Abstract
Morphologically, canine soft-tissue sarcomas (STSs) resemble human STSs. In humans, proper classification of STSs is considered essential to improve insight in the biology of these tumors, and to optimize diagnosis and therapy. To date, there is a paucity of data published on the significance of detailed classification of STSs in the dog. We revised a cohort (n = 110) of proliferative lesions obtained from a study in Golden Retrievers that were considered “soft tissue sarcoma, not otherwise specified or of uncertain subtype” in order to optimize the diagnoses of these lesions. The criteria according to the veterinary WHO classification, recent veterinary literature, and the WHO classification for humans were applied. Revision was initially based on morphologic characteristics of hematoxylin and eosin–stained histologic sections of the neoplasms. If considered necessary (n = 76), additional immunohistochemistry was applied to aid characterization. The diagnosis of STS was confirmed in 75 neoplasms (68%). Of this group, diagnosis of a specific subtype of the STSs was possible in 58 neoplasms. Seven neoplasms had morphologic characteristics that were suggestive for sarcoma subtypes only described in the WHO classification for humans. Seventeen neoplasms remained “unclassified STSs.” Thirty-one lesions (28%) were diagnosed “neoplasm, not being STS.” Four lesions (4%) were considered nonneoplastic. Because incorrect classification of a tumor could lead to inappropriate therapeutic intervention and prognostication, the results of our study clearly illustrate the importance of revision and further diagnosis of “unclassified STSs” in dogs.
Introduction
Soft-tissue sarcomas (STSs) form a group of complex diagnostic entities (subtypes)10,12,15 that originate from mesenchymal cells.15,44 Histogenetically, STSs are classified according to the adult mesenchymal tissue they most resemble.15,30,52 However (in dogs as well as humans), STSs that are considered to belong to a single subtype may display a spectrum of morphologic characteristics that show overlap with other subtypes.24,31,45 Some cases can actually appear too undifferentiated to be classified.2,30 This broad morphologic spectrum can cause problems in distinguishing STSs from nonmesenchymal neoplasms16,26 or even nonneoplastic proliferative lesions.45,52
In humans, it is accepted that correct identification of STSs is required in order to better predict the biological behavior of the individual sarcoma subtype and to choose the optimal therapeutic approach.2,7,12,14,30,38,44,46,48,49,52 Advances have been made in the characterization of STS subtypes by applying immunohistochemistry (IHC) in addition to morphologic analysis by examining hematoxylin and eosin (H&E)–stained sections.7,14,29 Revision is essential not only for studies of incidence and etiology but also to ensure that appropriate treatment is selected. 23 Major discrepancies can occur on revision, in particular in cases that have initially been diagnosed as “sarcoma or possible sarcoma” or poorly differentiated mesenchymal neoplasms. 45 These neoplasms may even sometimes be reclassified as nonmesenchymal neoplasms. 45 Discrepancies in diagnosis can sometimes have major therapeutic significance.1,45
Some canine STSs display morphologic characteristics that are very similar to STSs in humans4,11,19,33,40 and, as such, are considered to be a good comparative model.33,43,47 However, in contrast to human STSs, 44 the actual prognostic or therapeutic relevance of detailed subtyping of STSs has not yet been established in veterinary medicine. 8 Thus, detailed histopathologic classification of STS subtype in the dog is often not pursued in routine diagnostics in view of the extra costs of additional IHC. However, human,7,12,24 veterinary, and comparative oncology indicate that verification of the exact nature of a tumor (STS or any another cancer type) is deemed necessary for optimizing the choice of therapy and prognostication. 43 Furthermore, as is already shown in humans,28,37,42 additional classification may serve as a tool to compare results from genomic studies with histogenetic evolution. 40
We evaluated how many neoplasms initially suspected of being unclassified sarcoma would, at thorough revision, obtain a diagnosis with “major discrepancy,” defined as changes in diagnosis that could lead to a significant change in clinical management. It was hypothesized that the routine diagnosis of “presumable STS” is insufficient for proper prognostication, therapy choice, and pathogenetic studies.
Materials and methods
Initial case selection
Our study was performed on a series of archival formalin-fixed, paraffin-embedded samples originally diagnosed as mesenchymal neoplasm. Cases were obtained from the client-owned pet population of the Dutch population of Golden Retrievers submitted to the Veterinary Pathology Diagnostic Centre of the Faculty of Veterinary Medicine in Utrecht, The Netherlands during the period 1998–2004 by the Utrecht University Clinic of Companion Animals as well as by other referral hospitals and private practitioners. This breed was chosen as an annex to a study on the genetic background of STSs in Golden Retrievers. 6 All samples had been fixed in 10% neutral buffered formalin and were submitted as part of the diagnostic procedure in clinical patients. Therefore, no informed consent for the use of this material was required. No specific information on fixation time or of the ratio of tissue volume to volume of the fixative was available. After fixation, samples were routinely processed and embedded in paraffin wax from which 4–6-µm sections were cut and stained with H&E for microscopic examination.
Our earlier search through the archives of 1998–2004 identified 2,124 neoplasms diagnosed by means of histopathology derived from Golden Retrievers. The initial diagnoses of these neoplasms were performed by a number of different board-certified veterinary pathologists as presented in a previous study. 6 In a very limited number of cases, limited IHC was performed (3 cases were stained with S100 and 1 with Masson Fontana to exclude melanoma, 1 was stained with HHF35 anti–muscle actin antibody, and 1 was stained with toluidine blue to exclude mast cell tumor).
Of this group of neoplasms, 110 tumors that were considered (potential) STSs but not further classified were selected for the current study. Median age was 9.3 years; among the cases were 31 males, 19 neutered males, 21 females, and 23 spayed females. Sex of the remaining 16 cases was unknown.
Histopathology and immunohistochemistry
A thorough revision of microscopic morphology of the (potential) STSs (with review of patient data such as location and size of the neoplasm) was performed by a veterinary pathologist (E Hellmén) and a pathologist specialized in human soft-tissue tumors (H Willén). Interpretation was first and mostly based on cellular morphology and growth pattern according to the veterinary WHO classification, 27 recent veterinary literature, and, if considered necessary, with inclusion of the WHO classification for humans. 16 A detailed description of all neoplasms is considered beyond the scope of the present study.
In 76 cases (69%), additional IHC staining was considered necessary for subclassification. Instead of applying a panel of antibodies, specific primary antibodies were selected. The choice of antibodies was guided by the morphologic characteristics of the neoplasms in the H&E stain, and was thus based on commonly used indices such as the morphologic appearance and location or site of the neoplasm.
There is currently no consistent IHC stain or panel of stains that can accurately separate different STS types. 15 Therefore, various antibodies, reported in the literature as being useful in diagnosing STS subtypes in dogs as well as in humans 9 were selected. Selection was based on literature data, as being able to stain the following differentiation markers: S100, an unspecific marker of melanocytic neoplasms and peripheral nerve sheath tumors 25 ; CD31, a vascular endothelial marker 5 ; desmin, striated and smooth muscle cell marker 8 ; Myf4 (myogenin), a striated muscle cell marker 8 ; vimentin, a general mesenchymal cell marker 8 ; AE1/AE3, a cocktail of cytokeratins as an epithelial cell marker 17 ; smooth muscle actin (SMA), a smooth muscle cell marker 25 ; CD117 (c-kit), a stem cell factor receptor 25 ; melan A, a highly specific and low sensitive melanocytic marker 41 ; and antibodies staining CD34 (antibody C-18), a marker of hematopoietic progenitor and endothelial cells. 20 Also, Ki67, a proliferation marker, 20 and CD18, a general leukocyte marker, 34 were used. All antibodies used in the present study showed cross-reactivity with canine tissues.
IHC was applied on representative 4–6-µm sections of the selected cases. In all cases, 1 or more samples of appropriate canine positive control tissue was included for validation because information on cross-reactivity was not always available from the supplier or published literature. Parallel-stained sections without incubation with the primary antibody were used as negative controls.
Antigen retrieval pretreatment was carried out using antigen unmasking solution (Vector) or target retrieval solution, citrate pH 6 (Dako). For detailed information on each individual antibody, see the section “Sources and manufacturers.”
Immunohistochemical assessment
Immunoreactivity was assessed by light microscopy for each antibody. Immunoreactivity for S100, a CD31, b desmin, c vimentin, d AE1/AE3, e SMA, f c-kit, g melan A, h Myf4, i CD34, j and CD18 k was graded as follows: – negative, +/– (weakly) positive, or + positive in terms of number of positive cells. The staining intensity of the neoplastic cells was weak, moderate, or strong, and usually a continuum was seen within the positively labeled tumors. Cases that only had a nuclear staining for CD34 were considered to be negative. The percentage of cells revealing immunoreactivity for Ki-67 l was scored as low, intermediate, or high.
Results
In 34 neoplasms (17 of which were considered STS of a specific subtype), the review of H&E sections on the presence of sufficient morphologic features was considered conclusive, enabling a diagnosis based on the characteristics described in the veterinary literature, without the requirement of additional IHC. In all other neoplasms, additional IHC was required. The results of the revised classification of the 110 (potential) STSs are shown in Table 1. Supplemental Table 1 (available at http://vdi.sagepub.com/content/by/supplemental-data) provides detailed information on all diagnoses made.
Distribution after revision of 110 neoplasms “sarcoma, not otherwise specified or of uncertain subtype” (i.e., unclassified sarcomas) following veterinary WHO classification.*

STS = soft-tissue sarcoma.
As a final result of the revision, 58 neoplasms out of the total of 110 could be classified as a specific STS subtype. Within this group, the largest number of neoplasms (n = 20) was considered to belong to the group of “malignant tumors of fibrous tissue.” Five neoplasms within this group were considered fibrosarcomas, based on morphologic appearance at primary review or after negative staining for S100, desmin, CD31, and AE1/AE3. Seven were considered myxosarcoma, based on morphologic appearance, and, in 1 case, negative staining for S100 and CD34. One tumor was diagnosed as an anaplastic sarcoma with giant cells (previously named malignant fibrous histiocytoma [MFH] in the veterinary WHO classification). A group of 7 neoplasms in this fibrous tissue group could not be further subclassified based on the veterinary WHO classification or the veterinary literature. Still, these neoplasms did show typical morphologic characteristics that were suggestive of neoplastic entities described in humans. Therefore, the human WHO classification was used in an attempt to further classify these neoplasms, resulting in a diagnosis of dermatofibrosarcoma protuberans–like (n = 4; Fig. 1.1, 1.2). These tumors are regarded as well differentiated fibrosarcomas of the skin. The overlying epidermis is often thin. The growth pattern can be described as storiform. These tumors in our study were negative for CD34, and 1 tested tumor was intermediate positive for Ki-67. Desmoid fibromatosis (n = 1; Fig. 1.3, 1.4) is a slow-growing lesion that infiltrates the surrounding tissue but does not metastasize. The tumor in our study consisted of elongated cells with a fibrous stroma and with a low staining for Ki-67. Two neoplasms that stained negative for CD34 and negative for CD18 were regarded as belonging to the group of “tumors of fibrous tissue,” but could not be further classified.
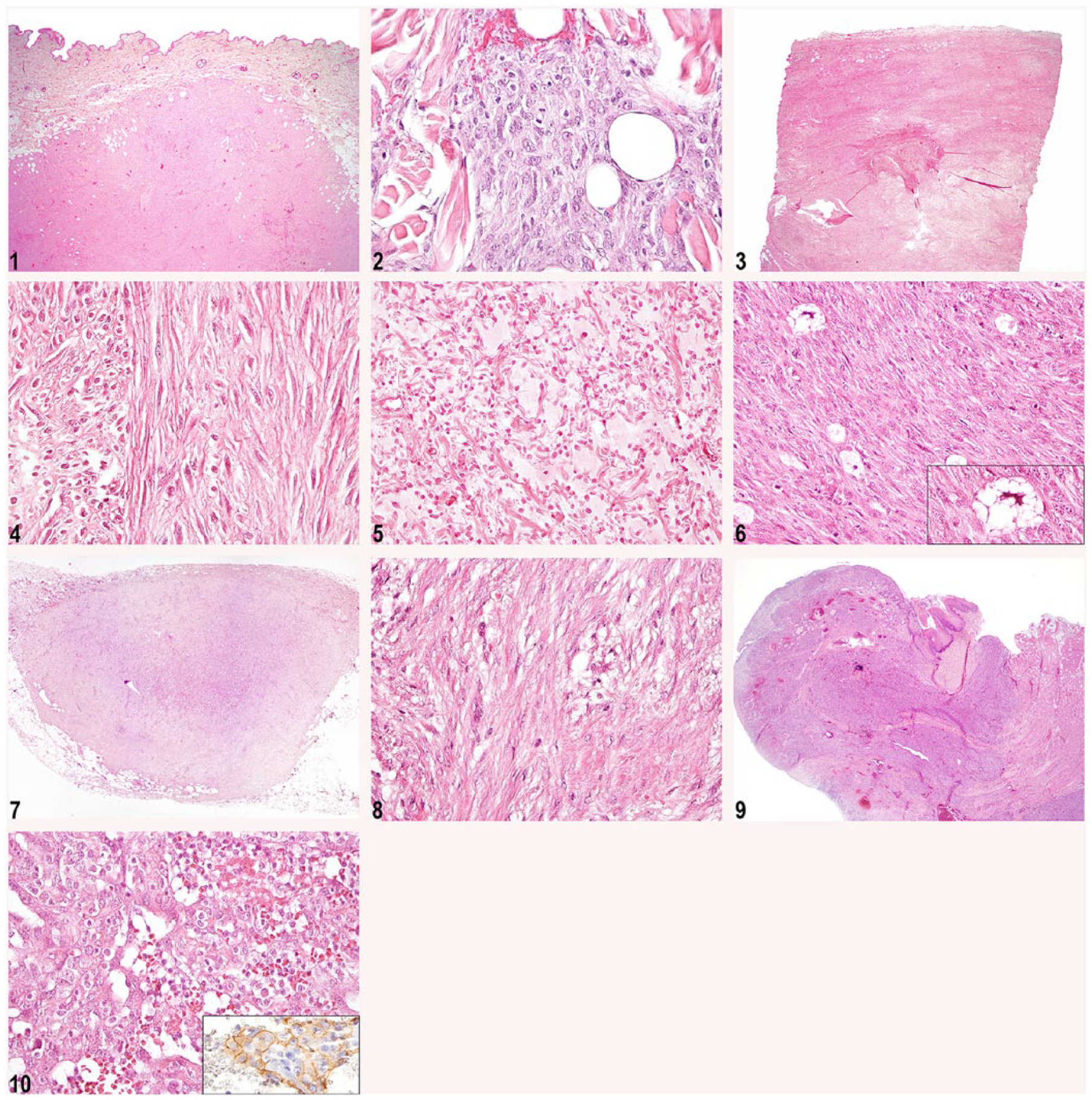
Exemplary neoplasms, in which subclassifications were possible, surpassing the current veterinary WHO classification of mesenchymal tumors.
A further group was categorized as “malignant adipocytic tumors” (n = 8). Further classification following the veterinary WHO classification 27 could be made into well-differentiated liposarcoma, myxoid liposarcoma (n = 2; Fig. 1.5), and pleomorphic liposarcoma (n = 1; Fig. 1.6). In addition to morphologic appearances, the diagnosis of pleomorphic liposarcoma was based on negative staining for desmin. The remaining liposarcomas (n = 4) could not be classified based on morphologic appearance as one of the above-mentioned sub-subtypes of liposarcomas currently recognized in the veterinary literature. One case was considered to be a round cell liposarcoma. This diagnosis is not included in the veterinary WHO classification, but has been described previously in dogs. 50
The remaining 3 neoplasms showed morphologic characteristics consistent with a (well-differentiated) sclerosing liposarcoma, a dedifferentiated liposarcoma, and a mixed-type liposarcoma, subtypes only described in the current human classification. 16 The sclerosing liposarcoma was characterized by rare multivacuolated lipoblasts in an extensive fibrillary collagenous stroma (Fig. 1.7, 1.8).
Five neoplasms were diagnosed as hemangiosarcomas (following the current veterinary WHO classification) based on morphologic characteristics. In 3 of these cases, additional staining for CD31 was performed, and all 3 neoplasms were found to be positive. One neoplasm was subclassified as an epithelioid angiosarcoma, a subtype that is not a part of the veterinary WHO classification, but which is already recognized in the veterinary literature. 51 The epithelioid angiosarcoma was characterized by large rounded epithelial-like endothelial cells with large nuclei. This tumor was also found positive for CD31 (Fig. 1.9, 1.10).
Even after extensive revision, 17 neoplasms of the total of 110 remained as “unclassified STS.” Six neoplasms, initially considered “undifferentiated,” were eventually diagnosed as histiocytic sarcoma (referred to as malignant histiocytosis in the veterinary WHO), based on histomorphologic hallmarks combined with positive immunoreactivity for CD18.
Thirty-one neoplasms out of the total of 110 were reclassified as “neoplasm, non-STS.” The most frequently encountered within this group were round cell neoplasms that showed morphologic characteristics of malignant lymphomas (n = 4), osteosarcomas (n = 10), malignant melanomas (n = 2), and 1 neoplasm that was suspected to be a melanoma. The cell of origin was confirmed in the melanocytic neoplasm only. No additional IHC was performed to substantiate the revised diagnosis of lymphomas and osteosarcomas. Furthermore, benign mesenchymal neoplasm was diagnosed in 4 cases, as was 1 case each of anaplastic carcinoma, complex adenoma, and carcinosarcoma. These 3 tumors most probably originated from the mammary glands. The complete list can be found in Supplemental Table 1. The following 4 proliferative lesions were reclassified as nonneoplastic: ulcerating inflammatory skin; lesion of uncertain type; granulation tissue; and autolytic, not diagnostic.
Discussion
In humans, diagnosing STSs can be difficult, and major discrepancies on review are known to occur.3,45 Major discrepancies are defined as changes in diagnosis that could lead to a significant change in clinical management, causing either under- or overtreatment. 45 These concern changes such as a change from neoplastic to nonneoplastic, from malignant to benign, or from mesenchymal to nonmesenchymal. Minor discrepancies are changes in which the discrepancy is not thought to provoke significant management change. 45 In general, STSs carry a high risk for diagnostic errors, 3 and some subtypes are even more prone to misdiagnosis than other subtypes.24,45 There are several reasons for the discrepancies to occur. In some cases, nonneoplastic mesenchymal neoplasms strongly resemble STSs.3,39 Furthermore, differences in interpretation of morphology can occur. 45 Also, the rare nature of a lesion 3 and difference in the degree of experience in differentiating sarcoma cases between pathologists can cause this discrepancy.3,45
Among the neoplasms originally diagnosed as unclassifiable STS and revised in our study, major discrepancies occurred. On revision, 31 of such neoplasms (28%) were identified as being “neoplasm, non-STS.”
In humans, revisions of STSs have resulted in discrepancy rates (including both major and minor discrepancies) of ~27%. 45 In the current study, the proportion of major discrepancies is substantially higher compared to human studies. This likely has to do with the type of lesion that was chosen for revision. In our study, focus was put on a select group of neoplasms that were considered either (potential) STS of undifferentiated type, or neoplasms for which several possible subtypes were indicated, whereas the discrepancy rate in the previous study was the result of a (retrospective) revision of a random group of STSs collected over a certain period, including differentiated subtypes, excluding second opinion cases. 45
In our series, there were 76 cases in which additional IHC needed to be performed to reach a diagnosis. Specific antibodies were used based on morphologic characteristics of the neoplasms and the location within the body, instead of a broad panel of antibodies applied to all neoplasms. This approach was chosen because of financial limitations, with the current approach for materials costing ~20 euros (~$22), whereas a broad panel applied to every case would have cost at least 5 times more. In human sarcomas, lack of familiarity of the pathologist with rare neoplasms or those with unusual morphologic appearances is probably more significant in explaining diagnostic discrepancies than absence of IHC.3,45 In these human studies, major discrepancies were identified solely based on H&E-stained slides; additional immunostaining had not been applied for this purpose. In order to be able to identify major discrepancies, other factors, such as the amount of experience in evaluating this specific group of lesions, appeared more important than additional immunostaining. 45
In the current study, even after extensive revision, a subclassification could not be made in 15% (n = 17) of the 110 neoplasms (i.e., 23% of the 75 STSs). Problems in subclassifying STSs are not uncommon in dogs or humans.10,11,15,19,21,36 In our study, the problem was most likely that these tumors were poorly differentiated and thus lacked the lineage markers that were used. The lack of morphologic and molecular characteristics can hamper classification into a subtype10,11,15,19,36,45 even when a large panel of markers is applied.4,11,21 However, the choice not to apply a uniform broad panel of antibodies for all suspected STSs could be considered to be a limitation of this study, as inclusion of a broad panel of appropriate antibodies might have led to subclassification of a larger number of neoplasms. However, because most of these tumors were poorly differentiated, this might not necessarily be the case.
The use of some of the selected antibodies yielded technical problems. These occurred either because of the absence of the expected staining in positive control tissues or because the stain was too unspecific because of the lack of interspecies cross-reactivity. This was the case when using HMB45, m an antibody that stains a glycoprotein present in premelanosomes (clone M634), 32 as well as MyoD1, n a marker of myogenic cells (clone 5.8A). 18 Despite testing these antibodies in several tissues (melanoma, rhabdomyosarcoma, striated muscle, respectively) no specific staining could be seen, likely because of lack of interspecies cross-reactivity.
At the time of writing, an antibody raised against canine CD34 has become commercially available (clone 1H6). However, this canine antibody was not available at the start of this study, which is why a different antibody that is known to cross-react with canine tissue was selected. 41
One neoplasm was presumed to be a gastrointestinal stromal tumor (GIST) based on morphologic features and the location of the tumor within the intestines. However, the c-kit staining was negative. Because immunoreactivity for c-kit is considered essential for a tumor to be diagnosed a GIST 25 and the neoplasm did show immunoreactivity for desmin, the tumor was diagnosed as a leiomyosarcoma.
Most STSs within this study belonged to the group of “malignant tumors of fibrous tissue” (n = 20). Additional subclassification following the veterinary WHO classification and more recent veterinary publications on this group of neoplasms proved difficult in approximately one-third of the neoplasms belonging to this group (n = 7). Four of these neoplasms showed morphologic characteristics that were considered similar to a human entity named dermatofibrosarcoma protruberans. According to current (human) WHO classification on the pathology and genetics of skin tumors, where this neoplasm is considered part of the fibrous, fibrohistiocytic, and histiocytic tumors, the diagnosis can be made on morphologic appearance and a positive staining for CD34. The identification of a characteristic fusion gene (t(17;22) (q22;q13)) translocation is, according to the (human) WHO, considered a hallmark of this tumor. In our study, the morphologic appearance was considered characteristic for a dermatofibrosarcoma protuberans. Because the identification of the presence of a possible fusion gene is beyond the scope of this study, neoplasms were tentatively classified as dermatofibrosarcoma protuberans–like. It would be of additional value to evaluate the presence of such a fusion gene within these neoplasms and possibly finding additional means for treatment that focuses more on the exact nature of this neoplasm.
One neoplasm within the group of “malignant tumors of fibrous tissue” was considered a MFH. In veterinary literature, the name MFH is considered controversial. A recent change to “anaplastic sarcoma with giant cells” 22 has been suggested, as the name MFH is considered a purely descriptive term.21,22
Within the groups “malignant adipocytic tumors” and “malignant vascular tumors,” the sclerosing liposarcoma and epithelioid angiosarcoma, respectively, are neoplasms that are not documented in the current veterinary WHO classification. These diagnoses were, however, previously described in the current veterinary literature. Atypical lipoma, characterized by sclerosing areas, has previously been described in dogs. 22 It is considered to be a low-grade malignant liposarcoma, recapitulating the features of an atypical lipomatous tumor of humans, resembling sclerosing liposarcoma of humans. 22 Sclerosing liposarcoma is reported as a variant of the atypical lipomatous tumor/well-differentiated liposarcoma in the WHO classification of soft-tissue tumors in humans. “Epithelioid angiosarcoma” has been described in dogs in previous studies.5,51 For this sarcoma subtype, as for the aforementioned MFH, 35 the Golden Retriever is likely to be predisposed. 51
Two tumors were diagnosed as synovial cell sarcomas (SCSs) based on expert consensus on morphologic features. One of these tumors stained positive for the cytokeratin antibody AE1/AE3, a staining known to often be positive in SCSs 13 ; however, the less differentiated tumor was negative. There can, however, be some concern about the diagnosis. Histiocytic sarcomas can sometimes be mistaken for a SCS. 34 Additional staining for CD18 would have been of additional value for classification of these 2 tumors. However, insufficient material had remained to perform this staining.
In total, 7 neoplasms could not be classified following the veterinary WHO classification. This was the case with the (well-differentiated) sclerosing liposarcoma, the epithelioid angiosarcoma, the 4 cases of dermatofibrosarcoma protuberans–like neoplasms, and the desmoid fibromatosis. To our knowledge, the latter 2 have not been described in recent veterinary literature. Although we noticed similarities with the human WHO classification, the low number of neoplasms and the absence of information on the biological relevance needs further investigation before introduction of new types of neoplasms for dogs is warranted. The cases are too few to form a separate report, and our aim is merely to draw attention to potential further diagnoses of canine STSs. These cases are new to the currently used classification, and are depicted in Figure 1.
Our study combined a revised morphologic analysis with IHC in a collection of 110 unclassified (potential) STSs. This approach allowed confirmation of the diagnosis of STSs and additional subclassification of 58 cases (53%) following the veterinary WHO histologic classification of mesenchymal tumors of skin and soft tissues of domestic animals. In 17 cases, (15%) revision did not result in subclassification and these neoplasms remained “unclassified STSs.” A large percentage (32%) of the remaining proliferative lesions appeared to be misdiagnosed, and these were considered to be “neoplasm, non-STSs” (31 cases), or even “nontumorous” (4 cases). This audit illustrates that a substantial percentage of major misdiagnoses were made within the original group of neoplasms that were initially diagnosed as STSs. To avoid an incorrect treatment, routinely applied additional diagnostic techniques such as IHC in this group of neoplasms is strongly advisable. Also, because of the potential of dogs as a translational animal model and the proven additional value of additional classification in humans, an improved classification of canine STSs can be beneficial for both research and clinics.
Footnotes
Acknowledgements
The staff of the UVDL (Universitair Veterinair Diagnostisch Laboratorium) is thanked for their help and expertise. Also, Lonneke Boon is thanked for her active participation in this study.
Authors’ note
Kim M. Boerkamp and Eva Hellmén contributed equally to this work.
Authors’ contributions
KM Boerkamp, GCM Grinwis, and GR Rutteman contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. E Hellmén and E Teske contributed to conception and design of the study, and contributed to analysis and interpretation of data. H Willén contributed to design of the study, and contributed to analysis and interpretation of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
S100, rabbit polyclonal, dilution: 1:1200, Z0311, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, Dako citrate (UltraVision LP system, Thermo Fisher Scientific BV, Breda, The Netherlands; as well as VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: peripheral nerve; lineage marker: cells derived from the neural crest.
b.
CD31 (PECAM-1, lot no. D2209), goat polyclonal, dilution: 1:200, sc-1506, Santa Cruz Biotechnology Inc., Santa Cruz, CA; pretreatment and technique: unmasking, Dako citrate (Dako Sweden AB, Stockholm, Sweden; VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: mammary gland; lineage marker: platelets, monocytes, neutrophils, some types of T cells and endothelial cells.
c.
Desmin, mouse monoclonal, IgG1, dilution: 1:500, M0760, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH (UltraVision LP system Thermo Fisher Scientific BV, Breda, The Netherlands; as well as VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: mammary gland; lineage marker: skeletal muscle, smooth muscle and cardiac muscle cells.
d.
Vimentin clone V9, mouse monoclonal, IgG1, dilution: 1:500, M0725, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH, VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: mammary gland; lineage marker: mesenchymal cells, myoepithelial cells.
e.
Cytokeratin (clones AE1/AE3); mouse monoclonal, IgG1, dilution: 1:25, M3515, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH (UltraVision LP system Thermo Fisher Scientific BV, Breda, The Netherlands; as well as VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: mammary gland; lineage marker: epithelial cells.
f.
SMA clone 1A4, mouse monoclonal, IgG2a, dilution: 1:200, M0851, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH (UltraVision LP system Thermo Fisher Scientific BV, Breda, The Netherlands; as well as VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: mammary gland; lineage marker: myofibroblasts, smooth muscle cells, myoepithelial cells.
g.
c-kit (CD117), rabbit polyclonal, dilution: 1:400, A4502, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH, VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: mastocytoma; lineage marker: hematopoietic stem cells, multipotent progenitors, and common myeloid progenitors.
h.
Melan-A (clone A103), mouse monoclonal, IgG1, dilution: 1:50, M7196, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH, VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: melanoma; lineage marker: protein antigen that is found on the surface of melanocytes.
i.
Myf4 (clone L026), mouse monoclonal, IgG1, dilution: 1:25, Novocastra NCL-L-Myf-4, Leica Biosystems GmbH, Wetzlar, Germany; pretreatment and technique: unmasking, high pH, VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: alveolar rhabdomyosarcoma; lineage marker: skeletal muscles.
j.
CD34 (C-18, lot no. F2111), goat polyclonal, IgG1, dilution: 1:400, sc-7045 Santa Cruz Biotechnology Inc., Santa Cruz, CA; pretreatment and technique: unmasking, Dako citrate (Dako Sweden AB, Stockholm, Sweden; VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: ovary (CL); lineage marker: early hematopoietic and vascular-associated cells.
k.
CD18, mouse monoclonal anti-CD18 (clone CA16.3C10), dilution: 1:40, Dr. P.F. Moore, Davis, CA; pretreatment and technique: VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: histiocytic sarcoma; lineage marker: leukocytes.
l.
Ki-67 (clone MIB-1), mouse monoclonal, IgG1, dilution: 1:1,000, M7240, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: unmasking, high pH (UltraVision LP system Thermo Fisher Scientific BV, Breda, The Netherlands; as well as VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA); positive controls: mammary gland; lineage marker: cell nucleus of cells in interphase.
m.
HMB45, mouse monoclonal, IgG1, dilution: 1:50–1:100, M634, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: LP; positive controls: melanoma; lineage marker: antigen present in melanocytic tumors.
n.
MyoD1 (clone 5.8A), mouse monoclonal, IgG1, dilution: 1:100, M3512, Dako Sweden AB, Stockholm, Sweden; pretreatment and technique: VECTASTAIN Elite ABC kit, Vector Laboratories, Burlingame, CA; positive controls: X; lineage marker: markers of myogenic commitment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly funded by the European Commission (grant no. LUPA-GA-201370); and by Foundation of Veterinary Research in Companion Animals (“Stichting DOG”).
