Abstract
Phosphatase and tensin homolog (PTEN) belongs to the group of gatekeeper tumor suppressor genes and is involved in multiple mechanisms leading to cellular defense against neoplastic transformation and progression. Twenty-four dogs and 17 cats were submitted to a 2-year follow-up study, and clinicopathologic features were recorded and compared with immunohistochemical PTEN staining. PTEN-negative status occurred in 33% of canine and 76% of feline mammary carcinomas. In canine mammary carcinomas, there was a significant (P < .05) correlation between loss of PTEN protein expression and simple carcinoma histotype, lymphatic vessel invasion, lymph node metastases, distant organ metastases, tumor dedifferentiation, tumor recurrence, and shorter overall survival. In feline mammary tumors, a significant correlation between loss of PTEN protein expression and lymphatic vessel invasion was found. Loss of PTEN expression could be a useful prognostic marker in canine mammary carcinomas.
Phosphatase and tensin homolog (PTEN) belongs to the group of gatekeeper tumor suppressor genes. 35 The protein product of this gene has multiple biologic activities: PTEN inhibits phosphorylation of phosphatidylinositol 2 phosphate to phosphatidylinositol 3 phosphate by antagonizing the activity of phosphatidylinositol 3 kinase and inhibiting the downstream signal cascade, thus leading to cell proliferation and survival via the protein kinase B pathway. 24 PTEN is also involved in stabilizing intercellular junctions that interact with members of the membrane guanylate kinase inverted family, 20,21,36 and loss of this function has been correlated with increased invasiveness of tumor cells. 20 Furthermore, PTEN interacts with p53 and increases the DNA binding and transcriptional activity of this protein. 14 Taken together, these functions are consistent with a primary role for PTEN in cellular defense against neoplastic transformation and progression. It has been demonstrated that the expression of PTEN is reduced or lost in varying percentages in many different human tumors, including brain tumors, 31 melanomas, 25 hepatocellular carcinomas, 42 thyroid carcinomas, 3 endometrial carcinomas, 37 lymphoid neoplasia, 33 and breast carcinomas. 2,27,38 Germline mutations in this protein have been observed in human patients with Cowden syndrome, characterized by hamartomas, along with a 25–50% lifetime risk of developing breast cancer. 34
The role of the loss of PTEN expression in human breast carcinomas has been investigated extensively, 2,6,27,32,34,38,41 and the reduction of its expression has been correlated with tumor grade, 2 estrogen and progesterone receptor negative status, 27,34 lymph node metastasis, 8,38 and worst prognosis. 8,38
The expression of PTEN in tumors of domestic animals has been investigated in canine osteosarcomas, 23 melanomas, 19 hemangiosarcomas, 10 and mammary gland tumors. 17,29,30 No data are available on the expression of PTEN in feline mammary carcinomas (FMCs), and only 1 recent study has investigated the expression of PTEN at the protein level in canine mammary carcinomas (CMCs). 30
The purpose of our study was to demonstrate the expression of PTEN protein in FMCs and CMCs and to explore morphologic and clinical correlations.
Materials and Methods
Thirty dogs and 18 cats that had undergone surgery at the Department of Veterinary Clinic, School of Veterinary Medicine, University of Pisa, from 2002 to 2007 were initially included in the study. All subjects received a total mastectomy, and surgical excision specimens were fixed for 48 hours in buffered formalin, routinely processed, and examined histologically. Sections were routinely stained with hematoxylin and eosin, and lesions were classified accordingly to the World Health Organization Histological Classification of the Mammary Tumours of the Dog and the Cat. 26 Subjects bearing mammary carcinomas (dogs, n = 26; cats, n = 18) were submitted to a 2-year postsurgery follow-up examination. Subjects bearing adenomas (dogs, n = 4; cats, n = 0) were excluded. Clinical examinations and tumor staging were performed 6, 12, 18, and 24 months after surgery. The presence of distant organ metastases and the recurrence of primary tumors were investigated by clinical and radiographic examinations. The dogs and cats that died during the follow-up period were necropsied. Animals that died during the follow-up period due to nontumor-related causes (dogs, n = 2; cats, n = 1) were excluded from the study. Twenty-four dogs and 17 cats bearing mammary carcinomas constituted the study population at the end of the follow-up period. Tumors were measured and divided into 2 groups: Group A (tumors with major axis <30 mm in cats or <50 mm in dogs) and Group B (tumors with major axis ≥30 mm cats or ≥50 mm in dogs). Sections of tumors, lymph nodes (dogs, 7/10; cats, 8/11), and distant organ metastases (dogs, 7/11; cats, 8/10) were stained using immunohistochemistry (IHC) for PTEN protein. Canine adenomas (n = 4) were submitted for PTEN IHC. For each block, 4-µm-thick sections were cut and mounted on Superfrost Plus slides (Thermo Scientific, Menzel GmbH & Co., KG, Braunshweig, Germany) and dried overnight at 37°C. Invasion of lymphatic vessels around the tumor, lymph node metastases, and mitotic index (total number of mitotic figures in 10 microscopic high-power fields) were evaluated, and tumor grading using the Elston and Ellis system 11 was performed.
For IHC, sections were dewaxed in xylene, passed through a graded series of alcohols, and rehydrated in deionized water. Antigen was retrieved with a citrate buffer pH 6.0 in a microwave oven for 15 minutes at 650 watts and cooled at room temperature for 20 minutes. Endogenous peroxidases were blocked with 0.5% hydrogen peroxide for 30 minutes followed by 3 washes in 0.05% Tween Tris-buffered saline solution at pH 7.6. Normal serum from the host species of the secondary antibody diluted 1/10 in Tween Tris-buffered saline solution was added to the sections and incubated for 30 minutes at room temperature. After 3 washes, the primary antibody, previously used on human mammary gland tissues 8,38 (mouse monoclonal anti-human/-rat/-mouse PTEN, clone A2B1, Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) diluted 1 : 50 in Tween Tris-buffered saline solution, was applied and incubated for 1 hour at room temperature. Alignment between canine (NCBI database, accession no. NP_001003192) and mouse PTEN (NCBI database, accession no. NP_032986.1) and rat PTEN (NCBI database, accession no. NP_113794.1) proteins showed 100% homology. Ninety-nine percent homology was found with the human PTEN (NCBI database, accession no. EAW50175.1).
After 3 washes, a secondary biotinylated antibody (Vectastain, Vector Labs Inc., Burlingame, CA, USA) was added and incubated for 30 minutes at room temperature. A peroxidase reaction was developed for 10 minutes using diaminobenzidine (Impact DAB, Vector Labs, Inc., Burlingame, CA, USA) and blocked with deionized water. Counter-staining was done with diluted hematoxylin. Negative controls were performed omitting the primary antibody and replacing the antibody with a murine subclass matched (IgG1) unrelated primary monoclonal antibody. Canine and feline renal glomeruli were used as positive controls in each experiment, and vascular endothelium was used as an internal positive control in each slide as previously described. 19 Canine and feline normal (n = 2 and n = 1, respectively) and hyperplastic mammary (n = 2 and n = 2, respectively) glands were selected from the archives of the Department of Animal Pathology of the University of Pisa and submitted to IHC as further positive controls.
PTEN IHC staining was judged positive by the presence of distinct brown cytoplasmic or both nuclear and cytoplasmic staining. Evaluation of PTEN expression was semiquantitative, based on staining intensity and distribution in accordance with a previously reported method for A2B1 antibody scoring on human mammary carcinomas 8 : 10 high-power fields (×400) were randomly selected in the tumor. Intensity was scored as strong, moderate, or weak compared with that of the normal surrounding tissues and normal mammary gland. Distribution was scored as diffuse (>50% tumor staining), regional (15–50% tumor staining), or focal (<15% tumor staining). Tumors with intense diffuse, intense regional, intense focal, and moderate diffuse stainings were considered positive for PTEN expression, whereas tumors with moderate regional, moderate focal, or weak staining with any distribution were considered negative.
Statistical analysis was performed using the statistical package SPSS Advanced Statistics 13.0 (SPSS Inc., Chicago, IL, USA). A chi-square test was used to investigate the significance of the relationship between PTEN protein expression and individual variables. Statistical significance was based on a 5% (0.05) significance level. Overall survival analysis was performed using the Kaplan-Meyer method, 13 and the Tarone-Ware test was used to investigate the relationship between PTEN expression and overall survival.
Results
At the time of diagnosis, the mean age of the bitches bearing mammary tumors was 10 years ± 1.5 years (range, 8–14 years), and the mean age of the queens was 10 ± 1.8 years (range, 7–14 years). Ten bitches (41%) had tumors ≥50 mm, and tumors ≥30 mm were observed in 7 queens (41%).
Canine and feline mammary lesions examined were histologically classified as follows: 24 (92%) CMCs, 17 (100%) FMCs, and 4 (8%) canine adenomas. No feline adenomas were observed. Carcinomas were morphologically of a complex (dogs, n = 9; cats, n = 0) or simple (dogs, n = 15; cats, n = 17) type. Simple carcinomas comprised tubulopapillary (dogs, n = 4; cats, n = 8), solid (dogs, n = 9; cats, n = 9) or anaplastic (dogs, n = 2; cats, n = 0) carcinomas. The Elston and Ellis grading system was used to divide tumors into well-differentiated carcinomas (WDCs) (dogs, n = 9; cats, n = 4), moderately differentiated carcinomas (MDCs) (dogs, n = 6; cats, n = 6), or poorly differentiated carcinomas (PDCs) (dogs, n = 9; cats, n = 7). At the time of diagnosis, invasion of lymphatic vessels around the tumor was observed in 8 (31%) canine and 9 (53%) feline primary carcinomas. Lymph node metastases were present in 10 (42%) dogs and 11 (65%) cats. Eleven (46%) CMCs and 10 (59%) FMCs generated distant organ metastases. For 2 CMCs and 3 FMCs, invasion of lymphatic vessels around the tumor was not observed despite lymph node and distant organ metastases. In 1 case of CMC, distant organ metastases were observed without invasion of lymphatic vessels or lymph node. The mitotic index of tumors ranged from 1 to 42 for bitches (median, 9; mean, 13.2 ± 11) and 2 to 50 for queens (median, 18; mean, 21.3 ± 15.3).
At the end of clinical follow up, 13 (54%) bitches and 7 (41%) queens were still alive, and 11 (46%) bitches and 10 (59%) queens had died from mammary carcinoma. Two FMCs invaded lymphatic vessels around the tumor and invaded lymph nodes without causing distant organ metastases during the follow-up period.
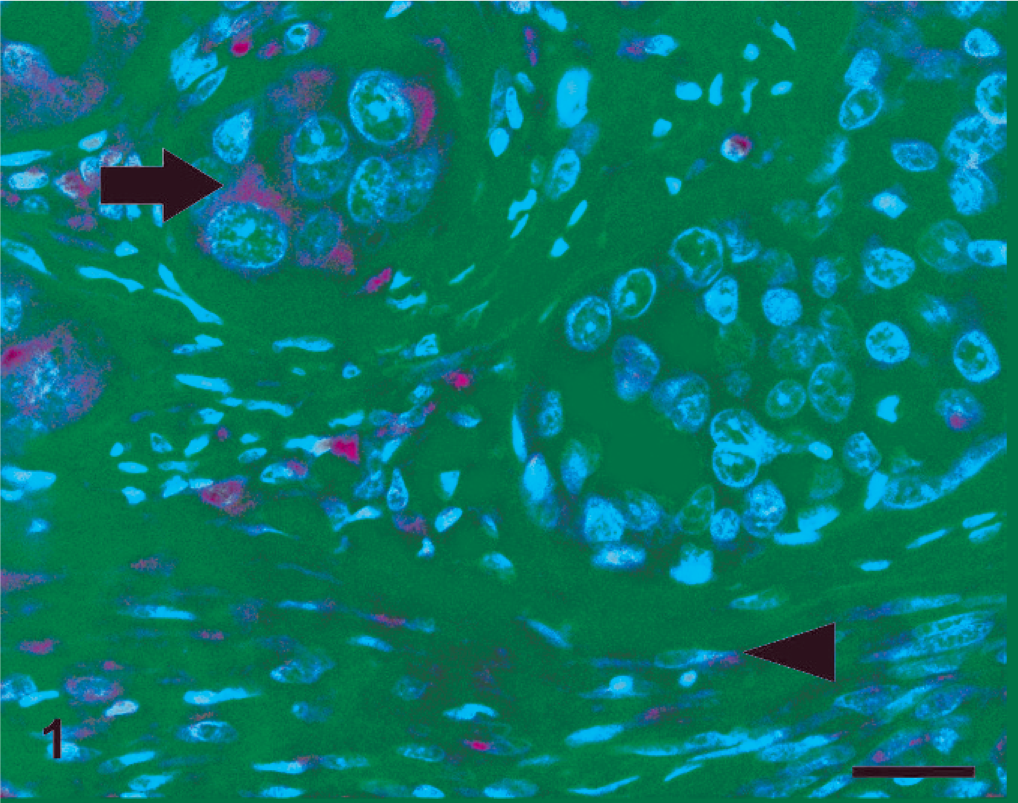
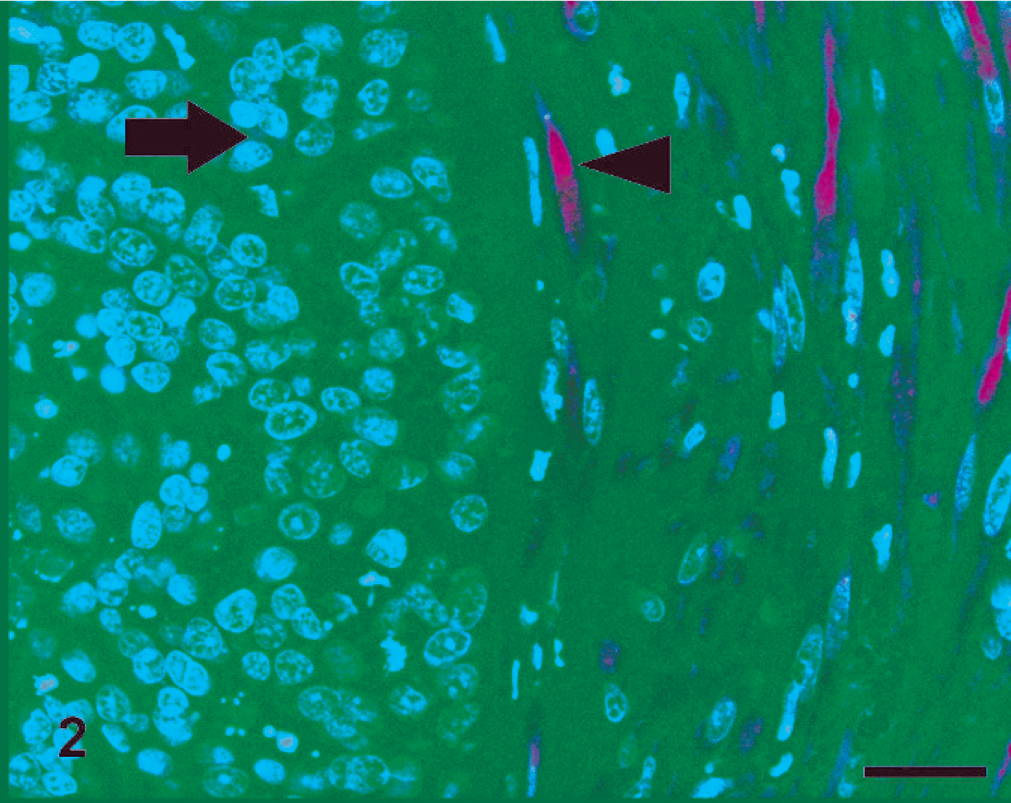
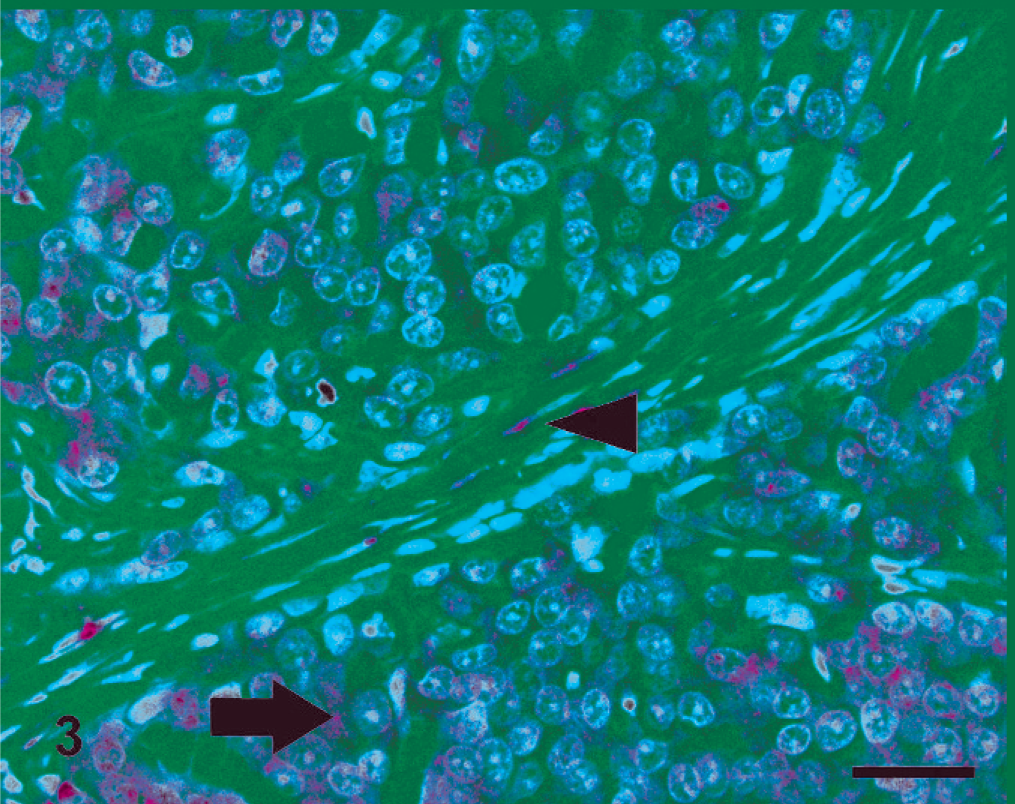
In both species, PTEN IHC staining was detected in the cytoplasm and nuclei of healthy and hyperplastic mammary cells. Nuclear staining was markedly evident in myoepithelial cells surrounding normal and hyperplastic mammary gland tissue. Nuclear staining was also occasionally found in some normal and hyperplastic glandular epithelial cells that showed mainly cytoplasmic stain. In PTEN-positive neoplastic tissues, the stain was predominantly found in the cytoplasm, even if in rare cases nuclear stain was detected. All CMCs and the majority of FMCs displayed a homogeneous distribution and intensity of staining among cells of an individual tumor. One FMC showed particularly intense PTEN staining in myoepithelial cells compared with positive epithelial cells. The surrounding mesenchymal cells were also strongly positive. PTEN expression in canine and feline mammary tissues is presented in Table 1. In dogs, all of the benign and 16 (67%) of the malignant tumors were PTEN positive (Fig. 1), and 8 (33%) of the malignant tumors were negative (Fig. 2). Of the FMCs, 13 (76%) were PTEN negative (Fig. 3), and 4 (24%) were positive (Fig. 4). Lymphatic emboli surrounding the primary tumor, lymph node metastases, and distant organ metastases, when present, showed the same IHC PTEN pattern as the primary tumors.
Phosphatase and tensin homolog (PTEN) immunohistochemical expression in neoplastic canine and feline mammary tissues.
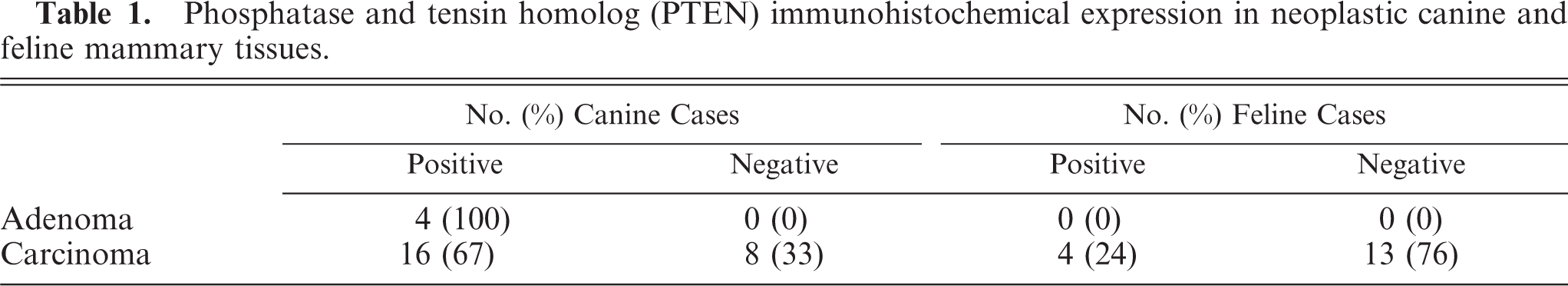
Dog, simple carcinoma; Positive phosphatase and tensin homolog expression in cancer cells (arrow) and in surrounding stromal cells (arrowhead). Streptavidin-biotin-peroxidase method, hematoxylin counterstain. Scale bar = 25 µ.
Dog, simple carcinoma; Lack of phosphatase and tensin homolog expression in the majority of cancer cells. Some cells demonstrate faint staining (arrow), whereas strong positive staining is evident in surrounding stromal cells (arrowhead). Streptavidin-biotin-peroxidase method, hematoxylin counterstain. Scale bar = 25 µ.
Cat, simple carcinoma; Positive phosphatase and tensin homolog expression in cancer cells (arrow) and in surrounding stromal cells (arrowhead). Streptavidin-biotin-peroxidase method, hematoxylin counterstain. Scale bar = 25 µ.
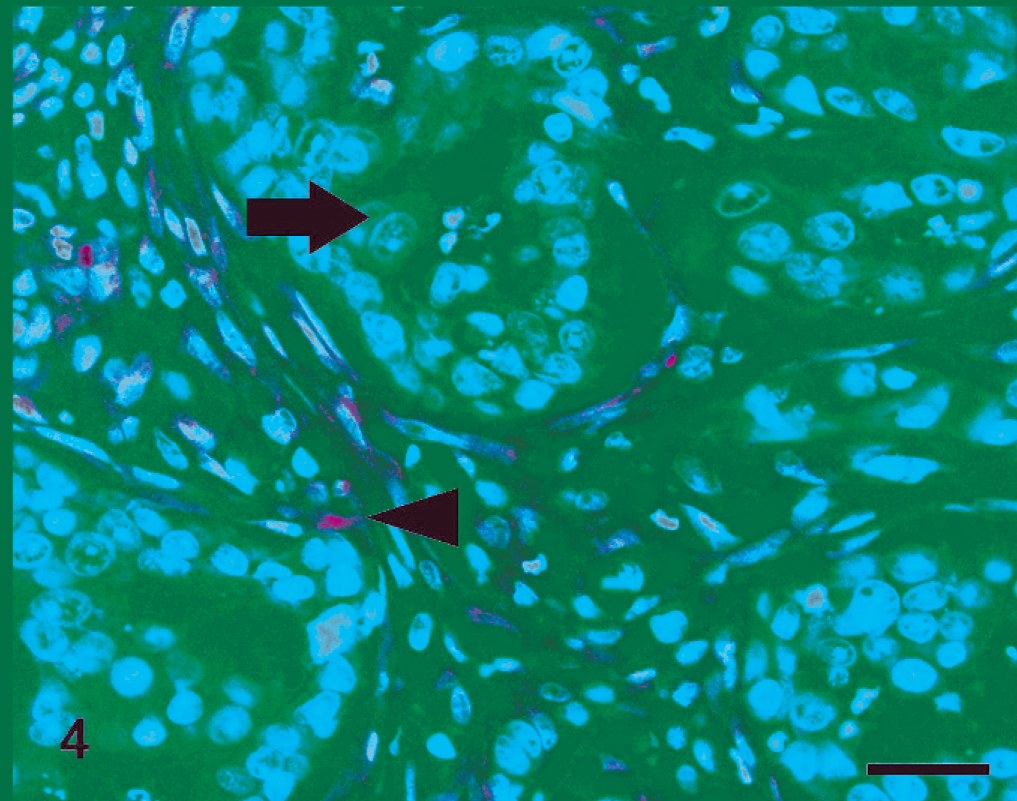
Cat, simple carcinoma. Lack of phosphatase and tensin homolog expression in cancer cells (arrow) and positive staining in surrounding stromal cells (arrowhead). Streptavidin-biotin-peroxidase method, hematoxylin counterstain. Scale bar = 25 µ.
The relationship between PTEN protein expression and clinicopathologic factors in CMCs and FMCs is shown in Table 2. There was a significant correlation between IHC loss of PTEN protein and simple carcinoma histotype, lymphatic vessel invasion around the tumor, lymph node metastases, distant organ metastases, dedifferentiation, tumor recurrence, and shorter overall survival in CMCs. In the dog, a significant difference was found between WDCs, MDCs, and PDCs (P = .017); WDCs and PDCs (P = .023); and WDCs and MDCs (P = .004) but not between MDCs and PDCs (P = .398). Significant differences were found grouping MDCs and PDCs and comparing them with WDCs (P = .007) but not grouping WDCs and MDCs and comparing them with PDCs (P = .371). In FMCs, a significant correlation between PTEN protein loss and lymphatic vessel invasion around the tumor was found. When animals with PTEN-positive and -negative mammary carcinomas were compared, overall survival was reduced in both species for those with PTEN-negative tumors (Figs. 5, 6), but the survival difference was significant for CMCs only (P < .05).
Correlation between phosphatase and tensin homolog (PTEN) immunohistochemical expression in carcinomas and clinicopathologic features.
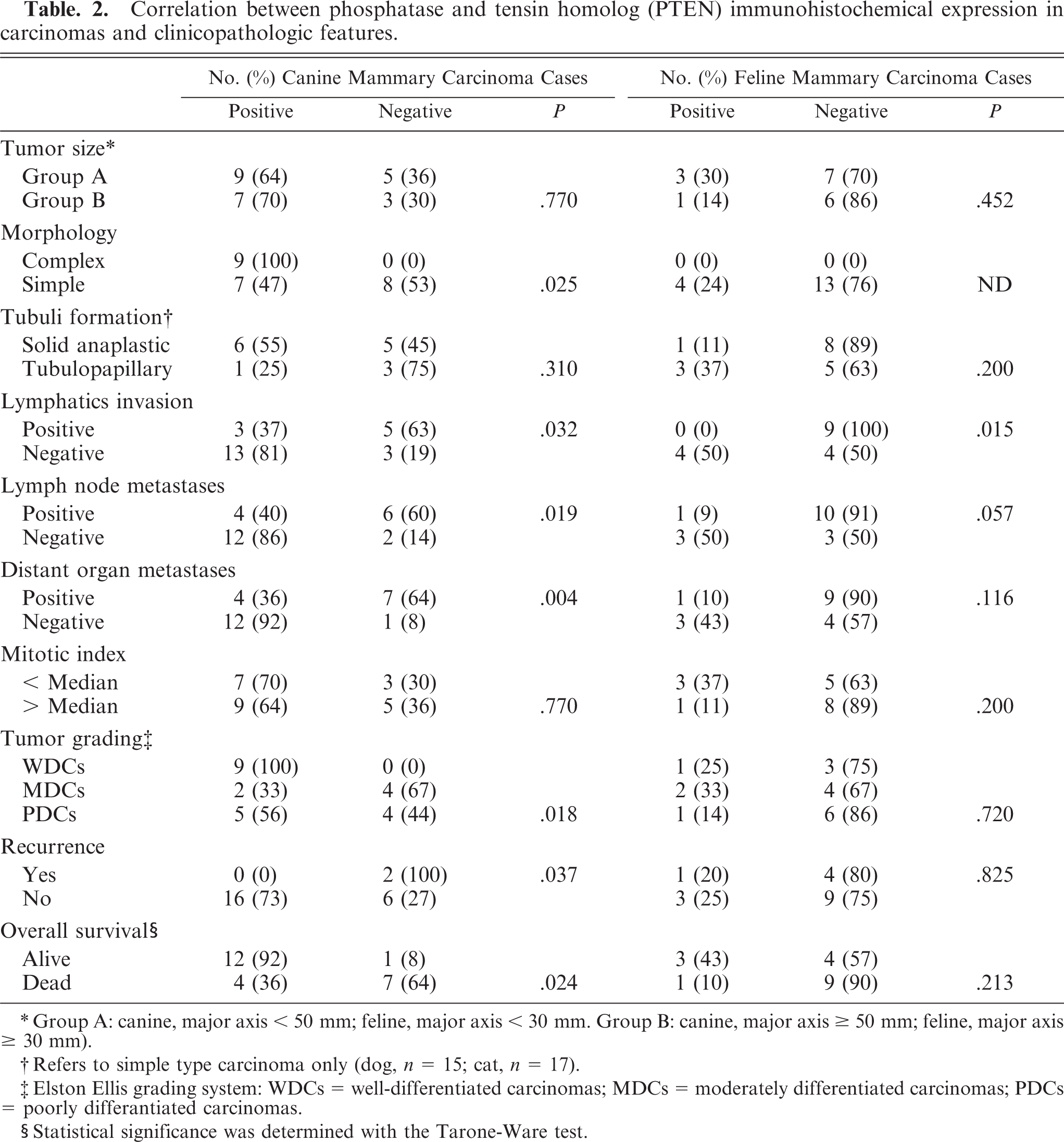
∗Group A: canine, major axis < 50 mm; feline, major axis < 30 mm. Group B: canine, major axis ≤ 50 mm; feline, major axis ≤ 30 mm).
†Refers to simple type carcinoma only (dog, n = 15; cat, n = 17).
‡Elston Ellis grading system: WDCs = well-differentiated carcinomas; MDCs = moderately differentiated carcinomas; PDCs = poorly differentiated carcinomas.
§Statistical significance was determined with the Tarone-Ware test.
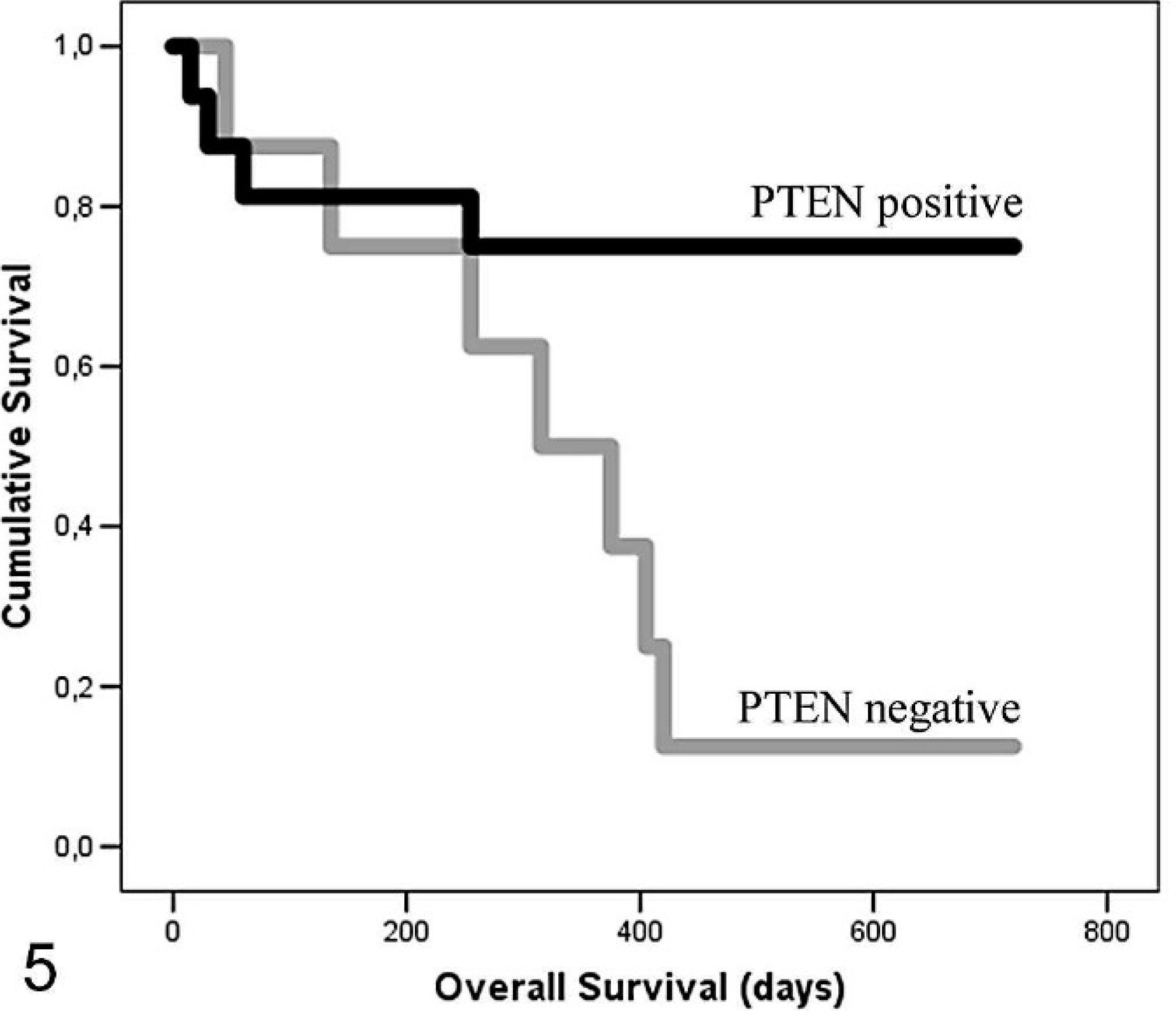
Kaplan Meier estimates of overall survival in phosphatase and tensin homolog–positive (black line) and –negative (gray line) 24 bitches bearing mammary carcinomas. P < .05.
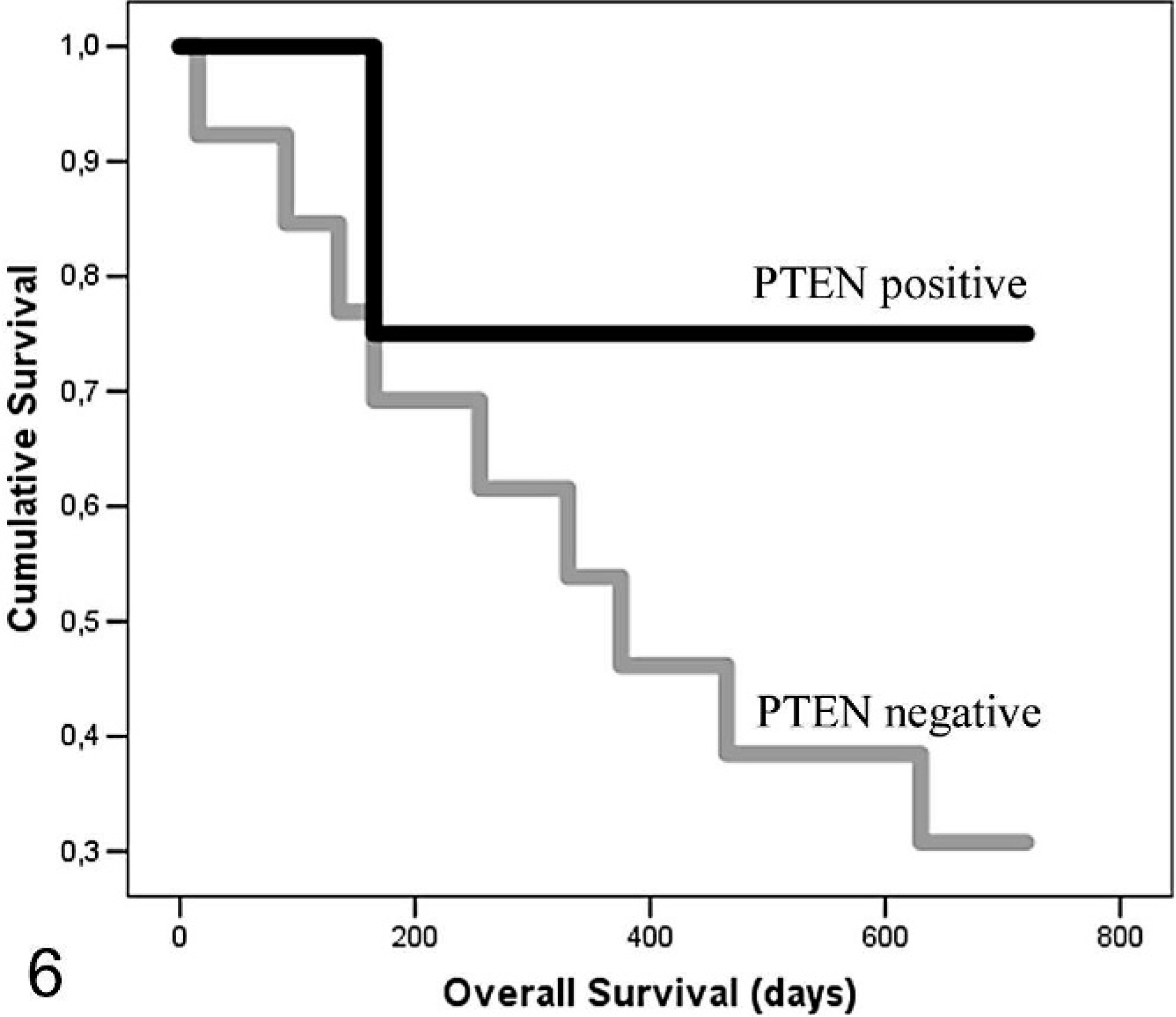
Kaplan Meier estimates of overall survival in phosphatase and tensin homolog–positive (black line) and –negative (gray line) 17 queens bearing mammary carcinomas. P > .05.
Discussion
This is the first report of PTEN protein expression analysis in FMCs and the first report of a correlation between PTEN expression and the clinical outcome of the neoplastic disease in CMCs. The IHC technique used in this study allowed an accurate localization of PTEN protein in CMC and FMC cells. PTEN had the same distribution pattern as previously described in human 2,8,27,38 and CMCs. 30
Nuclear positive staining was observed in normal, hyperplastic and occasionally in neoplastic mammary gland cells, as previously reported. 30
Although investigations of the role of nuclear PTEN are still limited, it has been demonstrated that nuclear localization of this protein is correlated with chromosome stability, DNA repair, and cell cycle arrest and negatively correlated with mitotic index. 28 In the present study, the predominantly cytoplasmic pattern of PTEN in neoplastic tissues, in contrast with both cytoplasmic and nuclear distribution in normal and hyperplasic glands, seems to confirm the nuclear PTEN role. Further investigations are needed to understand the role of PTEN cellular localization in CMCs and FMCs.
In human mammary carcinomas, despite the high incidence of the loss of heterozygosity affecting the PTEN gene, 15 mutation is identified in only a minority of human breast cancers. 12 Other mechanisms have been suggested to explain the results of IHC studies, including the silencing of the PTEN promoter through hypermethylation, 40 increased degradation, 7 or haploinsufficiency. 9 A similar finding of decreased PTEN protein expression despite a lack of PTEN gene mutations was observed in other tumors, such as melanomas 43 and prostate 40 and ovarian 22 cancers.
The IHC staining of PTEN protein was negative in 33% of CMCs and 76% of FMCs. Our study demonstrated a percentage of canine PTEN-negative tumors that is comparable to the percentage of PTEN negative human breast carcinomas (30–50%) 2,8,27,38 and CMCs (34%) 30 in previous studies. Previous investigations demonstrated a significant reduction in PTEN mRNA expression in a high percentage of CMCs versus normal mammary glands and benign mammary tumors. 29,17 To explain the discrepancy between gene and protein expression, mRNA and protein levels in the same samples should probably be investigated. This is the first report of PTEN expression in FMCs, and a high percentage of PTEN-negative tumors was observed. At present, PTEN protein expression cannot be compared with gene expression data in FMCs, and mRNA expression studies are needed.
Whether loss of PTEN protein expression is an early or a late event in human breast carcinogenesis is still controversial, although results of one study have suggested that it could be a late event due to the high percentage of PTEN loss in invasive carcinomas compared with in situ lesions. 2 In our study, IHC staining of PTEN was strong in canine adenomas, which is in accordance with a two- to fourfold increased PTEN mRNA expression in canine benign tumors in a previous study. 17 The loss of PTEN expression in invasive mammary carcinomas of both dogs and cats further indicates that the loss of PTEN protein could be a late event in these species. Tumor size was not found to be significantly associated with PTEN expression in either of the species examined, a feature identified in the previous CMCs study 30 and shared by human mammary tumors. 8
The simple type of CMCs was significantly correlated with PTEN-negative tumors, without a difference between subclassifications (tubulo-papillary versus solid and anaplastic). Simple carcinomas are more aggressive if compared with complex carcinomas 18 in dogs, and, interestingly, simple carcinomas showed a significantly higher percentage of PTEN-negative tumors than complex carcinomas.
Mitotic index was not found to correlate with PTEN expression in either animal species, matching the lack of correlation between proliferation markers and PTEN staining in human breast tumors. 38
Tumor grading was significantly correlated with PTEN-negative tumors in CMCs but not in FMCs. In the dog, a significant difference was found between WDCs, MDCs, and PDCs or grouping MDCs and PDCs and comparing with WDCs but not grouping WDCs and MDCs and comparing with PDCs. This finding is in accordance with a low predictive value of MDCs both in CMCs 18 and FMCs. 5 Studies on the relationship of IHC PTEN loss and tumor grading in human breast carcinomas gave discordant results. 2,8,38 Further investigations are needed on the lack of correlation between PTEN-negative status and tumor grading in the cat.
Tumor recurrence was found to be significantly associated with PTEN-negative status in CMCs, but the small number of subjects with recurrence does not allow a definitive conclusion. In our study, invasion of lymphatic vessels by the tumor was significantly correlated with PTEN-negative status in CMCs and FMCs, which is in accordance with previous IHC studies in human mammary cancers 2,8,38 and CMCs. 30 A strong relationship exists between lymphatic vessel invasion and short overall survival in CMCs, and lymphatic vessel invasion at the time of diagnosis is a strong prognostic factor in both species. 18,39 A significant association between PTEN expression and invasion of lymphatic vessels by tumor, lymph node metastases, and distant organ metastases is in accordance with PTEN protein functions regarding cell motility and proliferation in association with metastatic behavior and could help characterize the metastatic potential of the primary tumors in the dog.
There are no published studies correlating PTEN loss and outcome in CMCs and FMCs. A shorter overall survival was observed to be significantly associated with loss of PTEN expression in CMCs in this study, and similar findings have been observed in human breast carcinomas. 8,38 Overall survival in FMCs was not significantly correlated with loss of PTEN expression in our study, although queens with PTEN-negative tumors had a worse prognosis. The absence of significant relationships in FMCs studied may be due to the small number of tumors examined, and a larger population of cats with mammary tumors is needed to establish whether PTEN expression is a prognostic marker in the feline species as well.
The PTEN/phosphatidylinositol 3 kinase/protein kinase B pathway has recently been considered as a potential target for drug development against tumors displaying PTEN loss. 4,16 In human mammary cancer, PTEN loss has been suggested as explaining the fraction of Her2/neu-positive, Herceptin-nonresponsive tumors, 1 and interest is growing in developing therapeutic strategies to fight against phosphatidylinositol 3 kinase/protein kinase B pathway activation when the PTEN gatekeeper function is lost. In this respect, our findings seem to be relevant not only in terms of prognostic value but also for future therapeutic developments in CMCs and FMCs as animal models for human mammary cancer.
In conclusion, in our study we demonstrated that loss of PTEN expression revealed by IHC occurs in 33% of CMCs and 76% of FMCs. Loss of PTEN protein expression significantly correlated with simple carcinoma type, lymphatic vessel invasion, lymph node metastases, distant organ metastases, tumor grading, recurrence, and shorter overall survival in CMCs and with lymphatic vessel invasion in FMCs. Although a wider study is required to strengthen the predictive value of this protein, loss of PTEN expression could be considered useful as a prognostic marker in CMCs.
