Abstract
An aged Saanen doe was euthanized following repeated severe hemorrhage from the vulva. Necropsy examination revealed mural thickening of tubular genitalia with firm, protruding intralumenal masses containing blood-filled cavitations, and effacement of normal cervical architecture. Histologically, uterine and cervical masses comprised a variably dense population of mildly pleomorphic spindle cells forming interlacing streams supported by variably dense collagenous stroma. Immunoperoxidase staining of neoplastic cells was positive for muscle-specific actin, supporting the diagnosis of low-grade leiomyosarcoma. Months later, the doe's twin was likewise euthanized due to persistent bleeding from the vulva associated with a large vulvar mass having histopathologic features similar to those of the previous case. The clinical, gross, and histologic findings are similar to five cases of caprine genital leiomyosarcoma identified in retrospectively analyzed case material. Analysis of caprine tumor accessions over 20 years demonstrated a significantly higher incidence of genital leiomyosarcoma within the Saanen breed.
Reported neoplasms of the ruminant female reproductive tract are rare.1,6 An estimated 10–50% of such tumors are of smooth muscle origin, of which approximately 10% are considered malignant.6 Leiomyosarcomas in these species are reported to display a growth pattern of low-grade malignancy, with slow invasion and rare metastasis. Case reports of genital smooth muscle neoplasms in goats have been sporadic.4,10,11 In the present report, genital leiomyosarcoma was diagnosed on post mortem examination of two sibling does 18 months apart, each having presented for persistent vulvar hemorrhage. Retrospective analysis of case material revealed five additional cases with similar historic and pathologic features.
A 12-year-old Saanen doe (goat No. 1), presented to the New York State College of Veterinary Medicine (NYSCVM) Large Animal Teaching Hospital for evaluation of hemorrhage from the vulva of 1-day duration. The animal had a 10-year history of recurrent pseudopregnancy, including development of hydrometra with spontaneous expulsion of uterine fluids (“cloud-burst”) and spontaneous lactation. Masses were evident on vaginal examination, and vulvar hemorrhage persisted until euthanasia 1 week later. On necropsy examination, generalized pallor was present and the vagina, uterine body, and uterine horns were moderately distended with clotted blood. The wall of the cervix was effaced by a 4-cm-diameter cavernous mass replacing normal architecture, with multiple protruding intralumenal sessile to pedunculate polypoid masses (Fig. 1). Extensive irregular mural thickening of the vagina and uterine body was present. Dispersed cystic mucosal thickenings were present in uterine body and horns. Bilaterally, ovaries contained numerous follicles; mammary glands were in lactation.
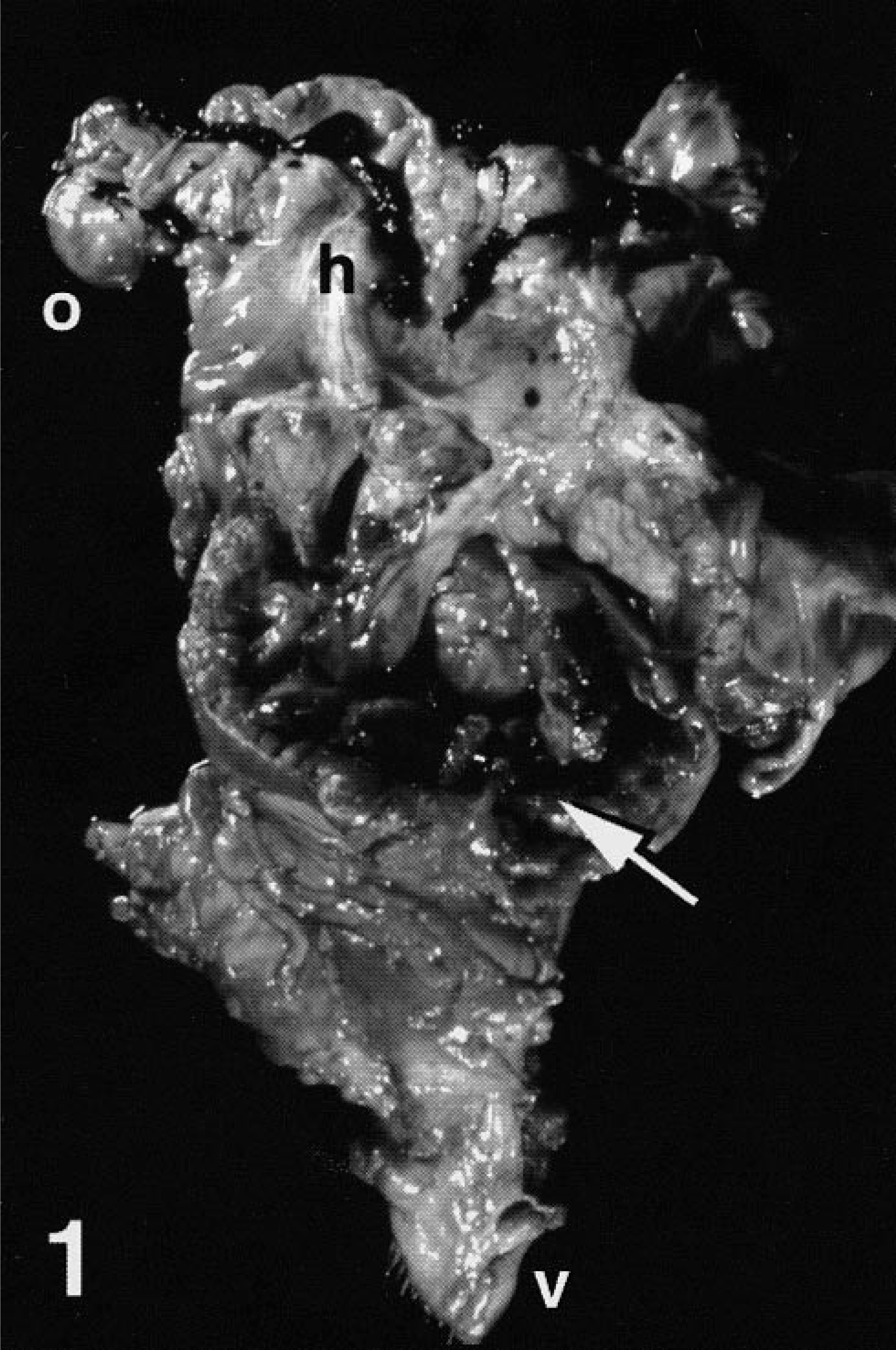
Reproductive tract, opened; Saanen doe, goat No. 1. Expansion and distortion of caudal uterine body and cervix by multiple intralumenal polypoid masses (arrow). Note hemorrhage within uterine horns (h). Ovary (o), vulva (v).
Tissues obtained at necropsy were fixed in 10% neutral-buffered formalin, paraffin-embedded, sectioned at 5–6 μm, and stained with hematoxylin and eosin (HE). Immunohistochemical (IHC) staining was performed by the strepavidin–biotin–peroxidase method (Zymed LAB-SA System, Zymed Laboratories, South San Francisco, CA) using primary murine monoclonal antibodies against muscle actin (Enzo Diagnostics, Inc., Farmingdale, NY), vimentin (Dako, Carpenteria, CA), desmin (Dako), and keratin AE1/3 (Boehringer Mannheim, Indianapolis, IN). Genital histology revealed tissue of dense cellularity composed of regularly interlacing and gently undulating bundles of elongate fusiform cells extensively infiltrating and expanding the uterine submucosa. The neoplastic tissue formed occasional sessile to papillomatous intraluminal projections, discontinuously covered by ciliated columnar epithelium, and engulfing and separating clusters of variably ectatic submucosal glands. The majority of neoplastic cells had elongate, mildly anisokaryotic nuclei with rounded ends, smooth margins, and finely stippled chromatin, and moderate amounts of finely fibrillar eosinophilic cytoplasm (Fig. 2). Variably dense collagenous matrix separated individual cells or streams. Neoplastic tissue contained areas of multifocal necrosis. Some sections displayed moderate nuclear atypia, including anisokaryosis, irregular nuclear contours, vesicular chromatin, and prominent single nucleoli. Increased numbers of mitoses were noted within areas of nuclear atypia. Overall, approximately six mitoses were present per 10 high-power fields (HPF). IHC staining of tissue against muscle actin revealed uniform moderate cytoplasmic staining of neoplastic cells, whereas staining against vimentin, desmin, and cytokeratin was negative. Criteria of tumor size, mitotic activity, cellularity, nuclear pleomorphism, and invasiveness supported a diagnosis of low-grade uterine leiomyosarcoma. Additionally, histologic changes consistent with cystic endometrial hyperplasia and suppurative endometritis were present.
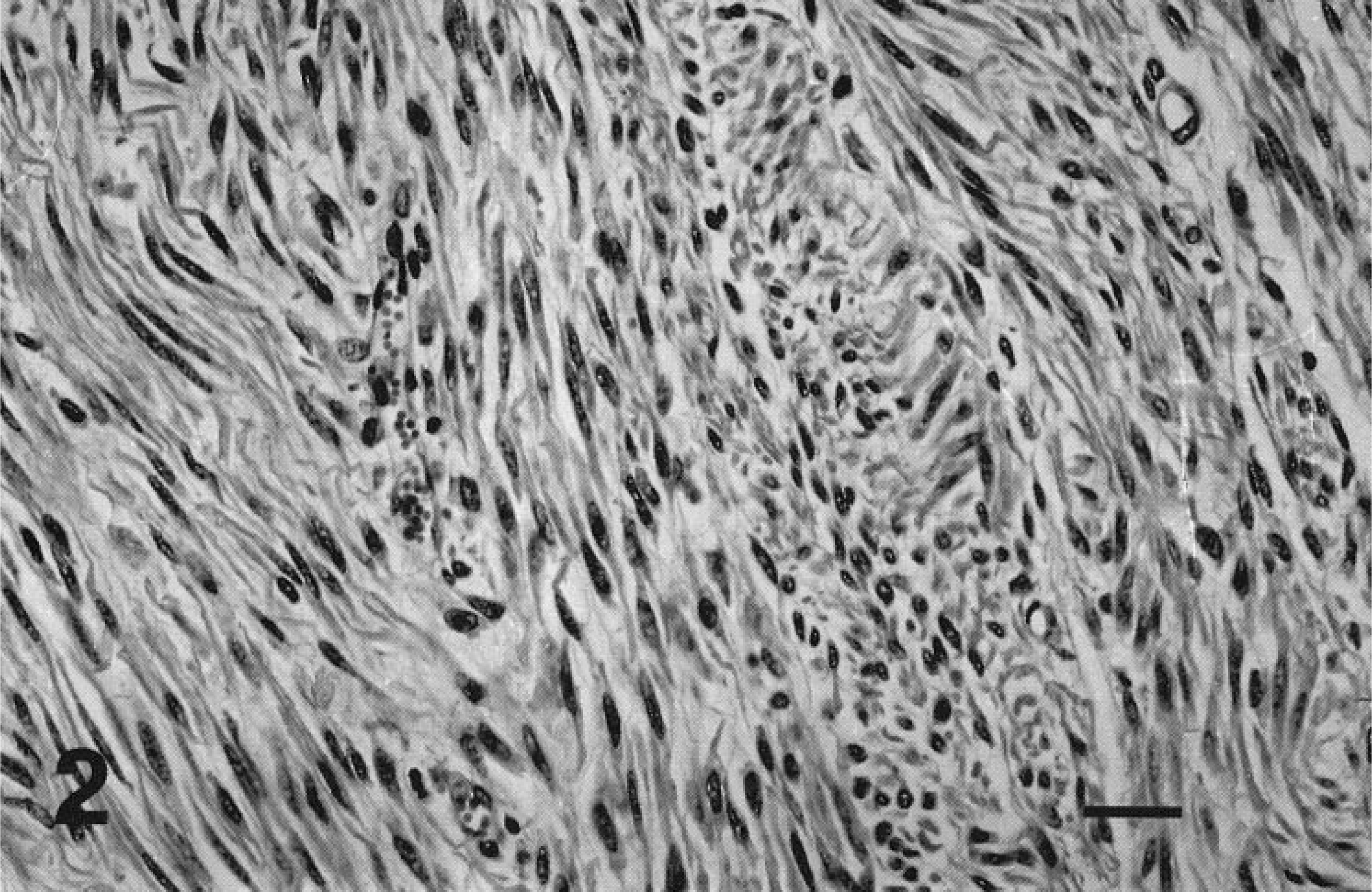
Uterus; Saanen doe, goat No. 1. Monomorphic, fusiform neoplastic cells form a regularly interweaving pattern in the submucosa. HE. Bar = 25 μm.
More than 1 year later, a 13-year-old Saanen doe (goat No. 2), from the same flock as the previously described animal, presented for pallor and history of straining. The doe, born co-twin to goat No. 1, likewise had experienced regular episodes of false pregnancy, and was euthanized after 3 weeks of vulvar hemorrhage. At necropsy of goat No. 2, a 23 × 13 × 10-cm pedunculated mass protruded through the cervix into the vaginal lumen, 5 cm distal to the external cervical os (Fig. 3). Extensive necrosis was evident on section.
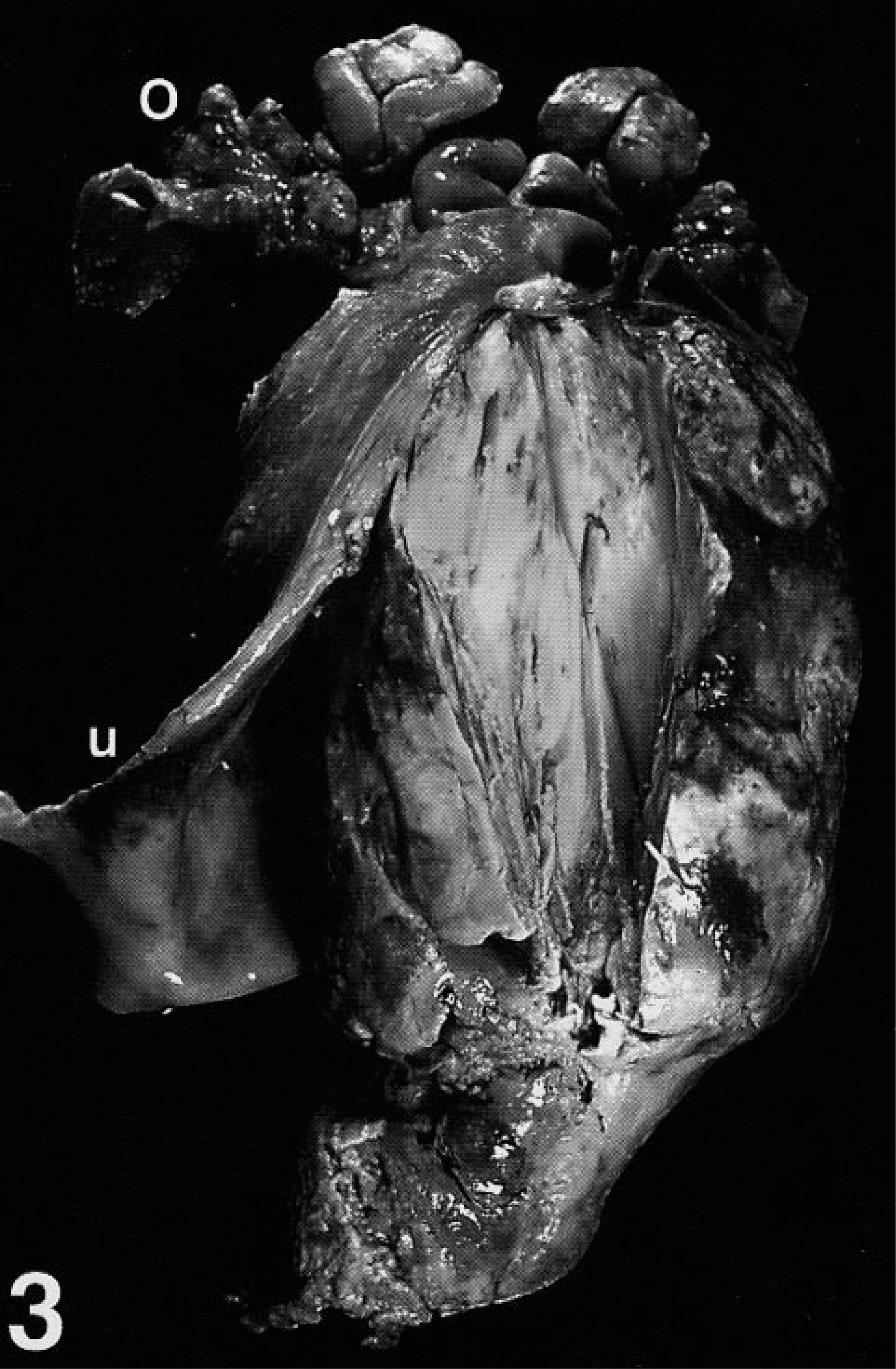
Reproductive tract, opened; Saanen doe, goat No. 2. A single large polypoid intralumenal mass with extensive areas of necrosis expands the uterine body (u) (reflected) and protrudes through the cervix into the vagina (not shown). Ovary (o).
Microscopically, the genital mass from goat No. 2 revealed extensive submucosal expansion by a spindle cell population of variable cellular density, forming fascicles and streams that separated and encircled variably sized ectatic glands lined by columnar epithelium. The neoplastic tissue formed irregular polypoid intralumenal projections and infiltrated variably between fascicles of the inner tunica muscularis. Spindle cells had moderate nuclear pleomorphism and were separated by variably abundant collagenous matrix (Fig. 4). More than 20 mitoses were present per 10 HPF. IHC staining against muscle actin was positive, with diffuse moderate cytoplasmic staining of most neoplastic cells, supporting a diagnosis of uterine leiomyosarcoma.
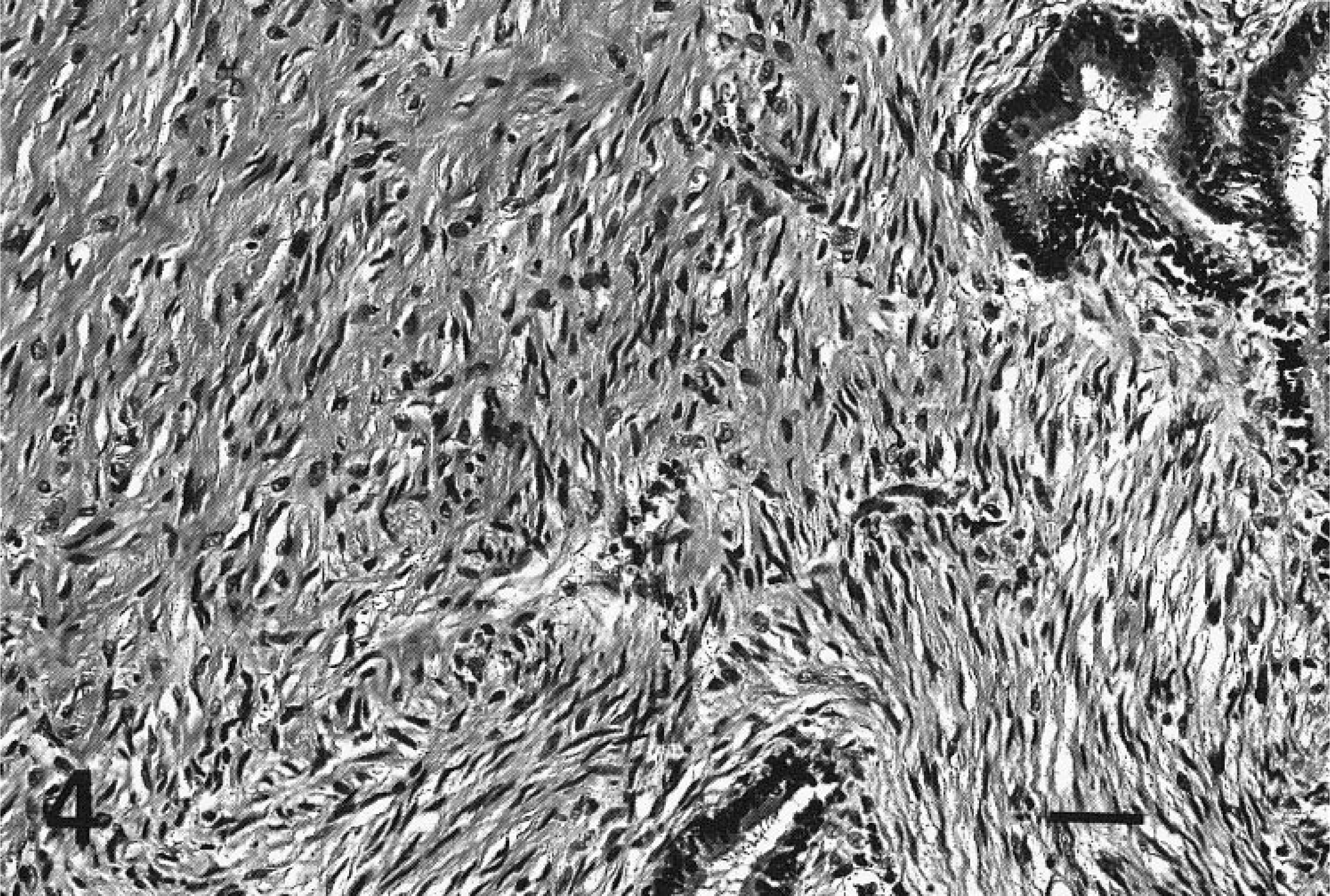
Uterus, Saanen doe, goat No. 2. Neoplastic cells engulfing submucosal glands are imbedded in variably dense collagenous matrix. HE. Bar = 50 μm.
A retrospective computer search of surgical biopsy and necropsy records from NYSCVM from 1977 to 1997 identified 12 additional caprine tumors of tubular genitalia for study. Available paraffin-embedded archival specimens were sectioned, stained by HE and anti-muscle actin IHC, and examined microscopically. Four cases of female genital spindle cell neoplasms (goat Nos. 3–6, Table 1) were identified, having morphology and IHC staining of lesions comparable to those previously described. Another (goat No. 7) diagnosed as vaginal leiomyosarcoma lacked tissues for review. Genital leiomyosarcomas comprised seven cases out of a total of 1,344 caprine accessions (0.52% incidence). In addition, single cases of uterine leiomyoma and vaginal undifferentiated sarcoma, both in Nubian does, were identified, as was a single case of scrotal leiomyosarcoma and two instances of uterine lymphosarcoma.
Caprine genital leiomyosarcomas: clinical presentation and gross pathology.

∗Siblings.
†Tissue not available for review.
Mitotic index among the caprine genital leiomyosarcomas examined varied from 5 to 40 mitoses per 10 HPF. Most neoplastic cells had mild to moderate diffuse cytoplasmic IHC staining against muscle actin, and had mild to moderate cellular pleomorphism. Tumors had occasional areas of atypia, multifocal zones of necrosis, and focal to extensive submucosal to transmural infiltration.
All affected goats for which ages were recorded were greater than 10 years old. In two cases (goat Nos. 4 and 6), the genital neoplasm was an incidental finding, but in the remaining cases it was considered the major factor in death or decision to euthanize. Three does (goat Nos. 3, 5, and 7) were reported to have multifocal involvement of the reproductive tract and one (goat No. 3) had localized peritoneal seeding and neoplastic tissue.
Computer search of pathology records identifying all neoplasms within the caprine study population yielded 101 tumors, for incidence comparison. Of these, only lymphosarcoma (20 cases) was more frequently diagnosed and thymoma (seven cases) was recognized with the same frequency as female genital leiomyosarcoma (Table 2). No nongenital caprine leiomyosarcoma was documented, consistent with the recognized site predilection in large animals.8
Caprine neoplasms diagnosed, 1977–1997, New York State College of Veterinary Medicine.
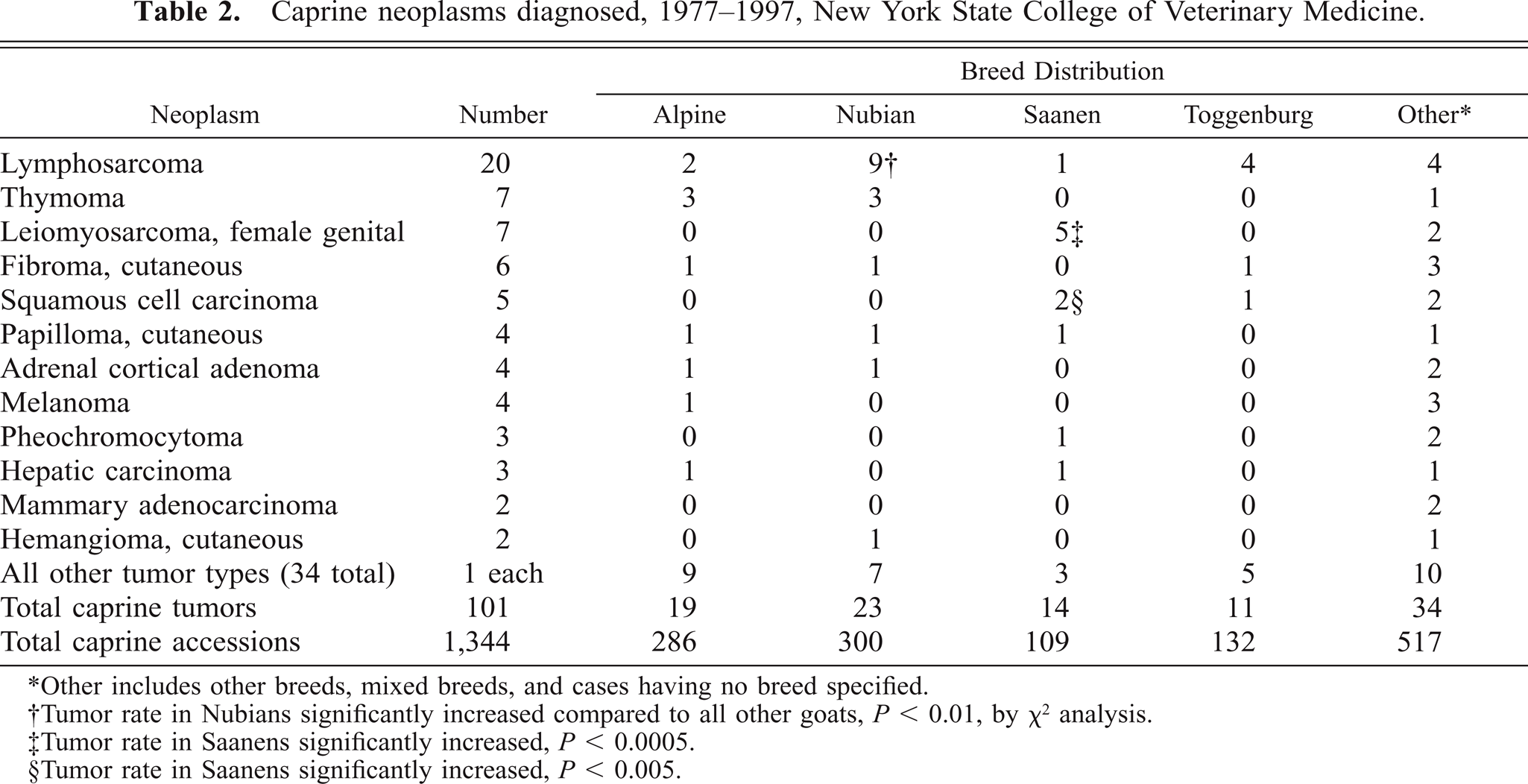
∗Other includes other breeds, mixed breeds, and cases having no breed specified.
†Tumor rate in Nubians significantly increased compared to all other goats, P < 0.01, by χ2 analysis.
‡Tumor rate in Saanens significantly increased, P < 0.0005.
§Tumor rate in Saanens significantly increased, P < 0.005.
Five of the seven cases of caprine genital leiomyosarcoma reviewed here occurred in Saanen does (Tables 1, 2). A total of 109 Saanen accessions were documented for the sample period, yielding a breed-specific incidence of 4.6% (5/109), and an odds ratio of 8.8 versus the incidence within the reference caprine population (0.52%). Chi-square (χ2) analysis showed a significant Saanen breed predilection for genital leiomyosarcoma (P < 0.0005). Interestingly, review of the veterinary literature identified two additional cases, also in Saanen does, having similar clinical and pathologic features.4,11 Chi-square analysis showed significant breed predisposition for specific tumor development in only two other instances. Nubian goats have an odds ratio of 2.0 (P < 0.05) for development of lymphosarcoma and Saanen goats are predisposed to squamous cell carcinoma, with an odds ratio of 4.9 (P < 0.005).
A retrospective search identifying neoplasms within the contemporary bovine pathology accession population yielded 26 spindle cell tumors of the female genital tract (comprising 10 vaginal fibromas; three each of vaginal fibrosarcoma, vaginal leiomyoma, and uterine leiomyoma; two each of vulvar fibroma and vulvar fibrosarcoma; and one each of uterine fibroma, vaginal fibroma, and vaginal leiomyosarcoma) among 17,896 total bovine accessions. Of these bovine genital neoplasms, five were of smooth muscle origin (0.03% incidence), one of which was diagnosed leiomyosarcoma (0.006% incidence).
Among the retrospective cases of caprine neoplasms, large tumor size (goat Nos. 1, 2, 5, and 7), infiltrative growth (goat Nos. 1, 2, and 3) peritoneal spread (goat No. 3), and tumor necrosis or hemorrhage (goat Nos. 1, 2, and 4) are gross features consistent with malignancy. The typical cellular morphology, with mild to moderate pleomorphism and often low mitotic rate, could be interpreted to imply benign or low-grade malignant behavior. Similar histologic appearance is described for genital smooth muscle neoplasms of women; however, clinical follow-up has shown aggressive behavior among tumors having more than five mitoses per 10 HPF selected from the most mitotically active sections.14 Analogous clinical data are lacking for goats. Mitoses within this series of neoplasms were frequently inconspicuous, and quantification was often complicated by accompanying inflammation or necrosis. Correlation of the histopathology with clinical outcome within this limited series suggested that the presence of even low numbers of mitoses and mild cellular atypia is consistent with malignant behavior in caprine genital smooth muscle tumors.
The seven cases of caprine genital leiomyosarcoma presented shared several historical, clinical, and gross pathologic features (Table 1). All cases involved aged does afflicted by a locally infiltrative submucosal smooth muscle neoplasm. One (goat No. 3) demonstrated transmural infiltration, vascular invasion, and peritoneal seeding with neoplastic tissue. Vulvar bleeding was noted ante mortem in three goats, and evidence of associated hemorrhage was found on necropsy examination in one other goat. Three does had a history of recurrent pseudopregnancy with lactation or hydrometra and spontaneous expulsion of uterine fluid (“cloud-burst”). Exposure to phytoestrogens is conjectured to contribute to the development of caprine hydrometra.13 Two published case reports of caprine vaginal leiomyofibromatosus4 and uterine leiomyosarcoma11 describe associated hemorrhage and numerous ovarian follicles11 or ovarian follicular cysts.4 The reported case of vaginal leiomyofibromatosis4 documented increased serum estrogen with clinical resolution after surgical resection and ovariectomy. Hormonal influence has been implicated in the pathogenesis of genital smooth muscle tumors of women.3,7,9,12 Immunohistochemical analysis of these caprine tumors for expression of estrogen and progesterone receptors would be of interest.
The myofibroblast has been implicated as the cell of origin for some genital smooth muscle cell tumors.2 The neoplastic cells within this series of caprine genital tumors were closely associated with lumenal epithelium and glandular structures (Fig. 4), had variably abundant production of collagenous matrix, and lacked evidence of derivation from smooth muscle of the tunica muscularis. These observations make plausible the consideration of endometrial stromal cells or myofibroblasts as origin of caprine genital leiomyosarcomas. Ultrastructural studies are necessary to better define the cell type. The possibility of myofibroblastic origin for the tumors presented here cannot be ruled out.
The observed Saanen breed predisposition for caprine genital leiomyosarcoma and diagnosis in two sibling does is intriguing. Several heritable mechanisms of increased neoplastic susceptibility offer explanations for apparent breed-related tumors. Increased incidence of an oncogene or tumor suppressor mutation within an inbred animal population can affect rate of tumorigenesis. Eker rats afford a well-characterized animal model of renal carcinogenesis. These animals have a germline mutation of a locus associated with tuberous sclerosis (Tsc2), encoding tuberin, a purported guanosine triphosphatase-activating protein having tumor suppressor activity.15 Affected rats also seem to have increased incidence of uterine smooth muscle tumors.5 A strikingly similar syndrome of presumed autosomal dominantly inherited neoplastic predisposition has been described in German Shepherd Dogs having multifocal renal cystadenomas and nodular dermatofibrosis.8 Affected bitches from tumor-prone lines also frequently have multiple uterine leiomyomas.8 Interestingly, two goats within our retrospective study group were reported to have concurrent cholangiocarcinoma (Table 1, goat Nos. 4 and 5). Saanen goats were also significantly (P < 0.005) overrepresented for incidence of squamous cell carcinoma (Table 2), although this tumor was not observed concurrently with genital neoplasia. Mutations affecting expression or activity of sex hormone receptors, signal transduction intermediates, DNA repair mechanisms, or enzymes responsible for xenobiotic activation, as well as additional diverse gene products could also result in heritable organ-specific neoplastic predilection. Investigation into an underlying genetic predisposition, if any, for this disease entity could be hampered by the advanced age at onset of afflicted goats.
Comparison of tumor incidence within the contemporary bovine and caprine populations of the retrospective study shows a lower incidence of bovine genital smooth muscle tumors. This observation is consistent with the reported relative rarity of this tumor type among cows. A report1 of a tumor incidence from an abattoir survey, encompassing more than one million animals, found five smooth muscle tumors among 302 genital tumors. Among 2,408 genital neoplasms in cows documented within a literature review included in the same paper, 10 were smooth muscle tumors, 3 of which were considered malignant. The decreased tumor incidence in bovine versus caprine populations is attributable at least in part to the difference in average age at inspection between species, due to husbandry practices.
Based on this case review, aged goats seem to have increased propensity for development of genital smooth muscle neoplasms compared to the incidence of such tumors in cows, as documented in the literature and reported here. Saanen does are significantly overrepresented compared to other caprine breeds within our study population for development of this malignancy. Bleeding from the genital tract is a common clinical presentation and this tumor is often concurrent with pseudopregnancy or increased ovarian follicular activity. The observation of this uncommon neoplasm in sibling does invites speculation on environmental or inherited predisposing factors.
Footnotes
Acknowledgements
We thank Joy Cramer for technical expertise in immunohistochemical staining.
