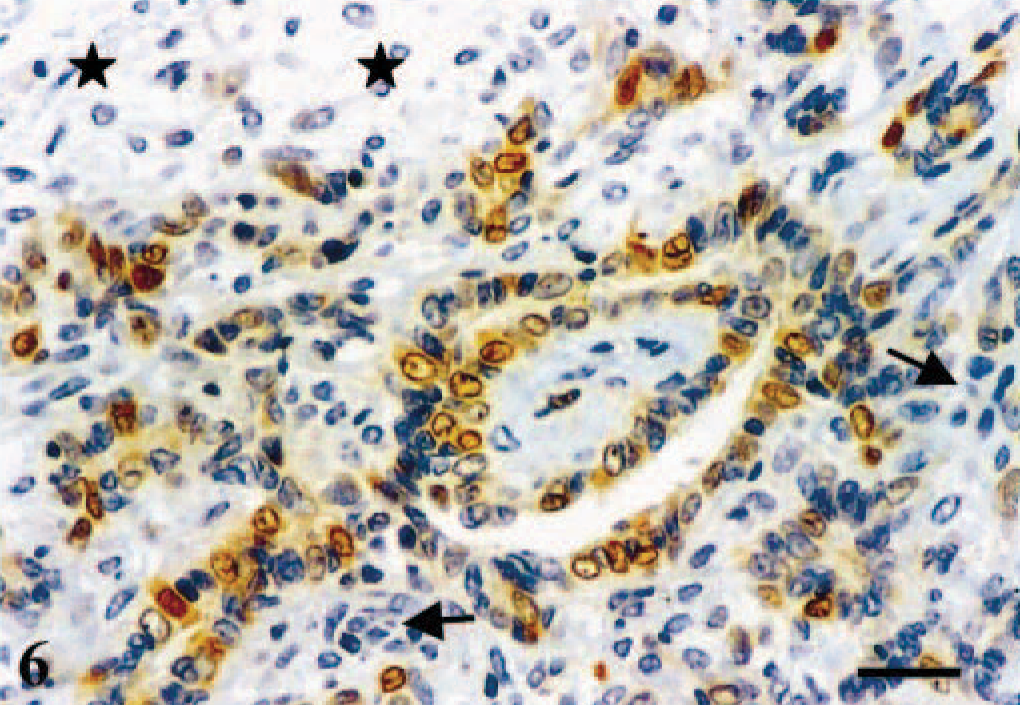
Mammary gland; dog. Complex carcinoma. Immunoreactive products to ERβ antibody are observed in the nuclei and the cytoplasm of some of the glandular epithelial-type tumor cells, whereas myoepithelial cells (stars) and stromal cells (arrows) are unreactive. ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 µm.
The polyclonal antibody anti-ERβ used in this study cross-reacted specifically with formalin-fixed and paraffin-embedded canine tissues in the same way that it reacted with cells and tissues known to contain the antigen in humans, 16 whereas there was no reaction in negative controls. As in human tissues, including breast carcinomas, ERβ immunoreactivity was found in the cell nucleus and, to a lesser extent, in the cell cytoplasm. The latter has been attributed to the production of both nuclear and cytoplasmic forms of the receptor by those cells. 8,16
In the mammary gland, ERβ was expressed in morphologically normal acinar and ductal epithelium and in one third of the tumors analyzed. In both cases, immunoreactive cells were abundant and of the luminal- or secretory-epithelial type, whereas stromal cells and the overwhelming majority of myoepithelial cells were unreactive. In humans, ERβ has been detected in normal breast epithelium and carcinoma cells by immunohistochemistry. 8,16 ERβ-positive tumors were more frequently benign (60%) than malignant (27%). Among the latter, tumors expressing ERβ were more frequently of complex and mixed histologic subtypes. 10 There are no data for comparison in the canine mammary tumor literature because, to our knowledge, our results are the first to address the issue of ERβ expression in either normal or neoplastic canine mammary gland. On the contrary, several studies have shown a similar differential expression of ERα in benign and malignant mammary tumors of the dog by using radioimmunoassay techniques, 13 which suggests that a similar progressive loss of hormone dependence with malignant transformation occurs with respect to ERβ.
The expression of ERβ in 26% of canine malignant mammary tumors is lower than the reported expression of ERα, which varies from 37 to 70% when using radioimmunoassay techniques 13 and from 10 to 87.5% when using IHC techniques. 4,5,12,14 In humans, western blot and IHC studies using ERβ antibodies have shown ERβ expression in 60% of breast carcinomas, 3,8 a figure similar to that of ERα expression in women. ERα expression is a favorable prognostic indicator in human and canine mammary carcinomas, 12,13 and our data point to similar biological implications for ERβ expression. Thus, the higher expression of ERβ in canine malignant tumors of complex and mixed histologic subtypes suggests that the expression of ERβ also may be a favorable prognostic factor because the grade of malignancy of complex carcinomas is lower than that of simple carcinomas. 10 Older reports point to lower grade of malignancy for the recently defined “carcinoma in benign tumor” histologic subtype. 10 However, the data with regard to ERβ expression in invasive tumors of the human breast and its relationship to prognosis are somewhat contradictory, with some groups reporting that the presence of this receptor is a good prognostic factor and others reporting the reverse. 2,8,15 Therefore, the role played by ERβ in mammary tumors is not clear. However, it is interesting to note that studies in ERα and ERβ knock-out animals suggest that ERα is the key mediator of estradiol action in the normal mammary gland. 6 Additional data suggest that ERα and ERβ play different roles in cell proliferation and carcinogenesis of breast cancer, partly by mediating the transcription of various genes via different types of deoxyribonucleic acid enhancers. 1 A negative correlation between the expression of ERβ (but not ERα) and the cyclin D1 gene (CCND1), a key cell cycle regulator that contains an AP1 element but not an estrogen-response element in its promoter, has been observed in human sporadic breast cancer. 1 In addition, ERβ and ERα seem to have different ligand-binding specificity, 9 and the results of experimental studies indicate that one role of ERβ is to modulate the ERα transcriptional activity. 7 Accordingly, the relative expression level of the two isoforms may be a key determinant of cellular responses to agonists and antagonists. Therefore, ERβ determination may provide additional information on the responsiveness of canine mammary carcinomas to different endocrine treatments. Taken together, these data suggest that the ratio between ERα and ERβ expression in canine mammary tumors may be useful to identify subgroups of ER-positive tumors. The clinical usefulness of the subgroups remains to be explored.
In conclusion, we have shown that normal ductal and acinar epithelium and approximately one third of canine mammary tumors express the second receptor for estrogens, the ERβ. The ERβ-positive tumors are more frequently benign than malignant. ERβ-positive malignant tumors are more frequently of the complex and mixed types than simple. The clinicopathologic associations of ERβ expression and the usefulness of examination of canine mammary tumors for ERβ expression over or in addition to ERα remain to be established.
Footnotes
Acknowledgements
This study was supported by Consejeria de Educatión y Ciencia, Junta de Andalucía, Spain (Grant number CVI-287).
