Abstract
Dilated cardiomyopathy and ascites in broiler chickens are frequently associated with rapid growth and pulmonary hypertension, but can be associated with some avian leukosis virus (ALV) infections. The novel subgroup J of ALV has a high cardiac tropism, but dilated cardiomyopathy has not been reported previously. We report a dilated cardiomyopathy incidence of 11.1% in broiler chickens congenitally infected with ALV subgroup J (ALV-J). Gross lesions included severe body weight suppression, cardiomegaly with biventricular dilation, right ventricular hypertrophy, visceral congestion, and ascites. Cardiac myocytes and Purkinje fibers contained 2- to 10-μm intracytoplasmic magenta inclusions that contained ALV-J-specific nucleic acid. Ultrastructurally, inclusions contained ribosomes and immature virions and were associated with myofibril disruption and disarray. Peracute centrilobular hepatic necrosis was present in most cases. ALV-J-associated cardiomyopathy may involve a direct viral effect on cardiac myocytes and Purkinje fibers.
Ascites is an economically important cause of mortality that continues to plague the poultry industry despite extensive research. Numerous etiologies contribute to the development of ascites in poultry and can be broadly placed in three categories: pulmonary hypertension, primary cardiac diseases, and cellular damage caused by reactive oxygen species. 5 Pulmonary hypertension is perhaps the most frequent cause of ascites in poultry. This pathogenesis most commonly relates to rapid growth and high metabolic rate inducing physiologic hypoxia, increased cardiac output, and subsequent pulmonary arterial hypertension. Compensatory right ventricular hypertrophy induces right atrioventricular (AV) valve insufficiency and right-sided heart failure. Factors contributing to physiologic hypoxia, such as high altitude, cold ambient temperature, poor ventilation, and respiratory infections, can accelerate this process. 5 Myocardial lesions may also contribute to the development of ascites. In avian species, the right AV valve is a muscular structure innervated by a separate recurrent branch of the bundle of His and a unique structure, the AV Purkinje ring, which encircles the right AV orifice. 11 Myocardial lesions in this conduction system would affect right AV valve closure and thus may contribute to valvular incompetence, heart failure, and ascites.
Cardiomyopathy associated with a retroviral infection is best documented for human immunodeficiency virus (HIV). Lymphocytic myocarditis and dilated cardiomyopathy (DCM) are the most frequent cardiac manifestations of HIV infection. 3,4 Retroviral cardiomyopathy is otherwise a rare entity, but has been observed in chickens infected with the subgroup A avian leukosis/sarcoma retroviruses (ALV). 8,9 During this infection, non–membrane-bound intracytoplasmic ALV inclusions consisting of ribosomes, viral glycoproteins, and immature virions are present in cardiac myocytes and Purkinje fibers, and mature retroviral particles are present in the interstitium. 7 Ultrastructurally, cardiac myocytes exhibit intracellular and organelle swelling with reduction in myofibril number and myofibril disarray. Lymphocytic myocarditis is consistently present in experimentally infected chickens. 8 Similar ultrastructural changes associated with the presence of a retrovirus morphologically resembling ALV have been documented in turkey flocks inbred for a high incidence of DCM, but the viral subgroup in these turkeys was not specified. 16
The novel ALV subgroup J (ALV-J) was first described in 1991, and some initial isolates came from commercial broiler chicken flocks experiencing high mortality due to ascites with retroviral particles in the myocardium. 13,15 ALV-J is best known for its induction of myeloid neoplasia, but it also has a significant cardiac tropism with the heart being one of the most frequently involved tissues during ALV-J infection. 1,2,18 However, DCM has not been described in experimentally infected chickens.
Our method of producing broiler chickens congenitally infected with ALV-J from infected broiler breeder flocks has been described. 17 Fifty-four congenitally ALV-J–infected (32 males, 22 females) and 51 ALV-negative (23 males, 28 females) broiler chickens were produced from eight separate hatches for study of their immune function. The ALV-J status of each chicken was confirmed at hatch using reverse transcriptase polymerase chain reaction for ALV-J genomic RNA isolated from plasma as previously described. 17 Of the 54 congenitally infected broiler chickens produced, 6 (2 males, 4 females) developed fatal biventricular DCM and congestive heart failure for a total cumulative incidence of 11.1%. In contrast, none of the 51 age- and breed-matched ALV-negative broiler chickens developed DCM. Assuming an expected DCM incidence of 1.4% in ALV-negative broiler chickens based on published data for commercial broiler chickens, ALV-J infection is associated with a statistically significant increase in DCM mortality using a chi-square test of independence (P < 0.05). 12 By using the same expected incidence in ALV negative broilers, the relative risk of developing DCM during ALV-J infection would be 8.05, and the expected odds ratio would be 8.93.
The case histories for all six broiler chickens with congenital ALV-J infection and DCM are summarized in Table 1. All had dramatic body weight suppression, as previously associated with congenital ALV-J infection, but to a greater degree than previously published in age- and gender-matched broiler chickens with congenital ALV-J infection. 17 The six affected chickens ranged from 3 to 12 weeks of age, lived at a range of ambient temperatures, and had no apparent seasonal trends. Three of these six chickens died while in Horsfall-Bauer isolation units under optimal temperature and ventilation conditions. Twice as many females as males died of DCM, but this difference was not statistically significant using a chi-square test of independence, possibly because of the overall low case number.
Case summaries for six broiler chickens congenitally infected with avian leukosis virus subgroup J that developed fatal dilated cardiomyopathy.

∗ Expected body weight for an age- and gender-matched broiler chicken congenitally infected with avian leukosis virus subgroup J (ALVJ). 17
† Expected body weight for an age- and gender-matched avian leukosis virus (ALV)–negative broiler chicken. 17
All six broiler chickens with fatal DCM had severe respiratory distress and cyanosis 24–48 hours before death. Gross pathologic changes invariably included cardiomegaly with severe biventricular dilation, moderate right ventricular hypertrophy, diffuse visceral congestion, thickening of the hepatic capsule, and severe distention of the body cavity and pericardial sac with clear, light yellow fluid (Fig. 1). Histologically, the myocardium and epicardium were mildly to severely congested with interstitial edema. Multifocal cardiac myocytes and Purkinje fibers contained single or multiple intracytoplasmic, round to oval, magenta inclusions that were 2–10 µm in diameter (Fig. 2). Inclusions were often arranged in long chains within myocytes, and in clusters within Purkinje fibers. Inclusions were present in cardiac myocytes and Purkinje fibers of both ventricular free walls, the interventricular septum, and the right AV valve. In situ hybridization specific for ALV-J with an antisense riboprobe constructed from the 5′ portion of the ALV-J env gene detected viral nucleic acid in both myocyte and Purkinje fiber inclusions. 18 Haphazard arrangement of fibers, anisocytosis, and karyomegaly with vesicular nuclei and rare nuclear polyploidy characterized cardiac myocytes and were consistent with hypertrophic changes (Fig. 3). Focally extensive acute myocyte necrosis with dystrophic mineralization in the left ventricular free wall was present in chicken No. 4. Five of the six affected chickens had moderate to severe, peracute centrilobular hepatic necrosis consistent with hypoxia (Fig. 4).

Cardiac ventricles; affected chicken No. 2 (right) and an avian leukosis virus–negative chicken (left). The cross section of the heart from chicken No. 2 shows severe biventricular dilation with hypertrophy of the right ventricular free wall and right atrioventricular valve compared to the heart from the age- and gender-matched leukosis–negative broiler. The myocardium of chicken No. 2 is diffusely dark, consistent with congestion and hypoxia.
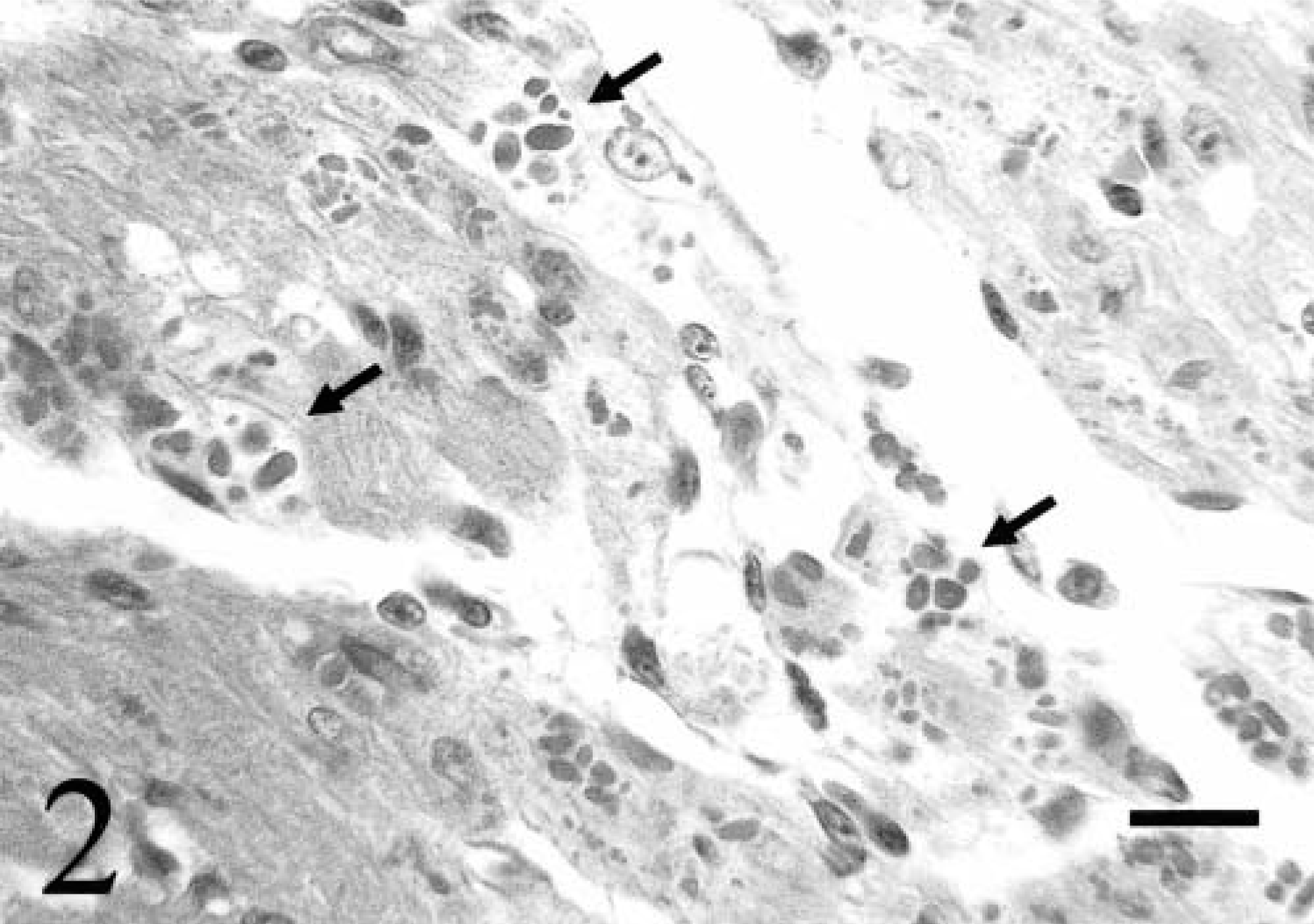
Right atrioventricular valve; chicken No. 6. Numerous round to oval intracytoplasmic inclusions (arrows) are present in cardiac myocytes and Purkinje fibers. HE. Bar = 24 µm.
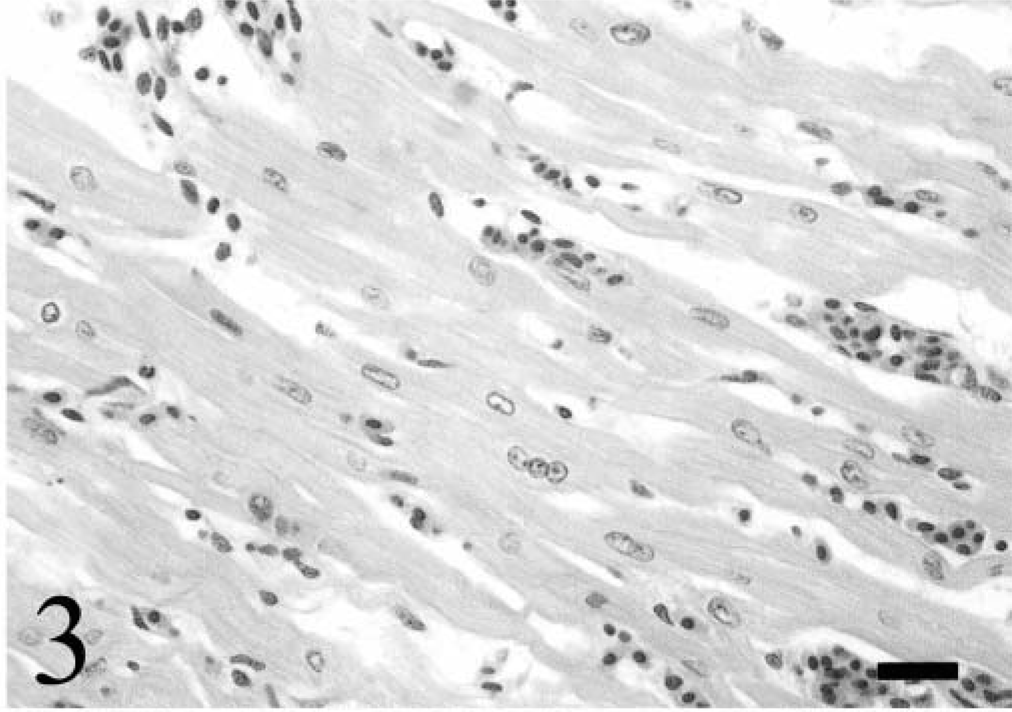
Right ventricular free wall; chicken No. 4. Cardiac myocytes exhibit anisocytosis, anisokaryosis, karyomegaly, and nuclear ploidy. The interstitium is congested and edematous. HE. Bar = 20 µm.
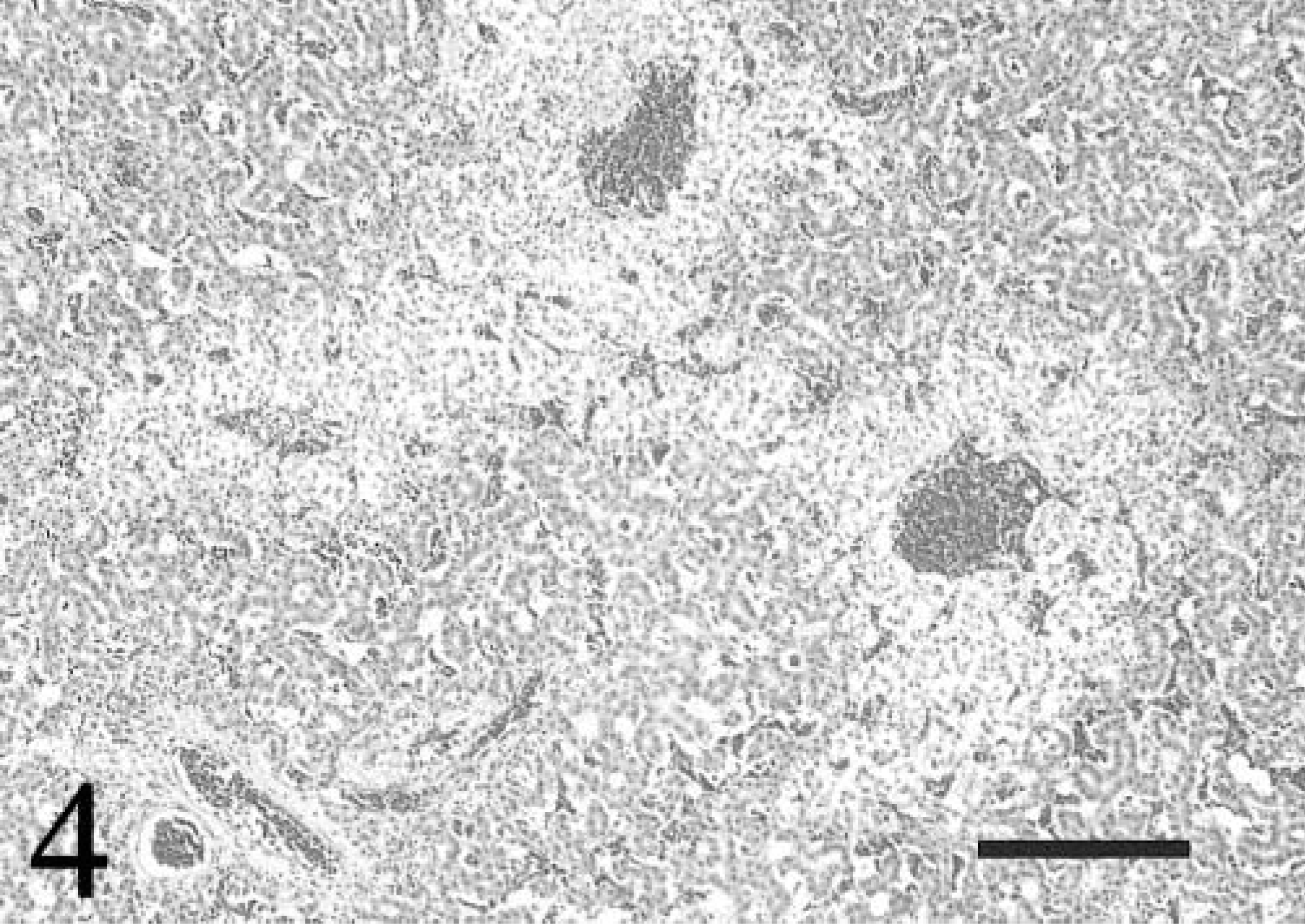
Liver; chicken No. 4. Peracute centrilobular necrosis is evident. The parenchyma surrounding an adjacent portal triad is unaffected. HE. Bar = 40 µm.
The mechanism of retroviral cardiomyopathy is uncertain. During HIV infection, HIV nucleic acid is commonly detected within cardiac myocytes and interstitial dendritic cells. 3,4 Lymphocytic myocarditis and other pathogens, such as coxsackie B virus, cytomegalovirus, and Epstein-Barr virus, are present in some cases. The mechanism of HIV cardiomyopathy may involve a direct viral effect on the myocardium, an immune-mediated reaction induced by the virus, synergistic effects of HIV and other cardiotropic viruses, or a combination of these mechanisms. Because lymphocytic myocarditis also develops during subgroup A ALV infection, the pathogenesis of DCM associated with this infection also may be due to similar mechanisms. 8,9 Lymphocytic myocarditis was not observed in any of the chickens in this study, indicating that ALV-J–associated DCM is more likely due to a direct viral effect on the myocardium and conduction system than to an immune mediated response. We have investigated the ultrastructure of the inclusions in congenitally ALV-J–infected 1-week-old chicks and found a similar composition to those described in subgroup A ALV infection. Inclusions consisted of variable numbers of immature virions surrounded by electron-dense granules consistent with ribosomes and granular, proteinaceous material (Fig. 5). Affected myocytes had intracellular edema, organelle swelling, myofibril fragmentation and disarray, and myelin figures. Accumulation of immature virions, viral proteins, and nucleic acid displaced and disrupted contractile fibrils. The pathogenesis of ALV-J–associated DCM may involve physical disruption of contractile elements within the myocyte. Alternatively, persistent high level synthesis of viral products may usurp cellular machinery and substrates in both myocytes and Purkinje fibers. Insertion of provirus within cytoskeletal or sarcomere protein genes may result in mutations that could manifest similar to the genetic defects in some human hereditary dilated cardiomyopathies. 10,14 However, the exact mechanism of cell injury is unknown. Chickens would likely be more sensitive to viral myocardial damage because the right AV valve is muscular with its own innervation. The presence of ALV-J in the right AV valve may directly impact both its innervation and contractility, resulting in impaired function and DCM.
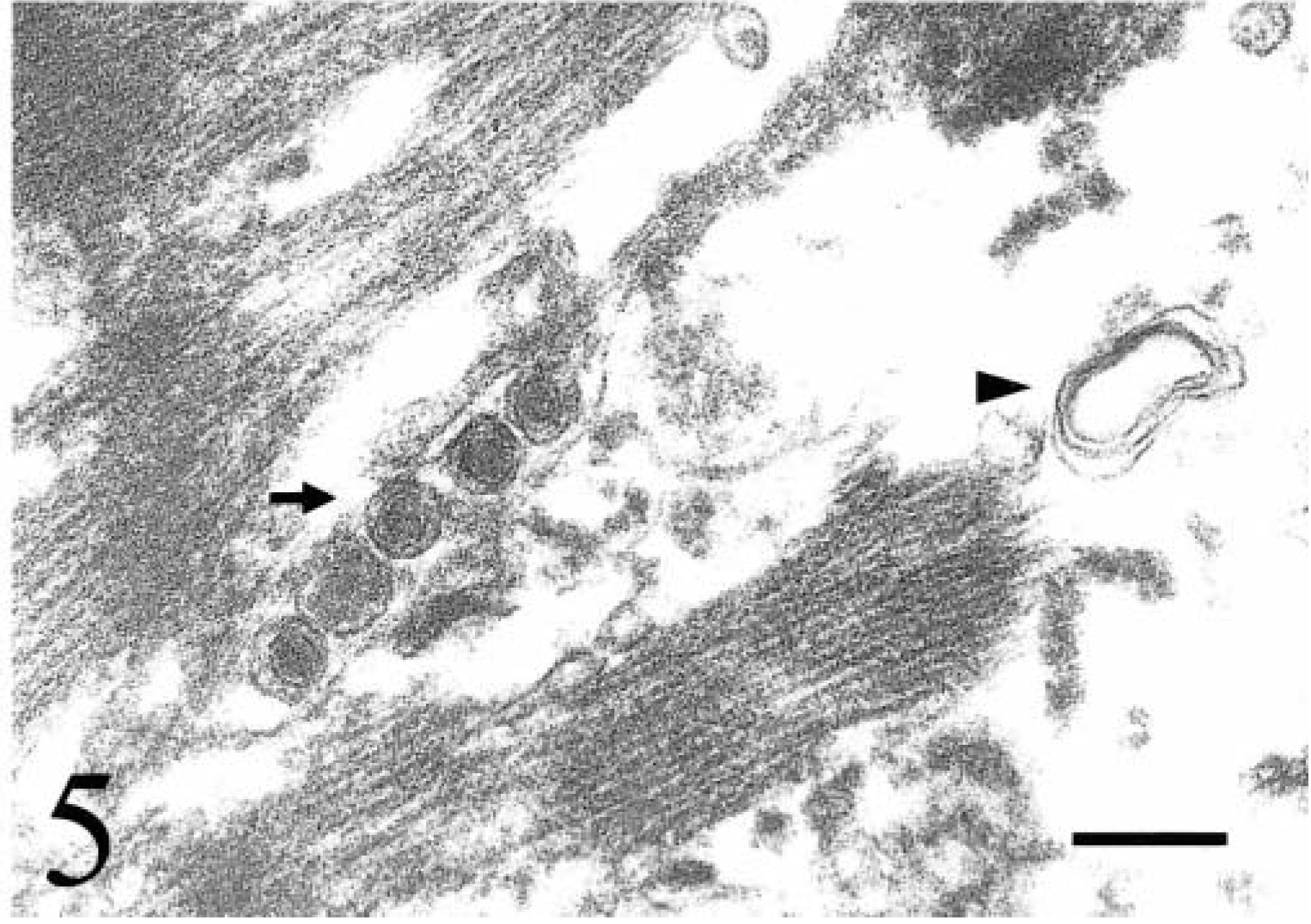
Myocardium; 1-week-old chicken congenitally infected with avian leukosis virus subgroup J. Five immature virions (arrow) with peripheral granular electron-dense material are present among fragmenting myofibrils. Intracellular edema and a myelin figure (arrowhead) are present. Uranyl acetate and lead citrate. Bar = 200 nm.
Subgroup A ALV–associated DCM has been experimentally induced by inoculating 1-day-old chicks. Why ALV-J–associated DCM has not been reported to be associated with experimental ALV-J infection of embryos and 1-day-old chicks is not known. Differences in strain of chicken or developmental stage at the time of exposure to ALV-J may account for this discrepancy. Interestingly, the ALV-J–infected chickens with DCM in this study had more severe body weight suppression than expected for congenital ALV-J infection. Impaired cardiac function may play a role in the pathogenesis of body weight suppression associated with congenital ALV-J infection, analogous to the body weight suppression associated with congenital heart defects. 6
