Abstract
A 6-year-old Simmental cow infected with Theileria buffeli had a clinical disease characterized by theilerial parasitemia, macrocytic normochromic anemia with acanthocytosis and spherocytosis, lymphoid hyperplasia (lymphocytosis, edematous lymphadenomegaly), dysproteinemia, evidence of liver disease, and a low serum antibody titer against T. buffeli. The cow was in a herd in which all cattle originated in Missouri; 22/75 (29%) of cattle had a theilerial parasitemia and 26/75 (35%) had titers to T. buffeli of ≥1:160. Classification of the Missouri bovine organism as T. buffeli was based on DNA sequencing and comparison to sequences for T. buffeli and Theileria sp. type A obtained from GenBank. Intraerythrocytic veils and piroplasms were seen during transmission electron microscopy. The organism was successfully transmitted to two splenectomized calves, which developed mild anemias while parasitemic. Blood from the second calf was used as the source of T. buffeli antigen for an indirect immunofluorescence antibody test. Theilerial isolates from a Missouri white-tailed deer were also sequenced and resembled Theileria sp. types F and G and were not consistent with the bovine organism.
Keywords
Hemoparasites known to infect bovine erythrocytes and cause anemia include organisms from the genera Anaplasma, Eperythrozoon, Babesia, and Theileria. Theilerial parasites infect a broad range of wild and domestic artiodactyls throughout the world with highest prevalence in tropical and subtropical climates of Africa, Europe, Australia, and Asia.4,13 East Coast fever, caused by T. parva, is the most severe theilerial disease of domestic cattle. The life cycle of Theileria spp. includes intralymphocytic schizonts and intraerythrocytic piroplasms (merozoites and trophozoites). Intralymphocytic schizonts are the major pathogenic stage for T. parva, erythrocytic piroplasms are the major pathogenic stage for T. mutans, T. orientalis, and T. sergenti, and lymphocytic and erythroid forms of T. annulata are considered pathogenic.13,27 Piroplasms of Theileria spp. are structurally very similar to those of Cytauxzoon spp. and small Babesia spp.4,23
The first two reports of Theileria sp. seen in bovine erythrocytes in the United States originated in Kansas (1950) and Texas (1975) and the organisms were identified as T. mutans.18,29 In both reports, intraerythrocytic piroplasms were not considered pathogens and were found in calves experimentally infected with Anaplasma marginale. More recently, cases of bovine theileriosis were reported in Texas and North Carolina. Infected cattle had parasitemias, but clinical signs were not reported.2 Based on small subunit ribosomal RNA gene analysis of four isolates, the agents were considered indistinguishable from or closely related to T. buffeli.
Here, we describe a severe case of bovine theileriosis in Missouri, characterize the organism's structural and molecular features, provide results of experimental transfer, and report a high herd prevalence of theilerial infection. This report is the most complete investigation of bovine theileriosis in the United States to date. Although bovine theileriosis had not been documented in Missouri, a theilerial agent is commonly seen in Missouri white-tailed deer (Odocoileus virginianus).17 Also, a closely related organism (similar to B. divergins, which infects cattle and people in Europe) is described in Missouri as the cause of a lethal case of human babesiosis.9
Materials and Methods
Animals
Index case
In mid-April, a referring veterinarian examined a 6-year-old pregnant Simmental cow that was lethargic and weak. She had separated herself from the rest of the herd for at least 2 days and recently weaned a 225-kg calf. The cow was in sternal recumbency but still eating, drinking, and chewing her cud (day 1). She could rise on her front legs but not on her rear legs. Other abnormalities included tachycardia and pale mucous membranes. Clinical signs were suggestive of anaplasmosis, which is common in the area. Blood was collected for hemoparasite detection and serologic assays. A dextrose solution containing calcium was administered intravenously, but no response was seen. Long-acting tetracycline was given intramuscularly. Hay, water, and grain were carried to the cow, and a portable corral was installed around her.
The cow continued to eat and drink over the next few days. On day 6, the referring veterinarian examined the cow, noted little change in the clinical status except more severe weakness, and collected blood for analysis.
On day 8, the cow was laterally recumbent, bloated, unable to stand, and moderately dehydrated. The rectal temperature was 105°F, heart rate was 110 beats/minute, the respiratory rate was 50 breaths/minutes, mucous membranes remained pale, and moderate scleral injection was present. Diarrhea was present, and the rectal wall felt thickened and edematous. External lymph nodes, most notably the prescapular nodes, were enlarged. The cow appeared hyperesthetic and her head and neck were pulled more dorsal than normal. Euthanasia was performed because of the cow's deteriorating condition, and a necropsy was conducted.
Splenectomized calves
Two male calves, 9 and 21 days of age, were considered clinically healthy based on physical, hematologic, and serum chemical data. Hemoparasites were not found during repeated blood analysis both prior to and after splenectomy. A splenectomy was completed on each calf by standard aseptic techniques.36
Herd of the index case
After the diagnosis of bovine theileriosis in one cow, the involved herd was quarantined by the Missouri Department of Agriculture (MDA), and cattle older than 4 months were examined by a consortium of private, MDA, US Department of Agriculture, and University of Missouri veterinarians. Other cattle in the cow–calf herd of about 45 cows and their offspring were not ill. The herd was located within 1.5 km of the Missouri River flood plain in central Missouri. All cattle in the herd were reared by the owner except for two purebred bulls that were purchased within the two previous years; both bulls were raised in Missouri.
Blood was collected from 75 cattle for hematologic and serologic evaluation. Blood films were made immediately after collection of the blood samples in tubes with ethylenediaminetetraacetic acid (EDTA). Later the same day, blood films were stained with a Wright-Giemsa stain (Fisher Diagnostics). Two people (a medical technologist and a clinical pathologist) examined a blood film from each animal for intraerythrocytic piroplasms for a minimum of 5 minutes for each film. If neither microscopist found intraerythrocytic piroplasms, the film was considered negative. To be considered a positive film, both microscopists had to find at least two parasitized erythrocytes.
Microhematocrits were measured by centrifugation of EDTA-treated blood. Serum was collected from clot tubes and sent to the School of Veterinary Medicine, University of California–Davis, for determination of titers to the T. buffeli and to the National Veterinary Services Laboratory (Ames, IA) for determination of titers to bovine leukemia virus (agar gel immunodiffusion test) and A. marginale (complement fixation test). While blood was being collected, veterinarians examined the cattle for evidence of lymphadenomegaly, anemia, and the presence of ticks. The herd was monitored by the MDA for additional cases of theileriosis for 4–5 weeks. No additional clinical cases were seen, and the herd was released from quarantine.
White-tailed deer
A 3-month-old white-tailed deer was presented to the University of Missouri Veterinary Teaching Hospital with diarrhea of 8 days duration, dehydration, and a heavy tick infestation. The fawn was from a farm near the Missouri River, about 50 km downstream from the cow–calf index herd. Blood was collected for routine analysis and for characterization of the intraerythrocytic piroplasms. Oxytetracycline and fluid therapy resulted in an uneventful recovery.
Transmission electron microscopy
Fresh whole blood from the index cow was processed for transmission electron microscopy using methods previously described.31
Attempts to isolate Theileria sp
In vitro cultivation of the Theileria sp. was attempted with whole blood samples from the index cow. Leukocytes were removed from EDTA-anticoagulated blood from a healthy cow by passage through a cellulose column. Erythrocytes were prepared as a 10% suspension in a solution containing 60% medium 199 with Earle's salts and 40% fresh bovine serum. The medium was buffered with TES [2-2(2-hydroxy-1-bis(hydroxymethyl amine)) ethanesulfonic acid], and pH was adjusted to 7.0. Blood from the index cow was added to the normal erythrocytes in culture medium at a ratio of one part test blood per four parts culture suspension. One-milliliter aliquots were dispensed into wells of 24-well culture plates and incubated at 37 C in an atmosphere of 2% O2 and 5% CO2. Cultures were checked daily for 14 days for parasite growth by examination of Giemsa-stained thin blood smears.
Balb/c mice were inoculated intraperitoneally with blood from the index cow. Giemsa-stained films of tail blood were examined daily for evidence of parasite growth. Mice were monitored for overt clinical signs of illness and hematologic evidence of infection for 21 days.
DNA and phylogenetic analysis of T. buffeli
Whole blood from the theileriosis cow was shipped to the School of Veterinary Medicine, University of California–Davis, for analysis. The Isoquick® Nucleic Acid Extraction Kit (Microprobe, Bothell, WA) was used as recommended by the manufacturer to extract DNA from two 100-μl aliquots of whole blood. The resultant DNA was pooled, and a 1.8-kb nuclear small subunit ribosomal DNA (nss-rDNA) amplification product was isolated using universal polymerase chain reaction (PCR) primers.11
DNA cycle sequencing was performed on the 1.8-kb product using the ABI Prism™ 377 DNA sequencer protocol (Perkin-Elmer, Foster City, CA). The cow isolate was sequenced directly from the PCR product.
The same universal primers were used to obtain nucleotide sequence data from either end of the template. Subsequently, internal primers were designed to allow for sequencing the entire product in both the 5′ to 3′ and 3′ to 5′ directions (primer sequences available upon request). The resulting chromatogram was read and analyzed using Sequencer™ 3.0 (Genecodes Corporation, Ann Arbor, MI).
The Missouri isolate was compared with 12 Theileria and Babesia sequences (Table 1) obtained from GenBank because of their genotypic similarity to the Missouri cow isolate or because they have been previously documented in animals in the United States.2 Two sequences of T. mutans obtained from cattle in Kenya and South Africa were also included in the analysis. Neospora canimum, an apicomplexan parasite also found in cattle in the United States,20 was used as an outgroup.
Isolate name and GenBank accession numbers of organisms compared by DNA analysis.
Sequences were initially aligned using Clustal W.35 Subsequent hand alignment using MacClade 3.019 followed the secondary structure of the 18S small subunit ribosomal RNA (ss-rRNA) molecules for Theileria and Babesia obtained from the ss-rRNA database.39 The final alignment included 1,850 positions, all of which were included in the analysis. Cladistic analysis was performed using PAUP 3.1.34 Transitions and tranversions were weighted equally, and gaps were treated as missing. Both a maximum parsimony approach using a heuristic method with 100 bootstrap replications and a branch-and-bound approach were used to generate phylogenetic trees.
DNA and phylogenetic analysis of Missouri's cervine theilerial agent
Whole blood from the fawn was submitted to School of Veterinary Medicine, University of California–Davis, for parasite identification. The 1.8-kb ss-rDNA from the white-tailed deer was cloned because high background resulted from initial sequencing. Amplicons for the 1.8-kb ss-rDNA were ligated into a plasmid pCR 2.1 vector and transformed into INVαF′ One Shot Escherichia coli, according to the manufacturer's instructions (TA cloning kit, Invitrogen, San Diego, CA). A kit (Quiaprep Spin Miniprep kit, Valencia, CA) was used to isolate cloned DNA. Presence of an insert was verified using an EcoR1 digest and visualization of the 1.8-kb product after agarose gel electrophoresis. Two clones were chosen for sequencing.
Experimental transmission to splenectomized calves
Whole blood from the index cow was diluted with Vega y Murguia solution (VYMS), prepared as reported previously,41 and centrifuged at 1,000 × g for 10 minutes. Supernatant was removed, and the pellet was combined at a 1:1 ratio with a 20% solution of polyvinylpyrolidone as cryoprotectant. One-milliliter aliquots of stabilate were frozen to −70 C prior to storage in liquid nitrogen.
Aliquots were removed from liquid nitrogen, thawed at 37 C, diluted in approximately 20 ml of 1× VYMS, and transferred to a syringe. Two aliquots were used for intravenous injection and one aliquot was used for subcutaneous injection into the first calf. Two aliquots were used for intravenous injection for the second calf. The inoculations occurred at 19 and 43 days of age and 4 and 19 days postsplenectomy for calf Nos. 1 and 2, respectively. The calves were kept in an indoor screened isolation facility after inoculation. The calves were monitored daily for clinical illness, and blood samples were taken frequently to search for a theilerial parasitemia and to measure hematocrits. Sera from postinoculation days 7, 18, 42, 53, and 66 were shipped to the School of Veterinary Medicine, University of California–Davis, for determination of titers to T. buffeli.
Preparation of antigen slides for indirect immunofluo-rescence antibody (IFA) test for antibodies against T. buffeli
Whole blood was collected from the second experimentally infected calf on day 59 postinoculation and shipped with ice packs overnight to California. At that time, 1–2% of the calf's erythrocytes contained intraerythrocytic piroplasms.
The IFA antigen slides were prepared as previously described for B. gibsoni 45 except that whole blood from experimentally infected calf No. 2 was used as the antigen source and rabbit anti-bovine fluorescein isothiocyanate conjugate (Jackson ImmunoResearch Laboratories, West Grove, PA) was used at a 1:300 dilution. Serum samples were diluted in serial two-fold dilutions from 1:80 to 1:10,240.
Results
Animals
Index case
Initial blood film examination revealed anemia, thrombocytosis, and leukocytosis. Nearly 25% of erythrocytes on Wright-Giemsa-stained blood films contained Theileria piroplasms, including cocci, rod, stick, comma, fusiform, racquet-shaped, signet-ring, and pear-shaped forms (Figs. 1, 3–8). Other erythrocytic abnormalities included spherocytosis (Figs. 1, 2), acanthocytosis (Fig. 2), basophilic stippling (Figs. 1, 8), Howell-Jolly bodies (Fig. 4), macrocytosis, and occasional schizocytes and keratocytes. There was a lymphocytosis with reactive lymphocytes. Structural defects were not detected in platelets.
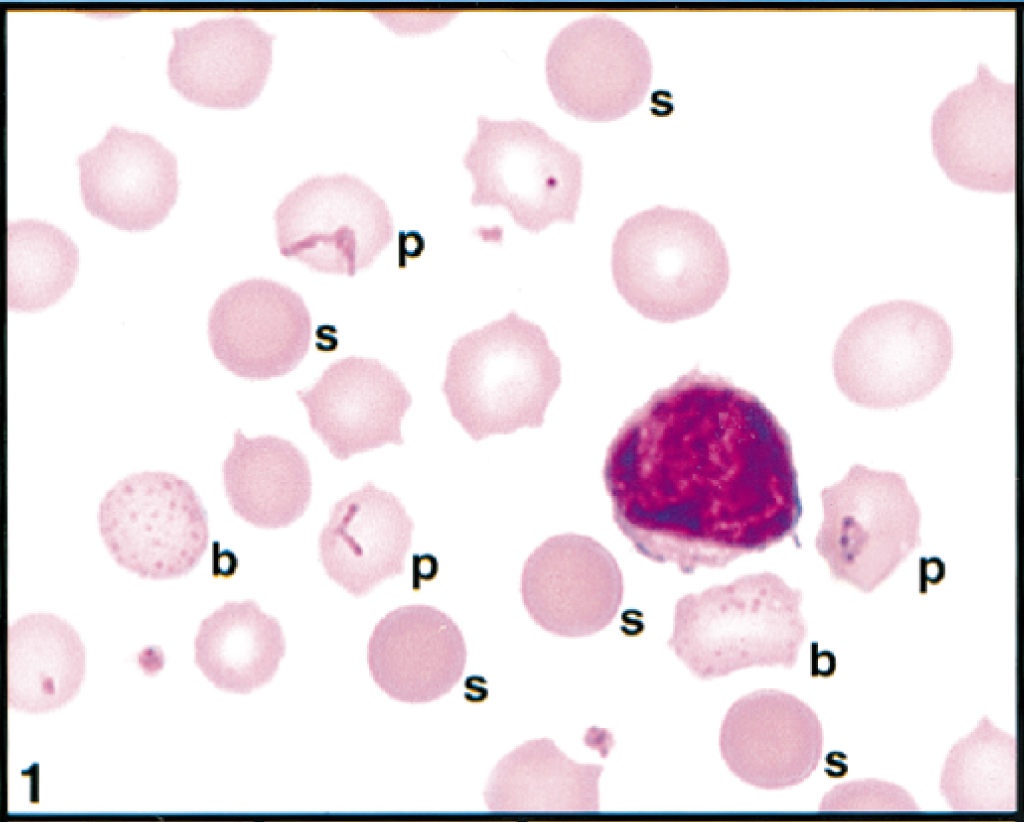
Erythrocytes, lymphocyte, and platelets in peripheral blood; cow. Three erythrocytes (p) contain rod and racquet piroplasms. Spherocytes (s) and erythrocytes with basophilic stippling (b) are present. Wright's-Giemsa stain. Same magnification as Fig. 2.
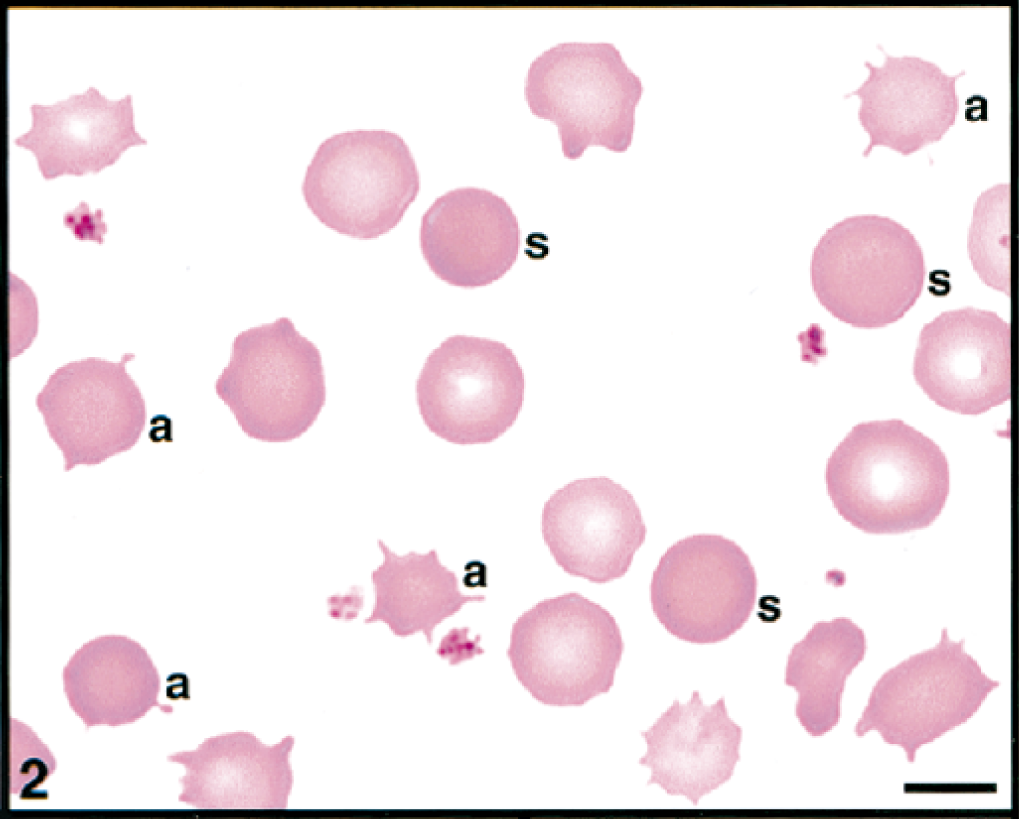
Erythrocytes and platelets in peripheral blood; cow. Spherocytes (s) and acanthocytes (a) are present. Wright's-Giemsa stain. Bar = 5 μm.

Erythrocyte in peripheral blood; cow. Erythrocyte contains a cluster of theilerial cocci. Wright's-Giemsa stain. Same magnification as Fig. 8.
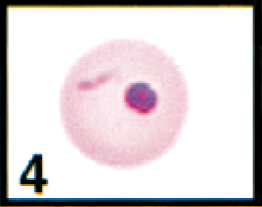
Erythrocyte in peripheral blood; cow. Erythrocyte contains a theilerial rod and a Howell-Jolly body. Wright's-Giemsa stain. Same magnification as Fig. 8.
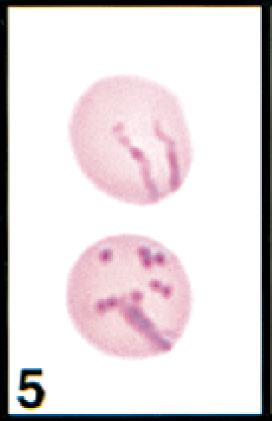
Erythrocytes in peripheral blood; cow. Erythrocytes contain the common linear or comma-shaped piroplasms (upper cell) and many coccoid forms (lower cell). Wright's-Giemsa stain. Same magnification as Fig. 8.
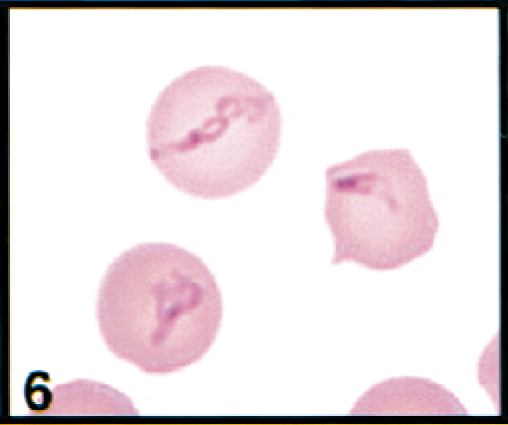
Erythrocytes in peripheral blood; cow. Erythrocytes contain larger, pleomorphic piroplasms. Wright's-Giemsa stain. Same magnification as Fig. 8.
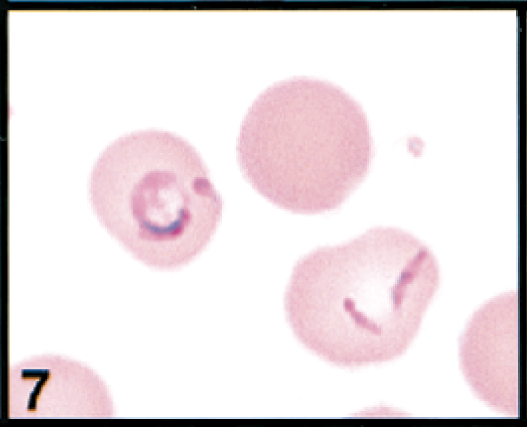
Erythrocytes in peripheral blood; cow. Erythrocytes contain a large signet-ring piroplasm (left) and small comma-shaped piroplasms (right); spherocyte also present (upper right). Wright's-Giemsa stain. Same magnification as Fig. 8.
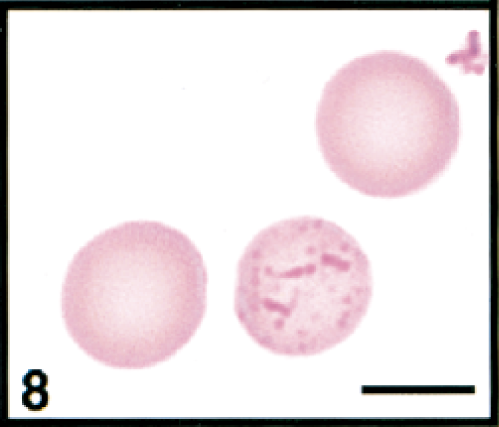
Erythrocytes in peripheral blood; cow. Three macrocytes are present; one contains small comma-shaped piroplasms and basophilic stippling. Wright's-Giemsa stain. Bar = 5 μm.
Complete blood cell count (CBC) results for a sample collected on day 6 included a macrocytic, normochromic, regenerative anemia, presence of many Theileria piroplasms, neutrophilia with mild left shift, lymphocytosis, monocytosis, and hypoproteinemia (Table 2). Abnormalities in erythrocyte structure were still present. Schizonts were not found in blood lymphocytes. Retrospectively, day 6 serum had a titer of 1:80 against the characterized T. buffeli using the IFA procedure developed during this study.
CBC results from a Missouri cow with bovine theileriosis (samples from days 6 and 8 of clinical evaluation).
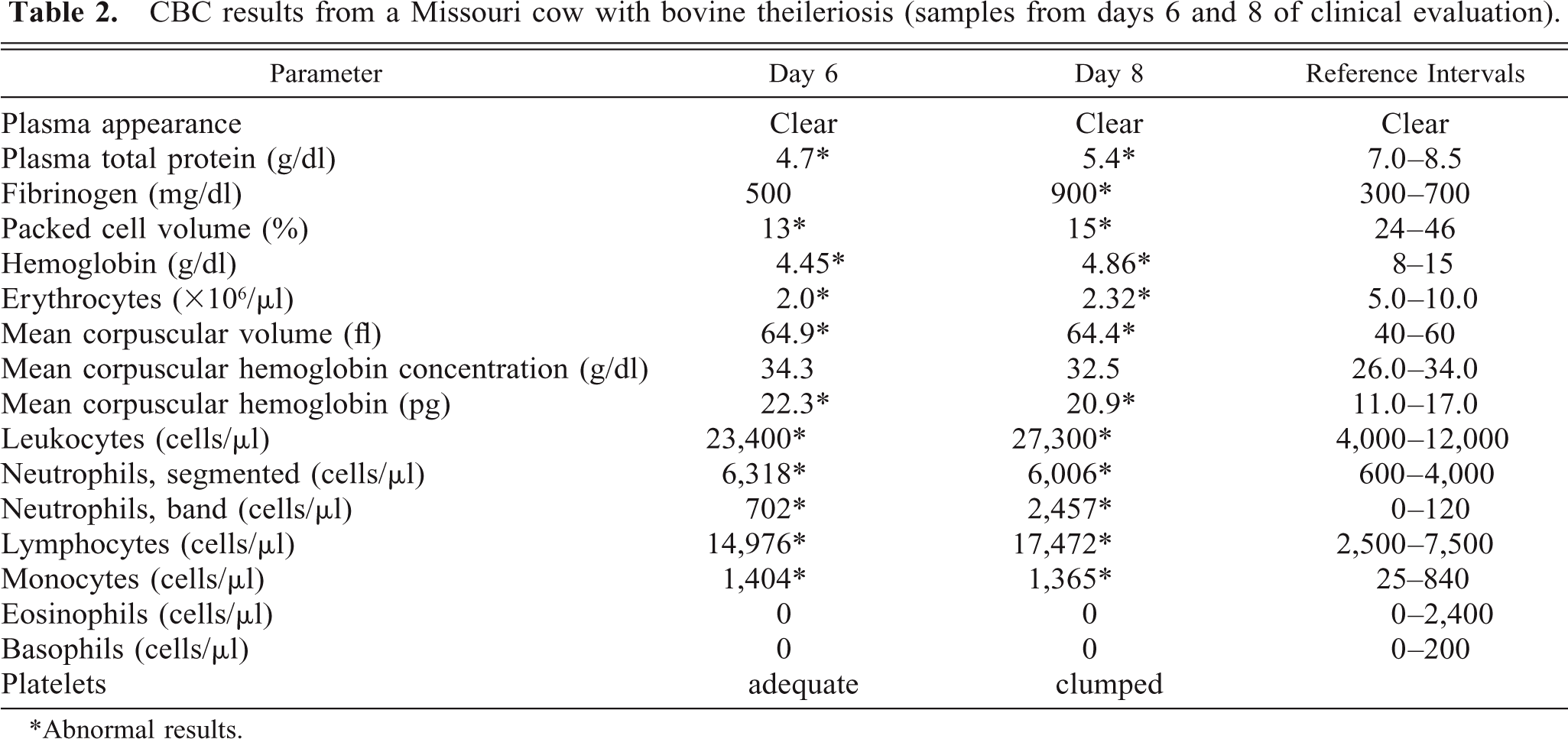
∗ Abnormal results.
In samples collected on day 8, intraerythrocytic piroplasms were present in 16% of erythrocytes, and CBC results revealed the persistence of anemia and an enhanced inflammatory response (Table 2). Abnormalities in erythrocyte structure were still present, and the degree of spherocytosis was increased. Serum chemistry assays revealed a hypoproteinemia (4.3 g/dl, reference interval [RI] = 5.8–7.5 g/dl), hypoalbuminemia (1.7 g/dl, RI = 2.4–3.5 g/dl), hypocalcemia (6.9 g/dl, RI = 8.2–10.0 g/dl), hypomagnesemia (1.4 mg/dl, RI = 2.0–2.8 mg/dl), hyperbilirubinemia (0.8 mg/dl, RI = 0.1–0.6 mg/dl), increased serum aspartate transaminase (AST), γ-glutamyl transferase (GGT), and creatine kinase (CK) activities (729 IU/L, RI = 58–100 IU/L; 225 IU/L, RI = 22–64 IU/L; 12,641 IU/L, RI = 56–1,236 IU/L; respectively). Concentrations of glucose, urea, creatinine, sodium, potassium, chloride, globulins, and inorganic phosphates and total carbon dioxide content were within reference intervals.
Bovine leukemia virus antigen was detected (enzyme-linked immunosorbent assay) and complement fixation antibodies against B. bigemina and B. bovis were not found in day 8 samples. Antibodies against A. marginale were not detected by a latex agglutination assay in a day 1 sample, but a weakly positive (1:5) complement fixation test for A. marginale was obtained for the day 8 sample. Marginal bodies of A. marginale were not found on any of the cow's blood films.
Necropsy findings included a thin cow in early pregnancy, edematous lymphadenopathy, splenic hemosiderosis, acute pneumonia, subcutaneous edema, and thoracic and peritoneal effusions. An engorged Amblyomma americanum (lone star tick) was found on the cow. Theilerial schizonts were not found in histologic sections or cytologic imprints of enlarged lymph nodes; however, postmortem autolysis interfered with analysis. There was no evidence of lymphosarcoma.
A diagnosis of bovine theileriosis was based on finding many theilerial intraerythrocytic piroplasms, macrocytic anemia, and lymph node hyperplasia.
Microscopic characteristics of bovine theilerial agent and infected erythrocytes
The pleomorphism of the theilerial agent as seen via light microscopy was also evident via transmission electron microscopy. Infected erythrocytes contained piroplasms and veils (Figs. 14, 15).
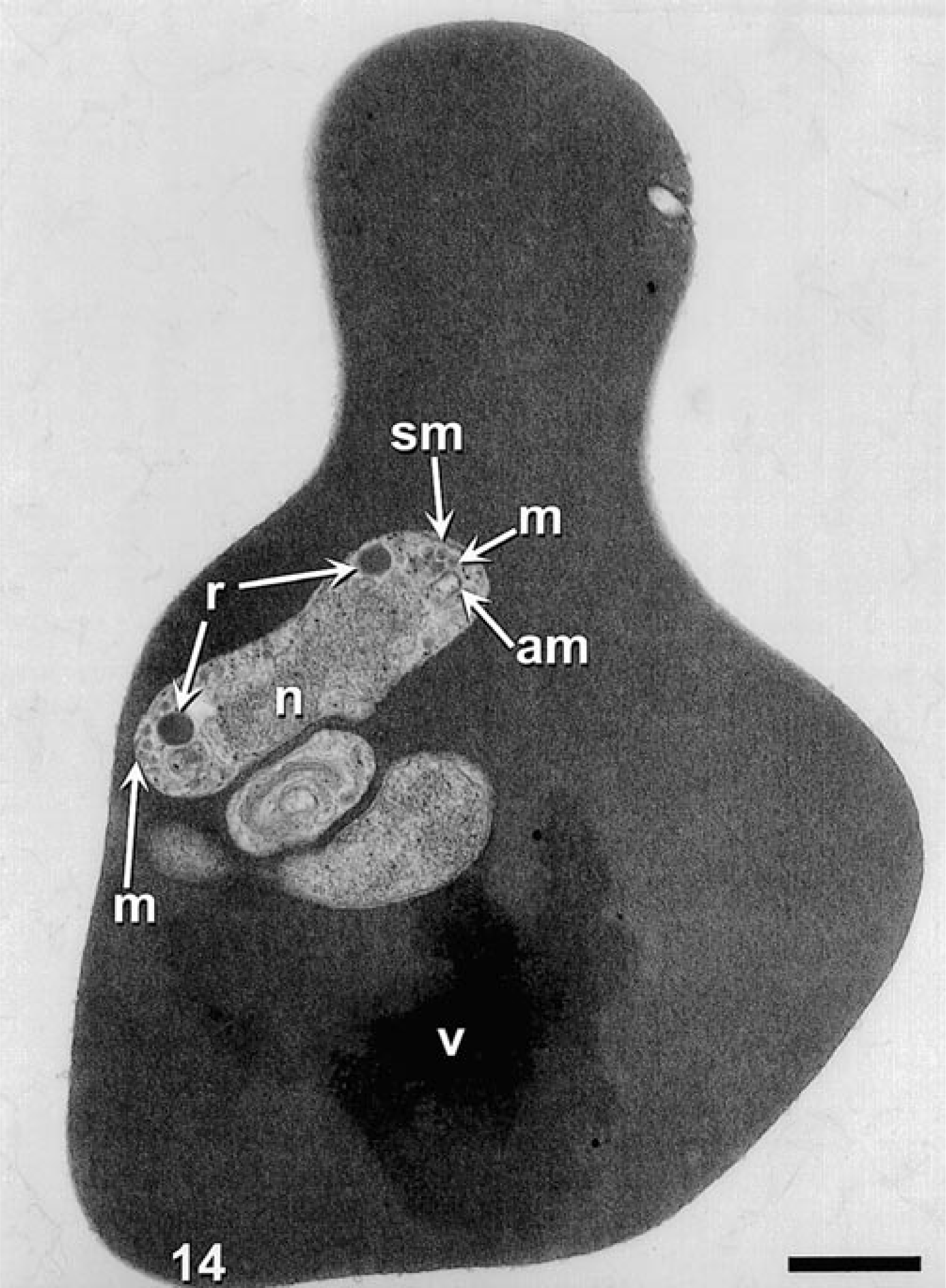
Transmission electron microscopy. Theilerial infected erythrocyte, cow. Erythrocyte contains a theilerial piroplasm with acristate mitochondria (am), micronemes (m), nucleus (n), rhoptries (r), subplasmalemal membrane (sm), and veil (v). Uranyl acetate and lead citrate. Bar = 500 nm.
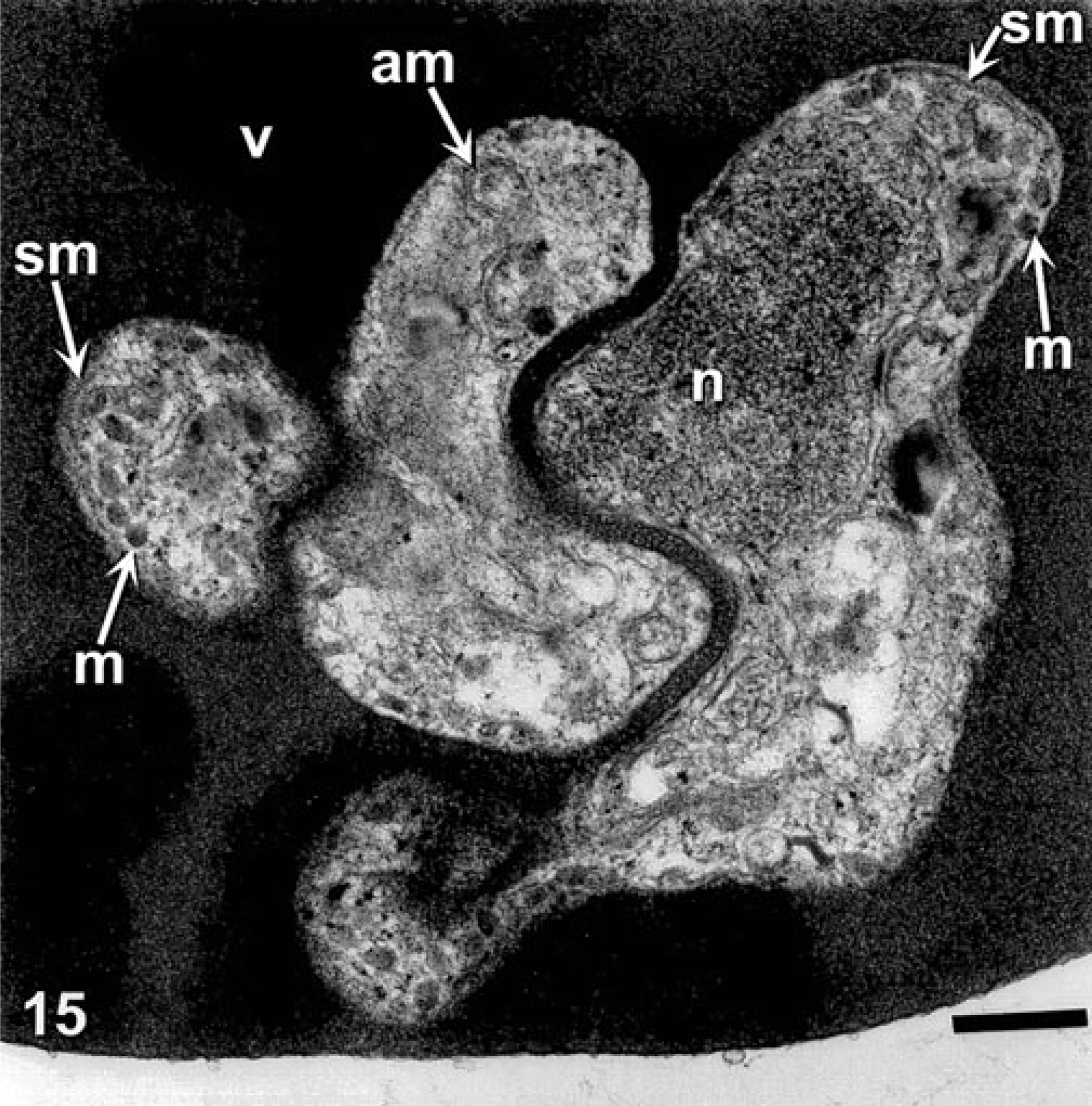
Transmission electron micrograph. Theilerial infected erythrocyte, cow. Erythrocyte contains a theilerial piroplasm with acristate mitochondria (am), micronemes (m), nucleus (n), subplasmalemal membrane sm), and veil (v). Uranyl acetate and lead citrate. Bar = 200 mm.
Attempts to culture Theileria sp
There was no in vitro propagation of organisms in bovine erythrocyte cultures as monitored by Giemsa-stained smears during a 14-day test period. Inoculated mice also showed no evidence of infection either by signs of clinical illness or hemoparasitemia.
Phylogenetic analysis of bovine agent
The relationship of the bovine isolate to other parasitic protozoa is shown by the maximum parsimony approach with bootstrap support in Fig. 16. The branch-and-bound method (results not shown) produced essentially the same relationship.
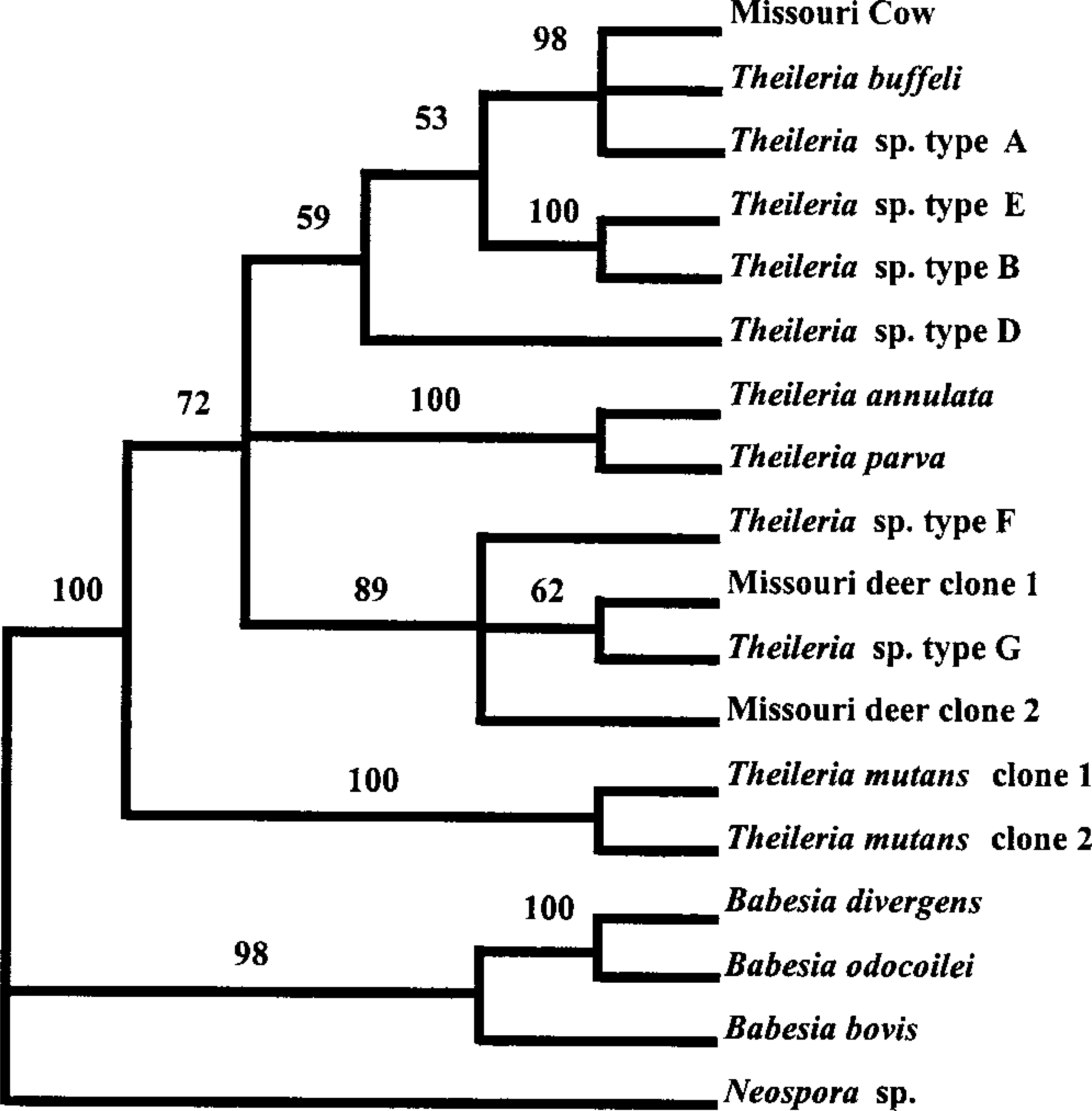
Dendogram showing evolutionary relationships of 18 Apicomplexan protozoal parasites. Branches are not drawn in proportion to number of substitutions inferred to have taken place. Numbers above the branches represent percentages of 100 maximum-parsimony bootstrap replications in which the current groupings were obtaned. Bootstrap values >70% are considered good support for the grouping. In this analysis, values <70% are a result of minor pattern changes (branch swapping) in the terminal branches and do not affect the major conclusions.
Theileria species, including the Missouri cow isolate, form a monophyletic group separate from Babesia. Thus, the Missouri cow isolate is not related to the potentially zoonotic Babesia sp. found in a Missouri man in 1992. The cow isolate is nearly identical to T. buffeli and Theileria sp. type A isolates. There is good statistical evidence to place T. annulata and T. parva (the African theilerial species) in the same clade as the North American theilerial species. Two clones of T. mutans are nearly identical to each other and are placed separately from the other theilerial species.
Experimental transmission to splenectomized calves
Theilerial piroplasms were found in the peripheral blood erythrocytes from days 39 to 66 postinoculation in calf No. 1 and from days 35 to 67 postinoculation in calf No. 2 (Fig. 9). Using the developed IFA test for antibodies against T. buffeli, titers were first detected in day 18 samples, and the serologic responses increased during the study (Fig. 17). Calf Nos. 1 and 2 were euthanized on days 66 and 67, respectively.
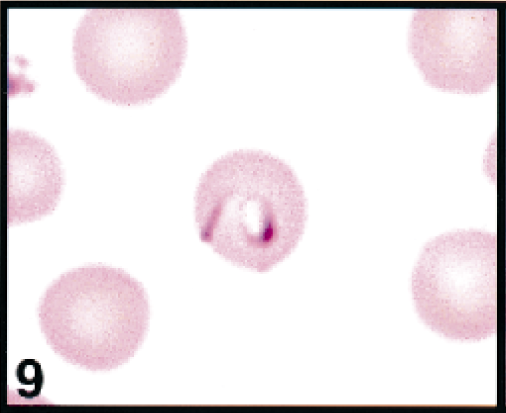
Erythrocytes in peripheral blood; calf. No. 2. Erythrocyte contains a large signet-ring and comma-shaped piroplasms. Wright's-Giemsa stain. Same magnification as Fig. 13.
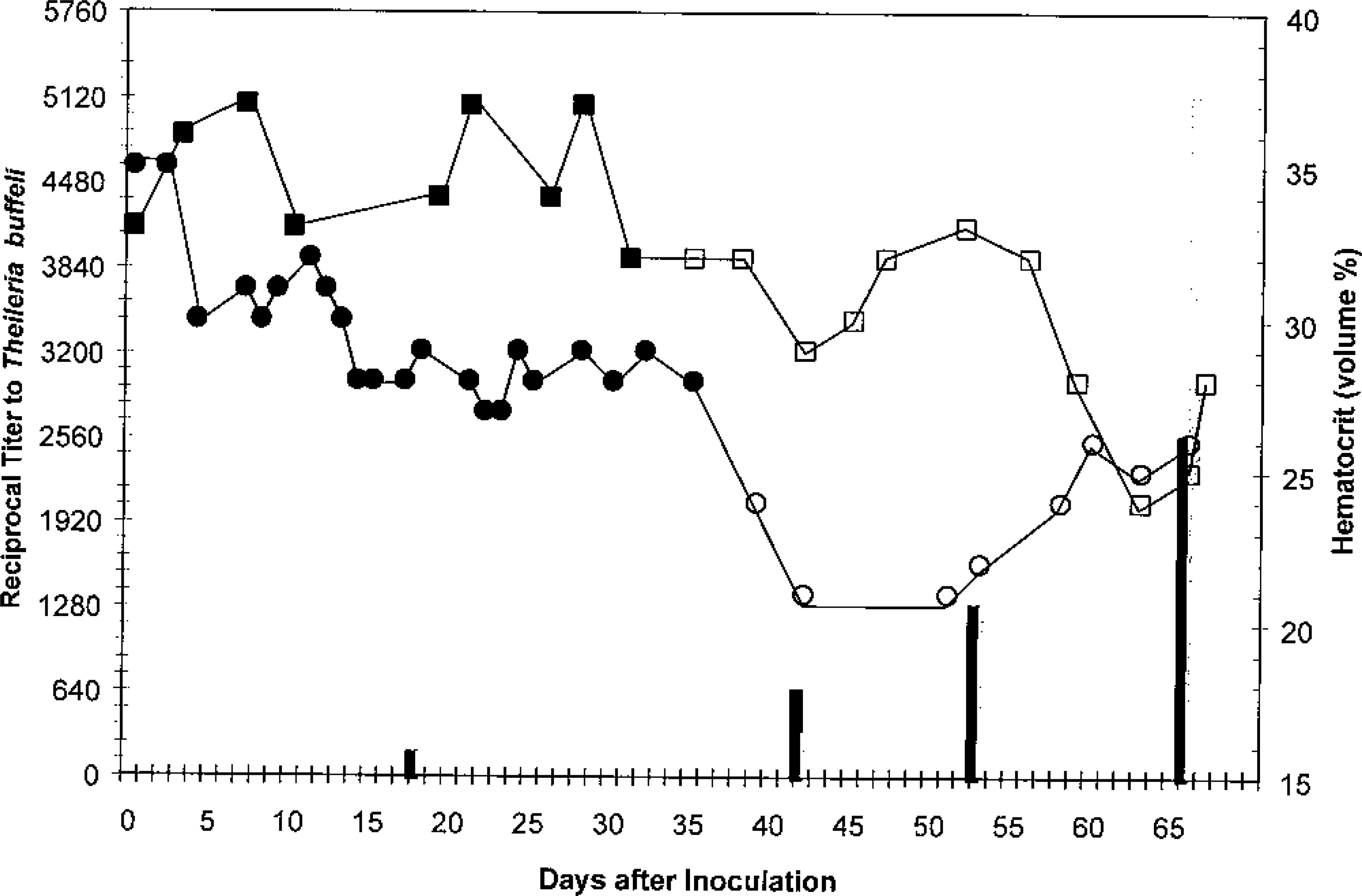
Reciprocal titers (left axis) and hematocrit values (right axis) in splenectomized calves inoculated with Theileria buffeli obtained from the index case. ▪ = PCV, calf No. 1, piroplasms not detected; □ = PCV, calf No. 1, piroplasms present; • = PCV, calf No. 2, piroplasms not detected; ○ = PCV, calf No. 2, piroplasms present; ▪ = titer to T. buffeli in calf No. 1; ▒ = titer to T. buffeli in calf No. 2.
Calf No. 1 developed a mild respiratory infection (viral or bacterial) soon after inoculation that progressed slowly in severity; ceftiofur sodium was administered on days 25–30, after which there was marked clinical improvement. Marginal bodies of A. marginale were found on days 30 and 32. Eperythrozoon cocci were so numerous on days 32 and 35 that they interfered with evaluation of erythrocytes for piroplasms. Marginal bodies and Eperythrozoon cocci were not seen during the remainder of the evaluation. Calf No. 1 developed a mild anemia (day 39) concurrent with the first detection of a theilerial parasitemia (Fig. 17).
Shortly after the first detection of theilerial parasitemia (day 35), calf No. 2 developed a slight anemia, which progressed in severity (Fig. 17). Calf No. 2 remained clinically healthy during the study, and parasites other than the theilerial piroplasms were not found except for rare marginal bodies of A. marginale in the day 35 sample. A peak theilerial parasitemia of 1–2% infected erythrocytes was found on day 56 postinoculation; blood from day 59 was used to produce antigen slides for the T. buffeli IFA test.
Herd investigation
Twenty-two of 75 (29%) cattle had intraerythrocytic piroplasms but infected cells were rare (<0.1% infected). One parasitemic cow had moderate lymphadenomegaly; theilerial schizonts were not found in several smears of a lymph node aspirate. In 22 parasitemic cows, the mean packed cell volume (PCV) was 33% (range, 26–39%); in 16 cows in which intraerythrocytic piroplasms were not found, the mean PCV was 32% (range, 27–39%). Dermacentor variabilis (American dog tick) and A. americanum were found on several cattle while blood samples were being collected.
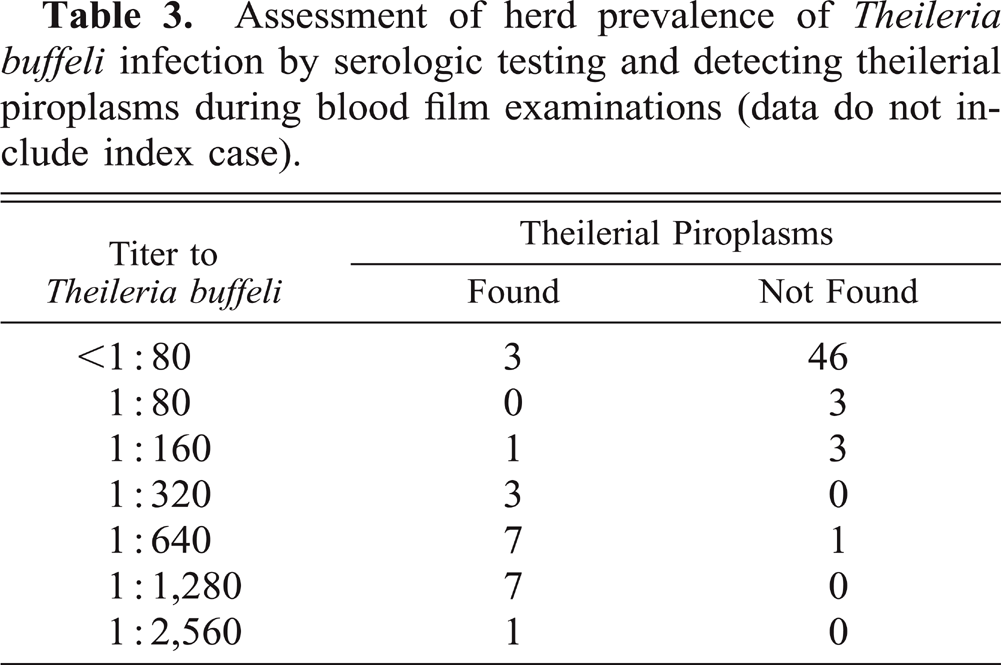
Serologic titers against T. buffeli and prevalence of theilerial parasitemia are reported in Table 3. A kappa value24 of 0.78, implying a substantial agreement beyond chance between microscopic detection of parasitemia and the IFA titer determinations, was derived using 1:160 as a positive cutoff. Of the 22 parasitemic cattle, 19 had titers ≥ 1:160. Conversely, of 53 cattle in which piroplasms were not detected, only four had titers ≥ 1:160.
Assessment of herd prevalence of Theileria buffeli infection by serologic testing and detecting theilerial piroplasms during blood film examinations (data do not include index case).
Fifty-nine of 71 (83%) cattle had positive titers for bovine leukemia virus (BLV) and six of 46 (13%) had a weak titer of A. marginale (four at 1:5, one at 1:10, one at 1:20). Of six cattle with positive titers to A. marginale, three had intraerythrocytic theilerial piroplasms. Of 59 cattle that had positive BLV titers, 20 had intraerythrocytic theilerial piroplasms. Of 12 cattle that had negative BLV titers, two had intraerythrocytic theilerial piroplasms.
Evaluation of white-tailed deer blood and DNA sequences
During the fawn's illness, it had a PCV of 45% and had numerous intraerythrocytic piroplasms (Figs. 10–13). The theilerial agent was not considered a contributing pathogen to the fawn's illness. The deer isolates were more closely related to Theileria sp. types F and G than they were to the cow isolate (Fig. 16).
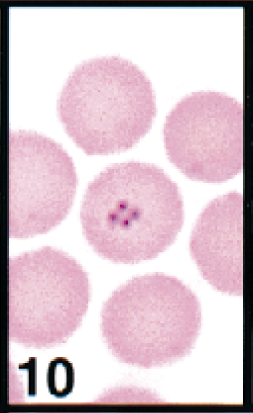
Erythrocytes in peripheral blood; white-tailed deer. Erythrocyte contains a cluster of four theilerial merozoites in a quadruplet or Maltese-cross formation. Wright's-Giemsa stain. Same magnification as Fig. 13.
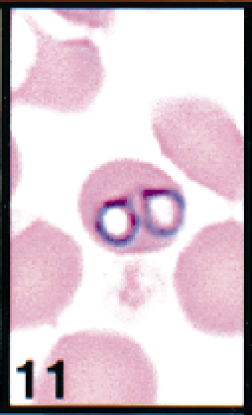
Erythrocytes in peripheral blood; white-tailed deer. Erythrocyte contains two large theilerial trophozoites. Wright's-Giemsa stain. Same magnification as Fig. 13.
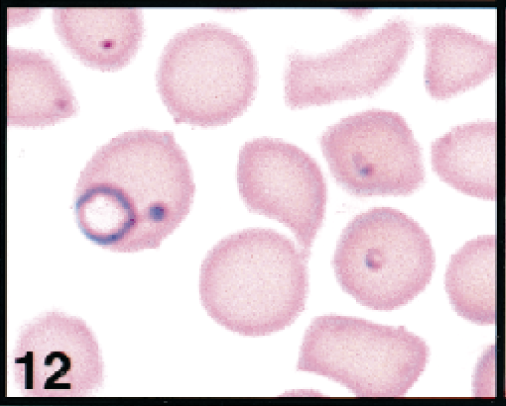
Erythrocytes in peripheral blood; white-tailed deer. Erythrocytes contains large ring, small ring, and safety-pin forms of theilerial piroplasms. Wright's-Giemsa stain. Same magnification as Fig. 13.
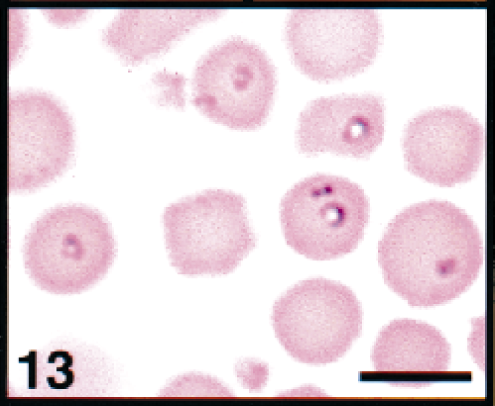
Erythrocytes in peripheral blood; white-tailed deer. Erythrocytes contains several ring-forms of theilerial piroplasms. Wright's-Giemsa stain. Bar = 5 μm.
Discussion
Theileriosis was considered the cause of the cow's clinical illness, macrocytic normochromic anemia, and lymphoid hyperplasia. Macrocytosis, polychromasia, and basophilic stippling indicated a regenerative response consistent with a hemolytic anemia. Splenic hemosiderosis and the mild hyperbilirubinemia supported the hemolytic concept. A combination of spherocytosis, acanthocytosis, schizocytosis, and keratocytosis indicated intravascular erythrocyte fragmentation, as can occur with vasculitis or intravascular thrombi. Spherocytes can also be formed by immune-mediated processes. Undoubtedly, the inflammatory disease also contributed to the anemia. There was no evidence of blood loss, but there was a hypoproteinemia.
The pathogenesis of anemia is not clearly established in theileriosis and may be multifaceted. Invasion of erythroid precursors by the merozoites of T. parva is associated with erythroid hypoplasia and severe anemia.22 Erythrocyte survival studies of calves infected with T. sergenti showed that both infected and uninfected erythrocytes have reduced survival.44 Studies with T. annulata in calves suggested an immune-mediated hemolysis as indicated by the presence of a hemagglutinin.12 However, T. sergenti infections in severely combined immunodeficient mice indicate that the erythrocyte destruction can occur without immunoglobulin or complement involvement.8 The finding that sera from cattle infected with T. sergenti and merozoite extracts both cause hemolysis led to the proposal that proteases are involved in erythrocyte destruction.8 Proteases are involved in the invasion of erythrocytes by malarial and theilerial organisms.33 Oxygen radicals may also be involved in the pathogenesis of the anemia.3
Spherocytosis and acanthocytosis indicated that at least part of the cow's anemia was due to erythrocyte fragmentation. Acanthocytes, keratocytes, and spiculated and spheroid cells have been reported in cattle infected with T. sergenti.10,43 Disseminated intravascular coagulation (DIC) can occur in theilerial and babesial infections,5,21 but it is unlikely that DIC was present in this index case because thrombocytopenia, hypofibrinogenemia, and microthrombi were not found.
Lymphoid hyperplasia was found in the index case and is consistent with bovine theileriosis. Intralymphocytic theilerial parasites transform host cells and lead to clonal growth of lymphocytes.26 The process by which this transformation occurs is not known but is associated with a growth factor produced by infected lymphocytes.7 Lymphocytic schizonts were not found in the index case; however, schizonts are considered uncommon in cows infected with T. buffeli.30
Serum chemistry abnormalities seen in the index case are also consistent with bovine theileriosis. Hypoproteinemia and hypoalbuminemia could be due to the extravascular accumlation of proteinaceous fluids resulting from diseased lymph nodes22 and thus explain the edema and body cavity effusions. Hypocalcemia and hypomagnesemia probably were due to the hypoproteinemia or decreased dietary intake. Increased serum GGT and AST activities, hypoproteinemia, and mild hyperbilirubinemia could indicate liver disease, as seen with some theilerial infections,42 but postmortem liver lesions were not found. At least part of the hyperbilirubinemia was caused by hemolysis or anorexia. Increased serum AST and CK activities reflected muscle trauma caused by prolonged recumbency.
Prior to molecular characterization, taxonomic classification of Theileria spp. was based on microscopic appearance of intraerythrocytic piroplasms, geographic location of the infected animal, apparent pathogenicity of the organism, and serologic testing.13,37 Most theilerial organisms of cattle that had low pathogenicity were called T. mutans.30 As more Theileria spp. were isolated and studied, some organisms were reclassified. For example, the US theilerial organism reported by Kuttler in 1975 to be T. mutans was renamed T. orientalis in 1985.38 These authors also considered some isolates of T. orientalis to be the same as some stocks of T. sergenti, the name given to a more pathogenic species found in southeastern Asia.
In the index case, intraerythrocytic piroplasms were highly pleomorphic, but rod forms were most common. Piroplasm shape may vary with stage of an infection and thus is not a reliable criterion for species differentiation.37,46 Veils were not seen on Wright-Giemsa-stained blood films, but veils were seen during electron microscopy. Electron-dense veils have been reported in erythrocytes infected with T. velifera, T. sergenti, T. buffeli, and T. orientalis.32 The veil contains hemoglobin40 and other proteins that appear to be of parasite origin.32
The Missouri cow and deer isolates are clearly part of the Theileria spp. complex that has been documented worldwide. The extreme genetic similarity of the cow isolate's 18S ss-rRNA gene to that of T. buffeli (99.88% or 2-bp differences) as well as its clinical and morphologic manifestations strongly support the characterization of this isolate as T. buffeli. This and other studies2 have demonstrated that T. buffeli is a clinical entity in North American cattle.
The role of wildlife as a reservoir for T. buffeli is not clear from this analysis. Our study showed that the isolates obtained from the Missouri white-tailed deer are more closely related to isolates previously found in deer and elk in the United States and Canada than to T. buffeli. Therefore, deer do not appear to be the source of infection for this herd. DNA sequences were not available for T. cervi, a commonly reported theilerial species in deer from the southern and southeastern areas of the United States.6,15 Thus, we cannot comment on the relationship of T. cervi to this bovine Theileria isolate. The North American cervid organism was classified as T. cervi in 1962,25 but failure of transmission to fallow deer16 indicated that the organism is not the same as the T. cervi originally reported in Europe.1 Bovine and cervid isolates from Missouri animals were grouped within the Theileria clade and thus are not related to the zoonotic Babesia divergens-type piroplasm found in a human patient from Missouri.9
T. buffeli was endemic in the cattle herd, as documented by the prevalence of parasitemia (29%) and the detection of T. buffeli antibodies (35%). The prevalence of this agent in other Missouri cattle is not known. The referring veterinarian had not seen similar cases in her practice area, and questioning of neighbors by an MDA veterinarian did not reveal suspect cases. However, the disease may be difficult to differentiate clinically from anaplasmosis without blood film examinations. The reason one cow developed a clinical illness while the other infected cattle in the herd remained clinically healthy is unknown. A combination of predisposing factors could have influenced the course of this disease; prednisolone and pregnancy have been associated with enhanced T. sergenti parasitemia and associated illness.14,28 The fact that the cow had a relatively low titer (1:80) against T. buffeli suggests either recent infection or a poor immunologic response.
Experimentally infected splenectomized calves did develop mild to moderate anemia. Interpretation of data from calf No. 1 is difficult because of the respiratory disease that was present during the evaluation period and the appearance of Eperythrozoon sp. and A. marginale shortly before detection of the theilerial piroplasms. Calf No. 2 remained clinically healthy, and A. marginale was seen only on 1 day during the study, suggesting its anemia may have been due to theileriosis. It was assumed that the inoculate contained all three hemoparasites, but the calves could have been infected with the nontheilerial parasites previously.
In summary, a cow infected with T. buffeli had a clinical disease characterized by theilerial parasitemia, macrocytic normochromic anemia with spherocytosis, acanthocytosis, and other erythrocyte abnormalities, lymphoid hyperplasia, dysproteinemia, evidence of liver disease, and a low titer against T. buffeli. The anemia's pathogenesis was not determined, but evidence suggested acanthocytic fragmentation hemolysis. The cow was in a herd in which all cattle had been raised in Missouri, and the T. buffeli was considered endemic based on serologic and microscopic blood examination results.
Footnotes
Acknowledgements
We acknowledge the cooperation and patience of the herd's owner during our investigations. We also thank Dr. John Hunt and his staff, MDA, and Dr. Glen Snider, US Department of Agriculture, for their cooperation and dedication during the investigation of the infected herd and for providing serologic testing for A. marginale and BLA. We thank Howard Wilson for assistance and guidance in creating photographic images, Preston Stogsdill for electron microscopy assistance, Erdal Erol and Craig Bailey for attempts to culture, isolate, and cryopreserve the theilerial agent, and the technical staff of the Clinical Pathology Laboratory for the analysis of blood samples. The work was supported in part by a grant from the Committee on Research, College of Veterinary Medicine, University of Missouri–Columbia.
