Abstract
Paeniclostridium sordellii is involved in enteric and histotoxic infections in several animal species. In humans, P. sordellii has been linked to gynecological disease, an association not previously investigated in animals. To unveil a potential association of P. sordellii with veterinary reproductive disease, a retrospective search of the database of the California Animal Health and Food Safety Laboratory System (1990–2020) was conducted and identified 9 cases of goats with P. sordellii-associated metritis or endometritis that were confirmed by immunofluorescence antibody test and/or bacterial isolation, and often co-colonized by Escherichia coli. Six of 9 does were also copper deficient. Polymerase chain reaction (PCR) on formalin-fixed, paraffin-embedded uterine tissue identified the sordellilysin gene in all 9 cases, and the lethal toxin gene in 4. Our findings suggest goats could be predisposed to P. sordellii-associated endometritis/metritis and toxemia when co-infected with E. coli. The role of mineral deficiencies influencing vulnerability to puerperal bacterial infections in goats is possible but remains undetermined. To our knowledge, this is the first report documenting the association of P. sordellii with veterinary gynecological disease.
Keywords
Introduction
During the peripartum period, most domestic species are susceptible to infection and disease, and this vulnerability may be exacerbated by extrinsic factors such as stress or nutrient deficiencies, many of which have been described in ruminants. One of the most significant peripartum diseases of animals is puerperal metritis, occurring as a sequela of retained fetal membranes and/or dystocia-associated uterine trauma resulting in infertility or, sometimes, septicemia. 3 In ruminants, the onset of puerperal metritis is usually 10 to 14 days after birth, and it may follow an acute or chronic course. Due to the relevance of reproductive diseases for the bovine dairy industry, the bulk of literature on puerperal endometritis or metritis has focused on cattle diseases,10,11,17,23 with fewer reports dedicated to small ruminants. 20 In goats, acute puerperal metritis (APM) is characterized by a dark red, malodorous uterine discharge, and complicated cases may result in peritonitis, involving agents such as Staphylococcus aureus, Pasteurella sp., and coliforms.14,23 As in other ruminants, APM is typically preceded by an abnormal parturition due to twin births, dystocia, placental retention, or trauma of the birth canal. 22 Most of the bacteria isolated in ruminant APM are gram-negative aerobes with fewer reports discussing the importance of anaerobes. 22 However, no reports have documented infection with Paeniclostridium sordellii in goats, a known cause of gynecological and umbilical disease in women and newborn children, respectively,1,5,10 and umbilical infections in foals. 18
We herein present a case series of P. sordellii-associated APM in goats, with special emphasis on the pathology of the condition and diagnostic workup.
Materials and Methods
Retrospective Database Search
The database of the California Animal Health and Food Safety Laboratory (CAHFS) was searched for all caprine cases of APM submitted between 1990 and 2020. The selected timeframe was based on availability of online records. Inclusion criteria included cases with (1) carcasses submitted for post-mortem examination and diagnosed with endometritis, metritis, and/or perimetritis; (2) history of peri-parturient death; and (3) identification of P. sordellii in uterine lesions by anaerobic culture or fluorescent antibody test (FAT). When available, data regarding epidemiology (eg, single case vs outbreak), viability of the offspring, and production class were recorded.
Gross Pathology and Additional Testing
Necropsies had been performed in every case, and in most instances, samples of lung, heart, trachea, thymus, thyroid glands, liver, spleen, adrenal glands, kidney, pre-scapular and mesenteric lymph nodes, esophagus, forestomachs, abomasum, skeletal muscle, ovary, uterus and/or placenta, and mammary gland were collected and fixed by immersion in 10%, buffered, pH 7.2 formalin for 24 to 72 hours, before being routinely processed to produce 4 µm-thick, hematoxylin and eosin–stained sections. Severity of inflammation on histologic sections was subjectively estimated based on the relative abundance of inflammatory cells, as follows: mild (scant numbers of inflammatory cells), moderate (modest numbers of inflammatory cells), and severe (abundant numbers of inflammatory cells associated with loss of tissue architecture). Brown and Brenn Gram stain was performed on sections of uterus of all animals.
Additional testing was completed following CAHFS standard operating procedures and included one or more of the following: aerobic culture of milk, liver, lung, and uterus; anaerobic culture of uterus; Brucella and Salmonella cultures of liver, uterus, and feces; FAT on uterine smears for Clostridium chauvoei, Clostridium novyi, Clostridium septicum, and P. sordellii. Numbers of bacteria on uterine smears were subjectively estimated based on the relative abundance of FAT-positive microorganisms per 400× high power field, as follows: mild (scant numbers of bacteria), moderate (clusters of bacteria), and high (excessive numbers impeding counting). Additional tests included fecal flotation and hepatic heavy metal screens, including lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, cadmium, and selenium.
Molecular Identification of P. sordellii
Formalin-fixed, paraffin-embedded (FFPE) sections of uterus from all cases were processed for polymerase chain reaction (PCR) detection of 3 relevant P. sordellii genes associated with virulence, including the sordellilysin (sdl), lethal toxin (tcsl), and hemorrhagic toxin (tcsh) genes, as previously described. 16 DNA extracted from P. sordellii strain JGS6382, which encodes all 3 toxin genes, was used as positive control for all reactions. Reactions in which DNA was replaced by FFPE uterine scrolls of an unaffected doe were used as negative controls. PCR amplicons were observed in 1.5% agarose gels stained with ethidium bromide.
Results
Clinical History and Epidemiology
Relevant epidemiological data are included in Table 1. Nine cases found in the database search fulfilled the inclusion criteria. In general, the clinical complaints indicated that the goats were in good nutritional condition, lacked significant premonitory signs, and died during or immediately after parturition. In only 3 instances, information about kidding was recorded; in 2 of these cases (cases 8, 9), fetuses were born alive and were clinically healthy at the time of the postpartum death of the doe. In 1 case (case 6), peripartum death of the dam resulted in delivery of a dead fetus. Epidemiological information was available in only 3 cases (cases 7, 8, 9), and indicated mortalities occurred as outbreaks involving 3 (cases 7, 8) or 4 goats (case 9).
Epidemiology of Paeniclostridium sordellii–associated reproductive disease in goats submitted to the California Animal Health and Food Safety Laboratory, 1990–2020.

Abbreviation: Unk, unknown.
Gross Pathology Findings
Subserosal petechiae were observed in the uterine horns and/or body in at least 1 case (case 8; Fig. 1a). The uterus was distended by approximately 300 to 500 mL of dark red, flocculent, malodorous fluid admixed with white friable strands (Fig. 1a) enclosed by a thickened uterine wall lined by an edematous and hemorrhagic endometrium (case 9; Fig. 1b). Uterine rupture occurred in 1 goat (case 3). Soft tissues in the pelvic area were edematous in 1 goat (case 2), and the cervix was noted to be open in another case (case 5). Mammary glands were unremarkable except in case 8, where the skin had a purple hue. In 3 cases, the external genitalia and perineum were edematous (cases 1, 2, 9) and the hindlimbs were soiled with a dried brown material presumed to be postpartum vaginal discharge (cases 2, 3, 9). In case 7, the right parotid lymph node and 1 retropharyngeal lymph node had central abscesses about 2 cm in diameter, that contained thick, pasty pale-yellow pus.
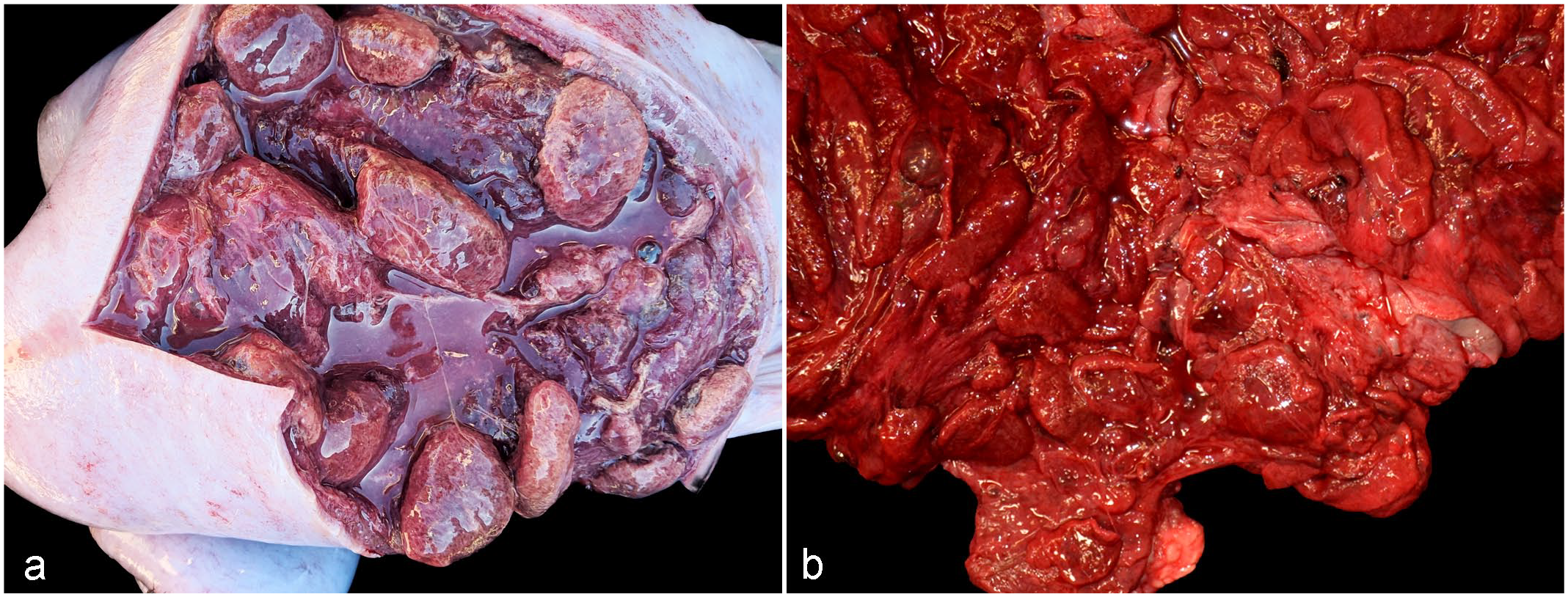
Paeniclostridium sordellii-associated metritis, uterus, does. Macroscopic findings. (a) Malodorous blood-tinged fluid admixed with fibrin strands fill the uterine lumen, and uterine walls are markedly edematous. Subserosal petechiae are also visible. Case 8. (b) Markedly edematous and hemorrhagic endometrium with caruncles still present. Case 9.
Histopathology of Reproductive Tissues
Histologic findings for each case are summarized in Table 2. Briefly, the uterine lesions included endometrial (cases 1, 2, 5, 7) or transmural inflammation (cases 3, 4, 6, 8, 9), and were recorded as endometritis or metritis, respectively. The severity of the inflammation was variable and was classified as mild (cases 3, 7, 9), moderate (case 8), or severe (cases 1, 2, 4, 5, 6). The lesions were necrotizing or necrohemorrhagic in 7 of 9 cases (cases 1, 2, 4, 5, 6, 8, 9), and the inflammation was suppurative (cases 2, 5, 6, 8, Fig. 2a) or a mixture of neutrophils, lymphocytes, plasma cells, and macrophages (cases 3, 7, 9) with thrombosis (cases 1, 5, 6, 8, Fig. 2b) and emphysema (cases 6, 7; Fig. 2c). Intralesional clusters of plump gram-positive, spore-forming bacilli and aggregates of fine, gram-negative cocci/coccobacilli (Fig. 2d) were observed in all cases. The mammary gland was inflamed in case 5, despite the lack of gross lesions, and was histologically normal in case 8 despite the cutaneous discoloration detected grossly. The only case of mastitis, found in case 5, consisted of neutrophilic inflammation without identifiable bacteria in the sections examined.
Main histopathological findings in goats with Paeniclostridium sordellii–associated reproductive disease submitted to the California Animal Health and Food Safety Laboratory, 1990–2020.
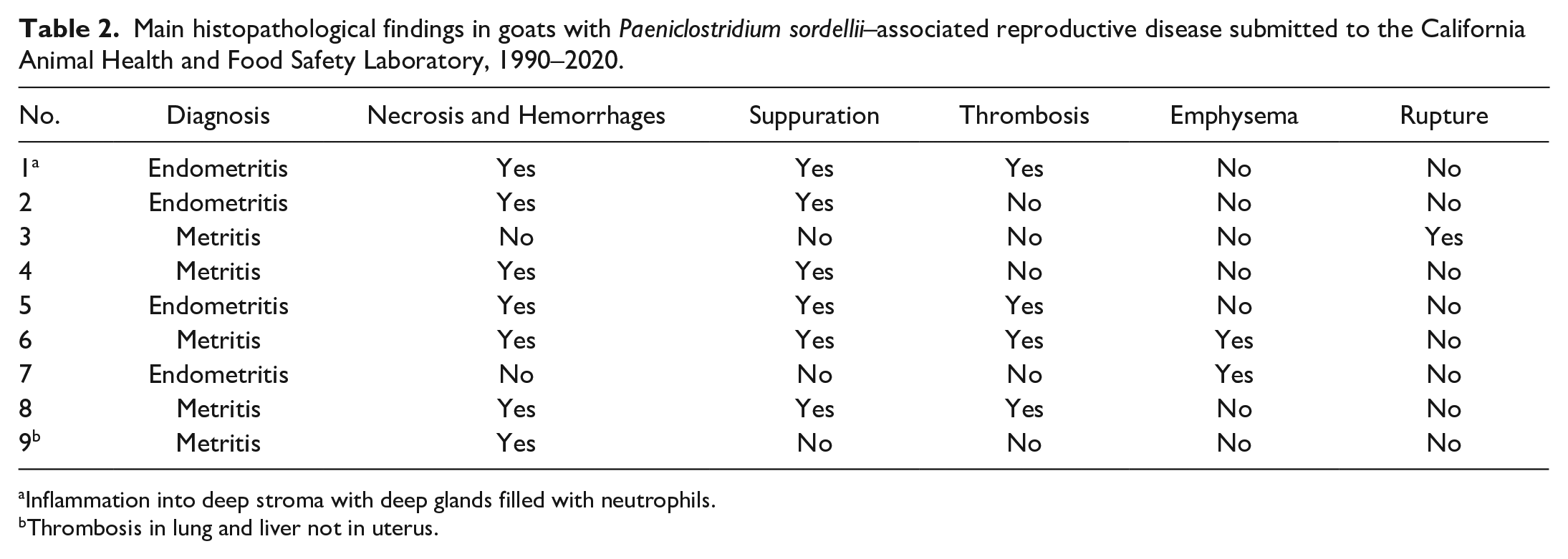
Inflammation into deep stroma with deep glands filled with neutrophils.
Thrombosis in lung and liver not in uterus.
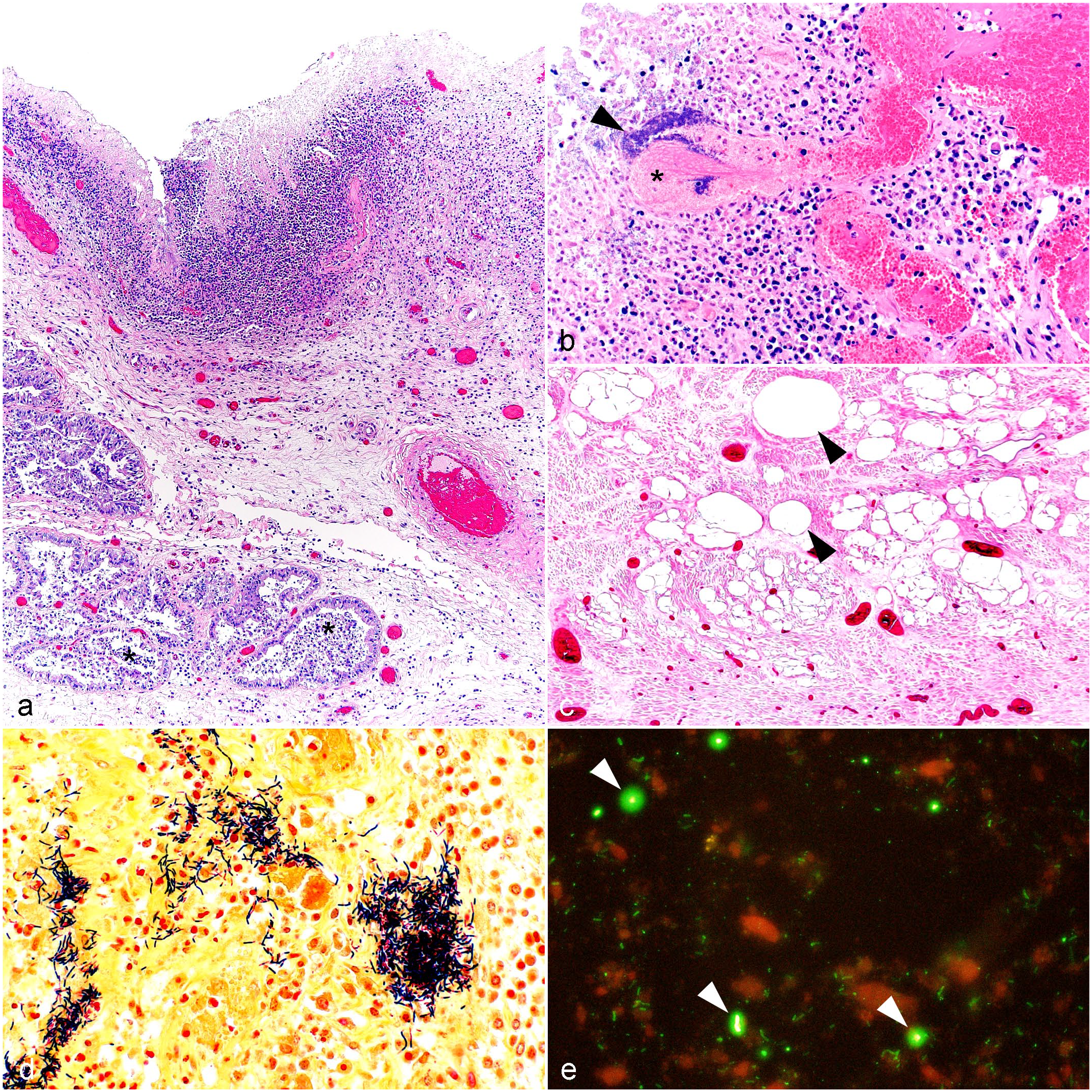
Paeniclostridium sordellii-associated metritis, uterus, does. Microscopic findings. (a) The endometrium is superficially necrotic and replaced by inflammation that often reaches endometrial glands (*). Case 6. Hematoxylin and eosin (HE). (b) The inflammatory infiltrate is composed predominantly of neutrophils. Bacterial colonies (arrowhead) are present within the necrotic debris replacing the endometrium and enmeshed in a thrombus (*). Case 5. HE. (c) Gas accumulation (arrowheads) is observed multifocally expanding and separating submucosal and muscular layers. Case 7. HE. (d) Bacterial colonies observed in Fig. 2b are predominately composed of gram-positive rods and fewer gram-negative rods. Case 8. Gram stain. (e) Uterine impression smears are positive for P. sordellii (arrowheads); bacteria are labeled green; cells and debris are labeled red. Case 8. Fluorescent antibody test (FAT) for Paeniclostridium sordellii.
Histopathology of Nonreproductive Tissues
Lesions suggestive of endotoxemia, exotoxemia, and/or septicemia were documented in 4 cases (cases 1, 5, 8, 9) and included pan-systemic circulatory leukocytosis (cases 8, 9) and the presence of fibrin thrombi in hepatic sinusoids and pulmonary capillaries (case 9). Case 8 also had random foci of hepatic necrosis, splenic congestion, and leukocytosis of lymph node sinuses. Cardiac lesions were reported in 1 animal (case 9) and included rare focal conducting cardiomyocyte necrosis, characterized by hypereosinophilia and fragmentation, and segmental hypercontraction and swelling of subendocardial myocytes. Mild to moderate centrilobular hepatic lipidosis was observed in 2 cases (cases 3, 7). Lymphadenitis with abscess formation in the right parotid and 1 retropharyngeal lymph nodes was diagnosed in case 7.
Detection of P. sordellii and Additional Testing
Results of bacteriology, molecular testing, and toxicology are summarized in Table 3. P. sordellii was isolated from the uterus in all cases, in combination with E. coli in most goats. C. perfringens (not typed) was also isolated from the uterus in 2 cases (cases 5 and 8). E. coli was isolated from multiple tissues in all cases except for case 4. From uterine samples, Streptococcus suis was isolated in case 4, and mixed flora in cases 2, 6, and 9. Corynebacterium pseudotuberculosis was isolated from the right parotid lymph node and 1 retropharyngeal lymph node of case 7.
Bacteriology, molecular testing, and toxicology results obtained from goats with Paeniclostridium sordellii–associated reproductive disease submitted to the California Animal Health and Food Safety Laboratory, 1990–2020.
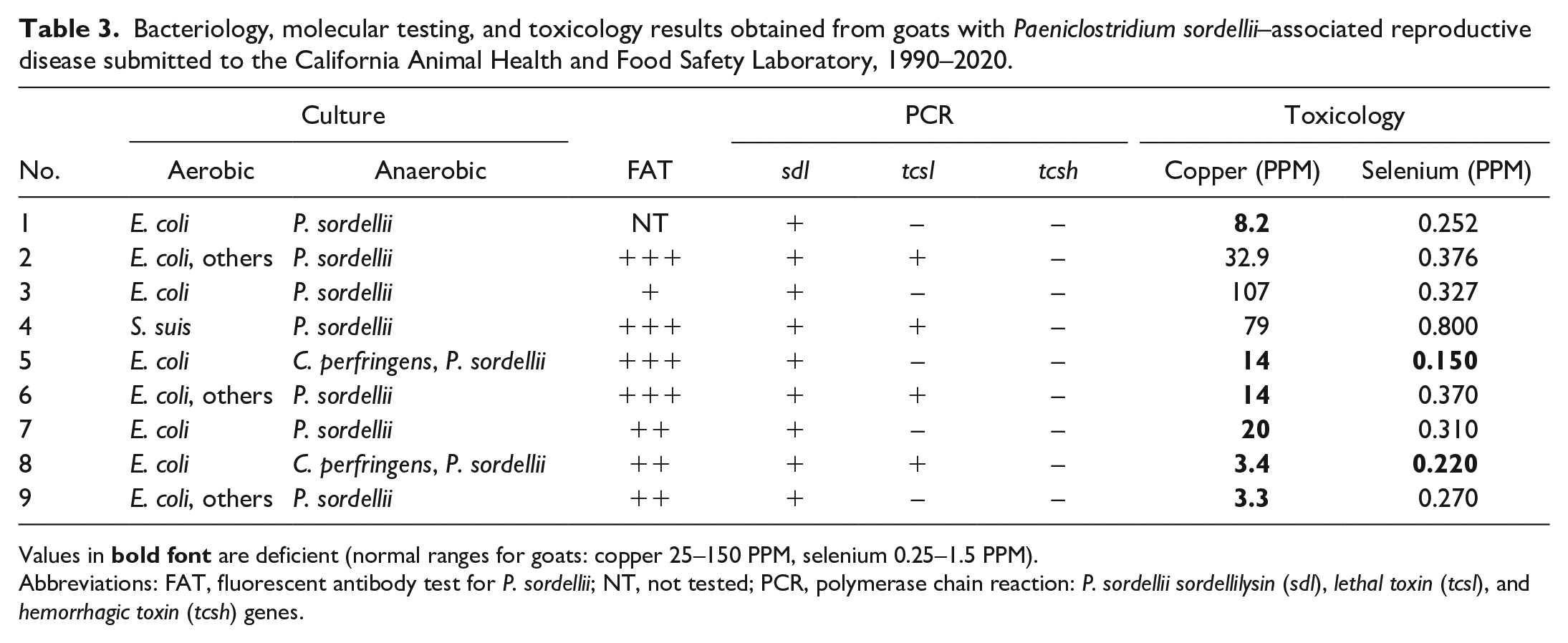
Values in
Abbreviations: FAT, fluorescent antibody test for P. sordellii; NT, not tested; PCR, polymerase chain reaction: P. sordellii sordellilysin (sdl), lethal toxin (tcsl), and hemorrhagic toxin (tcsh) genes.
Fluorescent antibody testing on endometrial impression smears was performed in cases 2 through 9, detecting P. sordellii in moderate to high numbers in all cases tested (Fig. 2e). Moderate numbers of C. chauvoei were detected in smears from case 9.
The sdl gene of P. sordellii was identified in all cases, whereas the tcsl gene was detected in 4 animals (cases 2, 4, 6, 8). The tcsh gene was not found in any case.
The concentration of copper in the liver of 6 animals (cases 1 and 5–9) was decreased, as was the hepatic selenium concentration of cases 5 and 8, and the zinc concentration in case 6 (Table 3).
Discussion
P. sordellii is an anaerobic, gram-positive, spore-forming rod, which is part of the gas gangrene complex. The bacterium is found in soil, sewage, and water, and is a rare inhabitant of the gastrointestinal tract in humans and animals. The bacterium is also considered a transient inhabitant of the genital tract in a small percentage of healthy women, 5 with infection following contamination of wounds from gynecological procedures. 12 It is also a documented cause of postpartum metritis and fatal toxic shock syndrome (TSS) in humans, often accompanied by a marked circulatory leukocytosis referred to as a leukemoid reaction, after full-term deliveries or medical/spontaneous miscarriages. 12
The main virulence factors of P. sordellii are tcsl and tcsh, both of which are part of the large glucosylating toxin (LCGT) family, 19 and are identified as the main virulence factors in human TSS. 25 Interestingly, even though non-toxigenic strains of P. sordellii induce reduced cytotoxicity in vitro and lower pathogenicity in animal models of infection, cases of non-TSS P. sordellii infection may occur in the context of polymicrobial inflammation. 27 Other strains are known to produce sdl, phospholipase C, and neuraminidase, 6 and although their role in animal diseases is not known, it is likely these toxins also play a role in human gynecological disease. The pathogenesis of P. sordellii-mediated histotoxic disease includes generation of localized hypoxia caused by trauma-related necrosis and pH alkalization, leading to increased vascular permeability, refractory hypotension, edema, 12 and accumulation of protein breakdown products, necessary for anaerobe proliferation and the development of gas gangrene in humans,2,13 ruminants, 28 horses, 21 and the umbilicus of foals. 18
Multiple species of clostridia have been implicated in reproductive infections in animals and humans. In APM, clostridial populations are usually represented by Clostridium perfringens and Clostridium tetani, which are known to colonize the uterus and lead to fatal toxemia. 3 Clostridiodes difficile has also been isolated from post-parturient sows with mastitis-metritis-agalactia following antibiotic administration, which is usually associated with low mortality. 7 Postparturient gas gangrene caused by Clostridium septicum has been described in cattle with necrotizing metritis and vulvo-vaginitis, and typically affecting first-calf heifers 1 to 3 days after parturition. 24 In goats, postpartum clostridial metritis is usually associated with retention of fetal membranes. This has prompted the development of anti-clostridial vaccination protocols designed to protect does after prolonged dystocia (failure to expel fetal membranes by 6 hours postpartum). 4
The non-gravid uterus is innately resistant to infection. This resistance varies throughout the estrus cycle, reaching its peak during the luteal phase. However, it has also been demonstrated that a uterus under the influence of progesterone is susceptible to bacterial infections. 22 During puerperium, uterine infections are typically the result of an abnormal parturition including placenta retention, twin births, dystocia, abortions, or traumatic lacerations of the birth canal. In goats, postpartum clostridial metritis has been associated with retention of fetal membranes. However, other factors such as deficiencies in trace minerals, including zinc, selenium, chromium, and copper, can negatively influence the doe’s health due to their roles in important physiological functions including immunity. 9 Copper, in particular, has an important immunomodulatory role as it assists with the regulation of humoral, cell-mediated, and innate immune function. Thus, copper-deficient animals are considered to be potentially immunosuppressed and increasingly vulnerable to infection with higher mortality rates when infection occurs. 26
The direct influence of copper deficiency in increasing susceptibility to bacterial puerperal infection in the 6 does in which it was found cannot be confirmed from this study, although this hypothesis should be further explored in future caprine APM cases. Zinc deficiency was detected in 1 case; however, artifacts from the type of container used to collect the blood sample frequently occur, so in this case, artifactual deficiency could not be distinguished from true deficiency. Two goats in this study were identified as selenium deficient, which is a documented predisposing factor with regard to post-parturient metritis. 15 It is therefore possible that selenium deficiency was also a contributing factor in these cases.
Even when clinical signs are not present, uterine involution is considered a septic process. Certain bacteria or their toxins within the involuting uterus during the postpartum period can interact with retained membranes resulting in the secretion of placental prostaglandin E2, further predisposing to uterine infection. 8 To date, it is unknown if P. sordellii is part of the reproductive tract microbiota in does, or if infection originated from fecal or environmental contamination, especially in the cases with retained fetal membranes. In summary, these results could suggest that the puerperal stress combined with specific mineral deficiencies facilitated P. sordellii proliferation in/on the endometrium and significant endometritis with subsequent polymicrobial infection, frequently represented as coliform endotoxemia in the present study.
Footnotes
Acknowledgements
The authors appreciate the assistance of Juliann Beingesser with FAT and histopathology, Dr Robert Poppenga with toxicology, and the numerous veterinary pathologists who contributed with coordination of the retrospective cases included in this study. We also thank Dr P. Wolff for providing one of the gross photographs (![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our study was funded by the California Animal Health and Food Safety Laboratory, UC Davis.
