Abstract
Epizootic hemorrhagic disease viruses (EHDVs) are orbiviruses transmitted by Culicoides biting midges to domestic and wild ruminants. EHDV-1 and EHDV-2 are endemic in the United States, where epizootic hemorrhagic disease is the most significant viral disease of white-tailed deer (WTD; Odocoileus virginianus) and reports of epizootic hemorrhagic disease in cattle are increasing. In 2006, a reassortant EHDV-6 was isolated from dead WTD in Indiana and has been detected each subsequent year over a wide geographic region. Since EHDV-6 is not a historically endemic serotype in the United States, it is important to understand infection outcome in potential hosts. Specifically, we aimed to evaluate the pathogenicity of the virus in 2 primary US ruminant hosts (WTD and cattle) and the susceptibility of a confirmed US vector (Culicoides sonorensis). Five WTD and 4 cattle were inoculated with >106 TCID50 EHDV-6 by intradermal and subcutaneous injection. All 5 WTD exhibited moderate to severe disease, and 3 died. Viremia was first detected 3 to 5 days postinfection (dpi) with surviving animals seroconverting by 10 dpi. Two of 4 inoculated cattle had detectable viremia, 5 to 10 dpi and 7 to 24 dpi, respectively. No clinical, hematologic, or pathologic abnormalities were observed. Antibodies were detected by 10 dpi in 3 of 4 cows. C. sonorensis were fed on WTD blood spiked with EHDV-6 and held for 4 to 14 days postfeeding at 25°C. From 4 to 14 days postfeeding, 19 of 171 midges were virus isolation positive and 6 of 171 had ≥102.7 TCID50 EHDV-6. Although outcomes varied, these studies demonstrate the susceptibility of ruminant and vector hosts in the United States for this recently emerged EHDV serotype.
Epizootic hemorrhagic disease viruses (EHDVs) are dsRNA viruses (genus Orbivirus, family Reoviridae) that are transmitted to susceptible ruminant hosts by certain species of Culicoides biting midges (Diptera: Ceratopogonidae). The EHDV serogroup consists of 7 provisional serotypes worldwide 5 that are generally considered to exist in temperate and tropical climates that support vector populations. Entomologic investigations of EHDV transmission are rare, and the studies needed to confirm an arthropod as a vector are lacking in most regions of the world. In the United States (US), the only confirmed vector of EHDV is Culicoides sonorensis 18,34 ; however, several studies suggest that other Culicoides spp are likely involved in the epidemiology of epizootic hemorrhagic disease (EHD) in certain regions. 22,45,62,64
Although EHDVs are distributed throughout much of the world and infect a variety of wild and domestic ruminants, EHD has been considered by some to be primarily a disease of wild ruminants in North America, where EHDV-1 and EHDV-2 have long been associated with outbreaks in white-tailed deer (WTD; Odocoileus virginianus). Although EHD can occur in several other wild ruminant species, WTD are arguably the most susceptible species in the world, and periodic outbreaks have occurred for decades in this species in the US during the late summer and early autumn. 29 Very little is known about EHDV in wild ruminants outside of North America, and while EHDV antibodies have been detected in numerous species, the only report of clinical disease has been from a captive pygmy brocket deer (Mazama nana) from South America. 16 Among domestic ruminants, cattle are the most important host for EHDV. Numerous sentinel animal studies and serologic surveys from many parts of the world suggest that infection of cattle with EHDV is common and widespread, 1,6,19,25,42,44,47,60 although global reports of EHD in cattle are historically rare. 7,27,43 The exception is EHDV-2 (strain Ibaraki), a distinct topotype that has caused periodic outbreaks of EHD in cattle in Japan and Korea. 38,48 However, in recent years, there have been numerous outbreaks of EHD in cattle in certain parts of the world 13,53,58,69,74 and associated production loss, 36 causing many to reevaluate EHDV as a pathogen of cattle, as well as question the role of cattle in the epidemiology of EHD.
In North America, EHDV-1 and EHDV-2 have caused cyclical epidemics among WTD populations for >60 years. 46,61,66 However, an exotic serotype, EHDV-6 (Indiana), was isolated from dead WTD from Indiana and Illinois in 2006, 2 and this virus has now been isolated from dead WTD each subsequent year over a wide geographic area in the central and eastern US. 3,4,53,65 This virus represents a novel EHDV reassortant of serotypes 2 and 6, with the L2 (VP2) and M5 (VP5) gene segments originating from EHDV-6 and the remaining gene segments originating from EHDV-2 (Alberta). 2 Comparative analysis of nucleotide sequences of the L2 and M5 genes from EHDV-6 (Indiana) with those of strains from Australia, South Africa, and Bahrain (strain 318) indicate that the gene segments in the US isolates are most closely related to the Australian strain. 3 Interestingly, EHDV-6 was also detected in the Caribbean in 2010, when it was isolated from a sentinel cow on the island of Guadeloupe. However, genetic analysis of the isolate indicates that it is the nonreassortant Australian strain of EHDV-6, which has not been detected in the US, so it is not the immediate parental strain of EHDV-6 (Indiana). 3 In 2011, EHDV-6 RNA was also detected in bovine blood from the South American French district of Guiana, suggesting that this serotype circulates in South America. 72 Partial nucleotide sequence analysis of segment 2 revealed 97% identity to the EHDV-6 strains from Australia, Guadeloupe, and the US. Unfortunately, other segments were not analyzed, so it is not known if this virus represents a reassortant.
Since EHDV-6 is not a historically endemic virus, there is a need to better understand the outcome of infection in potential hosts, as well as the significance of this novel virus to WTD populations and domestic livestock. Here we describe 3 separate experiments aimed to better understand the potential ruminant and vector host range of EHDV-6 in the US. Our specific objectives were to (1) fulfill Koch’s postulates for EHDV-6 in WTD, (2) determine whether domestic cattle are susceptible to infection with EHDV-6, and (3) determine if C. sonorensis are susceptible to infection with EHDV-6 after oral feeding.
Materials and Methods
Animals and Culicoides
Seven hand-raised WTD fawns were obtained from the University of Georgia’s Whitehall Deer Research Facility (Whitehall Experimental Forest, Clarke County, Georgia, US). Six fawns were used for the experimental infection of WTD, and 1 served as a positive control during the cattle trial. At the time of infection, the fawns were approximately 5 months old, and 4 were female (deer Nos. 1, 3, 4, 6) and 3 were male (deer Nos. 2, 5, 7). Four Holstein cattle (1 yearling male and 4 mature cows) were obtained from a dairy farm in Oglethorpe County, Georgia, US. All WTD and cattle tested negative for antibodies against EHDV and bluetongue virus (BTV) with agar gel immunodiffusion (AGID) test kits (Veterinary Diagnostic Technology Inc, Wheatridge, CO, US) and a serum neutralization test as previously described. 67 Positive and negative control animals were randomly assigned. All WTD were group housed except for the negative control animal for the WTD trial (deer No. 6) and the positive control animal for the cattle trial (deer No. 7). All cattle were housed in individual stalls. All animals went through a minimum 7-day acclimation period prior to inoculation. The research team was not blinded to infection status.
For the midge infection, laboratory-reared C. sonorensis free of BTV and EHDV were obtained from each of the 3 colonies maintained at the Arthropod-Borne Animal Diseases Research Unit (Agricultural Research Service, US Department of Agriculture, Manhattan, KS, US). Pooled 1-day-old male and female midges were shipped overnight to the University of Georgia, where they were kept in an insectary at 22°C ± 1°C on a light:dark cycle (12:12 hours) and provided 10% sucrose ad libitum until used.
Virus and Inoculum
The EHDV-6 isolate used for these studies was originally isolated on baby hamster kidney (BHK) cells (American Type Culture Collection, Manassas, VA, US) from the spleen of a WTD found dead in Henry County, Indiana, US, during October 2006 (Southeastern Cooperative Wildlife Disease Study accession CC304-06). This first passage virus stock was then used to prepare distinct inocula for each experimental infection by 1 (ie, WTD and cattle trials) or 2 (ie, Culicoides trial) additional passages on BHK cells. Briefly, a BHK monolayer propagated in maintenance media consisting of minimum essential media (MEM; Sigma Chemical Company, St Louis, MO, US) with 3% fetal bovine serum (Sigma), and antibiotics (100 U, penicillin; 0.1 mg, streptomycin; 0.25 µg/ml, amphotericin B; Sigma) was inoculated with EHDV-6 (first-passage BHK supernatant). After exceeding the 80% cytopathic effect, the flask was scraped and the contents centrifuged at 1200 × g for 10 minutes and the supernatant collected. The cell pellet was sonicated, centrifuged, and the supernatant combined with the original supernatant, which served as the virus inoculum. Each inoculum was stored at 4°C until used. The virus inocula were titrated with cattle pulmonary artery endothelial (CPAE) cells (American Type Culture Collection) in a 96-well plate format, and end point titers (median tissue culture infective doses; TCID50) were determined 52 : WTD, 6.1 log10 TCID50/ml; cattle, 7.0 log10 TCID50/ml; Culicoides, 7.4 log10 TCID50/ml. The final WTD and cattle inocula were second passage, whereas the Culicoides inoculum was third passage. The Culicoides inoculum was used to prepare an infectious blood meal to feed midges. Briefly, 100 ml of whole blood was collected via jugular venipuncture from a donor WTD and manually defibrinated.
Experimental Design
For the WTD trial, 5 fawns (deer Nos. 1–5) were experimentally infected with EHDV-6, and 1 served as a negative control (deer No. 6). For inoculation, deer were sedated with intramuscular xylazine (1–2 mg/kg; AnaSed, Lloyd Laboratories, Shenandoah, IA, US) and ketamine (2–3 mg/kg; Fort Dodge Animal Health, Overland Park, KS, US). The hair covering the lateral cervical region was shaved to facilitate inoculation. Each of the 5 experimental deer received 1 ml of inoculum intradermally and another 1 ml subcutaneously, for a total dose of 2 ml (6.4 log10 TCID50) divided over multiple sites on the neck. The negative control deer received a sham inoculum administered intradermally and subcutaneously, which consisted of 2 ml of uninfected BHK supernatant. Each animal was visually monitored twice daily for clinical signs during the first 12 days postinfection (dpi) and once daily thereafter. At approximately the same time each day, WTD were sedated as described above for physical examination, rectal temperature, and blood collection via jugular venipuncture on 0, 3, 5, 7, 10, 12, and 19 dpi. Sedation was reversed with intravenous tolazoline administered slowly (2–4 mg/kg; Lloyd Laboratories). Blood in sodium citrate was used for virus isolation and titration, and plasma was harvested for coagulation assays, including prothrombin time and activated partial thromboplastin time (APTT). Blood in calcium EDTA was used for complete blood count, including fibrinogen. Serum from additive-free blood tubes was used for serology, including AGID and serum-neutralizing titers, as well as serum total protein analysis. Deer that became moribund were euthanized with intravenous sodium pentobarbital (1 ml/5 kg; Beuthanasia-D Special, Schering-Plough Animal Health Corporation, Union, NJ, US) or on 19 dpi. Postmortem examinations were performed; representative tissues samples were collected at necropsy and fixed in 10% neutral buffered formalin; and hematoxylin and eosin slides were prepared routinely for histopathologic examination. Fresh samples of spleen, lung, popliteal lymph node, and skin were harvested for virus isolation and reverse transcriptase polymerase chain reaction (RT-PCR).
For the cattle trial, 1 Holstein steer (bovine No. 1), 3 Holstein cows (bovine Nos. 2–4), and a positive control WTD (deer No. 7) were experimentally infected with EHDV-6, and 1 cow served as a negative control (bovine No. 5). For inoculation, cattle were sedated with 0.1 to 0.2 mg/kg of xylazine intramuscularly, and the hair covering the lateral cervical region was shaved. Each of the 4 experimental cattle and the positive control WTD were inoculated by intradermal and subcutaneous injection (2 ml, 7.27 log10 TCID50) as described above, and the negative control cow received a sham inoculum. Similar to the WTD trial, the animals were closely monitored for clinical signs. Physical examination and blood collection occurred on 0, 3, 5, 7, 10, 12, 18, and 24 dpi. Blood was collected for virus isolation and titration, complete blood count, and total protein as described above. Animals were euthanized with intravenous sodium pentobarbital on 24 dpi, and a gross postmortem examination was performed. All of the above animal procedures were approved by the University of Georgia’s Institutional Animal Care and Use Committee (protocol A2008-10129).
For the C. sonorensis experimental infection, midges were fed manually defibrinated WTD blood spiked with EHDV-6 (5 ml, virus inoculum; 3 ml, blood). The virus titer of the infectious blood meal was 6.94 log10 TCID50/ml. Midges (female and male) were briefly anesthetized with CO2 and transferred to feeding cages in groups of 150 to 200 midges. Midges took a blood meal through a parafilm membrane stretched over the reservoir of an artificial feeding device. 10 Midges fed at room temperature in low light for approximately 30 to 40 minutes and were subsequently anesthetized and sorted according to feeding status (blood fed vs non–blood fed). To determine if midges ingested virus with the blood meal, 15 midges were immediately processed for virus isolation and titration on 0 days postfeeding (dpf). The remaining blood-fed females (n = 367) were kept at a constant temperature of 25°C, and ≥15 midges were harvested on 1, 2, 3, 4, 5, 6, 8, 10, 12, and 14 dpf for virus isolation and titration.
Virology and Serology
All virus isolation and titration from blood samples were performed on the day of collection via methods similar to those described previously. 51 For virus isolation from blood, plasma was removed from whole blood samples collected in sodium citrate and the blood resuspended with Dulbecco’s phosphate buffered saline (DPBS; Sigma) to the original blood sample volume. To isolate virus from erythrocytes and remove any possibly interfering antibodies, blood was washed 3 times in DPBS. Briefly, 1 ml of blood was washed in 9 ml of DPBS and centrifuged, with the wash removed and discarded. This wash step was repeated 3 times, and the 1 ml of washed erythrocytes was sonicated and centrifuged. Undiluted supernatant and supernatant diluted 1:10 in MEM were used to inoculate monolayered CPAE cells. Maintenance media consisted of MEM with 10% fetal bovine serum and 1× antibiotics and flasks were incubated in 5% CO2 at 37°C. Attempts at virus isolation from tissues were performed as described. 54 Briefly, tissues were homogenized with a Tissue Tearor (Biospec Products Inc, Bartlesville, OK, US), centrifuged at 1600 × g for 12 minutes, and the supernatant inoculated onto a CPAE monolayer. After 7 days, cells were scraped, and 100 μl of cell suspension was inoculated onto a new CPAE monolayer. Cultures exhibiting no cytopathic effect after 7 days in the second passage were considered negative. For cultures exhibiting cytopathic effect, RNA was extracted from supernatant via QIAmp Viral RNA Mini Kit (Qiagen, Valencia, CA, US). Virus isolates were confirmed as EHDV-6 by RT-PCR against VP2 through previously published primers. 2 Tissue homogenates from the virus isolation attempt were also tested via standard and/or nested RT-PCR targeting VP2 as previously described. 2 The viral titer of each blood sample (starting with a 1:10 dilution of washed and sonicated whole blood) was determined by endpoint titration on CPAE cells as described above.
For virus isolation and titration from Culicoides, individual midges were placed into 1.5-ml microcentrifuge tubes containing 500 μl of transport media (MEM with 10% fetal bovine serum and antibiotics (500 U, penicillin; 0.5 mg, streptomycin; 1.25 µg/ml, amphotericin B; Sigma). All midges were stored at 4°C until processed individually for virus isolation and titration within 24 hours as previously described. 55 Briefly, individual midges were manually homogenized, sonicated, and centrifuged. For virus isolation, supernatant (200 µl) was inoculated onto BHK cells in a 24-well plate format. 63 In addition, all midges were individually titrated for virus through BHK cells, 63 and endpoint titers (TCID50) were determined. 52 Because of the dilutions used, the minimum detectable titer was 102.3 TCID50/midge. The minimum EHDV titer of infected Culicoides that is suggestive of the ability to transmit the virus to a naive host has not been studied. However, in a previous study in domestic sheep with a related virus (BTV), C. sonorensis with virus titers ≥102.7 TCID50 were considered potentially competent because this was the virus titer of midges found capable of efficient transmission of BTV to susceptible sheep. 30 Per an extrapolation of these data, we are considering C. sonorensis with a virus titer ≥102.7 TCID50 to be potentially competent.
All serum samples were tested for precipitating antibodies against EHDV through AGID test kits (Veterinary Diagnostic Technology Inc) according to manufacturer instructions. For AGID-positive samples, including the preceding sample day, serotype-specific antibodies were detected by serum neutralization testing, and neutralizing antibody titers were determined as previously described, 67 except for the use of CPAE cells instead of BHK cells.
Results
WTD Infection
The viremia and serologic profiles of WTD are presented in Table 1. Viremia was first detected 3 to 5 dpi, and it lasted for the duration of the study (19 dpi) in animals surviving infection. Peak viremia occurred on 7 dpi, ranging from 105.93 to 106.8 TCID50/ml (geometric mean titer = 106.3 TCID50/ml). All surviving animals had precipitating and neutralizing antibodies by 10 dpi, and peak neutralizing antibody titers varied depending on last sampling day for the animal (20–1280). The negative control (deer No. 6) remained virus isolation negative and seronegative throughout the study.
Comparison of Viremia Profiles and Serologic Response of WTD Experimentally Infected With Epizootic Hemorrhagic Disease Virus 6 (Indiana).a
Abbreviations: SNT, serum-neutralizing titer; TCID50, median tissue culture infective doses; VT, virus titer; WTD, white-tailed deer.
aThe negative control deer remained negative by virus isolation and serology throughout the study.
bReported as log10 TCID50/ml.
cDashes indicate virus isolation negative after 2 passages.
dAnimal died or was euthanized due to severe disease
eReported as reciprocal of the dilution
fAnimal found dead 4 days postinfection; postmortem blood sample titer = 102.26 TCID50/ml.
All 5 infected animals exhibited moderate to severe clinical disease at 4 to 15 dpi. One animal (deer No. 5) was found dead on 4 dpi, and 2 (deer Nos. 1 and 2) were euthanized on 12 dpi because of severe clinical disease, including erythema of sparsely haired skin, lethargy, inappetence, and head tremors. Other commonly observed clinical signs included lameness, rough and dull hair coat, hyperemia of conjunctiva and oral mucosa, and fever. Hematoma formation at venipuncture sites and failure of the blood to clot normally in clot activator tubes (fragile clots or poorly formed clots with sedimentation of unincorporated red blood cells) also were observed 7 to 12 dpi. The mean ± SE baseline temperature of all deer in the study on 0 dpi was 39.6°C ± 0.08°C, with the peak rectal temperature of 40.8°C ± 0.44°C occurring on 5 dpi (Suppl. Fig. 1).
Clinical pathologic abnormalities in infected animals included leukopenia, lymphopenia, decreased total protein, prolongation of prothrombin time and APTT, decreased platelet count, and elevated fibrinogen (Suppl. Fig. 2). The mean ± SE total white blood cell count reached its nadir on 5 dpi (2175 ± 1220 cells/ml), a decrease from the baseline mean of 2933 ± 209 cells/ml, before recovering to near baseline levels by 7 to 10 dpi. The mean lymphocyte count markedly declined at 5 to 7 dpi, with a nadir of 153 ± 44 cells/ml (baseline mean 928 ± 84 cells/ml) occurring on 7 dpi. There was also a decrease in the segmented neutrophil count on 5 dpi (806 ± 261 cells/ml) in comparison with the baseline mean (1809 ± 284 cells/ml). Mean total serum protein began to decrease slightly on 3 dpi (5.8 ± 0.05 g/dl) and continued to decline until reaching its lowest value on 10 dpi (4.9 ± 0.2 g/dl), before returning to near the baseline mean (6.3 ± 0.16 g/dl) by 19 dpi. At each sampling day after inoculation, the mean platelet count declined from the baseline mean (718 667 ± 51 539/ml) and reached its lowest value on 12 dpi (406 250 ± 149 066/ml). The mean prothrombin time and mean APTT were prolonged at 7 to 12 dpi. The peak prolongation in mean prothrombin time occurred on 7 dpi (18.5 ± 1.0 seconds; baseline mean, 16.7 ± 0.7 seconds), although the timing of the peak varied from 7 to 12 dpi among individuals. Peak prolongation in APTT was 33.3 ± 2.9 seconds on 12 dpi, compared with a baseline mean of 23.7 ± 1.1 seconds. Mean fibrinogen decreased slightly from baseline mean (233 ± 21 mg/dl) between 3 and 5 dpi (lowest value, 220 ± 20 mg/dl on 3 dpi) before increasing 10 to 12 dpi (highest value, 475 ± 75 mg/dl on 10 dpi), then returning to near baseline by 19 dpi (200 ± 0 mg/dl).
Postmortem findings in the 3 WTD that died or were euthanized were consistent with peracute or acute EHD or bluetongue (BT) in this species (Figs. 1, 3, 5, 6, 8). 29 Deer 5 (4 dpi) had poorly clotted blood and widespread hemorrhages on the fascia, skeletal muscles, splenic capsule, pericardium, epicardium, adventitial surface of the pulmonary artery, left ventricular endocardium and myocardium, and rumen wall. Lymph nodes were enlarged and hemorrhagic. Severe pulmonary edema with hemorrhages was also present. Buccal papillae were blunted with hemorrhagic tips. Deer Nos. 1 and 2 (12 dpi) had similar findings along with mild subcuticular icterus, soft and yellow discoloration of the brain, as well as edema and hemorrhage of the spermatic cord, epididymis, and testes of deer No. 2. Deer euthanized at 19 dpi had few changes limited to tonsillar and thymic atrophy and enlarged and edematous lymph nodes.
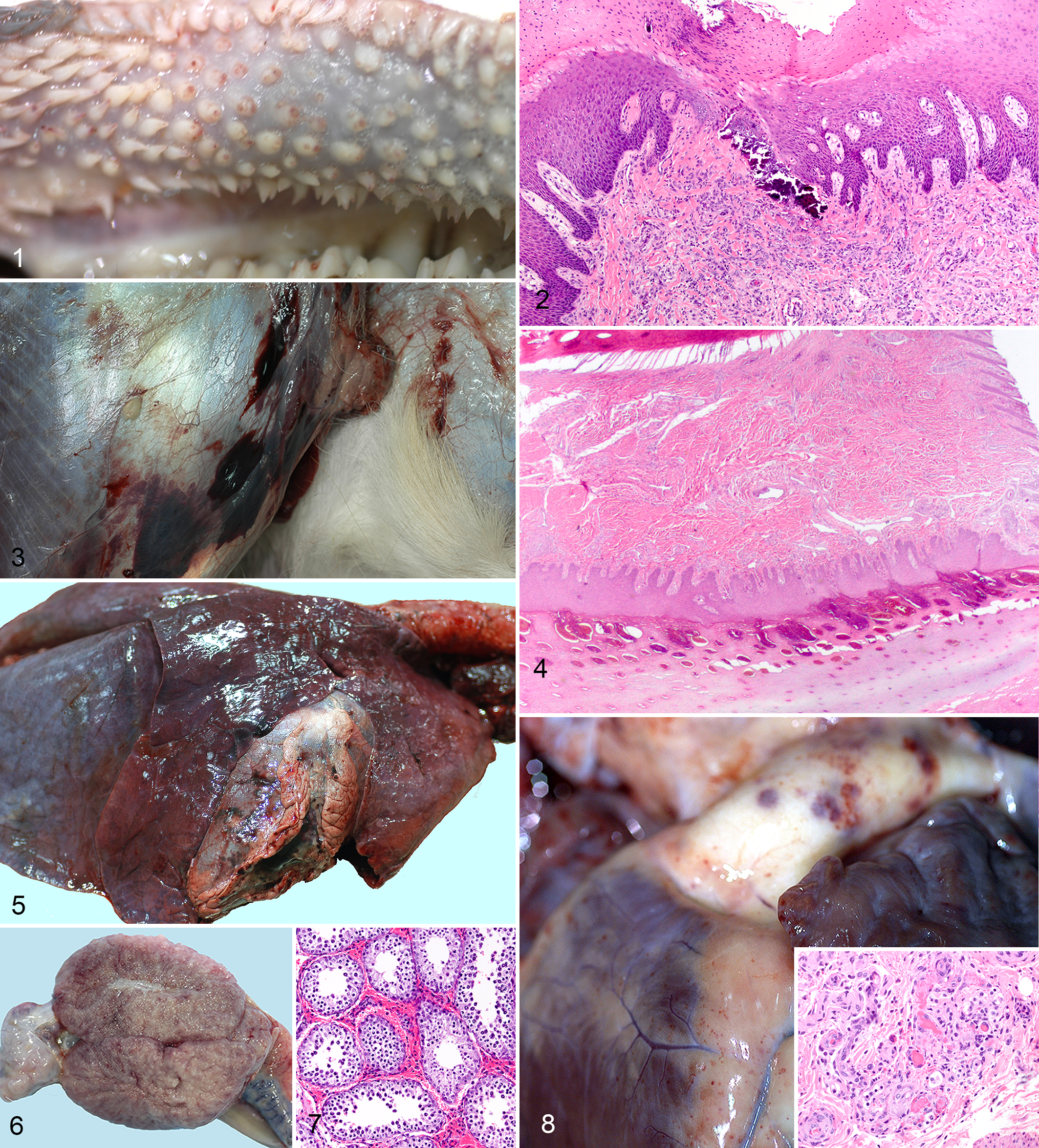
Representative gross and microscopic findings from 3 white-tailed deer experimentally infected with epizootic hemorrhagic disease virus 6 that died (4 days postinfection [dpi]) or required euthanasia (12 dpi).
Microscopically, changes were typical of EHD or BT (Figs. 2, 4, 7, 8). 29 Deer No. 5 (4 dpi) had pulmonary edema and hemorrhages in a variety of organs, including gastrointestinal tract, lung, spleen, pulmonary artery, thymus, heart, kidney, lymph node, testicle, bone marrow, and brain, with associated small vessels lined by necrotic or hypertrophied endothelium. Mild perivascular inflammation, mainly neutrophilic, was present in skin, gastrointestinal organs, testicle, and brain. Bone marrow had megakaryocytic hyperplasia. Changes on 12 dpi were similar to 4 dpi but with more extensive hemorrhage and perivascular inflammation that varied from being neutrophilic to lymphoplasmacytic. On 19 dpi, there were variable bone marrow changes (hyperplasia, including megakaryocytic hyperplasia or hyperplasia with serous atrophy of fat), mild hemorrhage in various locations (abomasum, pulmonary artery, lymph nodes), mild perivascular lymphoplasmacytic infiltrates in skin, gastrointestinal organs and brain, and mild multifocal glial nodules in the brain. Table 2 shows virus isolation and RT-PCR results from tissues collected at postmortem examination.
VI and nRT-PCR Test Results for Tissue Homogenates Collected After Death From WTD Experimentally Infected With Epizootic Hemorrhagic Disease Virus 6 (Indiana).

Abbreviations: dpi, days postinfection; nRT-PCR, nested reverse transcription polymerase chain reaction (against L2 gene); VI, virus isolation; WTD, white-tailed deer.
Cattle Infection
The viremia and serologic profiles of cattle were variable in this study (Table 3). Bovine 2 had detectable viremia at 7 to 24 dpi (ie, to the end of the study) with peak titer (103.6 TCID50/ml) occurring on 10 dpi. Bovine 1 had a transient low-titer viremia (<102.3 TCID50/ml) from 5 to 10 dpi. No viremia was detected in 2 cows (bovine Nos. 3 and 4), and one of these animals (bovine No. 3) failed to seroconvert during the study. Precipitating and neutralizing antibodies were detected by 10 dpi in the remaining cattle. Blood samples from the negative control cow remained seronegative and virus isolation negative. No clinical abnormalities consistent with EHD in cattle were observed during this study. The rectal temperature of all cattle in the study remained within the baseline range of all individuals in the study at 38.5°C ± 0.3°C. Although hematologic findings varied among individuals over the sampling period, no changes consistent with EHDV infection were observed during the study (data not shown). The mean total white blood cell count, lymphocyte count, platelet count, total protein, and fibrinogen all remained at or near the baseline mean of all individuals in the study during time points where alterations would be expected. At termination of the study (24 dpi), no significant necropsy findings were observed, and all attempts at virus isolation from tissue were negative. The positive control deer (deer No. 7) in this study had a detectable viremia 5 to 12 dpi, with a peak titer of 105.4 TCID50/ml on 7 dpi. The deer was euthanized on 12 dpi after exhibiting mild clinical signs of EHD, including elevated rectal temperature, erythema of sparsely haired skin, and lethargy. EHDV-6 was isolated from the lung and spleen.
Comparison of Viremia Profiles and Serologic Response of Domestic Cattle Experimentally Infected With Epizootic Hemorrhagic Disease Virus 6 (Indiana).a
Abbreviations: SNT, serum-neutralizing titer; TCID50, median tissue culture infective doses; VT, virus titer; WTD, white-tailed deer.
aThe negative control bovine remained negative by virus isolation and serology throughout the study.
bLog10 TCID50/ml.
cDashes (—) indicates virus isolation negative after 2 passages.
dReported as reciprocal of the dilution.
ePositive control white-tailed deer.
fStudy ended.
C. sonorensis Infection
The results of virus isolation and titration from whole-midge homogenates are presented in Table 4. The virus recovery rate from 0 dpf was 100% (15 of 15), indicating that all midges ingested virus in the blood meal, before falling to 0% (0 of 15) and 7% (1 of 15) by 2 and 3 dpf, respectively. From 4 to 14 dpf, the percentage of virus isolation–positive midges was 11% (19 of 171), while 4% (6 of 171) of midges had a virus titer ≥102.7 TCID50. When based only on virus isolation–positive midges from 4 to 14 dpf, 32% (6 of 19) of midges had a virus titer ≥102.7 TCID50. For the 6 midges in which virus replicated to ≥102.7 TCID50, the geometric mean virus titer was 103.01 TCID50, and the majority (5 of 6) were harvested on 12 to 14 dpf.
Virus Isolation and Titration Data From Culicoides sonorensis That Fed on White-Tailed Deer Blood Spiked With Epizootic Hemorrhagic Disease Virus 6 (Indiana) and Incubated at 25°C.
aDays postfeeding.
bVirus titer expressed as TCID50 (median tissue culture infective doses) per midge.
cTiter ≥102.7 TCID50/midge, considered capable of efficient transmission in a competent vector.
dIncludes data from 3 to 14 dpf.
Discussion
These results demonstrate that WTD, Holstein cattle, and C. sonorensis are susceptible to infection with EHDV-6, although the outcome of infection was variable. For WTD, the study fulfills Koch’s postulates, and the observed clinical signs, clinical pathology abnormalities, and postmortem findings are consistent with previous natural and experimental cases of other EHDV and BTV infections in this species. 29 WTD have now been experimentally infected with 4 (1, 2, 6, 7) of the 7 provisional EHDV serotypes, and clinical disease has been replicated or induced with each. 17,54,61 It is reasonable to speculate that this species is susceptible to clinical disease with all EHDV serotypes, although variation in susceptibility likely occurs among WTD subspecies. 21 Furthermore, confirmed fatal BT infections (natural or experimental) in WTD have been documented with multiple serotypes of BTV (1, 3, 8, 10, 11, 12, 13, 17). 15,28,31,51,66 It is well established that EHD and BT are clinically and pathologically indistinguishable in WTD. Accordingly, epidemic mortality in WTD during vector season should be investigated with diagnostic assays capable of distinguishing viruses not only at the serogroup level but among individual serotypes. The speed and sensitivity of RT-PCR assays for orbiviruses make these molecular assays very valuable in diagnostic laboratories 39,59 ; however, these assays must not entirely replace traditional virus isolation techniques, as having field isolates is invaluable for epidemiologic investigations, phylogenetic studies, and experimental trials.
The animal-to-animal variation in infection outcome observed among WTD during this study, ranging from moderate transient disease to peracute or acute death, is consistent with previous experimental infections. Furthermore, it has been shown experimentally and is supported in nature that there is significant variation in susceptibility to EHD among WTD. For instance, a northern subspecies, O. virginianus borealis, has been shown to be highly susceptible to fatal infection when compared with a southern subspecies, O. virginianus texanus. 21 The underlying mechanisms that explain this variation are not understood. In a related study, WTD previously infected with EHDV-2 were partially protected from disease upon challenge with virulent EHDV-1. 20 Whether these mechanisms of immunity in WTD (ie, innate resistance to EHDV in individual WTD or subspecies and heterologous serotype cross-protective acquired immunity) are also true for this emerging EHDV serotype remains undetermined.
Widespread microvascular damage due to viral replication in endothelium 70 was present microscopically, causing platelet consumption and thrombocytopenia, as well as activation of the clotting cascade, consuming clotting factors with prolongation of clotting times. The severity of the coagulopathy that developed manifested as hematoma formation at venipuncture sites, as failure of the blood to clot normally in clot activator tubes, and as gross and microscopic hemorrhages in multiple tissues. Along with endothelial damage, disseminated intravascular coagulation (DIC) likely contributed to the development of hemorrhage. DIC has been shown to occur in infection with the closely related BTV, 28 and although specific tests for DIC were not done in this study, microscopic fibrin thrombi in the microvasculature were present and would support its development. Despite widespread microvascular damage, inflammation in affected tissues was mild, which is typical for infection with both EHDV and BTV. 17,35 Ulcers, whether gross or microscopic, are also a common lesion in EHD, and they occurred here; they are the result of ischemia due to vasculature damage and not of direct epithelial damage. Such areas in the gastrointestinal tract often mineralize, 29 which can be seen microscopically, as in these deer, or be grossly evident. Cracked and sloughing hoofs are a common finding in animals that survive natural infection with EHDV. 29 While the animals in this study did not live long enough for this to develop, microscopically there was vascular damage and hemorrhage in the sensitive laminae of the hoof, which likely would have resulted in hoof lesions had the animals lived longer. Two animals (deer Nos. 1 and 2) that had to be euthanized developed tremors of the head. Neurologic signs and encephalitis are not common with EHD but have been reported. 29 Microscopically, damage and inflammation associated with small vessels were randomly scattered throughout the brain in these 2 animals.
The experimental infection of cattle with this reassortant strain of EHDV-6 failed to induce clinical signs or even consistent viremia. Virus could not be isolated from the blood of 2 of the 4 inoculated animals (bovine Nos. 3 and 4), and bovine 3 lacked detectable precipitating and neutralizing antibodies. The lack of seroconversion in bovine No. 3 is surprising; however, this has been documented in individual cattle during earlier EHDV infections. 11 It is also possible that this individual was improperly injected on 0 dpi, although at the time of inoculation, there was no observable difference, and the use of multiple sites of inoculation makes this possibility unlikely. Despite observations of cattle with EHD in the field, to date subclinical infection has been the predominate outcome of previous experimental infections of cattle with EHDV-6 (isolates from Australia, Morocco, Turkey, Reunion Island). 8,9,12,71,72 During an experimental infection of 18-month-old heifers with a Reunion Island isolate of EHDV-6, all animals remained afebrile and otherwise clinically normal during the trial; however, very subtle clinical manifestations were observed, such as conjunctivitis, epiphora, and a single oral erosion/ulcer (in 1 of 5 animals). 12 These subtle manifestations are far from those observed in some clinically affected animals in the field, and the inability to experimentally replicate EHD in cattle remains a significant scientific gap. Numerous factors have the potential to play a role in infection and pathogenicity but have not been explored experimentally, although many are difficult to address in a controlled study. For instance, the potential role of coinfections and secondary infections, harsh environmental conditions (eg, physiologic stress from heat, drought, biting insects), poor nutrition, production class, cattle breed, age, and Culicoides bites and salivary proteins needs to be explored. Furthermore, recent studies suggest that BTV population diversity decreases with passage in mammalian cell lines, which may affect clinical outcome during experimental infections. 14 This has not been evaluated with EHDV.
Outbreaks of EHD in cattle from South Africa, 7 Reunion Island, 13 Algeria, 41 Morocco, 58 and Turkey 69 were all presumably caused by a strain of EHDV-6 previously referred to as “strain 318,” indicating its virulence in cattle. However, this is in apparent contrast to other EHDV-6 strains. Confirmed reports of EHD in cattle associated with EHDV-6 strains from Australia (prototype strain) and the US (reassortant strain) are lacking in the literature. Strain 318 of EHDV-6, which has been detected in the Middle East and Africa, is genetically distinct from the prototype EHDV-6 that was isolated from sentinel cattle in Australia in 1981 68 and again from sentinel cattle from Guadeloupe in 2010. 3 This prototype strain of EHDV-6 has not yet been observed to cause natural disease in cattle, and an experimental infection failed to cause disease. 72 If real, this apparent variation in virulence among strains of EHDV-6 is interesting but has not yet been investigated. Regarding the reassortant EHDV-6 from the US, anecdotal reports of disease in cattle are rare despite the circulation of the virus for 7 years. However, during an intense EHD outbreak in the central and eastern US during 2012, EHDV-6 was isolated from dead WTD from numerous states and from the blood of a cow exhibiting clinical signs suggestive of EHD. 4,53,66 No cattle-focused serologic or virologic surveys for EHDV-6 have been conducted in the US, making interpretation of such isolated cases even more difficult. However, this and previous studies indicate that cattle are susceptible to infection with EHDV-6.
Although only a preliminary investigation, the results from the experimental infection of colonized C. sonorensis suggests that this species is poorly susceptible to infection with EHDV-6 (Indiana). Interestingly, the rate of infection and virus replication to ≥102.7 TCID50/midge was low as compared with historically endemic serotypes (EHDV-1 and EHDV-2). For instance, in the present study, EHDV-6 was isolated from 11% (17 of 156) of midges at 4 to 14 dpf, and only 4% (6 of 156) had virus titers ≥102.7 TCID50/midge. In a similar study, EHDV-2 was isolated from 75% (87 of 116) of midges at 4 to 14 dpf, and 36% (42 of 116) of midges had virus titers ≥102.7 TCID50/midge. 54 The apparently variable susceptibility of C. sonorensis to infection with different EHDV serotypes is interesting and has not been investigated. Previous studies of BTV infection in field and laboratory populations of C. sonorensis describe heterogeneity in response to oral infection among BTV serotypes, 32,33 although mechanisms of such variation remain unknown. Our findings indicate that the EHDV-6 infection rate in C. sonorensis is very low. Additional studies 73 are needed to determine the importance (if any) of this finding and whether this species has any role in the transmission of EHDV-6 in nature. Not always does a low infection rate in a competent vector species equate to poor vectorial capacity. For instance, C. brevitarsis is considered the most significant vector of BTV in Australia, yet it has a very low infection rate when compared with that of other BTV vectors in Australia. 9,42 C. brevitarsis overcomes this low vector competence by being widely distributed throughout Australia, existing in very high densities, and associating closely with hosts, which lead to high biting rates. 37,42,45 However, this scenario does not appear to be at play with C. sonorensis and EHDV-6 in the US, based largely on the known distributions of EHDV-6 and C. sonorensis. In the eastern half of the US where EHDV-6 has been repeatedly isolated from dead WTD during outbreaks, C. sonorensis is rare or absent, suggesting that other Culicoides species are involved with the transmission and maintenance of EHDV-6, as well as EHDV-1 and EHDV-2. 56 For instance, the following species have been implicated as suspect or potential vectors of EHDV and/or BTV in the US: C. debilipalpis, C. stellifer, C. paraensis, C. obsoletus, C. biguttatus, C. venustus, C. cockerelli, C. freeborni, C. neomontanus, C. hieroglyphicus, C. owhyeensis, and C. spinosus. 23,45,62,64 Entomologic investigations during future outbreaks may help to guide further research into other Culicoides spp aimed at identifying additional vector species. 73
In recent years, changes in the patterns of EHDV and/or BTV infection and disease among ruminants in the Middle East, Europe, Australia, and the US have highlighted some of the gaps in our understanding of the epidemiology of these orbivirus infections. 2,24,40,56,57,74 Since 1999, 10 historically nonendemic BTV serotypes, as well as EHDV-6, have been identified in the US, 2,24,50 a country in which certain serotypes of both viruses have been endemic since at least the 1950s. However, in light of the BT outbreaks in northern Europe, these introductions prompted uncertainties: Will these nonendemic serotypes persist in a new ecosystem and become endemic? Will these novel serotypes potentially result in more extensive disease among US ruminants? 24 For many of the BTV serotypes, the answers remain elusive; however, recent events have made the picture for EHDV-6 more clear.
The story of EHDV-6 offers insight into the emergence and subsequent establishment of an exotic orbivirus in the US. Although EHDV-6 was first identified in the US during 2006, a recent retrospective serologic survey detected neutralizing antibodies against EHDV-6 from WTD harvested in Louisiana in 2001. 26 This suggests that the virus may have circulated in the US for years before its initial detection in 2006. From 2006 to 2011, EHDV-6 was occasionally isolated from individual WTD or localized groups from 1 to 4 states per year. 3,49,65 Even during the 2007 EHD outbreak in the US—one of the most intense and geographically widespread on record—EHDV-6 represented <1% (1 of 281) of virus isolations made by the Southeastern Cooperative Wildlife Disease Study (SCWDS; University of Georgia). 56,66 However, during 2012, EHDV-6 represented 29% (55 of 189) of EHDV isolates made by SCWDS, 66,65 and the virus was isolated from ruminants in 16 states in the Great Plains, Midwest, and Southeast. 4,53,66,65 The underlying mechanisms for this dramatic shift in the transmission, incidence of disease, and geographic range expansion of EHDV-6 are unknown. These observations from nature support the findings of the experimental infections reported here: the major known EHDV hosts in the US are susceptible to infection, but additional controlled experiments and field investigations are indicated.
Footnotes
Acknowledgements
Funding for this research was provided by the support of Southeastern Cooperative Wildlife Disease Study member agencies. We thank Jim Kempert and William Yarnell (Arthropod-Borne Animal Diseases Research Unit, Agricultural Research Service, US Department of Agriculture) for providing midges, and we gratefully acknowledge the University of Georgia’s Department of Entomology for use of its facilities during portions of this work. We also gratefully acknowledge David Osborn, Karl Miller, and Robert Warren of the Warnell School of Forestry and Natural Resources (University of Georgia) for white-tailed deer procurement. We thank the University of Georgia’s Animal Resources staff and Sabrina McGraw, Justin Brown, and Andrew Cartoceti for assistance during animal trials. We thank D. Scott McVey, Richard Gerhold, and 2 anonymous reviewers for critical review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was made possible through the continued financial support from the member states of the SCWDS provided by the Federal Aid to Wildlife Restoration Act (50 Stat. 917) and through long-term cooperative agreements with the US Department of Agriculture, Animal Plant Health and Inspection Service.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
