Abstract
Previous studies showed that intercellular communication by gap junctions has a role in bone formation. The main connexin involved in the development, differentiation, and regulation of bone tissue is connexin (Cx) 43. In addition, Cx46 is also expressed, mostly localized within the trans-Golgi region. Alterations in the expression pattern and aberrant location of these connexins are associated with oncogenesis, demonstrating a deficient gap junctional intercellular communication (GJIC) capacity in neoplastic tissues. In this study, we evaluated normal and neoplastic bone tissues regarding the expression of Cx43 and Cx46 by immunofluorescence, gene expression of these connexins by real-time PCR, and their correlation with cell proliferation index and deposition of collagen. Fourteen neoplastic bone lesions, including 13 osteosarcomas and 1 multilobular tumor of bone, were studied. The mRNA levels of Cx43 were similar between normal and neoplastic bone tissue. In normal bone tissue, the Cx43 protein was found mainly in the intercellular membranes. However, in all bone tumors studied here, the Cx43 was present in both cell membranes and also aberrantly in the cytoplasm. Regarding only tumor samples, we determined a possible inverse correlation between Cx43 expression and cellular proliferation, although a positive correlation between Cx43 expression and collagen deposition was also noted. In contrast, Cx46 had lower levels of expression in neoplastic bone tissues when compared with normal bone and was found retained in the perinuclear region. Even though there are differences between these two connexins regarding expression in neoplastic versus normal tissues, we concluded that there are differences regarding the subcellular location of these connexins in normal and neoplastic dog bone tissues and suggest a possible correlation between these findings and some aspects of cellular proliferation and possibly differentiation.
Introduction
There is strong evidence demonstrating that direct interactions between cells can be involved in the maintenance of homeostasis in bone tissues.
53,67
Connexins are phylogenetically preserved proteins that compose gap junctions in different tissues, sharing a basic structure in which 6 subunits form a hemi-channel called the connexon.
45
Each one of these channels is supplied by an adjacent cell, allowing the direct exchange of molecules and ions weighing less than 1,000–2,000 Daltons.
62,64
Many substances such as ions, water, sugars, nucleotides, amino acids, fatty acids, small peptides, drugs, and carcinogens are small enough to move between cells through gap junction channels. However, proteins, complex lipids, polysaccharides, RNA, and other large molecules are not. Channel passage does not require ATP and appears to result from passive diffusion.
55
In mammals, a multigenic family consisting of 21 members of highly conserved genes (the connexin genes) code for the connexin proteins.
45
Many physiological roles have been proposed for gap junctional intercellular communication (GJIC), and some are briefly cited:
Maintenance of homeostasis. Gap junctions serve as electrical synapses in electrically excitable cells such as cardiac myocytes, smooth muscle cells, and neurons.
3,29
In these tissues, electrical coupling permits more rapid cell-to-cell transmission of action potentials than chemical synapses. In myocytes, this enables their synchronous contraction. Tissue response to hormones. GJIC may enhance the responsiveness of tissue on external stimuli.
24,36
. Regulation of embryonic development.
27
In bone tissue, cellular communication by gap junctions is associated with physiological processes such as formation, remodelling, and ossification as well as proliferation of osteogenic cells.
11,13,25,47
.
Connexins are also involved in carcinogenesis. 55,62,63,66 Trosko and Ruch 55 listed several major theories of carcinogenesis: 1) the idea that cancer is a disease of differentiation; 2) the stem cell theory of cancer; 3) the dedifferentiation theory of cancer; 4) the initiation/promotion/progression concept; 5) the nature versus nurture theory; 6) the mutagenic versus epigenetic concept; 7) the oncogene and tumor suppressor gene theory; and 8) the hypothesis that cancer was the result of dysfunctional gap junctional intercellular communication. The same authors postulated that dysfunctional GJIC theory can integrate all of the other theories because each of them can relate to GJIC. 55 Alterations in the expression pattern and aberrant location of connexins are associated with oncogenesis. 22,43,54,62,64 However, the aberrant GJIC of cancer or transformed cells can manifest itself in 2 distinct types of cell–cell interation, that is, homologous and heterologous. Lack of heterologous GJIC between cancer and normal cells was observed in vivo; some rat liver preneoplastic foci and hepatocellular carcinoma showed clear lack of communication with surrounding normal parenchyma cells. Both types of communication were decreased in cultured rat liver epithelial cells, rat liver tumors, and human liver tumors. 64 Several kinds of tumors showed absence or reduction of connexin expression and function. 30,32,52,62 One study in canine tissue, performed by our group, showed a reduction in the expression of connexin (Cx) 26 and Cx43, and aberrant localization of Cx26 in malignant mammary gland neoplasms. 52 Experiments performed in connexin knockout mice showed that these animals had a greater tendency to develop liver tumors (Cx32 knockout mice) 51 and lung tumors (Cx43 heterozygous knockout mice). 1 Transgenic mice that expressed a mutant Cx32 and present lower GJIC between hepatocytes also presented greater susceptibility to liver cancer. 9 HeLa cells lack the expression of connexins and consequently do not communicate through GJIC. When these cells were transfected with specific connexin genes, cell proliferation was decreased. 34 Therefore, it was suggested that connexin genes have a tumor-suppressing effect. 35,37,61,65,67 For example, the deletion of a single allele of Cx43 is associated with a reduction in the GJIC and increased cell proliferation of mouse lung type II pneumocytes. 1 Cell proliferation in no neoplastic situations may be also influenced by connexin deficiency or deficient GJIC capacity. 9 Corroborating these hypotheses, human U2OS osteosarcoma cells and other cell lineages transfected with Cx43 cDNA had decreased proliferation. 33,34,50,69 Most of these studies were performed with cell lineages and laboratory animals. There are few studies on connexins in normal and neoplasic tissues from domestic animals, 52 but no study so far with tumors originating in bone tissue in domestic animals has been done.
Osteosarcomas are the main type of primary bone tumors of the skeleton in dogs (representing 65–85%) and humans. 10,17 They represent 4% of all neoplastic diagnoses, and 98% of primary bone tumors originate in the appendicular skeleton, with very strong site preferences. In general, osteosarcomas originate in the metaphyses of the long bones of forelimbs, especially the distal radius and proximal humerus, and also can be found in the distal ulna, proximal tibia, and distal femur. Osteosarcomas usually arise in the medullary cavity, penetrate the cortex, and extend into the subperiosteum, causing the formation of a large soft tissue mass contiguous to the bone. Axial skeletal osteosarcomas are less common and can involve the ribs, vertebrae, and skull (cranial vault, zygomatic arch, and jaw, and may originate within the nasal cavity). When originating in the skull, osteosarcomas are usually of the multilobular type and are more commonly observed in middle-aged dogs. Axial osteosarcomas are more common in females. 17,40,60 In dogs, these tumors mainly affect middle-aged animals (with a mean age of 7 years) and large and giant dogs with no breed predilection. Osteosarcomas have a high metastatic potential, and although less than 5% of affected dogs present pulmonary metastasis that can be detected with radiographic studies when first seen by a veterinarian, 90% die or are euthanized because of complications associated with pulmonary metastasis. Survival of dogs with osteosarcomas in the axial skeleton is slightly better when compared with dogs affected in the appendicular skeleton. 5,14,26,29,59 Classification of bone tumors as benign or malignant is based on morphological findings. These groups are subdivided based on cell type, production of matrix, and localization in the affected bone (central or peripheral), with central tumors presenting a higher incidence. 39,40 An accurate diagnosis of bone tumors requires proper clinical and radiographic evaluations as a histopathological analysis. 4 Mesenchymal tumors from soft tissues and primary skeleton sarcomas are a large and heterogeneous group of neoplasms. Data from histomorphology, immunohistochemistry, and experimental data suggest that many, if not all, sarcomas derive from primitive pluripotential mesenchymal cells that can differentiate into one or more lineages during neoplasic transformation. 15 Histopathological aspects can differ in cases of osteosarcomas. However, definitive diagnosis is based on production of osteoid and/or bone by mesenchymal cells. 40 Survival and suspicion of malignancy in human osteosarcomas are associated with nuclear antigen of cellular proliferation (PCNA). 57
In this study, we evaluated the expression of connexins in neoplasic dog bone tissue and attempted to correlate the index of expression of these connexins with cellular proliferation index and production of collagen by cells from primary bone tumors from the skeleton.
Materials and Methods
Samples and histopathologic analysis
Normal bone tissue from 6 dogs was obtained from epiphyses and metaphyses of appendicular long bones (femur) from necropsies. Fourteen neoplastic bone lesions, including 13 osteosarcomas and 1 multilobular tumor of bone, were studied. These were obtained from female (5) or male (9) dogs from the Surgery Service of the Veterinary Hospital, School of Veterinary Medicine and Animal Science, University of São Paulo. Histological slides were stained with HE for histopathological analysis or picrosirius red 19 for evaluation of collagen fibers. All lesions were classified according to the criteria of the World Health Organization in 1994. 4
Histochemical staining for the histopathological analysis and detection of collagen
The normal and neoplastic bone tissues were fixed in methacarn (70% methanol, 20% chloroform, and 10% acetic acid) and embedded in paraffin. The 5-µm histological sections from normal and neoplastic bone tissues were first decalcified with formic acid solution (20%) and routinely stained with HE or picrosirius-hematoxylin. Histopathology was performed with light microscopy (Nikon Eclipse E-600; Nikon, Tokyo, Japan), and digital photomicrographs were captured by Image ProPlus software (Media Cybernetics, Silver Spring, MD). The picrosirius-stained sections were prepared as described previously. 18 The analyses were made with polarized light, quantifying the appearance (color, orientation, birefringence, thickness of collagen fibers and fibrils) of collagenic structures. Collagen type I (which is the predominant fiber type of osteoid) appeared as a three-dimensional network of randomly oriented, thin, short, weakly birefringent yellow or red fibers; collagen type III appeared as thin, weakly birefringent greenish fibers; and collagen type II did not form fibers and displayed a weak birefringent of a variable color. 20 The percentage of positive collagen area was obtained using Image Pro Plus software in 8 random fields.
Immunohistochemical staining for the detection of connexins, PCNA, cytokeratin, vimentin, osteocalcin, and osteonectin
All tumor samples were fixed in methacarn (70% methanol, 20% chloroform, and 10% acetic acid) and embedded in paraffin. The 5-µm histological sections from the normal and neoplastic bone tissues were first decalcified with formic acid solution (20%) and routinely deparaffinized, rehydrated, and incubated with a solution of methanol and hydrogen peroxide (10%) for 30 minutes. The slides were incubated with anti-Cx43 (710700, 1 : 500; Zymed, San Francisco, CA, USA) or anti-Cx46 (provided by Dr. Michael Koval, University of Pennsylvania, Philadelphia, PA) 1 : 3,000 polyclonal antibodies diluted in blocking buffer (TNB) at 48°C overnight. Anti-rabbit biotinylated antibody (E0432; Dako, Capinteria, CA) was used as the link (1 : 1,000 in phosphate buffered saline [PBS]) for 1 hour in a humidified chamber. The amplification was performed with a TSA kit (tyramide signal amplification fluorescein system, PerkinElmer, Waltham, MA, USA) and shown with fluorescein. The histological sections were analyzed with a Nikon E-800 microscope (Nikon) with immunofluorescence. Heart or lens samples from dogs were used as positive controls for the immunolabeling of Cx43 or Cx46, respectively. Negative control was performed by omitting the primary antibody in a tissue slide during the immunohistochemical reaction. Furthermore, samples of different tissues such as liver, skeletal muscle, and bone marrow 55 from dogs were present in the same slides with neoplastic tissues; these samples were also working as negative controls. Criteria for scoring the immunoreactivity of Cx43 and Cx46 were based on the following: 1+ (discrete immunolabeling), 2+ (moderate immunolabeling), 3+ (intense immunolabeling).
For the immunostaining of PCNA, cytokeratin, vimentin, osteocalcin, and osteonectin, additional sections were obtained from the methacarn-fixed, paraffin-embedded lesions, which were positioned on siliconized slides. The deparaffinized and hydrated histological sections were incubated with anti–osteocalcin antibody (1 : 200; Zymed), anti-osteonectin (1 : 200; Zymed), anti-vimentin (1 : 100; Dako), anti-pan-cytokeratin (1 : 100; Dako), and anti-PCNA (1 : 600; Dako) at 48°C overnight. Afterward, slides were incubated with secondary biotinylated antibody for 30 minutes, washed in PBS, and incubated with streptavidin peroxidase complex (LSAB; Dako), for 30 minutes. Positive cells became evident after revelation with DAB (3′3-diaminobenzidine tetrahydrochloride; Dako). To determine the PCNA-labeling index, 2,000 cells were counted in 8 random fields and separated into positive cells (which presented brown nuclei) and negative cells. The PCNA-labeling index (%) was determined by dividing the number of positive cells per total cells and multiplying by 100.
RNA extraction, cDNA construction, and real-time PCR analysis of Cx43 and Cx46 expression in normal and neoplastic bone tissues from dogs
Total RNA was extracted from tissue by a single-step technique with TRIzol Reagent (Invitrogen Life Technologies, Carlsbad, CA) according to the manufacturer's protocol. The quality of the RNA samples was determined by electrophoresis in 2% agarose gels and staining with ethidium bromide. 18S and 28S bands were visualized under ultraviolet light (not shown). Total RNA was then treated with DNase I (Invitrogen Life Technologies) before processing further. Oligo DT (1 µl) and dNTPs (1 µl) were added to the total RNA sample (500 µg) and incubated at 65°C for 5 minutes. After that, Buffer 5× (Superscript II) (4 µl), DTT (1 m, 2 µl), and RNAse OUT (1 µ) were added, and the mixture was incubated at 42°C for 2 minutes. Again Superscript II (1 µl) was added, followed by incubation at 42°C for 50 minutes, and subsequent incubation was performed at 70°C for 15 minutes. All reagents were purchased from Invitrogen Life Technologies. Real-time PCR analysis was performed in ABI Prism 7000 Sequence Detection Systems (Applied Biosystems, Foster City, CA) using TaqMan Universal Master Mix (item number 4304437; Applied Biosystems). PCR primers and the TaqMan probes for Cx43, Cx46, and 18 s (used as a housekeeping control) quantification were selected using the Primer Express software programs (Applied Biosystems) and checked by a basic local alignment search tool search of GenBank. The Cx43 assays were acquired from Applied Biosystems (Cf02690400_g1). Primers for Cx46 were designed from exons (GenBank XM_543178), 5′CTCATCTACCTGGGCCATGTG 3′ forward and 5′ TCGCGCTCCTTCTTCTTCTC 3′ reverse primers; and CF-CX46-RG6M2, FAM, CCATGCGCACGATGTG were constructed. The 18 s, a housekeeping control, was from Applied Biosystems (4319413E). The TaqMan probes carried a 5′-reporter dye 6-carboxy-fluorescein (FAM), a 3′-non fluorescent quencher dye (NFQ), and a minor groove binding (MGB). The primers and probes were used with 100% efficiency at the final concentrations of 0.9 µM and 0.25 µM, respectively. The thermal cycling conditions to cDNA quantification assays were established according to ABI Prism 7000 Sequence Detection Systems parameters (Applied Biosystems). Analysis of relative gene expression data was performed according to the CT method. 2
Statistical analysis
The difference and correlations between data were statistically analyzed by the Mann-Whitney nonparametric test (GraphPad Prism 3.01, GraphPad Software, La Jolla, CA, USA) and Spearman (P < .05) linear regression.
Results
General findings
As described in Materials and Methods, 14 dogs with bone tumors were studied. Among these animals, 64.3% (9/14) were male and 35.7% (5/14) were female. Regarding the age in which the tumors were diagnosed, we noted that all animals were adults ranging from 8 to 13 years and averaging 10.5 years. The age was not determined in 2 of the animals. Among animals with established breed (11/14), 91% (10/11) were from breeds considered large or giant and only 1 was from a breed considered small or medium (miniature poodle). Among animals with primary tumors from the appendicular skeleton (11/14), we observed a higher prevalence in the humerus (4/11), followed by the tibia (3/11), radius and cranium (both 2/11), scapula, and femur (each with 1/11). Primary tumors from the axial skeleton were located in the cranium (2/3) or ribs (1/3). The diagnosis of the histological type was performed considering clinical, radiographic, and macroscopic data. Subtypes of osteosarcomas found were productive osteoblastic (8/14), chondroblastic (3/14), nonproductive osteoblastic (1/14), and undifferentiated (1/14). One multilobular tumor of bone was also observed (1/14). Immunohistochemical reactions were performed in order to confirm the diagnosis of tumors found in this study and to confirm the mesenchymal origin of tumor cells (positivity to vimentin) and also the osteoblastic origin (positivity to osteocalcin and osteonectin) and to exclude possible carcinomas (by using pancytokeratin). All tumors studied were negative for cytokeratin and positive for vimentin, osteocalcin, and osteonectin (data not shown).
Cx43 expression in normal and neoplastic bone tissue
In order to verify expression of Cx43 in normal and neoplasic osteoblasts, the immunofluorescence reaction was performed, revealing positive immunolabeling. However, because of the difference in the distribution of cells in normal bone tissue in the Haversian system when compared with the anarchical distribution found in neoplasic bone tissue, it was difficult to compare the positive immunolabeling patterns. Nevertheless, it was possible to observe that cells in normal bone tissue presented positive immunolabeling with homogeneous distribution around cells. This suggests that the Cx43 was presented in the cytoplasmic membrane and also among the osteoid matrix, possibly in regions in which there are confluences of the osteocytes long dendritic processes (Fig. 1a, b).
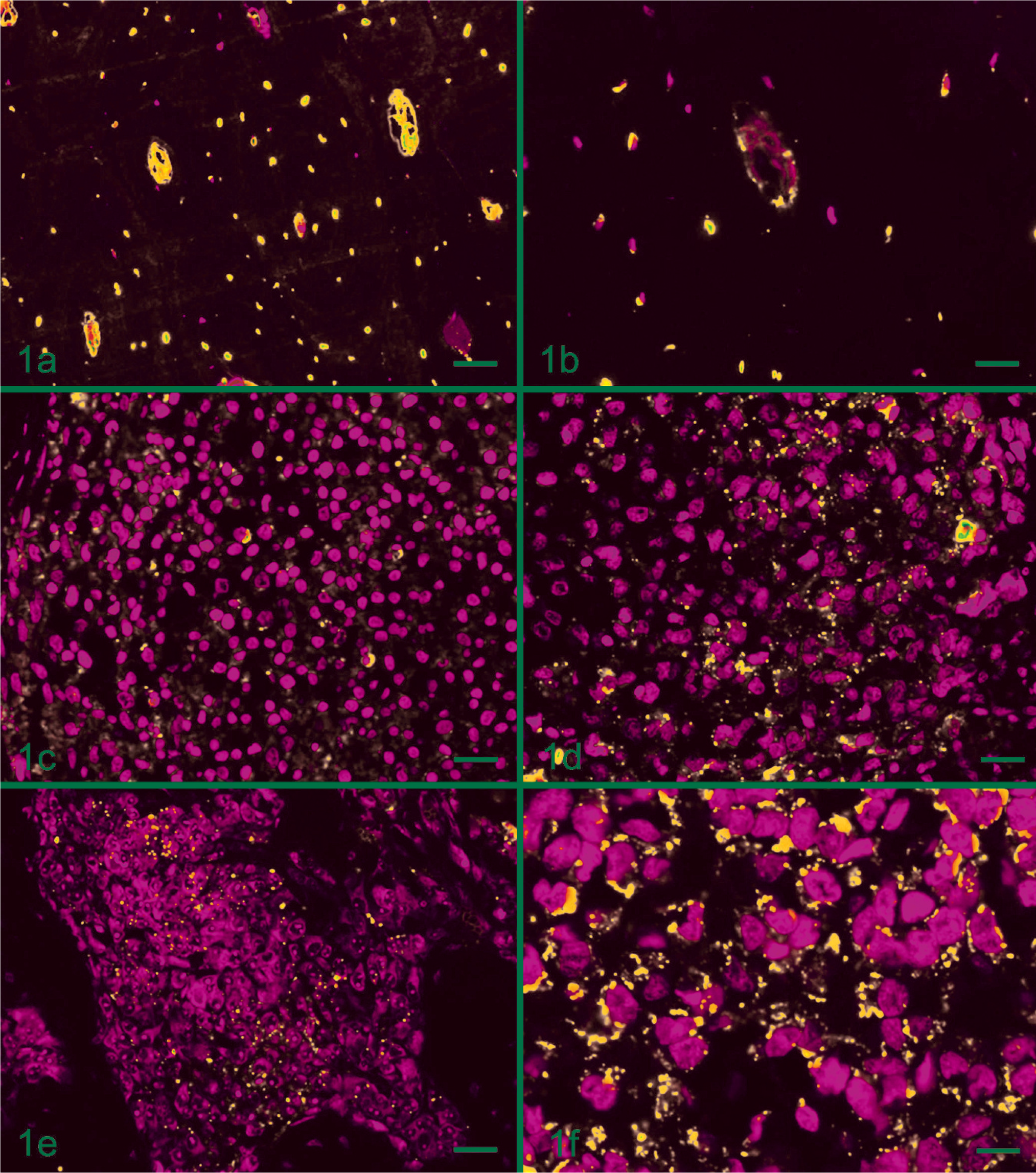
Immunolabeling of connexin 43 in canine normal and neoplastic bone tissue. Fig. 1a. Normal canine bone tissue, showing immunolabeling in the cell membranes and in formed long canaliculae, interconnect the osteocytes (bar = 40 µm). Fig. 1b. Magnified view of Fig. 1a showing positive immunolabeling for Cx43 in the cell membrane (bar = 20 µm). Fig. 1c, 1d. Canine osteosarcoma, showing punctate cytoplasmic and cell membranes immunolabeling of Cx43 (bar = 20 µm). Fig. 1e,
Intensity of immunolabeling of connexin 43 and 46.

∗Variation in intensity of immunolabeling of connexin 43 and 46 expression in different animals with neoplastic bone tissue. Criteria for scoring the immunoreactivity of connexin 43 and 46 were based on the following: + (discrete immunolabeling), ++ (moderate immunolabeling), +++ (intense immunolabeling). IHC = immunohistochemistry.
All bone tumors, including osteosarcomas and the multilobular tumor (animal T4; Table 1) expressed Cx43, generally presenting a heterogeneous appearance in the tissue, with some areas of intense positive immunoreactivity and others in which it was reduced or absent. The Cx43 was observed mostly among cells, indicating cytoplasmic membrane, and also was observed aberrantly as cytoplasm dots (Fig. 1c–e; Table 1). Variation in intensity and amount of marked cells was also observed. Data on mRNA levels of Cx43 demonstrated that they were not different among tumors and between tumors and normal bone tissue (Fig. 2a).
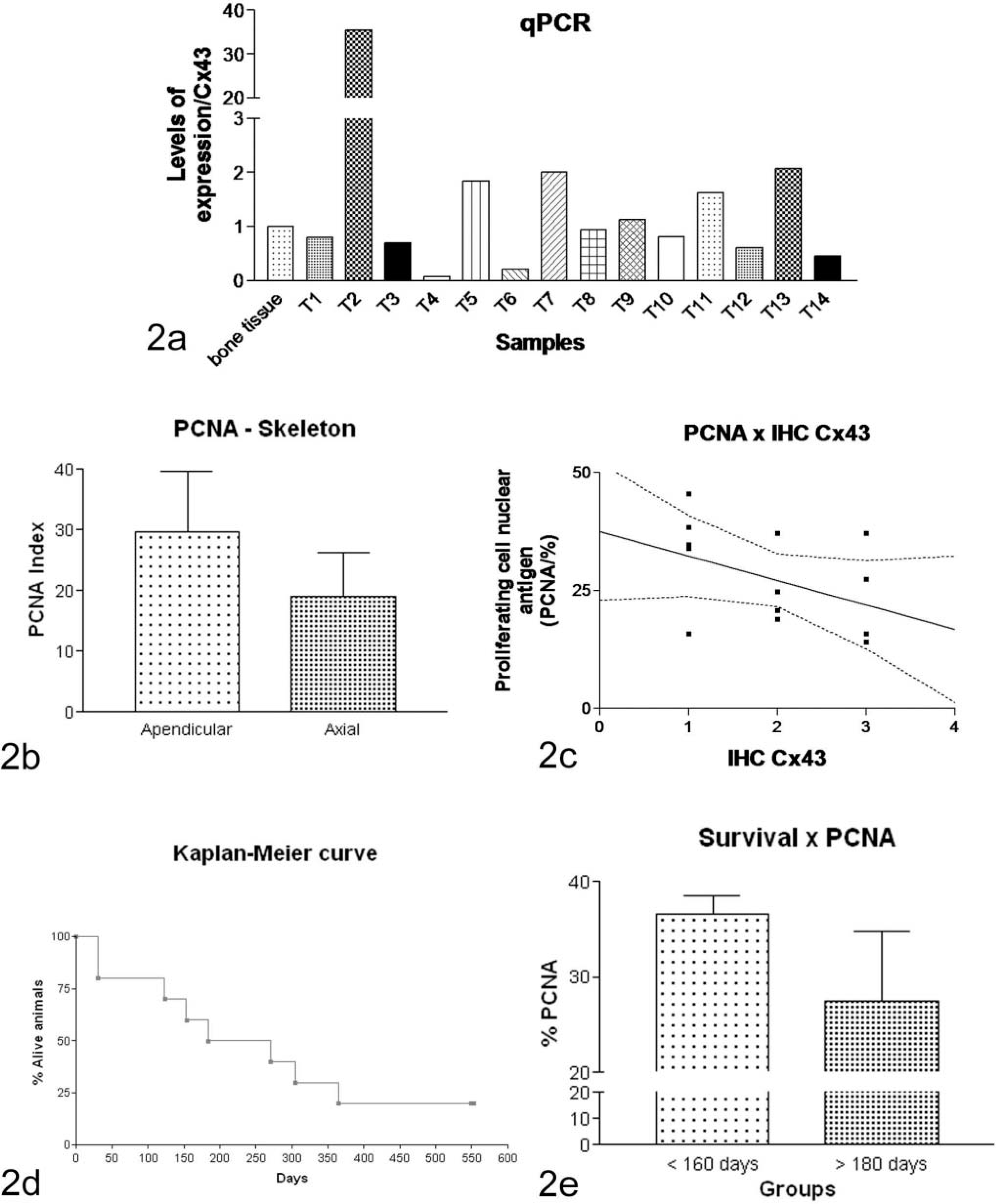
Cx43 real-time PCR and correlations of Cx43 with collagen and PCNA. Fig. 2a. No differences among tumors or between tumors and normal bone tissue were observed. Fig. 2b. Tumors of the appendicular skeleton had a higher cellular proliferation index when compared with tumors from the axial skeleton (Appendicular = 31.17 ± 7.20% PCNA-positive cells; Axial = 21.70 ± 5.77% PCNA-positive cells; P = .102). Fig. 2c. The PCNA index to Cx43 levels: it was possible to demonstrate a discrete inverse correlation meaning that higher amounts of Cx43 were associated with fewer PCNA-positive cells (Spearman r = −0.4039; P = .1521; linear regression r 2 = 0.1782; P = .1327). Fig. 2d. Survival period of osteosarcoma-affected dogs. Fig. 2e. PCNA was also compared with survival, finding a tendency of a lower cellular proliferation index in animals with higher survival when compared with animals that died earlier.
PCNA proliferation index correlates to levels of Cx43 and with location of spontaneous bone tumors
PCNA immunohistochemical reaction was performed in order to evaluate tumoral cellular proliferation. Nuclear PCNA marked cells were counted to evaluate cellular proliferation in the tumors. Tumors of the appendicular skeleton had a somewhat higher cellular proliferation index when compared with tumors from the axial skeleton (Appendicular = 31.17 ± 7.20% PCNA-positive cells; Axial = 21.70 ± 5.77% PCNA-positive cells; P = .102) (Fig. 2b).
By relating cellular proliferation index of bone tumors to Cx43 levels, it was possible to demonstrate a discreet inverse correlation, meaning that higher amounts of Cx43 were associated with fewer PCNA-positive cells (Spearman r = −0.4039; P = .1521; linear regression r 2 = 0.1782; P = .1327) (Fig. 2c).
Average survival time was calculated excluding euthanized animals after surgery. Then, the mean survival time of dogs with osteosarcoma after surgical removal was 256 ± 190 days (median = 227; interval: 30–553 days). This average survival is underestimated, given that 2 animals were still alive at the time of writing this manuscript (Fig. 2d).
Then, we assigned all animals into 2 groups, one in which the survival time was between 30 and 153 days (more aggressive tumors), and the other with a higher survival rate of between 184 and 553 days. When both groups were compared, it was possible to note a tendency for a lower cellular proliferation index in the group with higher survival (P = 0.11) (Fig. 2e). At last, no correlation was observed between survival time and other data evaluated in this study.
Cx43 correlates to collagen deposition
All bone tumors produce variable amounts of osteoid matrix consisting mainly of type I collagen, which was confirmed by the Picrosirius stain in optical microscopy and with polarized light. This collagenic matrix when deposited had irregular form, margins, and amount without interconnected trabeculae as observed in bone reactions in response to infection, inflammation, and fractures (Fig. 3a–e). In all sections studied, it was observed that the picrosirius polarization method stained only those structures containing collagen, and the intensity was proportional to the amount of collagen present. All birefringent structures formed by type I collagen were seen as a three-dimensional network of randomly arranged thin, short, red fibers against a dark background (Fig. 14;3b, d). This property differentiates the osteoids from other structures that show a similar appearance (collagen fibers show different structure because they appear as long, thick, strongly birefringent fibers of uniform aspect and thickness). 18 There was a tendency toward statistical significance when the amount of collagen deposition and the intensity of Cx43 positive immunoreactivity were correlated (Spearman r = 0.5201; P = .0566; linear regression r 2 = 0.2125; P = .0971) (Fig. 3f).
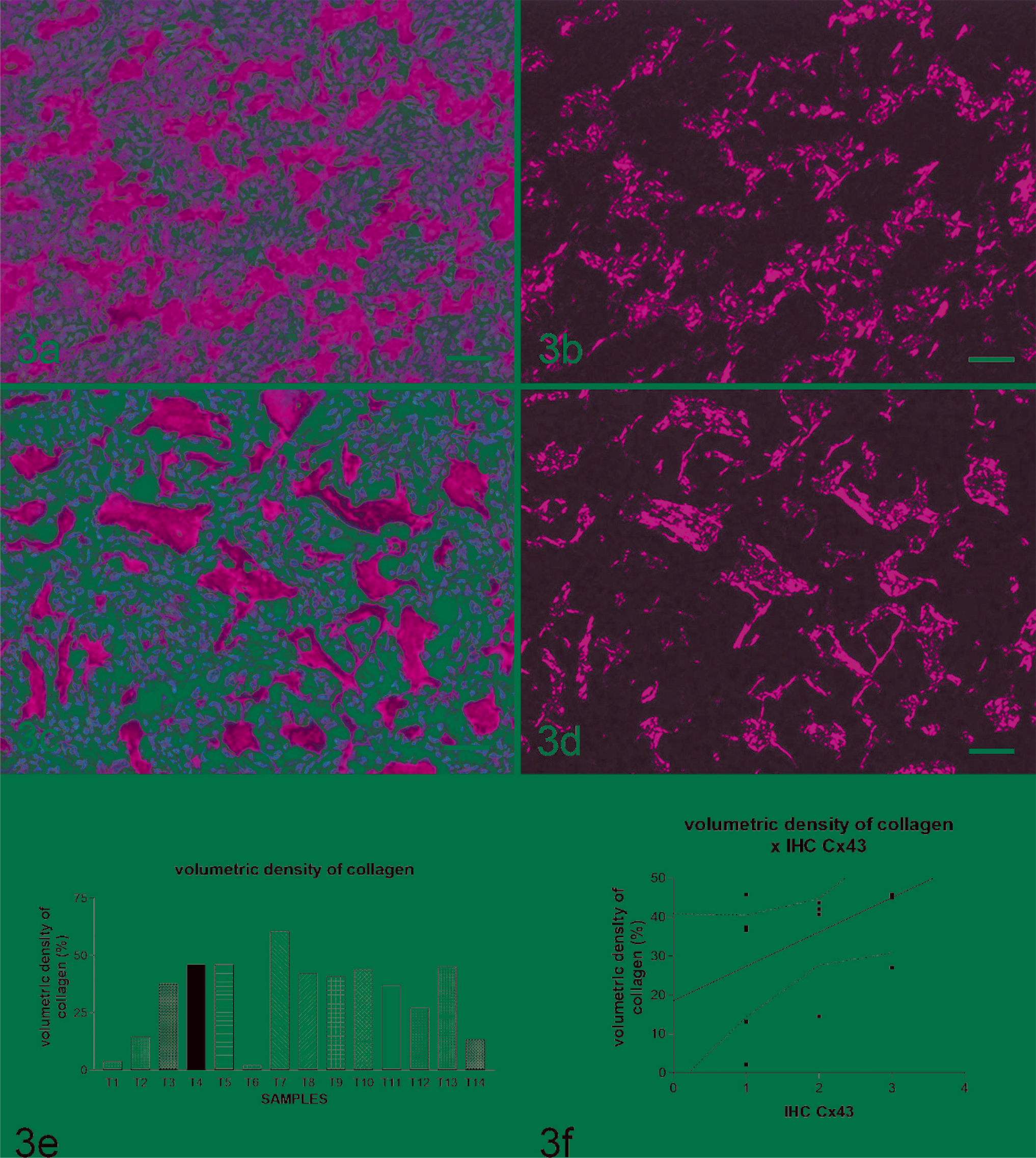
Neoplastic bone tissue (osteosarcoma); canine. Fig. 3a, 3c. Normal brightfield light microscopy showing variable amounts of red organic matrix secreted by neoplastic osteoblasts. Fig. 3b, 3d. Polarizing microscopy. Observe that in osteoid all birefringent structures are formed of type I collagen, seen as a three-dimensional network of randomly arranged thin, short, red fibers against a dark background. Picrosirius. Bar = 40 µm. Fig. 3e.Volumetric density of collagen from 14 different dogs with bone tumors. Fig. 3f. Amount of osteoid that was deposited compared with levels of Cx43 expression showed a tendency to positive correlation of these data (Spearman r = 0.5201; P = .0566; linear regression r 2 = 0.2125; P = .0971).
Cx46 expression in canine normal and neoplastic bone tissue
As mentioned in the Material and Methods section, normal bone tissue from 6 different dogs was evaluated. The Cx46 was found in normal bone tissues mainly around the nuclei (Fig. 4a). In all Osteosarcomas, it was found around the nuclei (perinuclear-positive immunolabeling) (Fig. 4b–d). This connexin also showed variation regarding regions and number of positive immunolabeling cells. In both cases, it was possible to evaluate the intensity of positive immunoreactivity in the whole tissue, showing variation between animals studied. Table 1 shows the index of positive immunoreactivity. There was more variability of Cx46 gene expression among the bone tumors than there was among the normal bone tissues evaluated. Eleven out of 14 animals had reduced Cx46 expression (Fig. 4b–e) compared with normal bone.
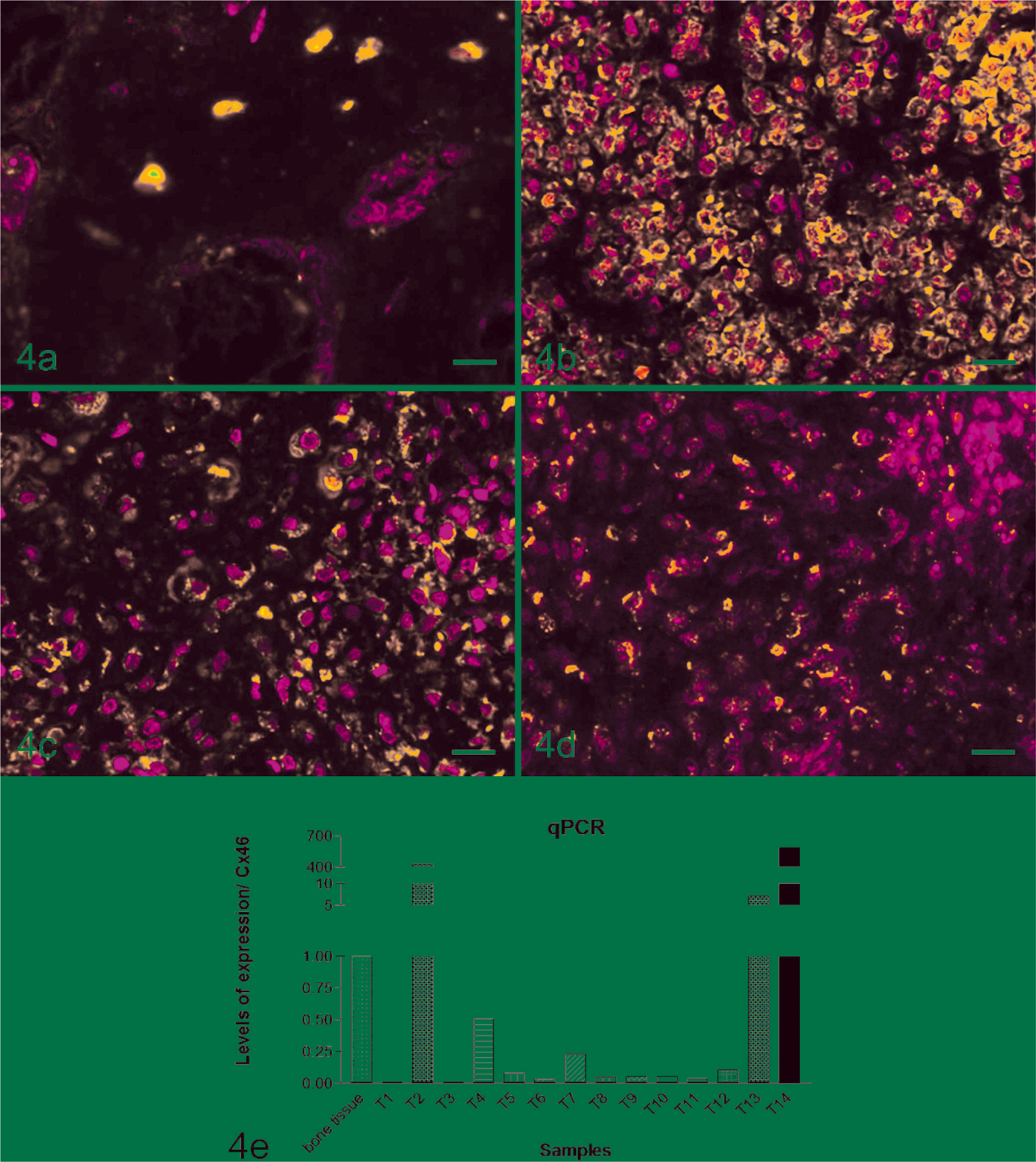
Immunolabeling and real-time PCR of connexin 46 in canine normal and neoplastic bone tissue. Fig. 4a. Normal canine bone tissue, showing immunolabeling of Cx46 (bar = 10 µm). Fig. 4b–4d. Cx46 is expressed by neoplastic osteoblastic cells localized to an intracellular perinuclear compartment (bar = 20 µm). TSA and fluorescein method, propidium iodide counterstain. Fig. 4e. Smaller amounts of mRNA levels of Cx46 were observed in a neoplastic bone tissue when compared with the expression of normal bones.
Discussion
The study of molecules involved in the process of differentiation, induction, promotion, and cellular proliferation and its correlation to normal and neoplasic tissues is extremely important considering the need for better prognostic factors, therapeutics, and prevention of neoplasms. Several studies observed that gap junctions could be involved in physiological processes of bone tissue such as ossification, modeling, and remodeling as well as in pathological processes such as neoplasia. 11,13,25,47 This study aimed to characterize the expression of Cx43 and Cx46 in primary bone tumors from dogs, comparing this expression with normal bone tissue and correlating Cx43 and Cx46 expression with the cellular proliferation index (PCNA), production of extracellular matrix, and postoperative survival.
According to many authors, normal cells communicate with each other through gap junctions, which helps maintain tissue homeostasis. Tumor cells, to the contrary, reject this communication and avoid the homeostatic control from neighboring tissues. 64 Many experiments in vivo and in vitro, in which connexin genes were transfected into cell lines with defects in gap junction communication, showed a reduction in tumor proliferation. 31,33,34,42 Thus, rat glioma, human rhabdomyosarcoma cells and chemically transformed mouse fibroblasts, which are deficient in Cx43 in comparison to their nontransformed counterparts, exhibit reduce growth and tumorigenicity following transfection with the Cx43 cDNA. 64 The ability to suppress tumors is related to specific connexin types. HeLa cells transfected with the Cx26 gene were completely nontumorigenic and showed a reduced growth rate in vitro, while the Cx40 and Cx43 genes did not have a significant tumor-suppressive effect on the same cells, despite an increase in GJIC. 34 These results suggest that connexin genes exert a differential cell growth control effect, depending on the cell type in which they are expressed. 64,65 Cells from Cx32 and Cx43 knockout mice have a high tendency to develop tumors when compared with naive lineages. 30,51 These observations make it possible to believe that Cx43 could have a reduced gene expression in canine bone tumors when compared with the expression in normal bone tissue. However, no difference in gene expression of Cx43 between normal and neoplasic bone tissue was observed. Dysfunctional GJIC could be the result of the connexin genes being silenced (e.g., HeLa or MCF-7 cancer cells) or the connexin proteins and gap junctions not being translated, transported, assembled or being functional because of a mutation, abnormal splicing of message, or being modified by oncogenes or chemical tumor promoters. 53,54 In the case of canine osteosarcomas, the aberrant localization of Cx43 was probably responsible for a dysfunctional GJIC capacity, because no difference in mRNA expression was noted.
Employing immunohistochemical reactions, we noted that Cx43 is also expressed in normal bone tissue in dogs, as reported by Stains and Civitelli 49 with electron microscopy and in vitro studies. Positive immunoreactivity was observed in a homogeneous and regular distribution around blood vessels of Haversian channels in 2 areas, the first located in the lacunaes where the osteocytes remain, around cell nuclei, suggesting cytoplasmic membrane. The second area was within the osteoid matrix equidistant from the nuclei, possibly in the regions where the long dendritic process or prolongations of osteocytes join inside through the canaliculi of bone. 6,58 Neoplastic tissue mostly presents an anarchical architecture, in contrast to the unaltered tissue. Even though the neoplastic cells seem to be in contact, our findings suggest that there is a reduction in Cx43 expression in the samples, independent of histological subtype of osteosarcoma. All osteosarcoma samples presented an irregular distribution of Cx43. Foci of absence or reduction of expression was found intermingled with other areas of tissue having cells with positive immunolabeling. In these foci we observed that Cx43 is located in cytoplasm and cytoplasmic membrane between adjacent cells in a punctual and disorganized manner. Table 1 shows that the variation in connexin expression was not restricted to only what was observed in each individual tissue, but that there was also a variation in pattern and intensity of staining of expression between the animals studied. Interestingly, the 1 multilobular tumor of bone (T4) only expressed Cx43 in neoplasic osteoblasts and chondroblasts, and no or discreet staining was found between fusiform cells around the central islands of cartilage and bone matrix. According to our findings and corroborating the literature data, many, if not all, tumors are associated with an absence or reduction of connexin function and expression. 30,51,52,64 Other in vitro experiments with cellular lineages of murine osteosarcomas showed variation in the connexins that were expressed. Lecanda et al. 26 showed that the ROS 17/2 8 (a rat osteosarcoma cells lineage) expressed Cx43 and consequently maintain the GJIC. Nevertheless, the UMR 106-01 lineage of osteogenic sarcoma, which predominantly expressed Cx45, also presented poor GJIC. Interestingly, one nontumoral lineage of rat osteocyte (MLO-Y4) expresses Cx43 but not Cx45, 25,68 a fact that agrees with the previously cited data. Two other U2OS lineages of human osteosarcoma and the MC3T3-E1 lineage of osteoblasts also express Cx43. 44,68
Osteoblasts are found on the surface of bones, where active bone formation takes place with secretion of a matrix called osteoid, which is later calcified. This matrix consists mainly of type I collagen associated with a mineral phase composed of hydroxyapatite but also 10–15% of noncollagen proteins, which include osteopontin, osteonectin (ON), syaloprotein, and osteocalcin (OC). A small amount of proteoglycan also constitutes the matrix. 12,41 During in vitro differentiation of osteoblasts, the expression of Cx43 increases. 11,47 If osteoblast differentiation is delayed by inhibition of communication by gap junctions, there is a reduction in the capacity of formation of mineralized extracellular matrix and in the expression of genes associated with osteoblasts differentiation. 11,25,47 Variable amounts of extracellular matrix were produced by the different subtypes of osteosarcoma found in our study. This matrix was constituted mainly by deposition of amorphous trabeculae, with birefringent structures formed by type I collagen, disclosed as a three-dimensional network of randomly arranged thin, short, red fibers against a dark background, as verified in polarized light microscopy. 18,19 By correlating the amount of osteoid that was deposited with levels of Cx43 expression, we observed a tendency toward a positive correlation of these parameters, suggesting the involvement of Cx43 in the processes of extracellular matrix production and differentiation. Even though osteonectin and osteocalcin were employed as a part of diagnostics on this study, positive staining for each was found in variable degrees in all tumors studied. Although it is more specific, osteocalcin is used as a marker for osteoblast differentiation.
Our findings comparing the index of cellular proliferation with levels of Cx43 demonstrate a discreet inverse correlation, meaning that higher amounts of Cx43 are associated with smaller percentages of proliferative cells. Survival and evidence of malignancy in human osteosarcoma are related to PCNA immunolabeling. 38,46,48,57 An interesting datum was the higher proliferative index in osteosarcomas from dogs affected in appendicular skeleton, with a tendency toward statistical significance, suggesting that tumors located in this region are more aggressive. The proliferative index was also compared with survival, finding a possible tendency of a lower cellular proliferation index in animals with higher survival when compared with animals that died earlier.
Koval et al. 21 showed that the UMR 160-61 lineage of osteosarcoma and calvarial rat lineage of osteoblast express Cx46 with a molecular weight of 53 kDa. Cx46 expressed by osteoblasts was mainly located in a perinuclear intracellular compartment, which, according to the authors, seems to be consistent with the trans-Golgi region, as determined by immunofluorescence. 21 A small amount of Cx46 was found on the cell surface. In the same experiment, the authors mentioned that Cx46 presented bands in the Western blot membrane with 2 molecular weights, 68 kDa and 53 kDa. The same data were observed by other authors. 16,20 The 68 kDa band could correspond to the phosphorylated form of Cx46. We observed that Cx46 is expressed in neoplasic osteoblasts around cell nuclei in variable degrees as described by Koval et al. 21 However, we found by qPCR that gene expression of Cx46 in the bone tumors of most animals was decreased when compared with its expression in normal bones. The process of oligomerization, the formation of a hemi-channel in an intracellular compartment before it can migrate to the cytoplasmic membrane, is necessary for GJIC formation. 23 These data suggest that there might be an accumulation of these proteins in a monomeric form as described by Koval et al. 21 The function of connexin and whether there is a correlation to tumor suppressing effects in normal and neoplastic tissues are still unknown. 21,44,69
In summary, our results indicate that Cx43 and Cx46 are expressed in normal bone and canine osteosarcoma. There is a trend toward increased Cx43 and osteoid deposition, and a trend toward lower Cx43 as PCNA increases. These suggest that high Cx43 expression correlates with a better survival outcome measured as a function of PCNA expression, but further functional and mechanistic studies need to be performed to confirm these data. Similarly, other authors have shown that reduction of proliferation was observed in osteosarcomas after the transcription of the Cx43 gene in association with the increase in p27 inhibitor levels, a protein involved in the control of cell-cycle progression. 7,69 Other authors such as Trosko et al. described an increase in antitumor activity of connexins when induced by antineoplasic agents as retinoids. 69 Thus, we believe that the study of agents that can modulate the expression of connexins in normal bone tissues, and maybe its transportation to cell membranes is important for the prevention of bone neoplasms in dogs. In this context, agents that increase the expression of connexins could be used as adjuvant in the treatment of canine osteosarcomas.
Footnotes
Acknowledgements
This work is part of the MS dissertation of D. S. Sanches in the Experimental and Comparative Pathology Program of the School of Veterinary Medicine and Animal Science of the University of São Paulo. D. S. Sanches was the recipient of a MS fellowship from the Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP), Process Number 05/57902-0. This research was supported by grants (Process Number 06/56512-6) from FAPESP.
