Abstract
Local recurrence of marginally excised subcutaneous soft tissue sarcomas is variable and difficult to predict. This study aimed to identify predictors of local recurrence after excisional biopsy. Medical records of 236 dogs from which tumors had been received between 2004 and 2007 were analyzed. Medium- to large-breed dogs, median age 10 years, were most commonly affected. A total of 139 tumors were graded histologically: 71 were grade 1 (51%); 59, grade 2 (42%); and 9, grade 3 (7%). Of these, 34 tumors (25%) were completely excised, and 104 (75%) were marginally excised. None of 30 completely excised tumors with follow-up information recurred. Three of 41 grade 1 tumors (7%), 14 of 41 grade 2 tumors (34%), and 3 out of 4 grade 3 tumors recurred after marginal excision. Kaplan-Meier survival curves were generated to evaluate survival and the tumor-free interval. The log-rank test and log-rank test for trend were used for comparisons. Tumor recurrence-free intervals for dogs with grade 1 and 2 tumors and for those with grade 1 and 3 tumors differed significantly (P = .0027 and .0001, respectively) and overall were inversely related to tumor grade (P = .0007). Kaplan-Meier survival curves, regardless of recurrence, for patients with grade 1, 2, or 3 tumors treated by marginal excision did not differ significantly, and none differed from the survival curves of patients treated by complete excision. In conclusion, histologic grade is a strong predictor for recurrence of marginally excised subcutaneous soft tissue sarcomas. Clean margins predict nonrecurrence. Tumor recurrence did not significantly reduce survival time.
Keywords
Subcutaneous soft tissue sarcomas (SSTS) comprise a heterogeneous group of neoplasms, which make up approximately 8% of skin tumors. 8 They include malignant peripheral nerve sheath tumors, hemangiopericytomas, liposarcomas, fibrosarcomas, myxosarcomas, perivascular wall tumors, and undifferentiated sarcomas. 1 The morphologic classification of the group is confusing, poorly correlated with histogenesis, and of little predictive or therapeutic value. Immunohistochemical studies have confirmed the complex histogenesis of many of these tumors, leading to the proposal that hemangiopericytoma be considered a diagnosis of exclusion. 1,5 Despite the new-found capability to recognize a variety of these tumors by immunohistochemistry, studies failed consistently to corroborate any morphologic distinction among them, leading to the recommendation that they be diagnosed simply as spindle cell tumors of canine soft tissue. 16
Spindle cell tumors share a number of clinical features, such as local spread along fascial planes, pseudoencapsulation, and slow growth rate; however, their clinical behavior varies widely, with postsurgical recurrence rates that range from 7 to 35%. 4,12 They are commonly treated by surgical excision, especially marginal excision, as a result of dissection through the natural cleavage path of the pseudocapsule and their common occurrence on the limbs, where there is less opportunity to take adequate margins.
No single characteristic of SSTSs has been shown consistently to predict recurrence or to assist materially in the management of these tumors after surgical excision. Mitotic rate and completeness of histologic margins were reported to be related to recurrence in 2 studies, but these findings were not corroborated in a third study. 3,4,11 Mitotic rate and tumor grade were found to be prognostic for the development of metastasis, and grade was prognostic for survival in one of these articles. 11 Immunohistochemical markers have some prognostic potential for survival. 9
Because of their variable recurrence rate and the fact that many SSTSs are marginally excised, they tend to be managed as locally aggressive tumors, which impacts patients in the form of re-excisions, amputations and poor prognosis.
This study examines the relationship between histologic grade, completeness of surgical excision, and excision site recurrence of SSTSs after primary excisional biopsy by surgeons in general practice, in a group of dogs with no adjunctive chemotherapy or radiation therapy and without limb amputation. The objective was to find a reliable and practical means of predicting excision-site recurrence that could be used to assist in the management of patients after marginal excision.
Materials and Methods
Animals from which primary excisional biopsies for subcutaneous soft tissue sarcomas (SSTS) were received by Gribbles Veterinary, Auckland (GVA), between January 2004 and July 2007, formed the base population of dogs for this study. None of the animals were known to have been treated by chemotherapy or limb amputation, and radiation therapy was not available. Tumors, such as hemangiosarcoma, histiocytic sarcoma, and synovial cell sarcoma, which are generally recognizable as a specific tumor type, were excluded because they are normally reported separately from SSTSs and were expected to have a higher rate of recurrence and metastasis. Apart from this, no attempt was made to identify specific tumor types.
Animal data
Age, sex, and breed of the dogs; grade of tumor; site of tumor; whether the tumor was single or multiple; whether or not the tumor recurred and, if so, how long after surgery; time and cause of death; and the type of excision as assessed by histology were recorded. Tumor margins were not inked and were assessed from lateral quadrant and deep sections of round biopsies, and from lateral and deep sections taken through the smallest diameter of elliptical biopsies. Information about tumor recurrence was obtained from computer records held by the veterinary practices that submitted the biopsy specimens for histopathology. Euthanasia was equated with death from the tumor if the reason for euthanasia was related to effects of the tumor.
Evaluation of mitotic index and margins
Histologic sections from all tumors were examined and graded by the author, according to a system adopted from human medicine and used in previous studies of SSTSs of dogs. 2,9,11 The grading system is based on tissue type and differentiation, mitotic rate, and percent necrosis. It was first described in 1984 and was shown to result in 75% agreement between 15 pathologists when tested on 25 cases. 7,15 See Appendix 1 for details. The differentiation score is an overall assessment of both tissue differentiation and histologic type or subtype. The mitotic index (MI) is the number of mitotic figures in 10 contiguous high power fields (hpf) (400×) from the most cellular part of the tumor. Occasionally, more than one area of a tumor with variable cellularity was scanned when selecting contiguous areas was not possible. Necrosis was carefully differentiated from mucinous or hyaline change; hemorrhage; and surgical trauma, e.g., fine needle aspiration biopsy or tru-cut biopsy tracts. The MI was split into 3 categories: 0–5, 6–9, >9. Tumors with histologic margins of ≤1 mm or those that did not include tissue outside the pseudocapsule were considered to have been marginally excised; those with ≥1 mm of tissue beyond the pseudocapsule were considered to have been completely excised. Most tumors fell into one of two categories: those with wide margins, which included connective tissue, fascia, or muscle external to the pseudocapsule, and those with narrow margins (marginal excisions). Four dogs had immediate scar revision to extend “dirty” margins, and those tumor margins were considered, for the purpose of this study, to be “clean” after the second surgery. Another dog had wide-margin excision after a tru-cut biopsy and was then considered to have clean margins.
Statistical analysis
The significance of the relative frequency of tumors for each breed was determined from the odds ratio, which was calculated from the total number of dogs from which specimens were received by GVA for any reason during the period of the study, the number of dogs of a particular breed from which specimens were examined during that time, the number of tumors for a particular breed, and the total number of tumors that occurred during the same time period. Graphpad Prism version 5 software (GraphPad Software, Inc., 2236 Avenida de la Playa, La Jolla, CA 92037, USA) was used to carry out all the statistical analyses. A null hypothesis probability level of .05 was set as the level of significance. All tests of significance were 2-tailed. The relative frequency of breed of dogs with SSTSs was subjected to chi-square analysis. The relationship between histologic grade and MI was analyzed by 1-way analysis of variance (ANOVA) and the Bartlett test for equal variances. The relationship of tumor grade and MI to tumor recurrence was analyzed by the Mann-Whitney test. Overall comparisons of recurrence and survival for tumors of different grades and MI were made by using Kaplan-Meier survival curve analysis. The log-rank (Mantel-Cox) test was used to assess the difference between survival curves, and the log-rank test for trend was used to test for a significant trend. When comparisons were made between 2 curves, the log-rank test and the Gehan-Breslow-Wilcoxon test was used to assess the significance of differences.
Results
SSTSs were most commonly found in medium to large dogs. The following 7 breeds were overrepresented from a total of 236 animals analyzed: Golden Retrievers (n = 15, P = .0001), Staffordshire Bull Terriers (n = 20, P = .0001), Rhodesian Ridgebacks (n = 6, P = .03), Labrador Retrievers (n = 51, P = .0001), Boxers (n = 18, P = .008), Kelpies (n = 6, P = .007), and German Shepherd Dogs (n = 19, P = .047). The median age of 198 dogs was 10.0 years (range, 5–17 years). The anatomical distribution of 143 tumors was equally split between trunk and limbs: 18 (12.5%) were found on the head, neck, and shoulder; 45 on the trunk (31.5%); 39 on the forelegs (27.3%); and 41 on the hind legs (28.7%). There was no anatomical site versus tumor grade interaction for 122 grade 1 and 2 tumors (P > .05, chi-square test). There were too few grade 3 tumors to analyze.
The majority of dogs, for which sex was reported, were females (142/223 [64%]); 101 were neutered. The remainder (81/223 [36%]) were males; 57 were castrated. Most primary surgical excisions (105/139 [76%]) were classified as marginal; 34 (24%) were considered to be complete. Four marginal excisions were revised within 2 weeks of the original surgery to extend the margins and, for the purposes of further analysis, were considered to be clean excisions. Five patients had more than one tumor at different anatomical sites. Three patients had contemporaneous tumors; 2 patients had sequential tumors (12 and 36 months after the first). The median time to recurrence of tumors at the same site was 12 months (n = 15; range, 1–25 months).
A total of 139 tumors were graded by using histology. Of these, 71 (51%) were classified as grade 1, 59 as grade 2 (42%), and 9 as grade 3 (7%). Clinical outcome was reported for 116 tumors excised from 112 dogs for which at least 6 months postsurgery history was available. A postsurgery follow-up period of at least 24 months was available for 50 of the tumors. None of the 30 tumors excised with clean margins recurred, irrespective of grade. Histologic grade was related to MI (1-way ANOVA, P < .0001) (Fig. 1), and both were significantly related to tumor recurrence (log-rank test, P = .0007 and .002; and log-rank test for trend, P = .0002 and .0007, respectively) for marginally excised tumors. The recurrence-free interval for dogs treated by marginal excision, analyzed by MI and histologic grade, is shown in the Kaplan-Meier curves in Fig. 2 and Fig. 3, respectively. The curves of recurrence-free interval for grade 1 and 2 tumors differ significantly (P = .0027). The recurrence-free interval curves for MI 0–5 versus 6–9, MI 6–9 versus ≥10, and grade 2 versus 3 tumors did not differ significantly. The curves for grades 1 and 3, and MI 0–5 versus ≥10 were significantly different (P = .0001 and .0004, respectively), as expected from the figures. Of those tumors treated by marginal excision, 3 of 41 grade 1 tumors (7%), 14 of 41 grade 2 tumors (34%), and 3 out of 4 grade 3 tumors (75%) recurred. The survival curves of patients with grade 1, 2, or 3 tumors treated by marginal excision did not differ significantly, and none of these differed from the survival curve of patients treated by complete excision (Fig. 4). There also was no difference in the survival time of dogs that died from SSTSs and those that died from other causes.
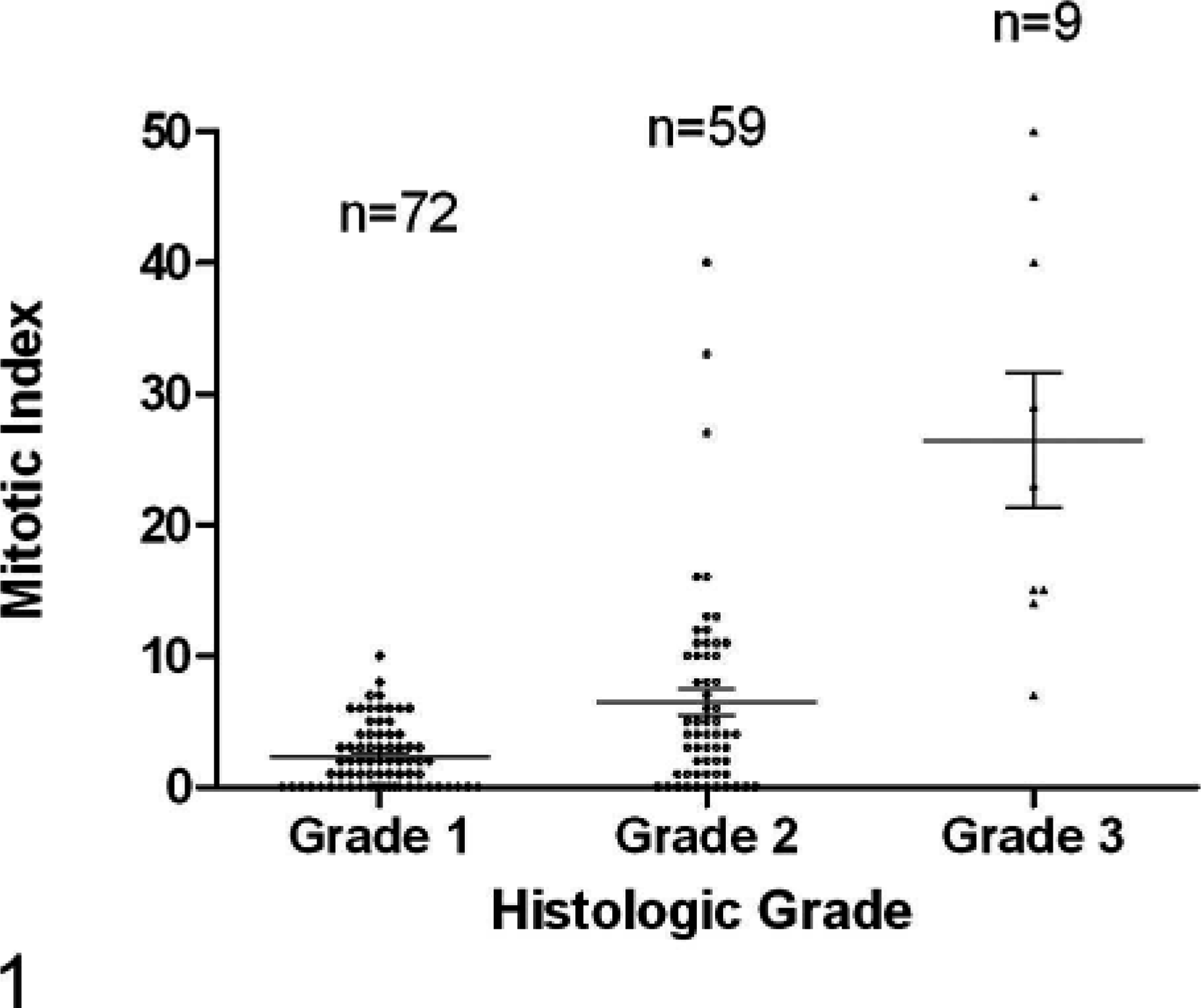
MI is correlated to tumor grade. The scatterplot shows the distribution of MI across the 3 grades. The mean MIs differed significantly (P < .0001) among grades: 1-way ANOVA and the Bartlett test for equal variances.
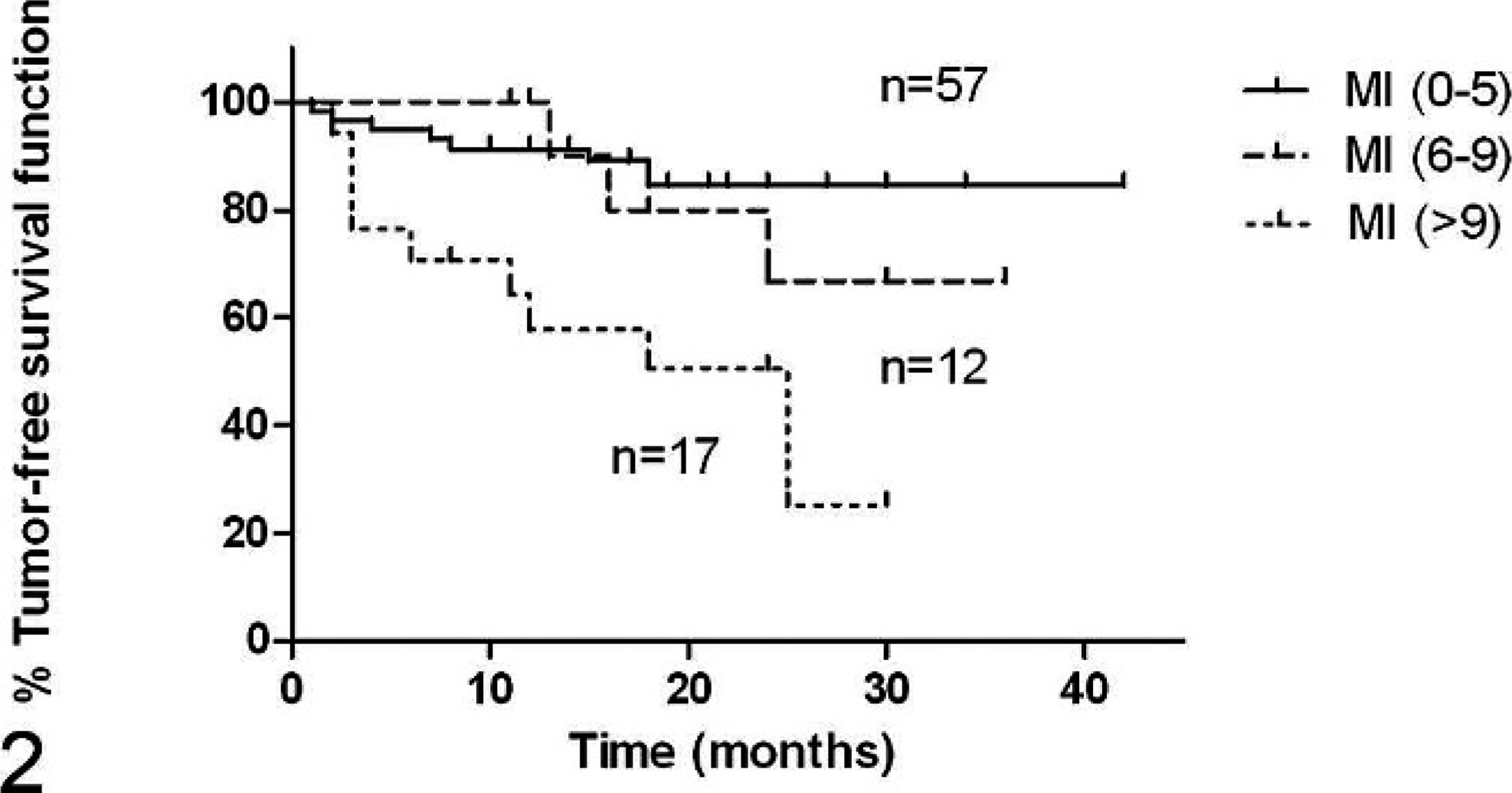
MI is correlated with recurrence-free interval. Kaplan-Meier survival function (P = .002). Paired comparisons: MI 0–5 versus 6–9; P > 0.05 (not significant): MI 6–9 versus >9; ns: MI 0–5 versus >9; P = .0004. A trend relationship was demonstrated (P = .0007).
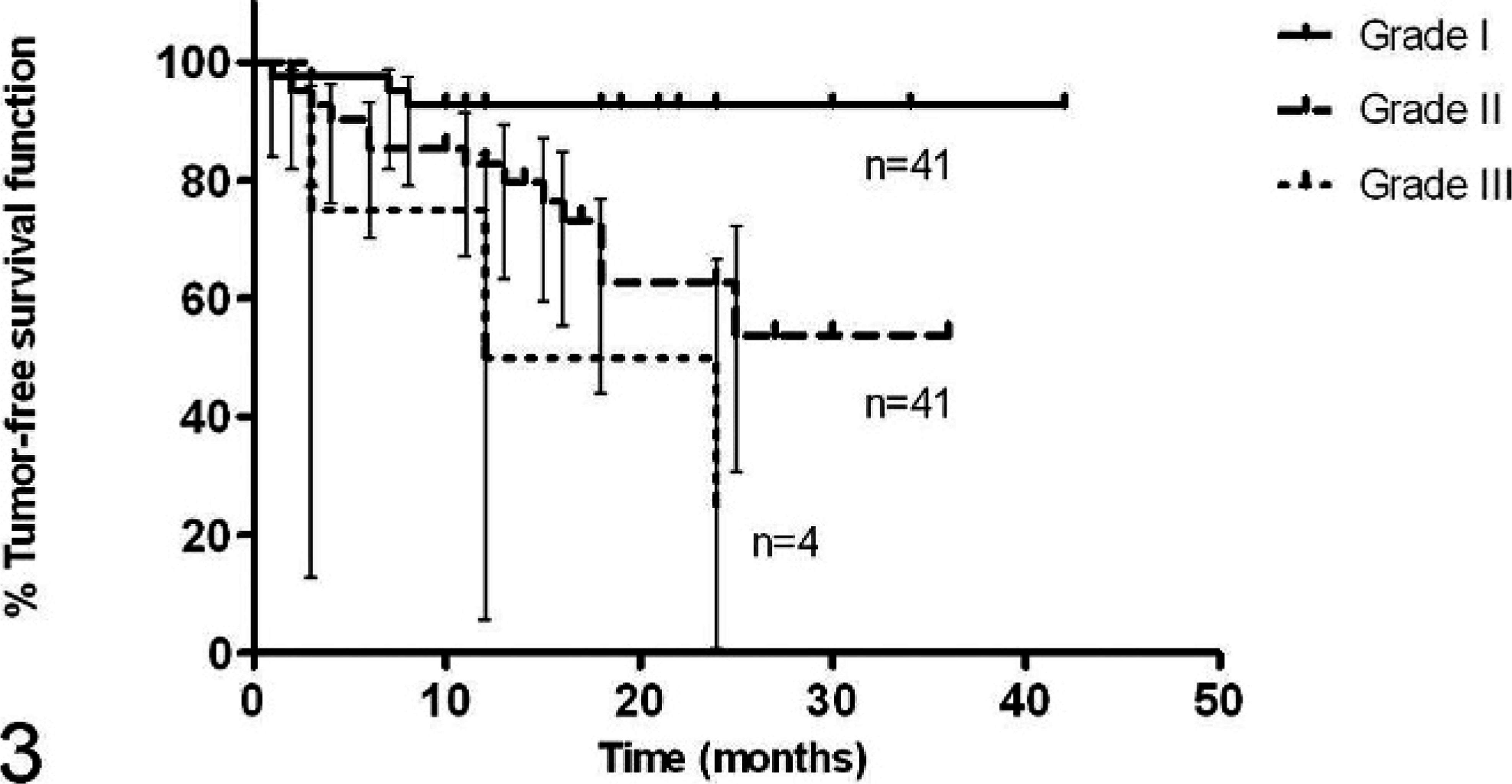
Grade is correlated with recurrence-free interval. Kaplan-Meier survival function (P = .0007). Paired comparisons: grades 1 versus 2; P = .003: grades 2 versus 3; ns: grades 1 versus 3; P = .0001. A trend relationship was demonstrated (P = .0002).
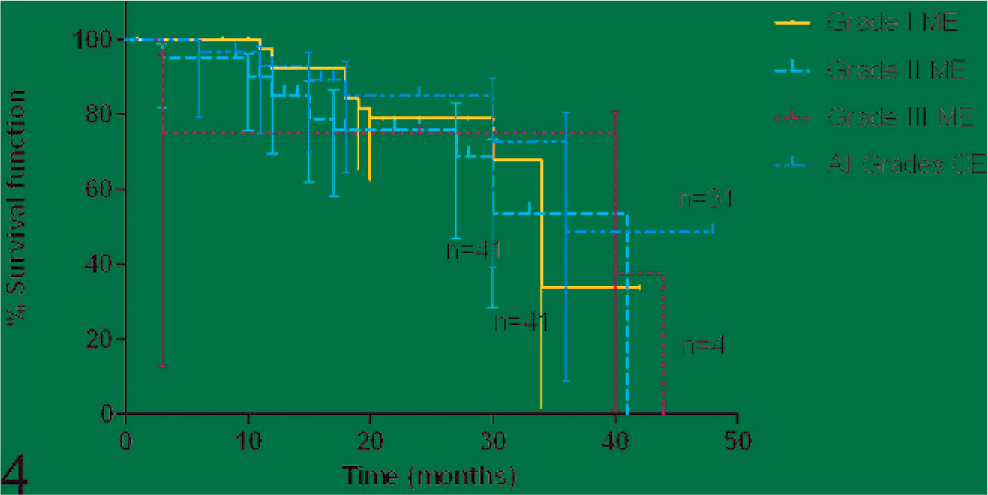
Kaplan-Meier survival function, showing survival of patients after marginal excision of grade 1, 2, and 3 SSTSs and survival of patients after complete excision. The difference among groups was not significant (P > .05). ME = marginal excision, CE = complete excision.
Discussion
The purpose of this study was to identify predictors of recurrence after excisional biopsy of SSTSs. Recurrence was chosen, rather than metastasis, because it is the most common clinical outcome encountered, producing a mass effect at the site of the original tumor or infiltrating to interfere with joint function for those situated over joints. Two predictors were identified. Clean margins were predictive for tumor-free survival, and histologic grade was predictive for recurrence, discriminating between tumors with high, low, and medium rates of recurrence.
Clean histologic margins were the most important prognostic factor for local recurrence. This is consistent with the results of other studies, where cure rates of 38 of 39 (97.4%) and 41 of 42 (98%) were reported after curative-intent surgical excision of primary and recurrent tumors, and 33 of 39 (85%) after scar revision. 2,9,11 Similarly, in humans, the quality of surgical margins was reported to be the most important factor for predicting local recurrence. 7 Whereas margins are accepted as the prime determinant of local recurrence in humans, tumor grade is also considered to be predictive, although not to the extent that it is prognostic for metastasis and survival. 7
Various systems for grading SSTSs have been used; however, apart from an early study in which the mitotic rate of 160 hemangiopericytomas and fibrosarcomas was found to be related to the rate of recurrence, tumor grade has not been significantly correlated with recurrence in animals, although it has been significantly correlated with metastasis. 4,11 This may be explained by the small number of patients with marginal excisions in previous studies, most of which were conducted in universities and specialist referral centers and were focused on curative-intent excision.
Because of the high proportion of marginal excisions in this and other studies that involved primary care surgery, it is important that prognostic indicators of recurrence are established to assist in the management of these cases. The advantages of radical surgery should be weighed against the risks of recurrence for these dogs, many of which are close to their expected life span and may have other health problems. Conservative treatment options are often more attractive to clients in these cases.
MI alone, although strongly correlated with recurrence, did not provide clinically useful discrimination between tumors of relatively low and medium recurrence rates. For example, it was not possible to distinguish the group of tumors with the lowest recurrence rate (9/57 [16%]) from that with the next highest recurrence rate (8/17 [47%]) based on MI, because the 2 recurrence-free survival curves did not differ significantly. However, histologic grade was predictive for recurrence, with grade 1 tumors having a significantly lower rate of recurrence (3/41 [7%]) than grade 2 (14/41 [35%]) or grade 3 tumors (3/4 [75%]). Many clients will not choose re-excision, if this were an option, or amputation, in the case of marginally excised grade 1 tumors, because the rate of recurrence is so low. Residual microscopic disease is no guarantee of local recurrence; distant metastasis is more likely to be related to tumor behavior than surgical margins. 2,10,11 Re-excision may be considered for grade 2 or grade 3 tumors, yet other factors, such as the age and overall health of the patient, site of the tumor, and effect of tumor recurrence on survival, should be considered.
The finding that survival curves for dogs with tumors of different grade are similar is perhaps surprising; however, it reflects the fact that no dogs were euthanatized because of tumor metastasis and that most recurrences were managed locally, irrespective of grade, some by repeated excision, others by debulking. With an average age at diagnosis of 10 years for SSTSs, 1 year beyond the peak of tumor incidence in dogs, and a predilection for tumors to occur in medium-to-large breed dogs with a shorter average life span, many of these dogs have other age-related problems that impact quality of life. 8 Clearly, management of a SSTS needs to be integrated into the overall health strategy for these aged patients. Subjecting a dog near the end of its natural life and with other age-related health problems to further surgery, when tumor recurrence may not materially affect its survival, is questionable.
In practice, many of the aged dogs with tumors are treated conservatively, and most die from nontumor-related causes. In this study, of the dogs known to have died, 7 of 31 (23%) died as a result of an SSTS. This compares with 5 of 20 (9.5%) in a study in Australia, and 25 of 75 (33%) and 12 of 54 (22%) in studies in the United States. 3,9,11
Although tumor grade is prognostic for recurrence of marginally excised tumors, it is not prognostic for patient survival, because many of the grade 1 and 2 tumors are relatively slow growing and can be managed conservatively, so that they do not impact significantly on the patient's quality of life. A previous study did not report on predictors for local recurrence-free interval but found that MI was prognostic for metastasis, and percent necrosis and mitotic rate predicted survival in multivariate analyses. 11
The proportion (105/139 [76%]) of primary surgical excisions in this study that were classified as marginal is similar to the 30 of 37 (81%) previously reported. 2 This high proportion may be attributed to the fact that most excisions (142/211 [67%] in this study and 24/41 [59%] in the previous study) were not preceded by fine needle aspiration, tru-cut, or incisional biopsy. 2 A similar situation exists in human medicine for soft tissue sarcomas, with one study of 100 patients reporting nonconformity to clinical practice guidelines for surgery and histologically positive margins in 48% and 29% respectively, while another reported histologically positive margins in 33/69 (48%) of patients after unplanned resection. 6,14 The findings probably also explain the higher rates of complete excision achieved at specialty practices and university hospitals, where prior biopsy is more common. 3
Other reasons for the high rate of marginal excisions include the ease with which many tumors “shell out” from the surrounding connective tissue through the cleavage planes of the pseudocapsule and the fact that many are on the extremities and cannot easily be excised with wide margins.
The grading system used here is relatively simple, reproducible, and easy to apply. In a recent review, the reproducibility of its 3 components, tissue differentiation, mitotic count, and necrosis score, was tested by 15 pathologists on 25 cases. The crude proportion in agreement for each was found to be higher than that for histologic type (74, 73, 81, and 61%, respectively). 7 Grading systems for SSTSs are not accepted by some pathologists because they are applied to the group, rather than to each particular type; however, subclassification of soft tissue sarcomas is not always feasible in daily practice in human medicine and is likely to be even less so in veterinary medicine. 7
Before grading a soft tissue tumor, it is important to confirm the diagnosis of SSTS and to eliminate such entities as amelanotic melanomas and non-neoplastic proliferative lesions. Each component of the grading score must be assessed carefully to ensure accuracy and consistency, and recounts should be made when indicated, e.g., when mitotic counts are near the cutoff score.
The low number of grade 3 tumors in this study compared with other studies is thought to be related to the fact that the tumors in this study were primary excisions. Many of the tumors in published studies had been previously excised, some more than once, and, perhaps because of their rapid growth and aggressive nature, they were more likely to be treated at university clinics or specialty centers and to be included in published reports.
The anatomical distribution of SSTSs, and the age, sex, and breed of the dogs reported here are consistent with the results of other studies. 3,4,12,13 The median time to recurrence (12 months) in this study compares with 12 months in another study, in which 11 tumors recurred, and 19.5 months in another study, which was based on only 4 animals. 3,11
In conclusion, histologic grade is predictive for recurrence of marginally excised subcutaneous soft tissue sarcomas and has the potential to assist in the management of patients that have been subjected to marginal excision. Complete histologic margins are prognostic for tumor-free survival. Grade 1 and 2 SSTSs do not materially affect the overall survival of patients, many of which die from nontumor-related causes. There were too few grade 3 tumors to draw conclusions with regard to their impact on survival.
Footnotes
Acknowledgements
This study was made possible with the help of the administrative staff of Gribbles Veterinary, Auckland, who assisted with the collection of data, and with the help of many veterinarians and practice staff who good naturedly responded to my persistent telephone calls and questioning. Thanks also go to my colleagues Catherine Harvey, Catherine Williamson, and Jon Meyer for advice and encouragement.
Appendix
Grading system for soft tissue sarcomas.∗
