Abstract
Introduction
Evidence for BVDV infections was found in a wide range of wild ruminant species. Persistent pestivirus infection was documented in a mouse deer (
Materials and methods
Virus propagation, titration and isolation, and determination of serum neutralizing titers
The virus (R03–20663) used in the present study was isolated from deer carcasses submitted to South Dakota State University for diagnostic testing.
5
This virus was noncytopathic, determined by a lack of cytopathic effect in cultured Madin Darby bovine kidney (MDBK) cells
10
and belonged to the
Housing, inoculation, and sampling of doe and fawns
Handling and treatment of the doe and fawns complied with the Animal Welfare Act as Amended (7 USC, 2131–2156). The pregnant doe was purchased from a commercial breeder and was tested for antibodies against BVDV by serum neutralization and the presence of replicating BVDV by virus isolation from buffy coat samples. Based on the date of contact with the buck, the doe was estimated to be between 6 and 7 weeks pregnant. The doe was housed in a climate-controlled barn, operated at a Biosafety Level (BL) 2 containment level, for the duration of the experiment, and was observed a minimum of twice daily.
On the day of inoculation, blood was drawn from the doe to determine its pregnancy status and the level of serum-neutralizing antibodies. A second blood sample was drawn at day 32 after inoculation. The limited number of blood samples collected was done to minimize stress on the doe, because this might negatively affect the pregnancy. The doe was inoculated with 5 ml of 1.0 × 10 6 tissue culture infectious dose per milliliter of R03–20663 by the oral/nasal route (viral titer determined as described above). This dose was similar to that used in previous studies in white-tailed deer fawns. 19
The pregnancy was confirmed at the time of inoculation and 32 days after inoculation by using an enzyme-linked immunosorbent assay (ELISA) that measures the presence of pregnancy-specific protein B in serum. a Serum samples were shipped to, and run at, the commercial laboratory that produces the test. b Blood and ear-notch samples were collected from live fawns. Buffy coat samples were isolated and tested by virus isolation, and serum samples were tested for neutralizing titers against BVDV as described above. Ear notch samples were tested for the presence of BVDV antigen by using a commercial antigen capture ELISA tested as per the manufacturer's protocol for testing bovine ear notches. Ribonucleic acid was prepared from viruses isolated from buffy coat samples, and the nucleotide sequence was determined as described. 19 The sequence of the BVDV isolated from fawns was compared with the inoculum virus given to the doe by phylogenetic analysis as previously described. 19
Histopathology and immunohistochemistry
Samples were collected from multiple fawn tissues, including lymphoid, endocrine, urogenital, and nervous tissue; gastrointestinal tract; respiratory tract; and skin. Tissues were fixed in 10% buffered formalin for 24 hr and then transferred to alcohol for 1 day before processing. Paraffin-embedded tissue blocks were sectioned at 5 μm and stained with hematoxylin and eosin.
To characterize the lymphocytes present in tissue, sections were stained by using CD3 e and CD79a/mb-1 f antibodies as T-cell and B-cell markers, respectively. The CD3 was used at a dilution of 1:75 and the CD79a/mb-1 antibody at 1:20. Reactions were conducted in a Ventana NEXES Auto Immunostainer c with a hematoxylin counterstain. Positive tissue control slides were made by using a section of lymph node. Negative control slides were sections of test tissue with the primary antibody removed and replaced with an irrelevant, isotype similar, mouse antibody. c
Results
The doe did not have detectable antibodies against BVDV at the time of inoculation. At 32 days after inoculation, the doe's serum antibodies had a 7.1 log2 titer against BVDV-2 but no titer against BVDV-1. Two fawns, 1 male and 1 female, were delivered live, 163 days after inoculation. Both fawns were undersized but otherwise appeared normal. The 2 fawns were positive for BVDV by virus isolation from buffy coat, positive for BVDV antigen-by-antigen capture ELISA test, and negative for BVDV antibodies based on serum-neutralizing titers. Based on phylogenetic analysis, the viruses isolated from the 2 fawns were identical and matched the inoculum virus. During the first night after birth, the doe apparently killed the 2 fawns.
Histopathology
Samples of 34 tissues were collected from one or both fawns. Tissues from the female fawn showed evidence of mild autolysis, most prominent in the brain, whereas the remaining sections were in good postmortem condition. Sections of the gastrointestinal tract evaluated included oral mucosa, salivary gland, esophagus, rumen, duodenum, jejunum, ileum, cecum, colon, rectum, pancreas, and liver. No microscopic lesions were observed in the oral mucosa, salivary gland, esophagus, cecum, colon, rectum, and adjacent anal gland. Lymphoid tissue throughout the small intestine was reduced and present as a diffuse band within the lamina propria, most prominent in the ileum. Enterocytes of the jejunum were diffusely vacuolated. The ruminal papillae and pancreatic islet cells were morphologically consistent with fetal or perinatal tissue. Hepatocytes often contained a single intracytoplasmic glycogen-containing vacuole, and there was a moderate amount of extramedullary hematopoiesis within sinusoids.
Representative sections of the respiratory tract, including the nasal orifice, nasal turbinates, trachea, and lung, were collected. In both fawns, the pulmonary parenchyma was incompletely inflated. The nasal orifice, nasal turbinates, and trachea were histologically unremarkable. Lymphoid organs evaluated included the mesenteric, submandibular, retropharyngeal, ileocecal, and parotid lymph nodes; tonsil; spleen; and thymus. There was a paucity of lymphocytes within all lymph nodes and medullary rays were prominent; evidence of necrosis or apoptosis was absent. Lymphoid depletion was most prominent within the germinal centers of follicles (Fig. 1A). Follicles were also markedly depleted in the tonsil and spleen. Splenic lymphocytes were most common in the periarteriolar lymphoid sheath. The cortex of the thymus appeared thin in some areas. The amount of extra medullary hematopoiesis in the spleen was adequate for a young animal.
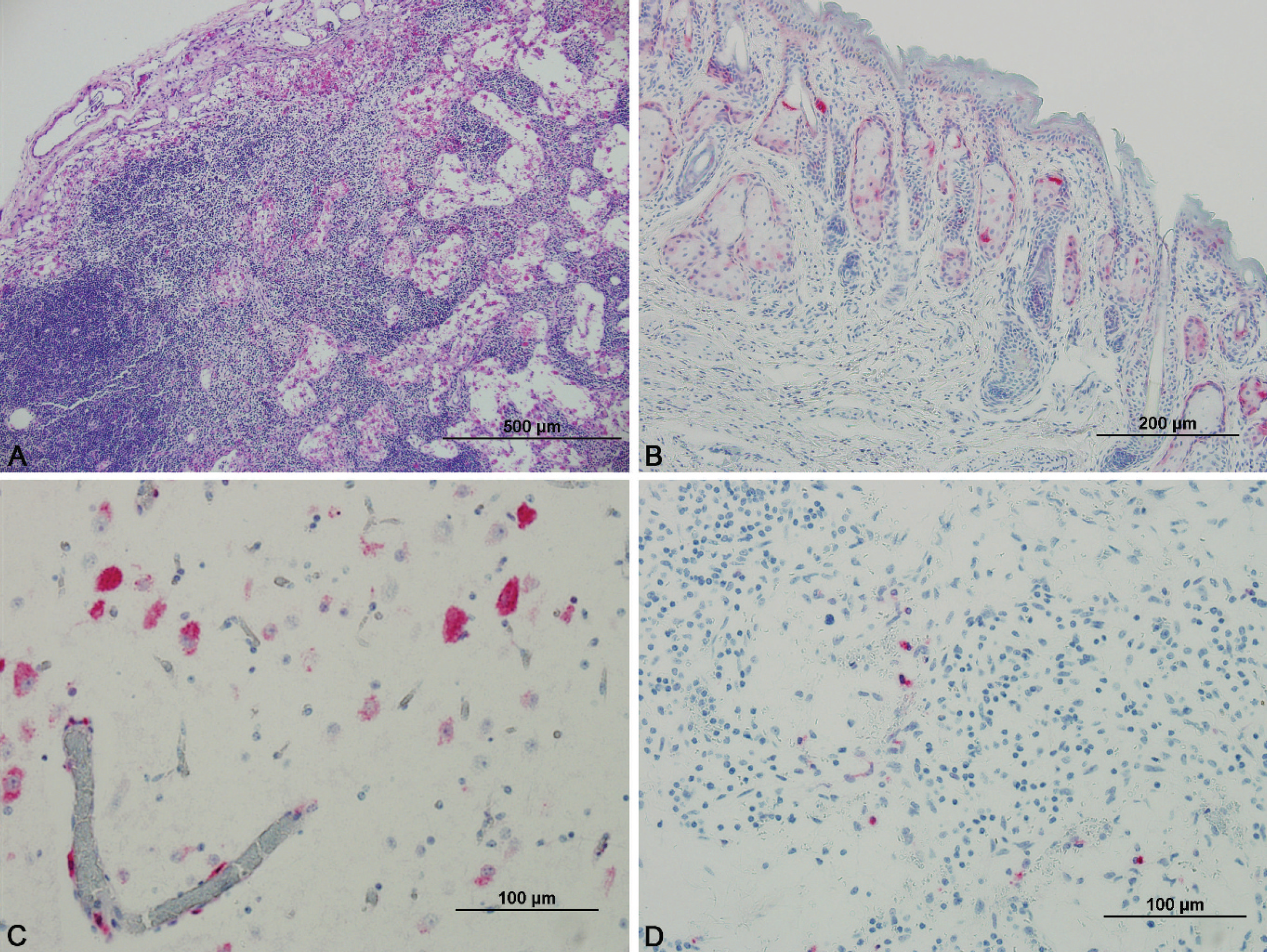
Sections of the cerebrum were taken from the middle of the parietal lobe, including a portion of the thalamus. Vacuolation of rare individual neurons was noted in the brain of the male fawn. Purkinje cells were occasionally observed within the granular cell layer of the cerebellum of both fawns; the remaining cells were normally placed. Sections from the female reproductive tract examined included ovarian tissue, fallopian tube, fimbriae, and broad ligament. No microscopic lesions were observed in the ovary, which was composed of many primordial follicles and stroma. The testes were immature but did not contain any microscopic lesions. The kidney of 1 fawn was mildly congested.
Myocardial cells of both animals were diffusely thin. There were no microscopic lesions in the skin or skeletal muscle of the diaphragm. Adipocytes adjacent to other organ tissue were moderately to markedly hypoplastic in both fawns. Diffuse vacuolation of follicular cells was present in the thyroid gland of both fawns. The adrenal glands were histologically unremarkable.
Distribution of

+ = weak, faint, or minimal antigen in the cytoplasm; ++ = moderate, uniformly distributed, cytoplasmic staining; 0 = no detectible antigen; +++ = intense cytoplasmic staining; NA = not applicable, tissue or cell type was not present in examined sections.
Immunohistochemistry
Neurons of both the cerebellum and cerebrum (Fig. 1C) were diffusely positive; however, more intense staining was noted in the neuronal cell bodies of the cerebrum (male: ++, female: +++) relative to the cerebellum (Purkinje and granule cells, both animals: +). Only rare staining was observed in the macroglia or microglia of either animal. Strong antigen staining (+++) was present in the endothelial cells of vessels in both the cerebrum and cerebellum of both animals. In addition, vasculature in the meninges overlying the cerebrum (+) and cerebellum (+++) was antigen positive in both animals. No antigen was identified within the neuropil.
The distribution of antigen in the gastrointestinal tract, by location and cell type, is presented in Table 2. Weak staining for BVDV was present in the epithelium throughout the gastrointestinal tract of both fawns. Antigen was rarely detected in blood vessels, leukocytes, or myocytes of the intestine. Hepatocytes were strongly and diffusely antigen positive, and hepatic vasculature exhibited more positive staining than blood vessels in other tissues. Kupffer cells, randomly distributed throughout the liver, were the most intensely positive cells in the liver of both animals.
Both follicular (male: ++, female: +) and parafollicular cells (male: ++, female: +) of the thyroid gland stained positive for BVDV antigen. Adrenal cortical secretory cells and medullary cells were positive (+) in the male fawn; however, no antigen was detected in the adrenal gland of the female fawn. The urogenital system showed mild staining in both animals. Renal tubular epithelium and vascular endothelium was weakly positive (+) in both fawns. Rare epithelial cells of the fallopian tube were positive (+) in the female; no positive staining was noted in the male reproductive system.
Distribution of viral antigen in lymphoid tissue is presented in Table 3. In lymph nodes, only rare cells stained positively for BVDV antigen (Fig. 1D). These positive cells may represent lymphocytes or macrophages. In the tonsil, rare round cells were positive, whereas surrounding epithelium was diffuse and strongly positive. Epithelial cells and rare round cells were positive in the thymus.
Distribution of
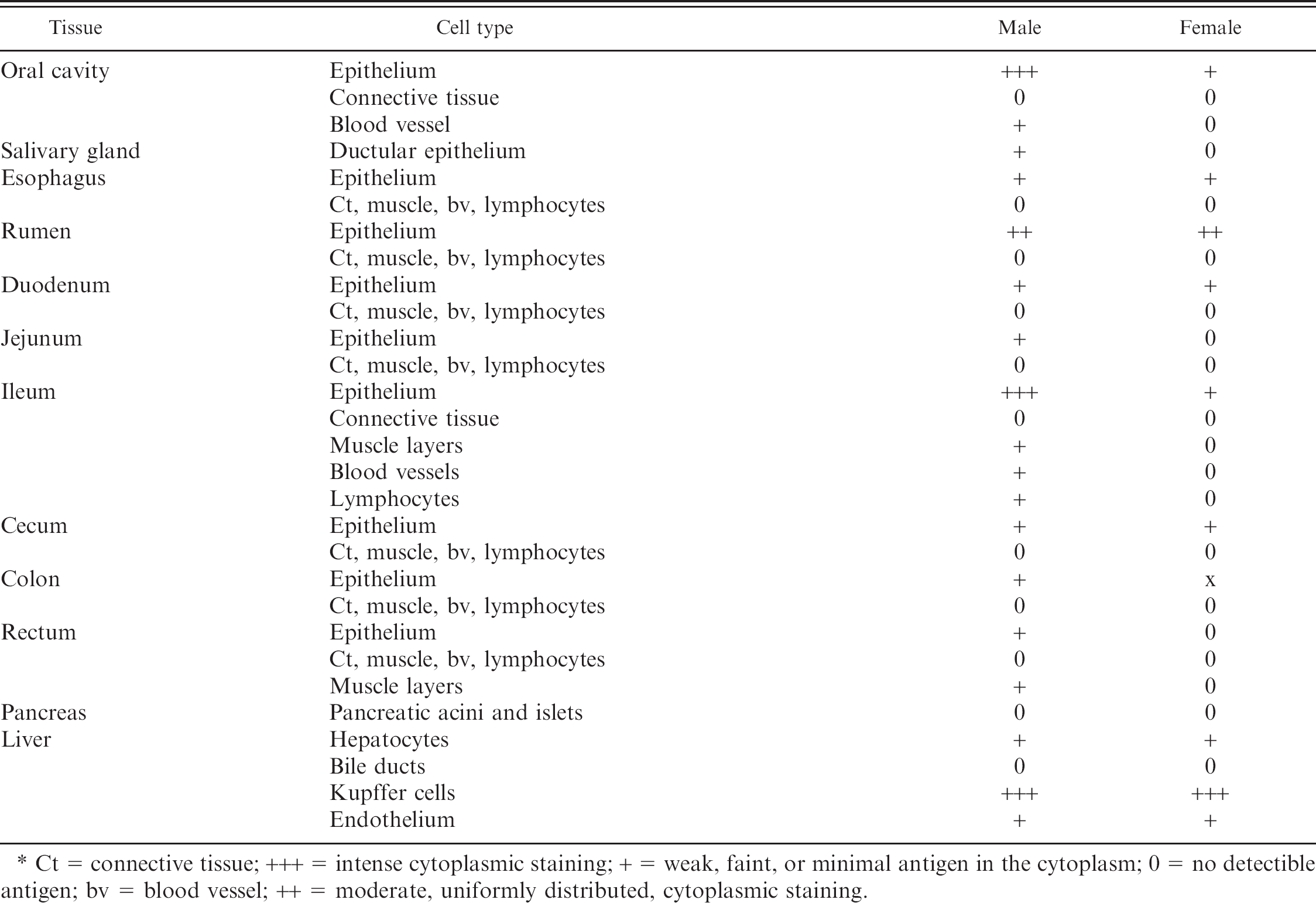
Ct = connective tissue; +++ = intense cytoplasmic staining; + = weak, faint, or minimal antigen in the cytoplasm; 0 = no detectible antigen; bv = blood vessel; ++ = moderate, uniformly distributed, cytoplasmic staining.
Distribution of
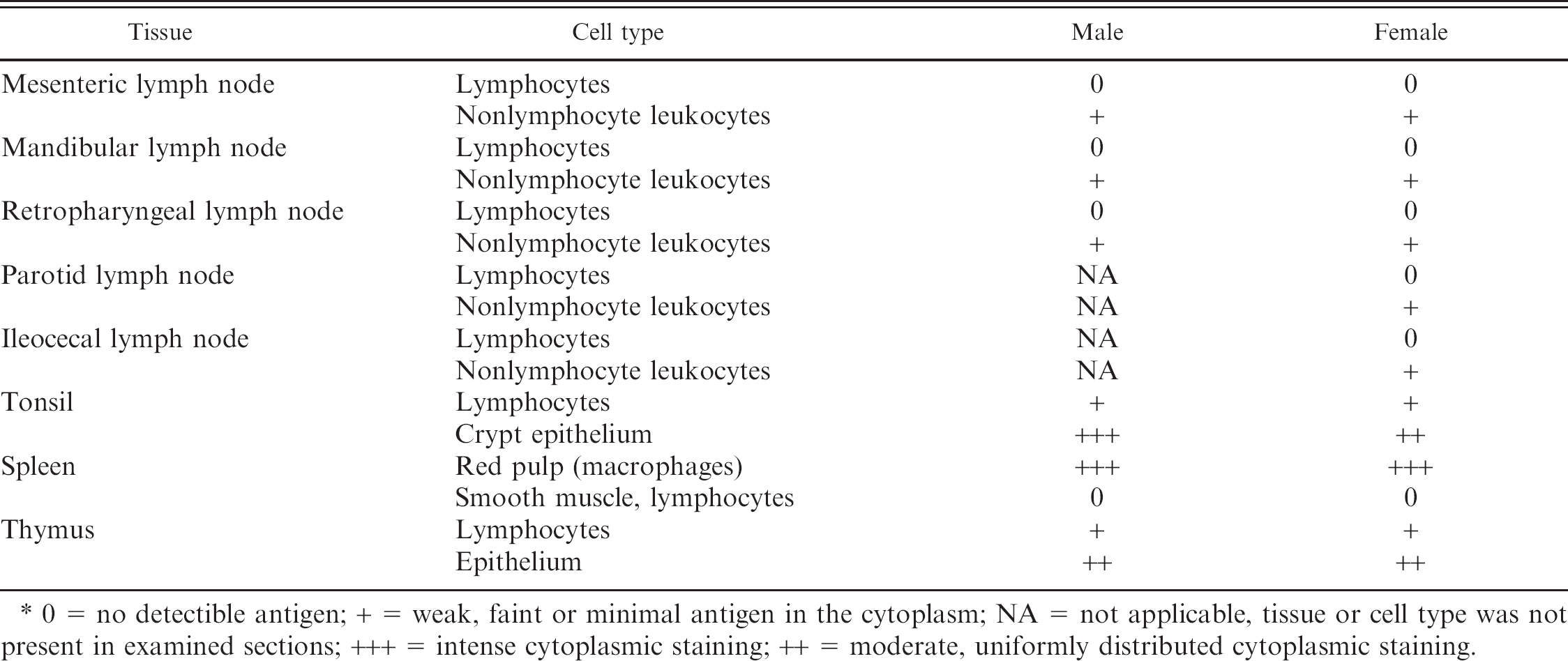
0 = no detectible antigen; + = weak, faint or minimal antigen in the cytoplasm; NA = not applicable, tissue or cell type was not present in examined sections; +++ = intense cytoplasmic staining; ++ = moderate, uniformly distributed cytoplasmic staining.
Discussion
The 2 fawns were classified as PI given the stage of gestation at the time of inoculation, isolation of virus identical to the inoculum strain, and the presence of antigen in skin and serum in the absence of BVDV antibodies. Whereas, a temporal pattern of viremia could not be documented in these animal given their premature death, diagnostic test results were consistent with previously reported experimental induced persistent infection in white-tailed deer 18 and cattle.
The histopathologic and immunohistochemical changes observed in the 2 PI fawns examined are similar to those seen in PI cattle, with some subtle differences. Lymphoid tissue of the fawns contained less antigen than is commonly reported in cattle; lymphocytes and macrophages were strongly positive in bovine studies. 16,17 The spleen of both fawns had abundant, strongly positive cells present in the red pulp; similar cells were observed in lymph nodes. On morphologic examination, these cells were consistent with macrophages, as has been reported previously; however, the paucity of staining of lymphocytes in the deer fawns conflicts with previous reports in cattle. 3
In the gastrointestinal tract, faint staining was observed in the epithelial cells from the oral cavity to the rectum. Blood vessels were rarely positive for viral antigen. In PI calves and adult cattle, BVDV antigen has been detected in epithelial cells and rare mesenchymal cells in the gingiva, tongue, esophagus, abomasum, omasum, rumen, and ileum; however, no positive staining was noted below the ileocecal orifice. 3 Strong positive staining was observed in the hepatocytes and Kupffer cells of the fawns consistent with reports in cattle. 22
Viral antigen was present in neuronal cell bodies of the cerebellum and cerebral cortex, consistent with that reported in cattle. 12,34 In contrast to reports in cattle, 12 however, antigen was identified in blood vessels and meninges, along with Purkinje and granule cells of these 2 fawns. The cerebral cortex and hippocampus have been proposed as predilection sites for BVDV antigen in cattle, with approximately 90% of neurons staining positively. 12 Mapping of viral distribution within the brain was not done in these fawns, so predilection sites within the central nervous system remain unknown.
In bovines, the distribution of BVDV antigen in the respiratory track has been extensively studied, and virus has been identified in epithelium, glands, vessels, circulating leukocytes, and chondrocytes. 6 A similar pattern was observed within the deer fawns; however, staining was only rarely noted in chondrocytes and lymphocytes.
The most prominent histologic finding in the 2 fawns was lymphoid depletion. Although healthy, age-matched controls were not available for inclusion in the current study, lymphoid tissue examined contained significantly fewer lymphocytes than deer fawns previously examined by the authors. In clinically normal PI cattle, histologic lesions are also rare. 3,12 Lymphoid depletion is present in calves with mucosal disease 33 but is not commonly reported in healthy PI animals. Lymphoid depletion observed in these 2 fawns was consistent with a paucity of B-cells. These findings suggest that there may be a difference in the clinical expression of persistent infection between cattle and deer.
Tissue sections available for evaluation may not fully represent the distribution of virus within all PI deer; however, findings suggest that antigen is broadly distributed throughout many organs and cell populations consistent with the pathogenesis in cattle. Overall, epithelial cells were most often positive, and immunohistochemical staining of skin biopsy specimens, as is commonly done with cattle, may be an effective test for persistent infection in white-tailed deer.
The prevalence of BVDV in wild populations and the subsequent risk to domestic livestock is unknown. Seroprevalence studies of numerous North American wild cervid populations, including mule deer (
Given the similarity of pathologic lesions observed in PI deer relative to cattle, it may be hypothesized that PI deer also represent a significant risk of disease transmission. Persistently infected mouse deer have been shown to infect cattle without direct contact. 28,29 The role of wildlife in the epidemiology of BVDV should be considered if BVDV is to be effectively managed in cattle populations.
Footnotes
a.
BioPRYNTM, Biotracking LLC, Moscow, ID.
b.
Biotracking LLC, Moscow, ID.
c.
Ventana Medical Systems, Inc., Tucson, AZ.
d.
IDEXX, Westbrook, ME.
e.
Dako North America, Inc., Carpinteria, CA.
f.
Biocare Medical, Concord, CA.
