Abstract
In a 2-month-old female savannah kitten that died unexpectedly, the pathologic findings of significance were restricted to the heart and included abnormal Purkinje fibers and biventricular myocardial trabeculation or noncompaction. The Purkinje fibers were large, angular, and tightly packed. They contained few disorganized myofibrils among a rarified cytoplasm. The fibers were distinct from adjacent myocytes and were immunohistochemically positive for desmin, muscle actin, myoglobin, sarcomeric actin, and chromogranin A. These findings are identical to those that occur in children with histiocytoid cardiomyopathy, a fatal genetic mitochondrial disorder of Purkinje fibers. Ventricular noncompaction likely has a multifactoral cause that results from fetal arrest of ventricular organizational development that might occur in conjunction with, or independent of, histiocytoid cardiomyopathy.
The savannah cat was created in 1986 by cross-breeding an African serval with a variety of domestic cat breeds. Thus, savannah cats have a variety of phenotypes. The International Cat Association first recognized savannahs as a separate breed in 2000. The gestation period of servals exceeds that of domestic shorthairs by 10–14 days. Hybrid inviability is a problem in low F breedings. F1 females are generally fertile, but male savannahs are usually sterile until the F4 or F5 generation. Therefore, the distribution of serval genes among savannahs varies.
Purkinje fibers are modified cardiomyocytes that play an essential role in electrical conduction and depolarization in the heart. In conjunction with the His system, Purkinje fibers are necessary for efficient, coordinated ejection of blood starting at the base of the ventricles. 21 Inductive effects such as those of endothelin-1 from the nearby coronary vasculature and migration of epicardium-derived cells are necessary for Purkinje cell/fiber development, at least in the chick. 11,21 In mammals, Purkinje fibers develop early during embryogenesis, before the development of cardiac septation and the coronary vessels. 21 Purkinje fibers vary by species in the number of T tubules they contain; they have a paucity of lateral cell junctions and they lack particular isoforms of gap junction proteins (connexins). They have modified intercalated discs, few myofibrils, and abundant glycogen, at least in larger mammals. 9 Human stem cells xenografted into fetal sheep hearts indicate that, in mammals, Purkinje fibers develop from single cells rather than homing of multiple cells. 7
Background
Congenital Purkinje fiber dysplasia is generally known as histiocytoid cardiomyopathy in the pediatric medical literature. The abnormal appearance of the Purkinje fibers as sharply demarcated groups of large, pavemented cells resembling histiocytes with abundant clear to granular to foamy cytoplasm is linked to functional abnormalities in the cardiac conduction system, leading to early death. Immunohistochemically and ultrastructurally, it is clear that these cells are not histiocytes, but the term histiocytoid (histiocyte-like) remains entrenched in the literature. Histiocytoid cardiomyopathy in children results in cardiomegaly (dilated cardiomyopathy), ventricular tachycardia, and death by 2 years of age. 15,26 These deaths are sometimes misdiagnosed as sudden infant death syndrome (SIDS).
In children, histiocytoid cardiomyopathy is usually an autosomal recessive or X-linked disorder. 14 Four times as many female infants are affected as males. 2 Some evidence suggests that histiocytoid myocytes (Purkinje fibers) are undergoing apoptosis and therefore, the condition is a degenerative change rather than a developmental abnormality. 10 In a few reported cases, clusters of mast cells, lymphocytes, and macrophages are associated with the lesions. 10,17 Most cases, however, are free of inflammation. Currently, histiocytoid cardiomyopathy is considered a genetic mitochondrial disease on the basis of the mitochondrial deoxyribonucleic acid mutations that many patients have and the histiocytoid cardiomyocytes (Purkinje fibers) that have morphologically abnormal mitochondria and decreased mitochondrial enzyme activity. 13,20,27
Ventricular noncompaction is a hyper-trabeculation of the endomyocardium. The recesses/trabeculae communicate with the ventricular lumen and represent an arrest of normal myocardial development and compaction. Early fetal trabeculation is necessary for myocardial nutrition before development of the coronary arteries. In cases of ventricular noncompaction, the left ventricle is always affected; right ventricular involvement (biventricular noncompaction) is variably present. The endothelium-lined channels/trabeculae are contiguous with the endocardial endothelium, and the myocardium is arranged in a honeycomb, or spongy, configuration that resembles the myocardial structure of nonmammalian vertebrates (ontogeny recapitulates phylogeny). The channels do not communicate with the coronary vasculature. 6,8 Ventricular hyper-trabeculation is increasingly reported in humans and likely has a multifactoral cause, 3 including genetic heterogeneity. 20 A variety of genetic mutations encoding sarcomeric proteins have been demonstrated in affected persons. 19 Familial and nonfamilial cases have been described, as well as “isolated” ventricular noncompaction and ventricular noncompaction concurrent with other congenital heart anomalies. 20 The adult genetic form of this condition is believed to be autosomal dominant, whereas the pediatric form is X-linked, at least in some cases. 18 Interestingly, one of the reported associations of ventricular noncompaction is with mitochondrial disorders. Mitochondriopathies can be inherited or sporadic and present with a wide variety of clinical signs and organ involvements. 12 Some cases of pediatric histiocytoid cardiomyopathy are associated with left ventricular noncompaction, endocardial fibroelastosis, or both. 4,10,16
History
An 8-week-old female savannah kitten was presented for necropsy. The dam was an F1 savannah (progeny of a male serval mated to a female domestic cat) queen. The sire was an F5 savannah tom. Therefore, the kittens from this litter were considered F2 savannahs. The cattery had a history of abortions, malformed fetuses, juvenile deaths, and positive polymerase chain reactions to calicivirus and coronavirus, as well as exposure to Tritrichomonas sp. Spirochetal colitis had been diagnosed post mortem in a female littermate 1 week before submission of the propositus kitten.
Pathologic Findings
The propositus kitten weighed 284 g and was in very thin condition with virtual absence of body fat. A bifid gall bladder was present, as was hepatic sinusoidal congestion. The proximate cause of death was severe pulmonary congestion and edema. There were no gross lesions of the heart.
Hematoxylin and eosin–stained 5-µm-thick sections of selected tissues were prepared with the use of standard methods. The microscopic lesion of note was the presence of numerous, discrete, irregular aggregates of abnormal cells/fibers in both ventricles (Fig. 1). The cells/fibers were polyhedral to round and tightly apposed to each other. They had sparse, somewhat fibrillar, poorly stained, eosinophilic, vacuolated cytoplasm with radiating striations (myofibrils) resembling a starburst. Along the striations were thicker, denser, parallel, basophilic areas resembling Z bands (intercalated discs). Nuclei were elongate and euchromatic to hyperchromatic. The cells/fibers were distinct from the surrounding myocytes although the myofibrils of the histiocytoid cells blended with those of adjacent myocytes. No inflammatory cells were present. Also noncompaction of the left and right ventricular myocardium was present (Fig. 2). The littermate presented for necropsy a week earlier had histologically normal Purkinje fibers and normal compaction of the ventricular myocardium.
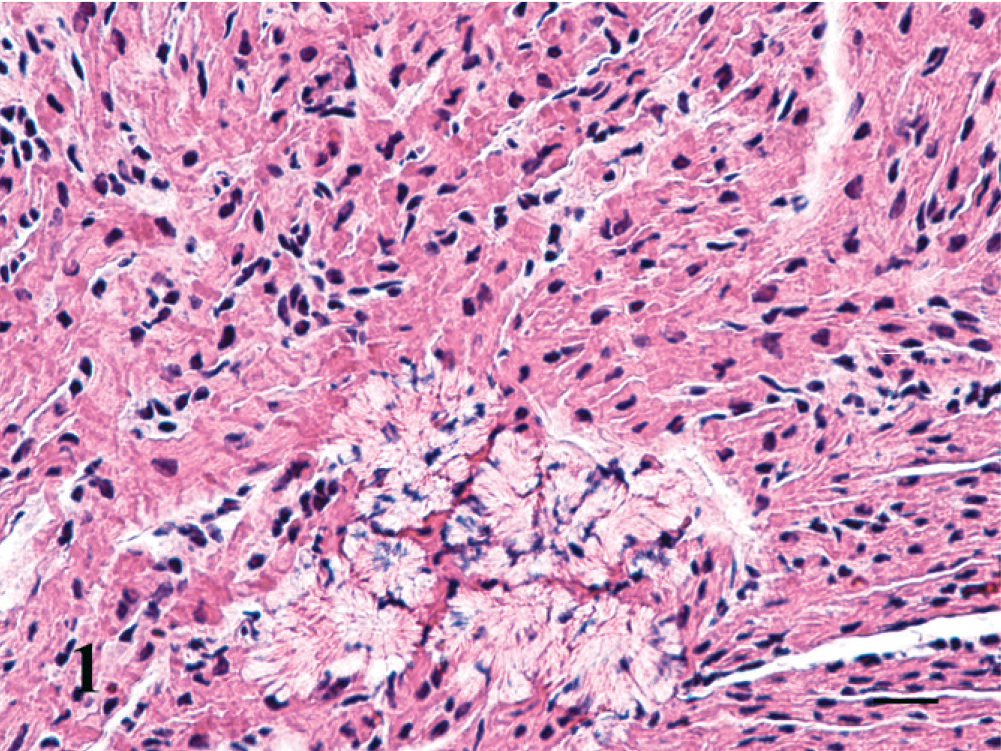
Right ventricular myocardium; kitten. Abnormal Purkinje fibers (histiocytoid cells) are distinct from adjacent myocytes. The histiocytoid cells have rarified cytoplasm and reduced numbers of disorganized myofibrils. HE. Bar = 20 µm.
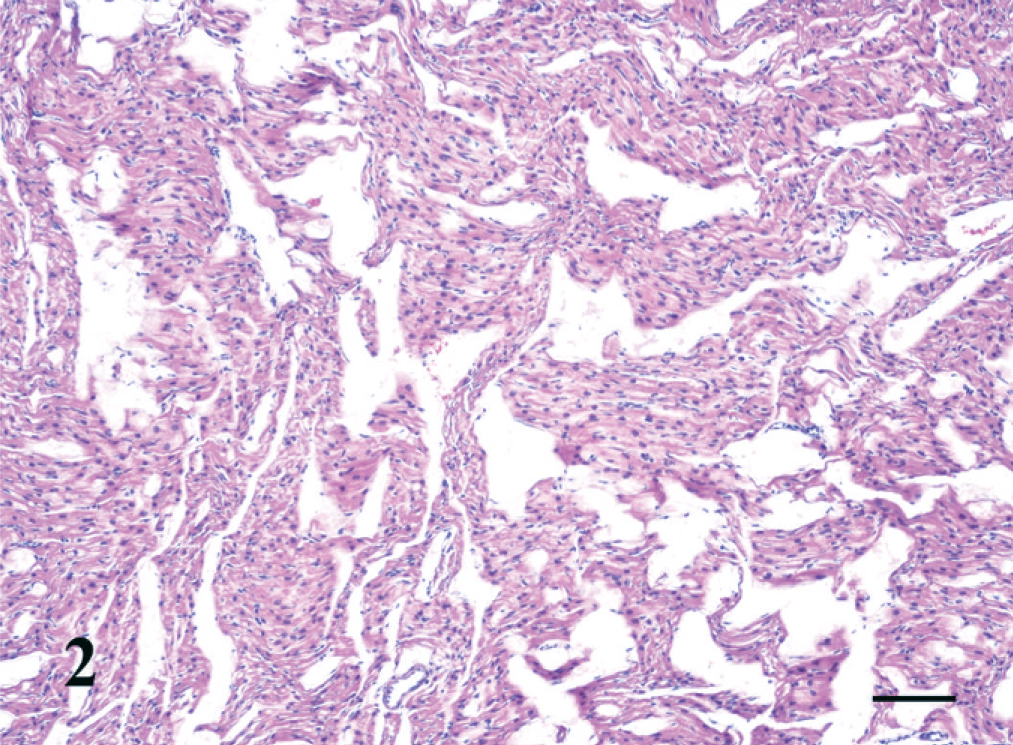
Left ventricular myocardium; kitten. Noncompaction (trabeculation) of the ventricular myocardium is indicated by the interconnecting clear spaces lined by endothelium that separate myocardial fiber bundles. HE. Bar = 100 µm.
Paraffin-embedded myocardium was sectioned at 3–5 µm and placed on positively charged glass slides. Immunohistochemical staining was performed with an automated staining device (Dako Autostainer; Dako, Carpinteria, CA) with Nova Red (Vector Labs, Burlingame, CA) as the chromogen and a hematoxylin counterstain. Antibodies tested were polyclonal rabbit anti-human chromogranin A (1 : 500), monoclonal mouse anti-swine desmin (1 : 50), monoclonal mouse anti-human muscle actin (1 : 50), polyclonal rabbit anti-human myoglobin (1 : 600), and monoclonal mouse anti-rabbit sarcomeric actin (1 : 50), all from Dako. High-temperature antigen retrieval 5 was employed for chromogranin A, desmin, and sarcomeric actin, whereas proteinase K retrieval (5 min) was an added step for muscle actin. Positive control tissues for chromogranin A included normal feline adrenal medulla and intestine (enterochromaffin cells) from the study kitten. Positive control tissues for the muscle antibodies were internal, as well as canine heart, canine intestine, and canine rhabdomyosarcoma. The negative control for all tissues and antibodies was the universal negative control provided by Dako.
Discussion
The kitten's myocardium was immunohistochemically positive for all muscle markers. The abnormal Purkinje fibers were also positive to varying degrees compared with the surrounding myocardium (Fig. 3). The control adrenal medulla and the chromogen cells of the intestine developed dark, granular, cytoplasmic staining with chromogranin A (Fig. 4), whereas the negative controls remained unstained. The histiocytoid cells developed faint but distinct cytoplasmic staining with chromogranin A (Fig. 5); surrounding myocytes were negative.
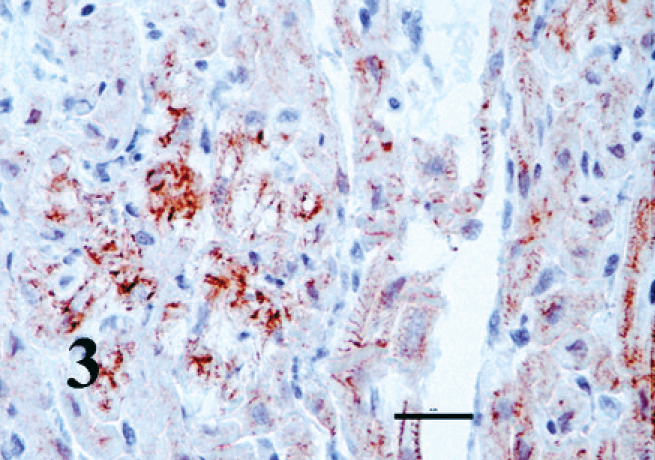
Right ventricular myocardium; kitten. Purkinje fibers (histiocytoid cells) are strongly positive for desmin. EnVision + System HRP. Bar = 20 µm.
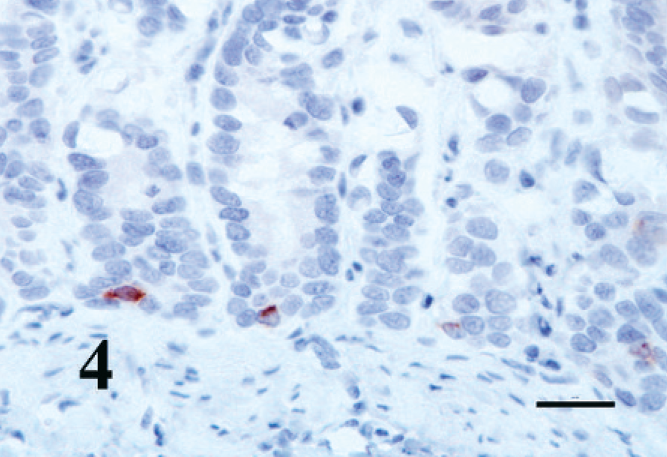
Small intestine; kitten. Enterochromaffin cells in the crypts are positive for chromogranin A. EnVision + System HRP. Bar = 20 µm.
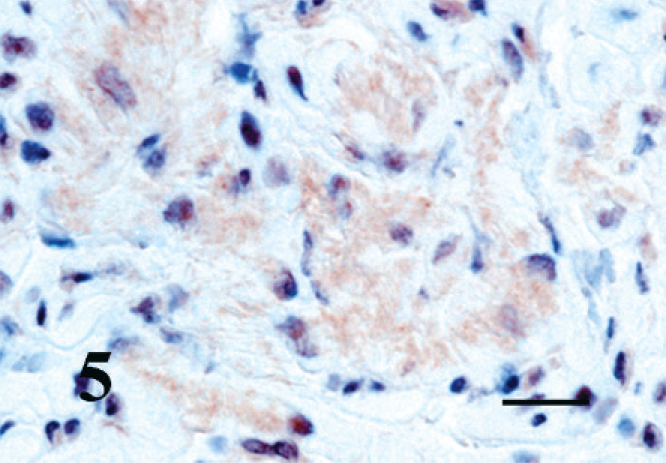
Right ventricular myocardium; kitten. Purkinje fibers (histiocytoid cells) are weakly-positive for chromogranin A. EnVision + System HRP. Bar = 20 µm.
Cubic samples (1 mm) of formalin-fixed ventricular myocardium were postfixed in 2.5% glutaraldehyde and 1% paraformaldehyde in 0.1 M cacodylate buffer, treated with OsO4, dehydrated in an acetone series, infiltrated, and embedded in Spurr's epoxy resin. 24 The blocks were sectioned at 70 nm, placed on 300-mesh copper grids, stained with lead acetate/uranyl citrate, and examined with a Philips CM12 transmission electron microscope (Philips Electron Optics BV, Eindhoven, The Netherlands).
The myocardium was marginally acceptable for ultrastructural evaluation as a result of autolysis. The Purkinje fibers contained few disorganized myofibrils among a rarified cytoplasm. Organelles could not be conclusively identified.
In children, histiocytoid cardiomyopathy is recognized by the presence of multifocal tan nodules or plaques, often within the subendocardial myocardium. In some cases in children, as in this kitten, the lesions are not macroscopic. 22 The histologic appearance of the abnormal cells in this kitten match that described in children. 17,23 These features include a histiocytoid appearance of the Purkinje fibers with few disorganized myofibrils and vacuolated to finely granular cytoplasm. The fibers are immunohistochemically positive for sarcomeric actin, desmin, muscle actin, and myoglobin. Nuclei are condensed and the affected cell aggregates are distinct from the surrounding myocytes. Ultrastructurally, myofibrils are few and disorganized. Other cellular features of histiocytoid cardiomyopathy in children include the presence of numerous mitochondria with abnormal cristae in some cases, prominent Z bands (intercalated discs), and few intercellular junctions. In children, treatments include surgical or chemical ablation of macroscopic, subendocardial, histiocytoid nodules and cardiac transplantation. 23,28
Evidence has accumulated establishing the heart as an endocrine organ. Among the nonneuronal cells within the heart, the atrial and ventricular Purkinje fibers store and release chromogranin A and atrial natriuretic peptides. 25 Thus, the presence of myofibrils and chromogranin A within the histiocytoid cells of this kitten indicates that they are components of dysplastic Purkinje fibers.
Descriptions of histiocytoid cardiomyopathy are absent from the veterinary and comparative literature. However, aggregates of histiocytoid cells have been described and illustrated from a 7-month-old male beagle as an incidental finding in a toxicology study. The authors speculate that this dog might have had histiocytoid cardiomyopathy, and the illustrations are consistent with that supposition. 1 The current report is the first documentation of both histiocytoid cardiomyopathy and noncompaction of the ventricular myocardium in the veterinary literature. Although electrocardiograms were not available from this kitten, it seems likely that death occurred because of a conductive abnormality of the heart, probably ventricular tachycardia, similar to that which occurs in affected children with this syndrome.
Footnotes
Acknowledgements
I sincerely thank Kay Fischer and the personnel of the histopathology section of the Veterinary Diagnostic Laboratory and Teresa Sawyer of the OSU Electron Microscope Facility for their excellent technical assistance. Drs. Christiane Löhr and Beth Valentine are thanked for data regarding the associated submissions, as well as Dr. Kristin Patton and Jill Bartlett for assistance with photomicroscopy.
