Abstract
A meningeal osteosarcoma was diagnosed in a dog displaying neurologic signs compatible with a space-occupying cerebellar lesion. Gross lesions, restricted to the brain, consisted of a solitary, compressive mass attached to the dura mater overlying the left cerebellum. The mass was composed of single and multinucleated, atypical polygonal cells that lined or rested within lacuna surrounded by eosinophilic, mineralized matrix. The matrical component stained dark green-yellow to blue with Movat's pentachrome stain, deep blue to red with Heidenhain aniline blue stain, and brown-black with Von Kossa stain. Results of these stains were interpreted as tumor osteoid. Foci of dural mineralization and osseous metaplasia were present at the point of tumor attachment. The microscopic observations were interpreted as an osteosarcoma of extraskeletal origin. To our knowledge, these findings represent the first documented case of a meningeal osteosarcoma in a domestic animal species.
Extraskeletal osteosarcomas are bone-producing tumors of mesenchymal origin that lack an association with either bone or periosteal tissue. In dogs, these tumors have been reported at cutaneous (including mammary gland), muscular, gatrointestinal, visceral (spleen, liver), endocrine (thyroid, adrenal gland), urinary tract (kidney, bladder), reproductive (testis, vagina), and ocular sites. 8 11 Osteosarcomas of meningeal origin have been documented in humans and in an albino rat. 3 9 In this report, we describe a canine meningeal osteosarcoma located in close spatial association with foci of dural mineralization and metaplastic bone formation.
A 10-year-old, neutered male English Pointer dog presented to a private veterinary practitioner with a history of an unsteady gait. On physical exam, the dog was ataxic and had nystagmus. A primary brain disorder was suspected, but financial constraints restricted further diagnostic workup. The dog was discharged on a course of novabiocin, tetracycline, and corticosteroids (Delta Albaplex Upjohn, Kalamazoo, MI), with orders for the owner to monitor any change in the animal's condition. Over a 10-day period, the animal developed a progressive hypermetric gait and periodic grand mal seizures. The owner declined further treatment and the dog was euthanized (Beuthanasia, Shering, Union, NJ) immediately before necropsy.
Gross lesions were restricted to the brain. The anterior half of the left cerebellum was compressed by a firm, expansive, multilobate, white–tan mass measuring 2.75 × 1.5 × 1.75 cm (Fig. 1). The bulk of the mass had a fibrous consistency with a few gritty areas. The mass was attached to thickened portions of superior cerebellar dura mater and extended rostrally to incorporate portions of tentorium cerebelli. The mass was distinctly separate from the dorsal calvarium and tentorium osseum. Gross evaluation of the appendicular skeleton revealed no primary bone tumors. No evidence of distant metastasis was present within the lungs or any abdominal viscera. The axial skeleton was not evaluated.
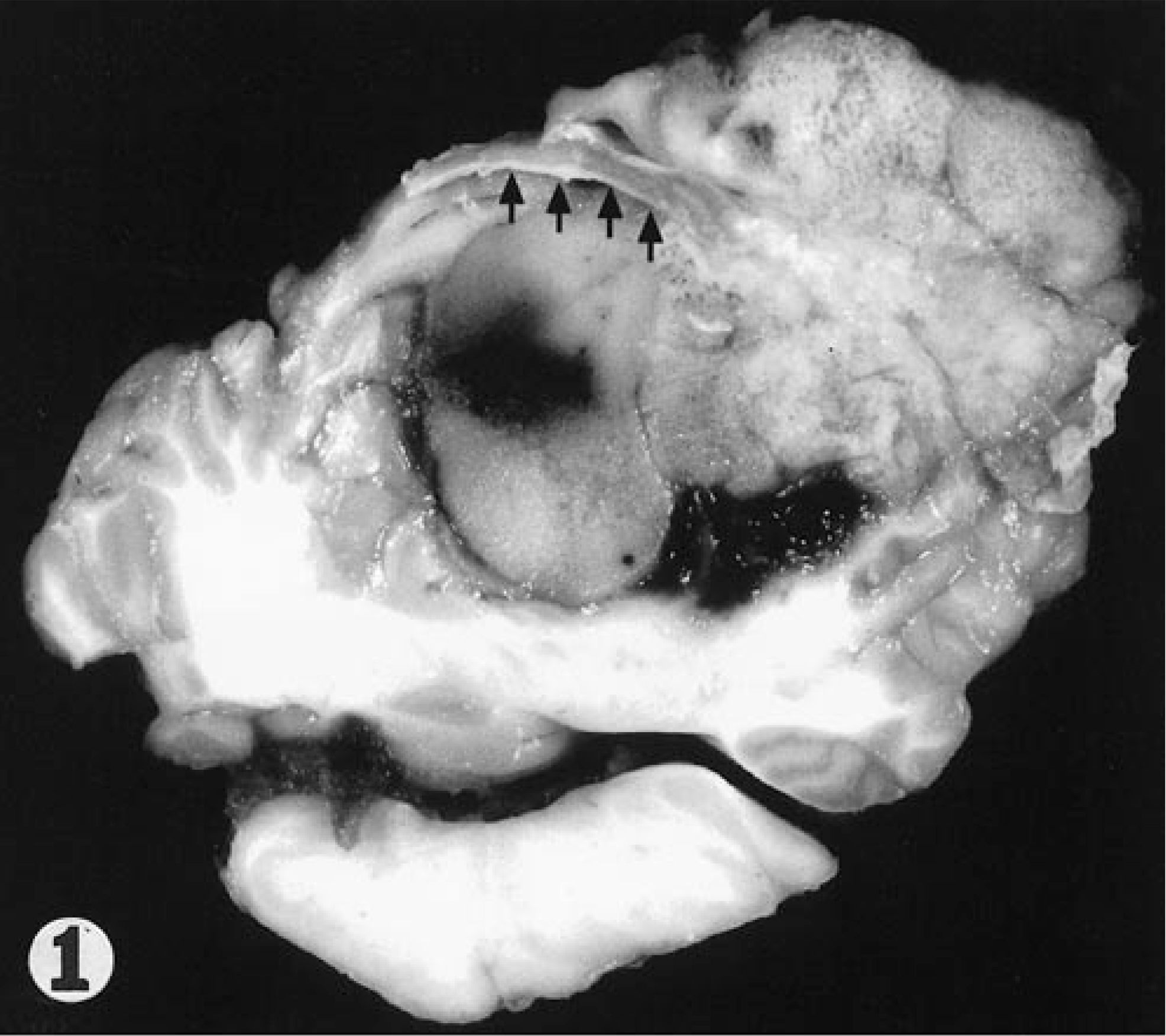
Cerebellum; dog. Extending from the dura mater is a multilobate, expansive mass causing compression of the adjacent cerebellum. Note the thickened dura mater (arrows) in the region adjacent to the tumor.
Tissues were fixed in 10% neutral buffered formalin, decalcified, and processed for histopathologic evaluation using a standard procedure. Microscopically, the mass was of moderate cell density and consisted of large, atypical polygonal cells that varied in size and morphology. Cells of the mass lined, or were interspersed between, irregular islands of hyalinized, slightly fibrillar and commonly mineralized matrix (Fig. 2). The matrical component stained dark green–yellow to blue with Movat's pentachrome stain, deep blue to red with Heidenhain aniline blue stain, and brown–black with Von Kossa stain. Results of these stains were interpreted as tumor osteoid. Fewer atypical cells were housed within lacuna of osteoid and mineralized matrix. Individual cells had distinct cell borders, increased nuclear to cytoplasmic ratios, and moderate quantities of pale, amphophilic cytoplasm. Faint cytoplasmic stippling with eosinophilic granules was present in scattered cells. Anisokaryosis and nuclear pleomorphism were prevalent. The majority of nuclei were large, hypochromatic, and contained a vesicular pattern of chromatin dispersion. A single, irregular, and variably sized, central to paracentrically situated nucleolus was most commonly present. Bi- and multinucleated cells were frequent throughout the mass (Fig. 3). Mitotic figures ranged from zero to three per field at 400× magnification. The affected portions of cerebellum and dura mater were step sectioned, with similar cell populations present in all slides examined. Cartilage differentiation, hemorrhage, and necrosis were minor features within the mass. Immunohistochemical stains exhibited moderately strong, diffuse cytoplasmic stain uptake with antibody against vimentin intermediate filaments and no observable cellular staining against cytokeratin antibody. Microscopic changes in sections of cerebellar folia and peduncle white matter tracts adjacent to the mass included foci of hemorrhage, necrosis with gitter cell accumulation, diffuse astroglial proliferation, generalized endothelial hypertrophy, and neovascularization. Thickened portions of dura mater bordering the mass contained proliferating fibroblasts and foci of mineralization. Heterotopic (metaplastic) bone containing fat and a few hematopoetic cells was multifocally present in thickened dural regions. A diagnosis of meningeal osteosarcoma was made based on morphologic criteria for malignancy, regionally elevated mitotic activity, hydroxyapatite crystal precipitation within a foundation of collagen matrix, and lack of lamellar remodeling. Psammamatous meningioma, the primary differential diagnosis for tumors arising in this location, was ruled out based on lack of cellular features or architecture patterns consistent with a meningioma. 1 Focal positive staining against epithelial markers (epithelial membrane antigen and cytokeratin) have been described in human meningiomas. 1 Based on our immunohistochemical results, no evidence of epithelial differentiation was suggested in this case. To our knowledge, expression of epithelial markers in canine meningiomas have not been definitively characterized, nor has the specificity of epithelial membrane antigen been reported for canine tissues. Therefore, immunohistochemical results offer little, if any, additional information for addressing the histogenic origin of this tumor.
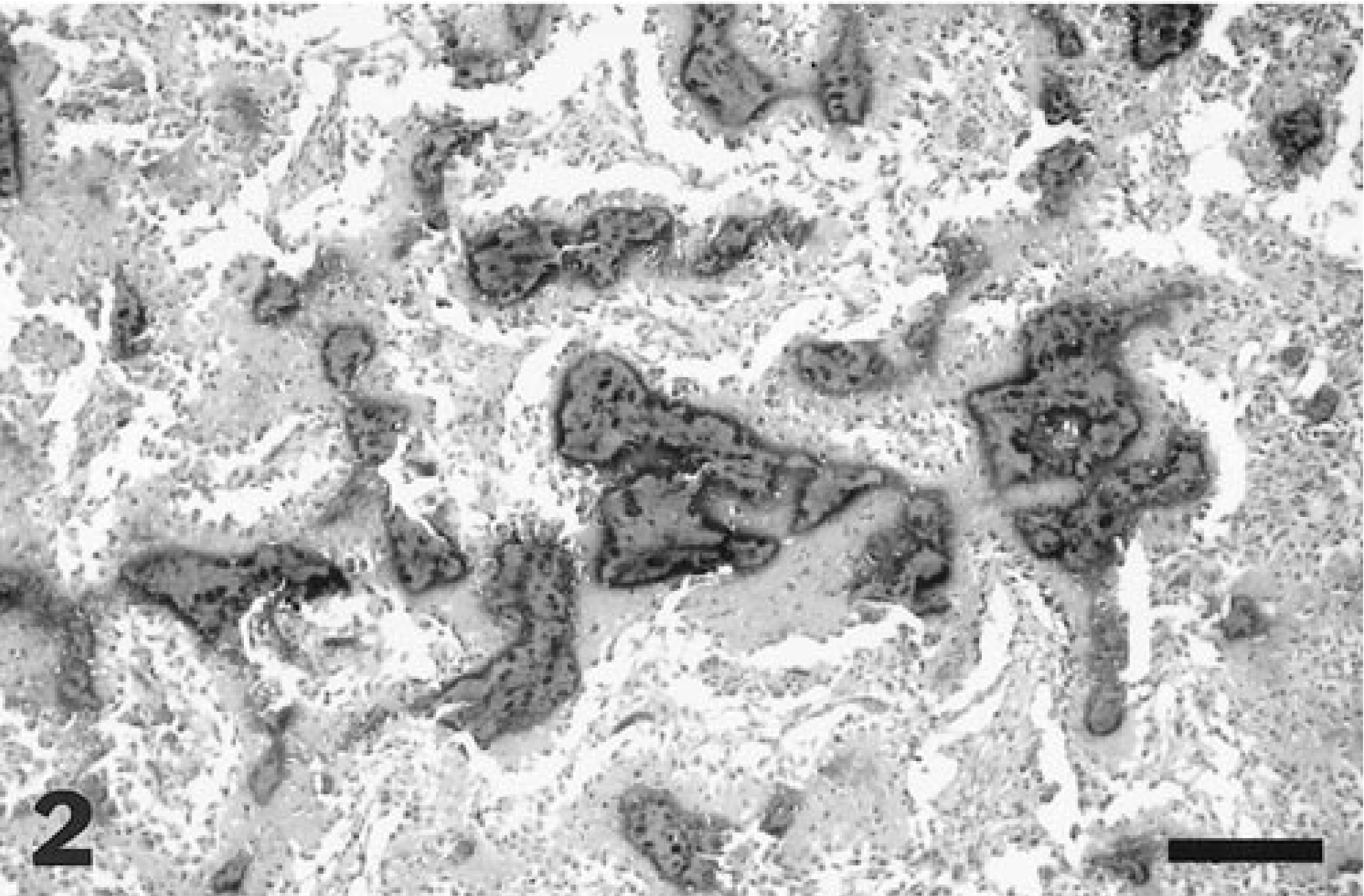
Cerebellar mass. The mass is composed of a pleomorphic population of atypical polygonal cells lining and interspersed between irregular islands of hylanized, often mineralized matrix. Bar = 160 µm.
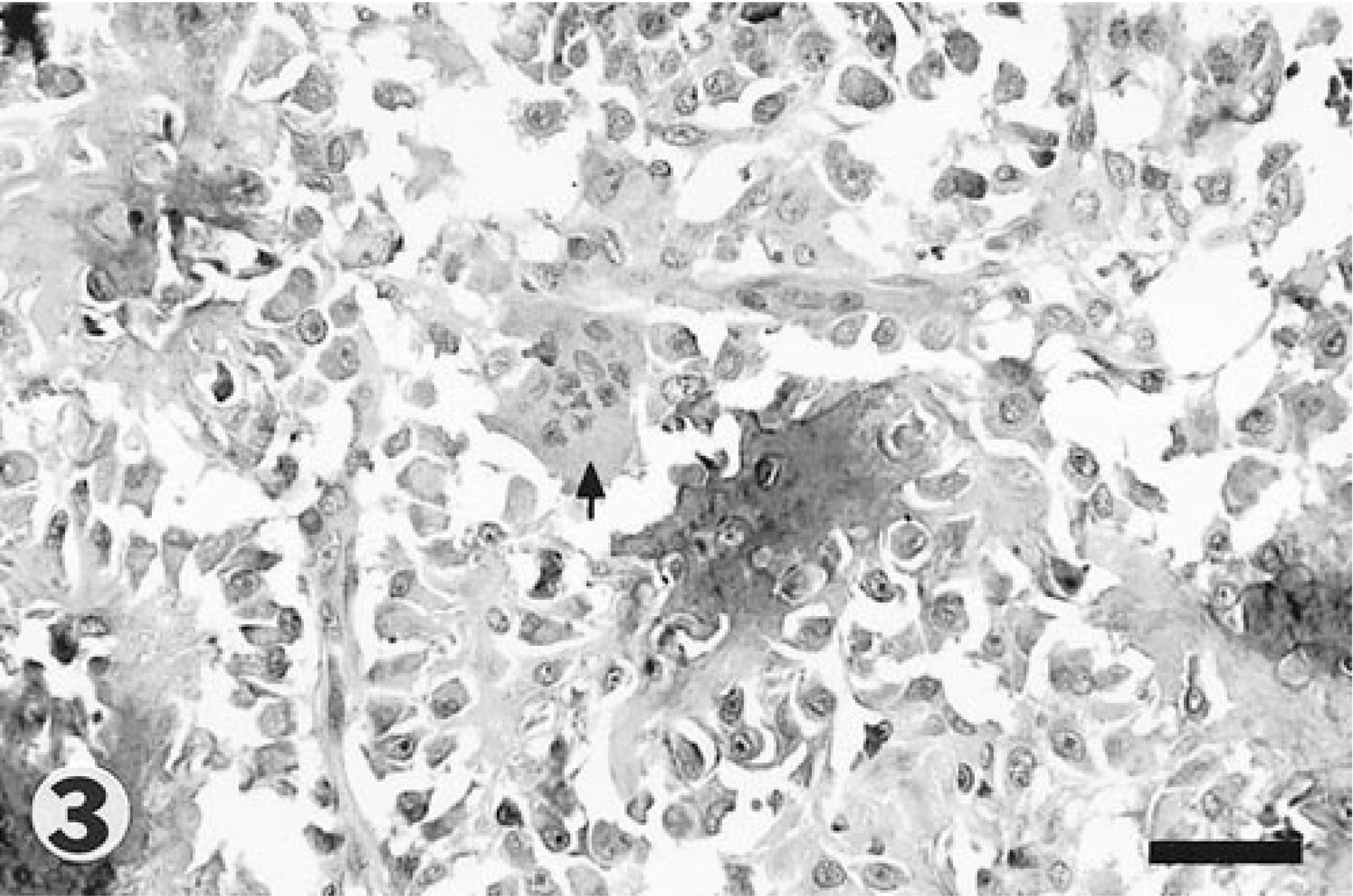
Cerebellar mass. Higher magnification of the mass, with single and multinucleated cells (arrow) displayed. A small proportion of the cells are housed within osteoid lacunae. Bar = 80 µm.
The changes in the dura mater (mineralization, heterotopic ossification, and neoplastic bone formation) could be coincidental. Alternatively, these findings may represent a causal association, related sequentially by initial mineralization of the dura mater, followed by heterotopic ossification and culminating with malignant transformation of the heterotopic bone.
Tissue mineralization, a common occurrence detected in a variety of normal and disease conditions, is a relatively well-understood process. 4 In extracellular calcification, nucleation commences with calcium binding to phospholipids on membrane-bound vesicles of degenerating and aging cells. Activation of membrane phosphatases leads to a repetitious cycle of calcium/phosphate binding. When threshold ion concentrations are reached, calcium deposits form. Structural rearrangement of calcium and phosphate groups within these calcium deposits results in microcrystal formation. Ultimately, the extent of microcrystal propagation is determined by ion concentrations, inhibitors, and regulators in the local, extracellular matrix.
Ectopic (heterotopic) ossification within mineral deposits is a predictable, metaplastic event in certain tumors and lesions containing a background of longstanding inflammation. Formation of ectopic bone is considered a common, incidental lesion in the cerebral and spinal dura mater (ossifying pachymeningitis) of aged dogs. 7 16 These metaplastic changes arise from resident populations of undifferentiated mesenchymal cells with osteogenic potential, termed inducible osteogenic precursor cells (IOPCs). 2 These IOPCs are distinguished from determined osteogenic precursor cells (DOPCs) of marrow origin based partially on initial dependency and continued persistence of appropriate inducer(s) in the tissue. Endowed with multipotentiality, both precursor cell types possess the capacity to establish all marrow lines (Fig. 4). A host of cytokines (transforming growth factors β1 and β2), bone morphogenetic proteins (BMP1, BMP2, and BMP3), BMP regulators (noggin, chordin, cereberus, dan and gremlin), and matrix constituents (decorin, bone sialoprotein, osteopontin, osteonectin, and osteocalcin) influence the continued activity and extent of bone formation. 4–6, 10 13 15 17 Transforming growth factor (TGF)-βs and related bone morphogenetic proteins (BMPs) are particularly important in osteoinduction, stimulating differentiation of primitive mesenchymal cells into bone-forming tissues. 15

Origin of determined osteogenic precursor cells from stromal progenitor cells. Osteogenic precursor cells in extraskeletal tissues (inducible osteogenic precursor cells [IOPCs]) follow an analogous pattern of differentiation. Adapted from Puzas et al. 12 ∗CFU-F = colony-forming unit–fibroblastic.
Although malignant transformation of heterotopic bone is not a specifically described pathologic process, it remains a conceptually plausible explanation for the changes occurring in the dura mater of this animal. A similar mechanism may apply to other extraskeletal osteosarcomas, such as feline vaccine-induced osteosarcomas and feline trauma-induced lenticular osteosarcomas. Both of these latter tumors often originate in a background of chronic inflammation. The continued persistence of cytokines and growth factors that perpetuate metaplastic tissue formation provides a fertile environment for malignant transformation. 4 Direct evidence for such a mechanism is provided in humans, where pulmonary carcinoma is a well-documented consequence of transformed metaplastic tissue (squamous metaplasia) in lungs of chronic cigarette smokers. 4 The potential for neoplastic transformation of heterotopic bone is supported by experimental evidence demonstrating disparate behavior between osteoblasts derived from normal versus heterotopic bone. 14 Compared to cultivated osteoblasts of normal bone, osteoblast-like cells of heterotopic origin have increased metabolic activity coinciding with a three- to fourfold increase in cell density. Utilizing in vitro clonogenic assays, these differences have been shown to be partly attributable to an increased fraction of mitotically active cells in heterotopic bone.
In conclusion, we describe the gross and histologic features of a meningeal osteosarcoma in a dog. Although the axial skeleton of this animal was not examined, the animal displayed no clinical signs or spinal deformations suggestive of an advanced primary tumor of the vertebral column. In addition, the origin of the mass was identified in the dura mater at gross and histologic examination. Thus, the meningeal mass was considered to be a primary neoplastic lesion. With respect to the biologic behavior of this tumor, too few cases exist to permit a prognostic generalization. The absence of cranial vault involvement or metastasis are findings similar to reported cases in humans and in the rat. The additional presence of colocalized dural mineralization and ectopic ossification is an interesting association, although the nature of this relationship is unclear.
