Abstract
Gap junctions are the only communicating junctions found in animal tissues and are composed of proteins known as connexins. Alterations in connexin expression have been associated with oncogenesis; reported studies in rodent and human mammary glands, which normally express connexins 26 and 43, confirm these alterations in malignancies. Mammary neoplasms represent the second most frequent neoplasm in dogs, and since there are no reports on the study of connexins in canine mammary glands, the present study investigated the expression of connexins 26 and 43 in normal, hyperplastic, and neoplastic mammary glands of this species, to verify if altered patterns of connexin staining are related to higher cell proliferation and malignant phenotypes. A total of 4 normal, 8 hyperplastic mammary glands, 9 benign, and 51 malignant mammary gland neoplasms were submitted for the immunostaining of connexins 26 and 43, E-cadherin, and proliferating cell nuclear antigen (PCNA). Normal, hyperplastic, and benign neoplastic mammary glands showed a punctate pattern for connexin 26 and 43 staining and an intercellular E-cadherin staining. Malignant neoplasms, especially the most aggressive cases with high cell proliferation rates, presented either fewer gap junction spots on the cell membranes or increased cytoplasmic immunostaining. Malignant tumors also expressed a less intense immunostaining of E-cadherin; the expression of this adhesion molecule is important for the transportation of connexins to cell membranes and in forming communicating gap junctions. Deficient expression of E-cadherin could be related to the aberrant connexin localization and may contribute to the malignant phenotype. In conclusion, the expression and distribution of connexins and E-cadherin are inversely correlated to cell proliferation in malignant mammary neoplasms of dogs and may well be related to their more aggressive histologic type and biologic behavior.
Gap junctions are the only communicating junctions found in animal tissues, in all species, which are responsible for the direct traffic of molecules with molecular weights less than 1,000–2,000 d. The gap junctions are not identical in all tissues but share a basic structure with subunits of six proteins, the connexins, that form a hemi-channel, the connexon, in each plasma membrane and are phylogenetically preserved proteins. 11, 39, 40
Because of the importance of intercellular junctions in the maintenance of the cellular homeostasis, the modulation of intercellular junctions and of connexin expression seems to be involved in carcinogene-sis. 8, 10, 12, 14, 16, 36, 39, 41 Evidence for the participation of gap junctions and connexins in tumorigenesis is based on several in vitro studies that have demonstrated a prominent reduction in gap junction intercellular communication (GJIC) capacity in most human and rodent cell lines. 2, 18, 32, 42 Besides this, a large group of tumor promoters inhibit the GJIC capacity in cell cultures. 36 More recently, the role of gap junctions in cancer has been studied in vivo, with the help of rodent animal models. 16, 22, 23, 28, 38 However, to our knowledge, there are no reports about the study of connexins and gap junctions in domestic animal tumors.
In rodent mammary glands, two connexin types, the Cx43 and the Cx26, form gap junctions. In human mammary glands, the expression of Cx26 has been described in epithelial cells, and the expression of Cx43 has been described in myoepithelial cells. 6, 15, 22, 23, 28, 38
Mammary neoplasms are very frequently diagnosed in female dogs of several breeds. Their classification is based on the histologic characterization of epithelial cell proliferation, pleomorphism, invasion, typical and atypical mitosis, and the accompanying proliferation of myoepithelial cells. The proliferation index, evidenced by the proliferating cell nuclear antigen (PCNA), has been correlated with the histologic type of neoplasm as well as with the tumor prognosis. 3, 17, 20, 29
One of the mechanisms that seems to be involved in the regulation of the intercellular junctions is the relationship between connexins, gap junctions, and adhesion molecules, particularly E-cadherin. These play a role in the adhesion between individual epithelial cells. 4, 5, 7, 9, 35 The expression of E-cadherin is also related to the histologic tumor types. It has been suggested that the reduced expression and abnormal distribution of E-cadherin are related to the differentiation of epithelial neoplasms 31 and to metastasis. 26
In this study, the histologic tumor types of dog mammary glands were related to the expression of connexins 26 and 43, and these were compared with the expression of E-cadherin and the proliferation index detected by PCNA. The relationship of these parameters with the biologic behavior of the neoplasms was also investigated.
Materials and Methods
Samples
A total of 68 mammary lesions, including 8 hyperplasias, 9 benign neoplasms, and 51 malignant neoplasms, were studied. These were obtained from 33 female dogs from the Surgery Service of the Veterinary Hospital, Faculty of Veterinary Medicine and Zootechny, University of São Paulo. All lesions were classified according to the criteria of the World Health Organization in 1999. 19
The dogs were monitored for 18 months to ascertain the outcome of the neoplasms. Recurrence was observed in five cases of malignant neoplasms, and metastases were present in six dogs. Malignant tumors metastasized to the inguinal lymph node, to the lungs, or both. Three animals died due to malignant neoplasms.
Immunohistochemical staining for the detection of connexins, E-cadherin, or PCNA
All tumor samples were fixed in methacarn (70% methanol, 20% chloroform, and 10% acetic acid) and embedded in paraffin. The 5-μm histologic sections from the mammary glands were routinely deparaffinized, rehydrated, and incubated with a solution of methanol and hydrogen peroxide (10%) for 30 minutes. The slides were incubated with anti-Cx26 (Zymed-71-0500) or anti-Cx43 (Zymed-71-0700) polyclonal antibodies diluted 1 : 1,000 in blocking buffer (TNB) at 4°C overnight. Anti-rabbit biotinylated antibody (Dako—E0432) was used as the link (1 : 1,000 in phosphate-buffered saline [PBS]) for 1 hour in a humidified chamber. The amplification was performed with a TSA kit (Tyramide Signal Amplification Fluorescein System) and shown with fluorescein. The histologic sections were analyzed with a confocal microscope (Axiovert 100M—Zeiss, LSM 510 system). Liver or heart samples from dogs were used as positive controls for the immunostaining of connexins 26 or 43, respectively. Negative controls omitted the primary antibody in a histologic section while performing all the other immunostaining steps.
For the immunostaining of E-cadherin, additional sections were obtained from the methacarn-fixed, paraffin-embedded lesions, which were positioned in silanized slides. The de-paraffinized and hydrated histologic sections were incubated with monoclonal anti–E-cadherin antibody (clone G-10, monoclonal—Santa Cruz 1 : 4,000) at 4°C overnight. After incubation with the secondary anti-mouse antibody, the reaction was amplified with a CSA kit (Catalyzed Signal System—DAKO) and detected by staining with diaminobenzidine. Negative controls were obtained by performing the same procedure while omitting the primary antibody.
For the staining of PCNA, histologic sections were incubated with monoclonal anti-PCNA (clone PC10, DAKO M 0879) antibody (1 : 4,000, 4°C, overnight). After the incubation with the secondary biotinylated antibody (1 : 100 in PBS), the sections were incubated with the streptavidin–biotin–peroxidase complex (DAKO, 1 : 100 in PBS), and the reaction was detected with a solution containing diamino-benzidine and hydrogen peroxide.
Scoring of immunoreactivity
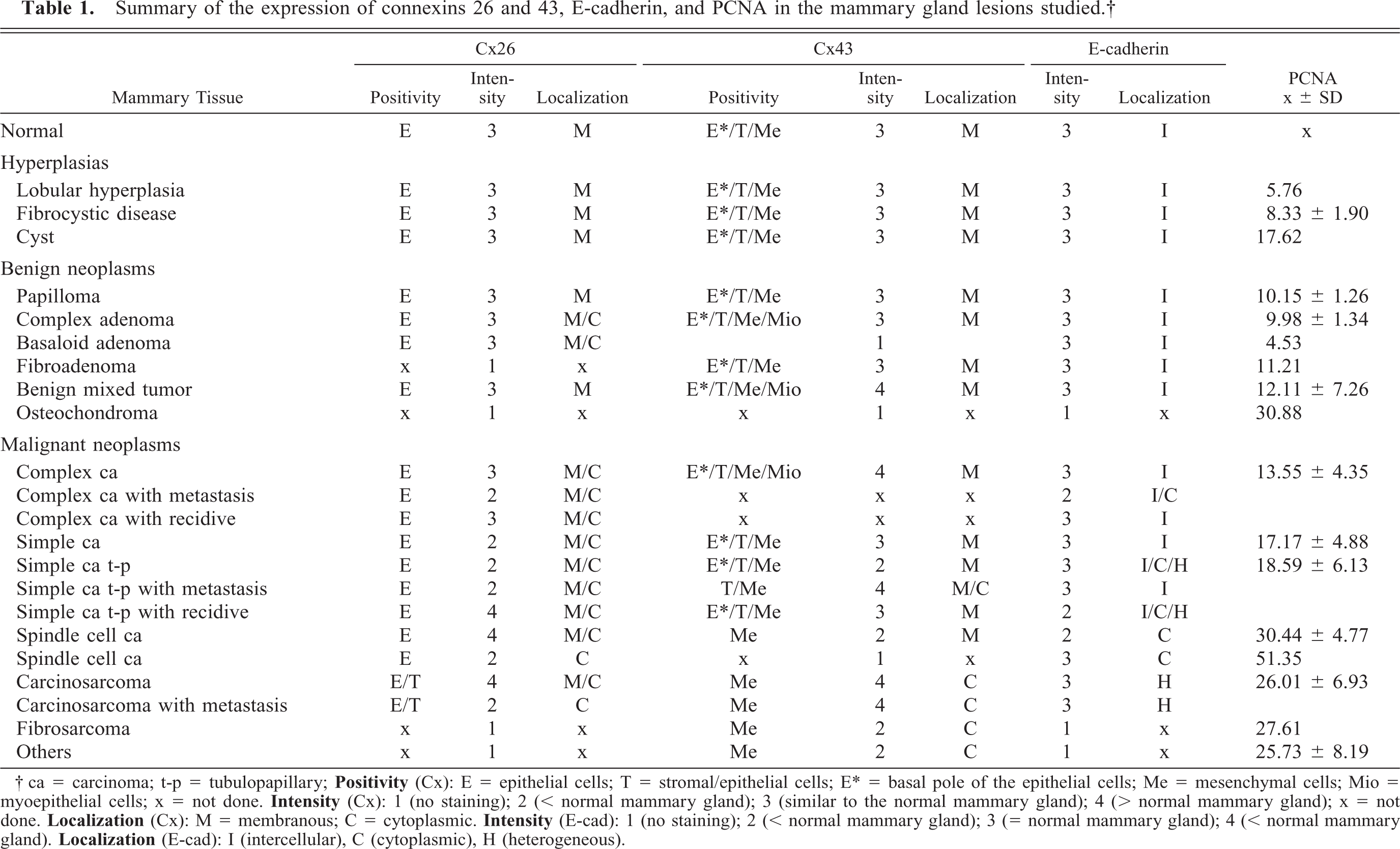
Criteria for scoring the immunoreactivity of connexin 26 or 43 were based on the following: 1) cell type that presented positivity (E = epithelial cells; E∗ = basal pole of the epithelial cells; T = stromal/epithelial cells; Me = mesenchymal cells; Mio = myoepithelial cells); and 2) intensity of immunostaining: 1 (no immunostaining), 2 (less intense than a normal mammary gland), 3 (similar to a normal mammary gland), and 4 (more intense than a normal mammary gland); subcellular localization of the staining: M (membranous) and C (cytoplasmic).
Criteria for the scoring of the immunoreactivity of epithelial cells to E-cadherin in mammary tissue included: E-cad + or score 3 (positivity similar to normal mammary tissue), E-cad ± or score 2 (less intense than normal mammary tissue) or E-cad—or score 1 (no positivity to E-cadherin). The distribution of E-cadherin in mammary tissue was classified as: I—intercellular (intercellular positivity), H—heterogeneous (heterogeneity of staining: some cells exhibit a strong immunostaining pattern, among other weakly stained ones), and C—cytoplasmic (positivity in the cytoplasm of neoplastic cells).
Counting 1,000 PCNA-positive nuclei (objective, 40×), recording the positive and the strongly positive nuclei, was used to assess cell proliferation.
Statistical analysis
The differences between the means of the PCNA-labeling index values in hyperplasias, benign neoplasms, and malignant mammary neoplasms were statistically analyzed by the Kruskal-Wallis nonparametric test (P < 0.05). The intensity of immunostaining of the connexins 26 and 43 and E-cad-herin in normal, hyperplastic, or neoplastic (benign and malignant) mammary glands (scored as described above) was correlated to the cell proliferation indexes by the Pearson linear correlation index.
Results
Immunostaining of Cx26 and Cx43
The immunostaining of connexins 26 and 43 is presented (Figs. 1, 2, Table 1).
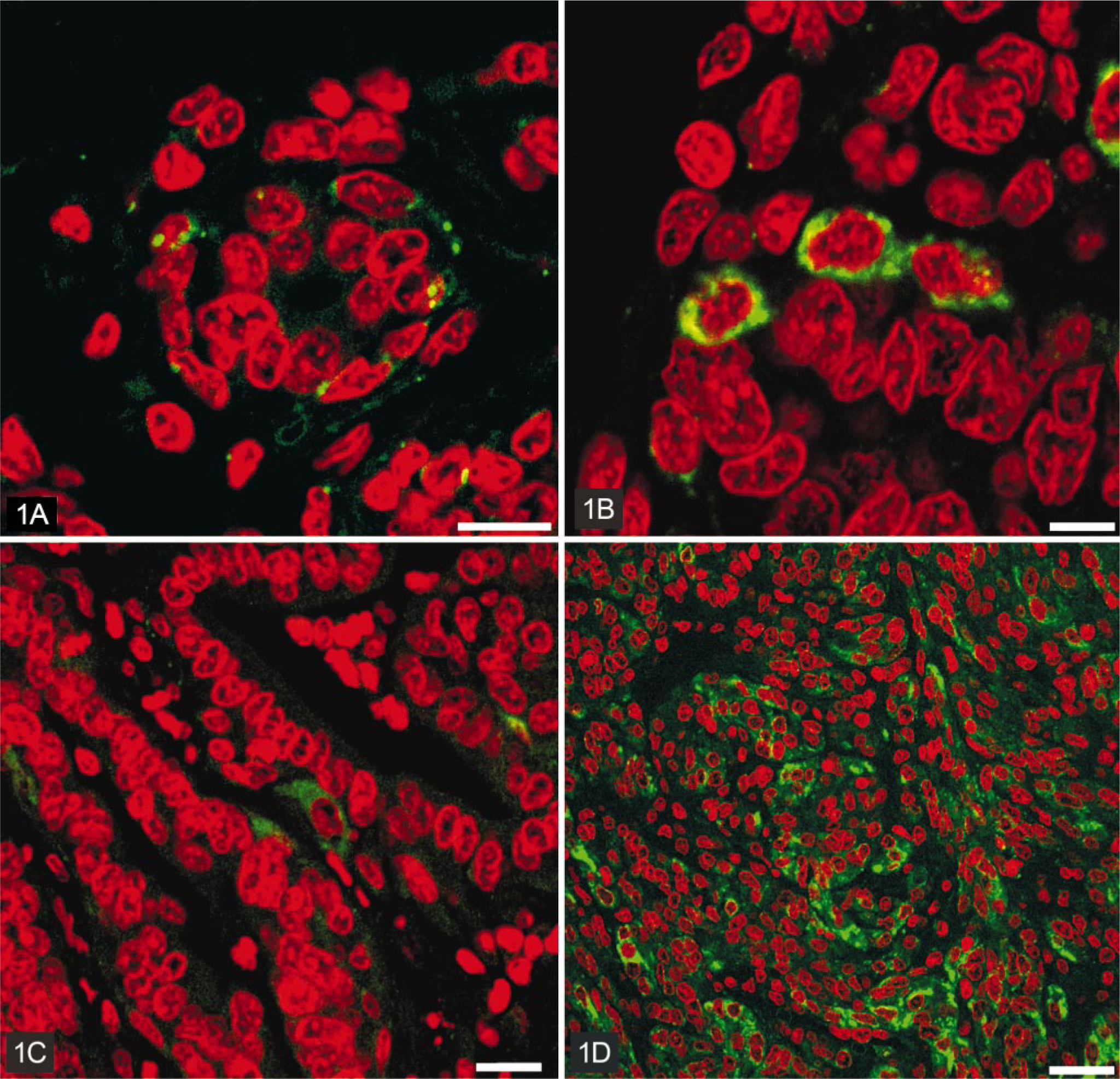
Immunostaining of connexin 26 in canine mammary gland.
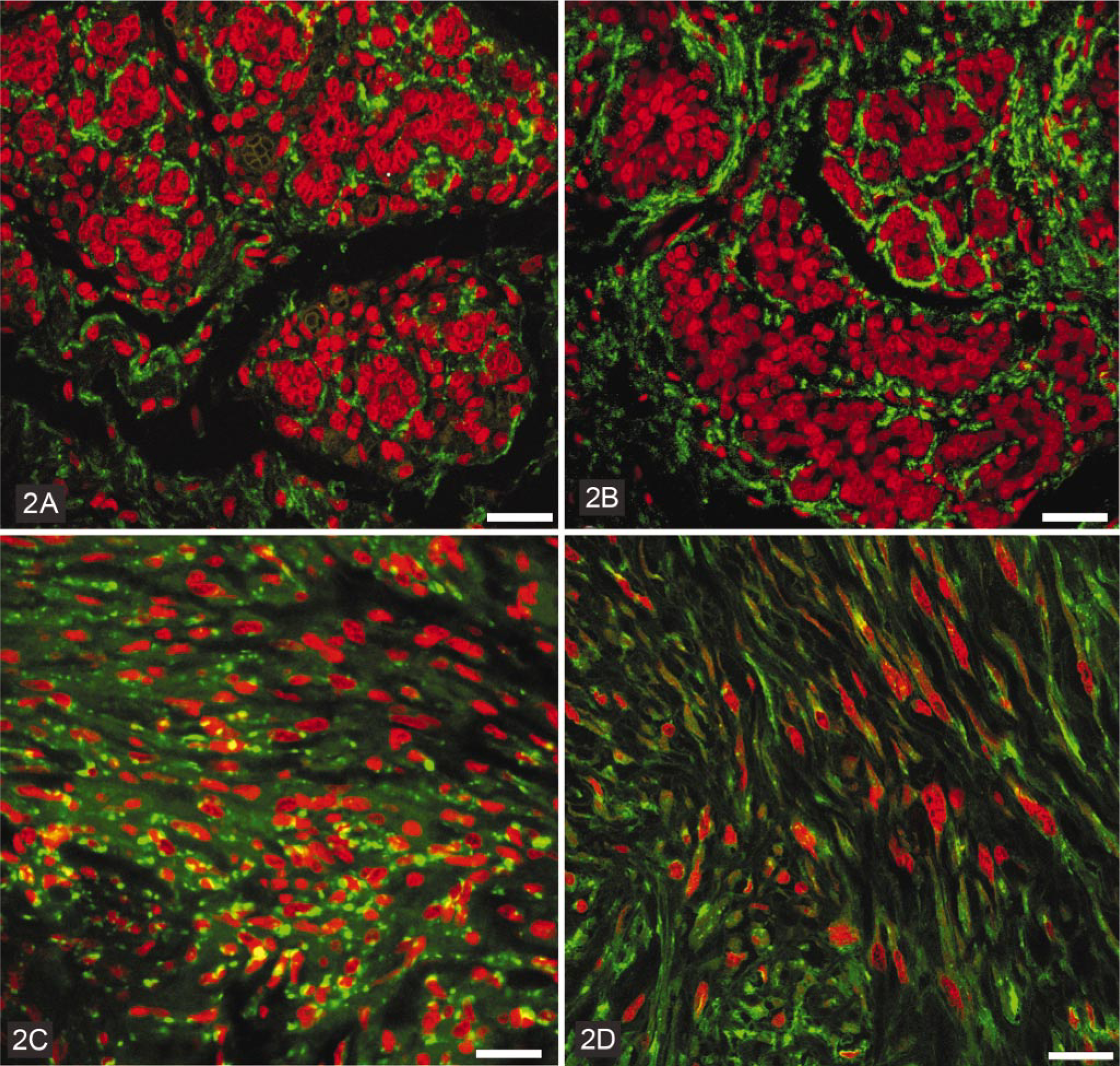
Immunostaining of connexin 43 in canine mammary gland.
Summary of the expression of connexins 26 and 43, E-cadherin, and PCNA in the mammary gland lesions studied.†

† ca = carcinoma; t-p = tubulopapillary;
In hyperplastic mammary tissues, the pattern of immunostaining of both Cx26 and Cx43 was similar to that of normal mammary glands. Epithelial cells were highly positive for Cx26, and the immunostaining was strictly localized in the cell membranes (Fig. 1). The basal poles of epithelial cells and mesenchymal cells were strongly positive for Cx43, which was localized in the cell membranes (Fig. 2).
Most benign mammary neoplasms showed the same pattern of immunostaining when compared to normal and hyperplastic mammary glands. However, complex and basaloid adenomas also showed some cytoplasmic staining for Cx26. Fibroadenomas and osteochondromas did not present any positivity for Cx26. Most benign neoplasms stained for Cx43 in a manner similar to hyperplastic/normal mammary glands. Some Cx43-positive myoepithelial cells were observed in complex adenomas and in mixed benign tumors of the mammary glands; these tumors exhibited a higher intensity of immunostaining for Cx43 when compared to normal/hyperplastic mammary glands. Neither the basaloid adenomas nor the osteochondromas showed any positivity for Cx43.
Most malignant mammary neoplasms demonstrated positivity for Cx26 in epithelial cells. The intensity of Cx26 staining varied from no immunostaining, in fibrosarcomas and sarcomas of uncertain origin, to a strong immunostaining for Cx26 in simple carcinomas, spindle cell carcinomas, and in carcinosarcomas. We observed that most malignant tumors exhibited cytoplasmic and/or membranous staining for Cx26, although squamous cell carcinoma and metastatic carcinosarcoma presented only a cytoplasmic staining (Fig. 1). The pattern of immunostaining of Cx43 was quite variable in malignant mammary gland neoplasms. Some of them did not demonstrate any immunostaining (complex carcinoma and squamous cell carcinoma) for Cx43, and many presented strong staining only for Cx43 in mesenchymal (stromal) cells. The intensity of immunostaining was higher than in normal/hyperplastic mammary glands in simple and complex carcinomas and in both metastatic and nonmetastatic carcinosarcomas. Positive spots of Cx43 staining were localized in cell membranes, but in carcinosarcomas and fibrosarcomas, this was only seen in the cytoplasm.
Immunostaining for E-cadherin
The immunostaining of E-cadherin in hyperplastic and benign neoplasms from the mammary glands was similar to normal mammary glands (Fig. 3). The immunostaining was localized between the epithelial cells, confirming the normal localization of this adhesion molecule. In malignant mammary gland neoplasms, the pattern of immunostaining of E-cadherin was variable. In metastatic simple and complex carcinomas, as well as in spindle cell carcinomas, the immunostaining was less intense, and in the mammary gland fibrosarcoma, there was no immunostaining for E-cadherin at all. The localization of the immunostaining of E-cadherin was also quite variable in malignant neoplasms. Many of them stained not only intercellularly, but also in the cytoplasm; some cells exhibited a strong immunostaining pattern, among other weakly stained ones, referred to as a heterogeneous pattern (Fig. 3).
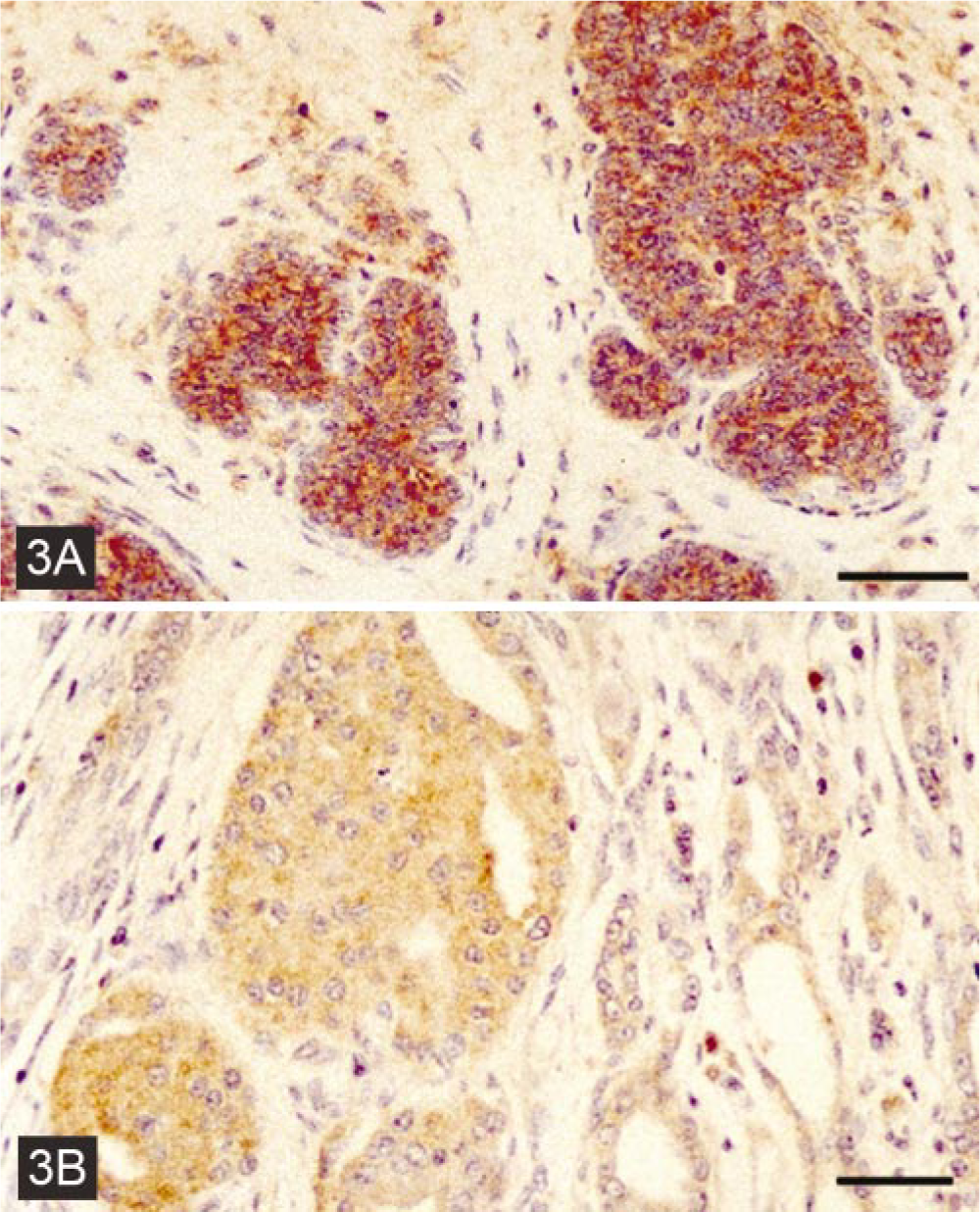
Immunostaining of E-cadherin in mammary ducts.
Immunostaining of PCNA and quantification of S-phase cells
The results of PCNA-positive cell quantification counting 1,000 PCNA-positive nuclei (objective, 40×), recording the positive and the strongly positive in normal and hyperplastic, benign and malignant neoplasms from canine mammary glands, are shown in Table 1. Malignant mammary neoplasms demonstrated more PCNA-positive cells when compared to normal, hyperplastic glands or benign mammary neoplasms.
Statistical analysis and Pearson linear correlation
Table 1 shows all the results together, and Table 2 shows the means of the immunostaining intensity scoring values, which were used to calculate the Pearson correlation index. Malignant neoplasms presented significantly higher PCNA-labeling indices when compared to benign neoplasms and hyperplasias. The Pearson linear correlation coefficient value (r) was negative for the correlations between Cx26 and PCNA (r = −0.2675), Cx43 and PCNA (r = −0.4792), and E-cadherin and PCNA (r = −0.4383). The Pearson linear correlation coefficient value (r) was positive for the correlation between Cx26 and E-cadherin (r = 0.5691).
Linear correlation between the expression of connexins 26 and 43, E-cadherin, and PCNA according to the lesion type. Values are represented as mean ± standard deviation of the scoring system.†.
† Linear correlation coefficient: Cx26 × PCNA r = −0.2675; Cx43 × PCNA r = −0.4792; E-cad × PCNA r = −0.4383; Cx26 × Ecad r = 0.5691.
∗ P = 0.0204, considered significant, Kruskal-Wallis test (nonparametric ANOVA).
Discussion
The goal of this study was to describe the pattern of immunostaining of Cx26 and Cx43 in dog hyperplastic and neoplastic mammary glands, in comparison with normal canine mammary glands, in an attempt to verify if a correlation with cell proliferation and the malignant phenotype could be found. Because of the lack of studies focusing on connexins in domestic animal neoplasms, we consider this a pioneer work in the field.
The results obtained in this study show that the immunostaining of connexins 26 and 43 in normal mammary glands is comparable to the results obtained by Jamieson et al. 6 with normal human mammary glands. The immunostaining of Cx26 and Cx43 in hyperplastic mammary tissue and in benign mammary tumors showed no differences when compared to normal tissue.
One general finding was a reduction in the expression of connexins 26 and 43 in malignant mammary gland neoplasms. However, the expression of the same connexins was increased in the histologically more aggressive mammary gland neoplasms or in the cases in which metastases were detected. Strong immunostaining of connexin 26 was also described in the human invasive mammary carcinomas studied by Jamieson et al. 6 In addition, the presence of intense immunostaining of connexins in the cytoplasm of more aggressive neoplastic cells raises some questions about the role of connexins that are accumulated in the cytoplasm, in relation to the capacity of cell-to-cell communication as well as on cell proliferation. This suggests that the deficiency in connexin expression and/or the erratic localization of these proteins can cause a deficiency in the formation of communicating gap junctions. This, in turn, due to deficient GJIC capacity, could contribute to a higher cell proliferation and to a more aggressive phenotype of these neoplasms. In fact, many authors have reported evidence of this relationship. 16, 32, 36, 39, 40 According to Trosko and Ruch, 37 gap junction size and number, connexin expression, and cell-cell coupling (GJIC) have been studied in many neoplastic cells using ultrastructural, biochemical, and immunologic means and by introduction of fluorescent or radioactive tracers and the determination of tracer passages into adjacent cells. The vast majority of neoplastic cells have fewer and smaller gap junctions, express less connexins, and have reduced GJIC when compared to their nonneoplastic counterparts. It has been pointed out 13, 16, 24, 43 that cells can have defective GJIC at several levels. First, they may lack functional (permeable) gap junctions. Second, they may have functional gap junctions among themselves (homologous GJIC), but they are incapable of gap junction formation with nontransformed cells (heterologous GJIC). Third, they may form gap junctions but are insensitive to the gap junction signals that control growth and phenotype.
Regarding Cx43 staining, a strong staining of stromal cells has been detected in some more aggressive cases as well as in cases in which metastases were present. These findings have been described. 6 The increased staining of Cx43 in mammary neoplasms in vivo or in mammary cell lines has also been observed by others. 33
Because of the variability of connexin expression in canine mammary gland neoplasms, which has also been observed in human mammary tumors, the role of cell communication by gap junctions in the development of mammary neoplasms seems to be complex. The decreased staining of connexins in carcinomas, mainly in those that recur or metastasize, and the increased expression of Cx26 with cytoplasmic distribution in histologically more aggressive neoplasms, associated with an increase in the expression of Cx43 in the basal portion of epithelial cells, characterize the more aggressive phenotype.
The transportation of connexins to the cell membrane is dependent on E-cadherin. 4 In an attempt to explain why connexins were strongly stained in the cytoplasm of more aggressive tumors, the expression of the adhesion molecule E-cadherin has been investigated in the same neoplasms. In fact, a strong immunostaining of E-cadherin has been detected in the normal, hyperplastic, and benign neoplasms of the mammary gland. However, the expression of E-cad-herin was variable in the malignant mammary neoplasms. Most malignant cases showed a decreased immunostaining of E-cadherin, associated with an erratic subcellular distribution. In the study cases, it seems that the reduced expression and abnormal distribution of E-cadherin are related to the degree of differentiation of the neoplasms. Also, the progressive loss of adhesion molecules may contribute to the detachment of the tumor cells, the subsequent invasion of the adjacent tissue, and metastasis. 27
The significance of finding E-cadherin in the cytoplasm is unknown. We believe that it may represent a disruption in the transportation or anchoring of E-cad-herin in the cell membrane 30 or that it may be the consequence of a defect in the cytoplasmic portion of the E-cadherin. This impairs the binding with the intracellular catenins, resulting in the inability to form intercellular connections. 1
In the analysis of these data, it seems that the cytoplasmic distribution of E-cadherin is related to the cytoplasmic distribution of connexins.
Recently, it was shown that the methylation of 5′CpG islands in the promoter region of E-cadherin suppresses the GJIG in endometrial cancer cells. 25 It has been observed that this effect was due to the aberrant localization of the connexins in the cytoplasm. Once more, the relationship between connexin transportation and E-cadherin has been shown.
Among the malignant neoplasms, squamous cell carcinomas showed the highest cell proliferation index, followed by spindle cell carcinomas, fibrosarcomas, carcinosarcomas, poorly differentiated sarcomas, tubulopapillary carcinomas, simple carcinomas, and complex carcinomas.
The inverse relationship between the expression of connexins and cell proliferation has been shown. 40 In some of the cases of mammary neoplasms, it was possible to observe a higher expression of the connexins and a higher cell proliferation.
This study showed that there are alterations in the expression of connexins 26 and 43 as well as in the E-cadherin and in the proliferation index in malignant canine mammary gland tumors. It is known from the literature that both the connexin expression and the GJIG capacity decrease, coinciding with the advancement of the carcinogenic process. On the other hand, cancer chemopreventive agents may increase the expression of connexins and avoid the decrease in GJIC. Recently, a study showed that the Bowman-Birk Inhibitor up-regulates the expression of connexin 43 in a mammary epithelial cell line (MCF10). 34 We believe that finding agents that could increase the expression of both connexins and E-cadherin as well as improve their transportation to the plasma membranes would be an important contribution in the prevention of mammary tumors in female dogs.
Footnotes
Acknowledgements
This work is part of the MS dissertation of L. N. Torres in the Experimental and Comparative Pathology Program of the Faculty of Veterinary Medicine and Zootechny of the University of São Paulo. This research was supported by grants (Nos. 00/04225-7) from the Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP). L. N. Torres was supported by a fellowship from FAPESP 00/02521-8. We thank the Laboratory of Cell Biology of the Department of Histology of the Institute of Biomedical Sciences of the University of São Paulo for the use of their confocal microscope.
