Abstract
Rhabdomyomas, defined as rare benign tumors of striated muscle, predominantly occur in the myocardium. In veterinary medicine, cardiac rhabdomyomas have rarely been reported but most frequently in pigs as an incidental finding. In the present case report, multiple circumscribed nodules found in the myocardium of 2 pigs were composed of large, vacuolated, glycogen-loaded cells. They expressed vimentin and desmin, neuron-specific enolase, and atrial natriuretic peptide. In addition, these cells expressed protein gene product 9.5, a marker for neuronal tissue and Purkinje fiber cells, in contrast to adjacent myocytes. The present findings—especially, the protein gene product 9.5 immunoreactivity—provide further evidence for the Purkinje fiber cell origin of the porcine myocardial proliferation. Therefore, the term purkinjeoma or purkinjeomatosis is proposed for cardiac rhabdomyoma in pigs.
Keywords
Rhabdomyomas, defined as rare benign tumors of the striated muscle, predominantly occur in the myocardium. They represent the most common primary cardiac neoplasm in children, and spontaneous regression is frequently observed. 10 Although cardiac rhabdomyomas have rarely been reported in veterinary medicine, they occur relatively frequently as an incidental finding in pigs, with red wattle pigs having a predisposition. 7
Regardless of the species, cardiac rhabdomyomas usually occur as multiple circumscribed nonencapsulated nodules consisting of large, vacuolated cells up to 100 μm in diameter. The cells are filled with glycogen and contain some myofibrils. Some cells display features of so-called spider cells, characterized by abundant, centrally located, fine, granular cytoplasm surrounding the nucleus and by thin, elongated projections radiating to the cell periphery. 2,9 Mitotic figures are usually not present. 9
Protein gene product 9.5 (PGP 9.5), also known as ubiquitin carboxyl-terminal hydrolase-1, is frequently expressed by neuronal cells. PGP 9.5 also serves as a reliable marker for Purkinje fiber cells, 1 as shown in various studies investigating the conductive system of the heart in several species, including sheep, camels, and pigs. 1,3
So far, it remains unclear whether cells of the porcine cardiac rhabdomyoma derive from the striated myocardium or Purkinje fiber cells. 2,9 The present case report provides evidence that porcine cardiac rhabdomyoma probably originates from proliferating Purkinje fiber cells; it also proposes new terminology for this cardiac mass based on the provided evidence for the cell of origin.
Case Presentation
Two meat hybrid pigs of about 30 kg, originating from 2 independent fattening units, were euthanized and necropsied. Both animals displayed multiple tan, firm, and discrete nodules up to 2 cm in diameter within the left ventricular wall of the myocardium (Fig. 1). For histology, tissue samples were routinely processed and stained with hematoxylin and eosin (HE). The nodules consisted of large cells, up to 80 μm in diameter, arranged in bundles and streams, with distinct cell borders and an abundant, pale, eosinophilic, partly vacuolated cytoplasm. In addition, some cells exhibited thin, peripherally orientated cytoplasmic streams from a centrally located, finely granular cytoplasm surrounding the nucleus. The cytoplasm was separated by vacuoles, giving the cells a spider cell–like appearance (Fig. 2). Mitotic figures were not observed. In addition, substances interpreted as glycogen were found in the cytoplasm, as based on a positive reaction to periodic acid–Schiff. Phosphotungstic acid–hematoxylin revealed the presence of cytoplasmic cross-striations similar to normal-striated myofibers; few myofibrils were found within the cytoplasm.
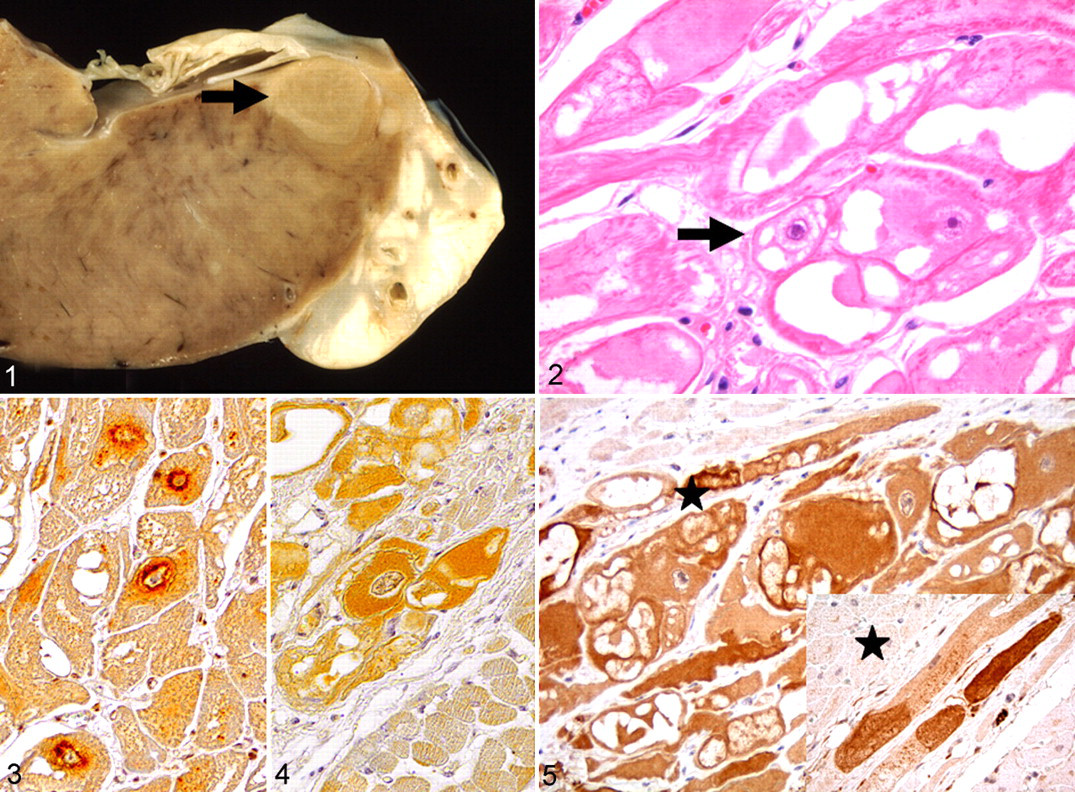
Immunohistochemistry was performed with antibodies directed against desmin, vimentin, neuron-specific enolase, atrial natriuretic peptide and PGP 9.5 (Table 1). The antigen–antibody complex was visualized by the avidin–biotin–complex method with 3,3′-diaminobenzidine as substrate. 8 The rhabdomyoma cells exhibited a strong cytoplasmic reactivity for desmin and a weak one for vimentin, similar to the expression of adjacent myocardial and Purkinje fiber cells. The latter reflects the development of Purkinje fiber cells from multipotent working cardiomyocyte precursors during embryogenesis, from which cardiomyocytes also originate. 1,9 Expression of atrial natriuretic peptide was found predominantly in perinuclear cytoplasmic granules (Fig. 3), whereas immunoreactivity for neuron-specific enolase was found more diffusely, as expected, in rhabdomyoma and Purkinje fiber cells (Fig. 4). 2,3 Furthermore, an intense, homogeneous, diffuse cytoplasmic PGP 9.5 immunoreactivity was observed in rhabdomyoma cells (Fig. 5). Similarly, subendocardial Purkinje fiber cells and myocardial nerve fibers revealed cytoplasmic reaction in the affected animals and control pigs (Fig. 5).
Immunohistochemical Details for the Characterization of the Rhabdomyoma Cells
a Hamburg, Germany.
b Cambridge, UK.
c Hofheim, Germany.
A Purkinje Cell of Origin?
Investigations of the conductive system in the developing heart of camels and sheep have shown that adult and fetal Purkinje fiber cells express PGP 9.5. 1,3 These findings provide substantial evidence for the specificity of PGP 9.5 and for its suitability to identify Purkinje fiber cells. Moreover, features of the rhabdomyoma cells—such as cytoplasmic glycogen storage, sparse cross-striated myofibrils, and immunoreactivity for atrial natriuretic peptide and neuron-specific enolase—resembled characteristic features of Purkinje fiber cells. These findings indicate a Purkinje fiber cell origin of the investigated cell proliferations.
Most investigators have proposed that cardiac rhabdomyoma represents a hamartoma of fetal cardiac myocytes. 5 Other interpretations have included neoplasm of cardiac myocytes or Purkinje fibers, with the suggested name purkinjeoma for the latter, 4 as well as glycogen storage disease, cellular gigantism, and dysplasia. 10 However, a clear distinction among these processes may be difficult. The enlarged and proliferating cells in the present cases displayed common features of Purkinje fiber cells, most notably demonstrated by the widespread immunoreactivity for PGP 9.5. We therefore propose the term purkinjeoma for cardiac rhabdomyoma. Alternatively, the term purkinjeomatosis may be more appropriate, given the similarity to other cell proliferations characterized by multiple benign tumors—lesions such as neurofibromatosis or schwannomatosis in human beings, bovine, and horses. 6
In summary, porcine cardiac rhabdomyomas may be distinguished from other proliferative myocardial lesions by the expression of PGP 9.5 as a specific marker of Purkinje fiber cells, thereby allowing a more precise conclusion about the cell of origin. However, further investigations of additional cases of porcine cardiac rhabdomyomas are needed to confirm the specificity of the PGP 9.5 expression and the proposed terminology.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
