Abstract
A fibroblastic osteosarcoma with epithelioid and squamous differentiation in the distal femur of a 9-y-old spayed female Greyhound dog is described. Grossly, the tumor consisted of a pale-white, firm-to-hard mass that replaced the medullary and cortical areas of the distal end of the right femur. Histologically, the mass was composed predominantly of spindle cells admixed with areas of mineralized and non-mineralized osteoid matrix that were surrounded by stellate osteoblasts and scattered multinucleate giant cells, consistent with the diagnosis of a fibroblastic osteosarcoma. In addition, well-demarcated clusters of neoplastic epithelioid cells and foci of squamous differentiation with keratin pearls were present throughout the neoplasm. The spindle cells, epithelioid cells, and areas of squamous differentiation expressed cytoplasmic immunostaining for osteocalcin and osteonectin. The spindle cells and epithelioid cells were also immunopositive for vimentin. Epithelioid cells also expressed occasional cytoplasmic immunostaining for pancytokeratin (PCK) Lu-5, and areas of squamous differentiation were immunoreactive for PCK Lu-5 and high molecular weight CK; these areas were inconsistently immunoreactive for CK 5-6 and immunonegative for low molecular weight CK. Foci of squamous differentiation were not located within blood or lymphatic vessels, given that no immunoreactivity for factor VIII−related antigen was observed around these areas. A thorough autopsy and an evaluation of the medical history excluded a primary carcinoma or other neoplasm elsewhere in the dog. The findings were consistent with a diagnosis of fibroblastic osteosarcoma with epithelioid and squamous differentiation.
Osteosarcoma is the most common primary bone neoplasm of dogs.1,4,9,13,14 Tumors are aggressive and typically occur in adult, large- and giant-breed dogs.4,6,9,13 The appendicular skeleton is more commonly affected than the axial skeleton, and the thoracic limbs are affected twice as often as the pelvic limbs. Preferred primary appendicular skeleton sites include the distal radial metaphysis, proximal humeral metaphysis, and the distal ends of the tibia and femur.2,14,16 Metastases are frequent and usually occur in the pulmonary parenchyma and regional lymph nodes, but other organs can also be affected. 13 The classification of osteosarcoma in veterinary medicine is determined by the predominant morphologic features of the neoplastic cells and includes osteoblastic, chondroblastic, fibroblastic, telangiectatic, giant cell–rich, and poorly differentiated osteosarcoma.5,14 Less common tumor subtypes have also been described in dogs, including myxoid, round cell, and epithelioid osteosarcoma. 10 Epithelioid osteosarcoma has been reported in humans as a typical osteosarcoma exhibiting areas where osteoblasts morphologically resemble epithelial cells, which can rarely undergo glandular or squamous differentiation.3,8 In dogs, epithelioid osteosarcoma has been described involving the bones of the skull, but no glandular or squamous differentiation within osteosarcomas has been reported in the veterinary medical literature. 10 We describe herein a fibroblastic osteosarcoma with epithelioid and squamous differentiation in the distal end of the right femur of a dog.
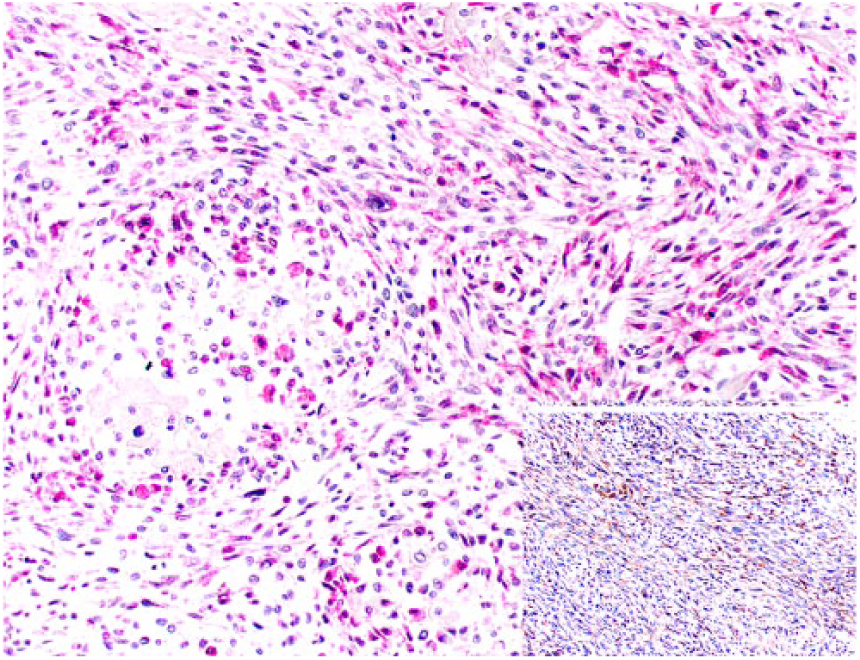
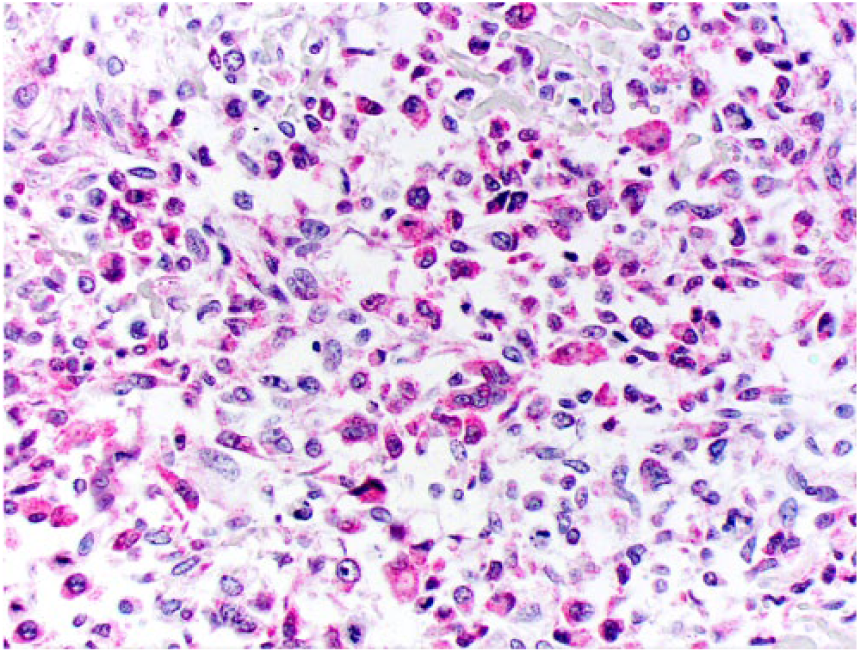
A 9-y-old spayed female Greyhound dog was presented with a 3-day history of right pelvic limb lameness. Radiographic examination revealed an osteolytic lesion affecting the right distal femoral epiphysis. The owner elected to have the dog euthanized and authorized autopsy. At autopsy, gross changes consisted of a pale-white, firm-to-hard mass that partially replaced the distal marrow and cortical areas of the femoral metaphysis and epiphysis (Fig. 1). Histologically, the mass was composed predominantly of spindle cells admixed with multiple, irregular areas of mineralized and non-mineralized osteoid matrix surrounded by neoplastic osteoblasts and scattered multinucleate giant cells (Fig. 2). Neoplastic spindle cells were arranged in closely packed interweaving bundles. These cells had abundant, eosinophilic cytoplasm with indistinct margins and elongate nuclei with dense chromatin and 1–4 nucleoli. Osteoblasts had stellate, eosinophilic cytoplasm with distinct margins and round nuclei with finely stippled chromatin and 1–2 nucleoli. Additionally, there were occasional sheets of polygonal epithelioid cells (Fig. 3) and well-defined areas of squamous differentiation with keratin pearls (Fig. 4).
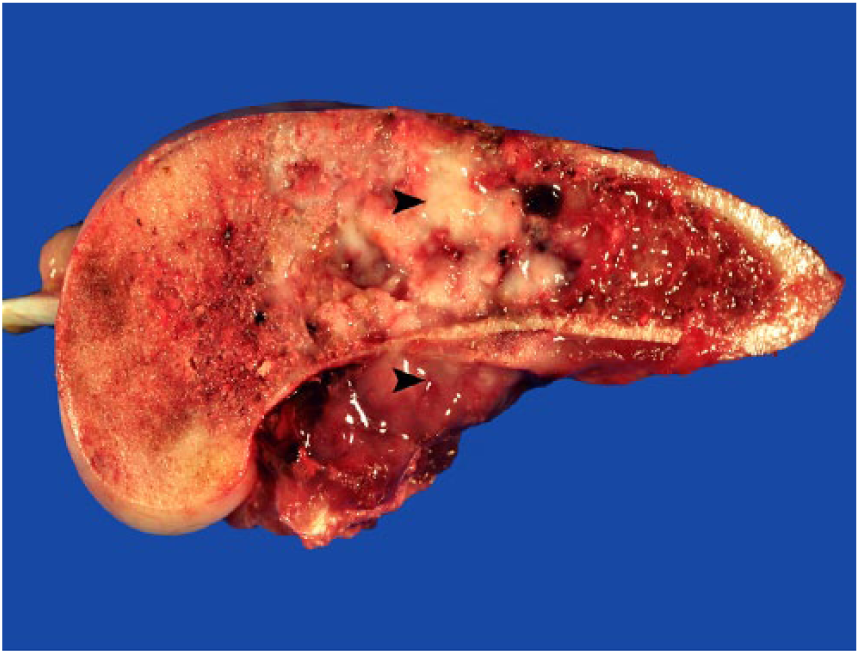
Longitudinal section of a fibroblastic osteosarcoma of the right distal femur exhibiting firm and hard pale-white areas (arrowheads) that partially replace the marrow spaces and cortical bone.
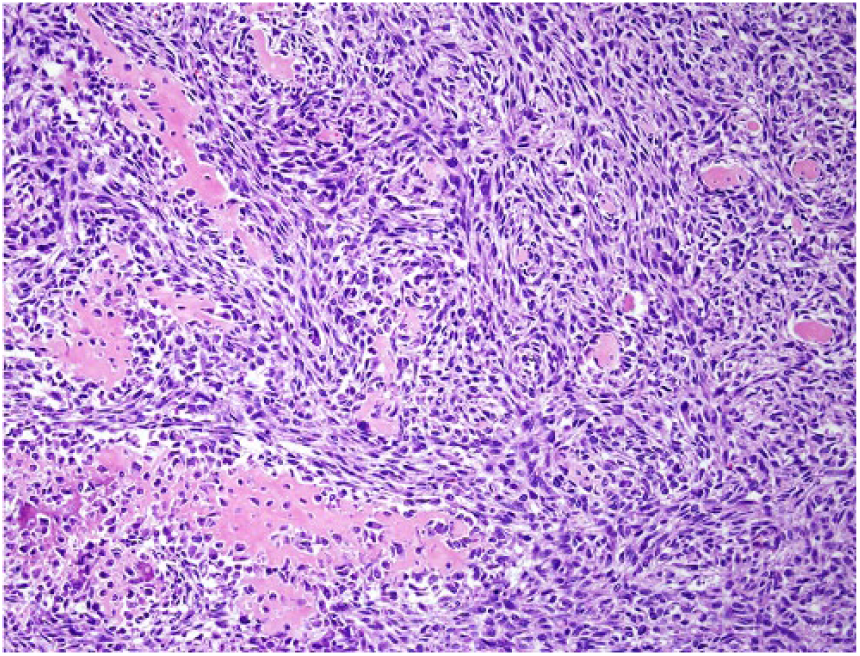
Closely packed interweaving bundles of neoplastic spindle cells surround areas of osteoid matrix in a fibroblastic osteosarcoma. H&E.
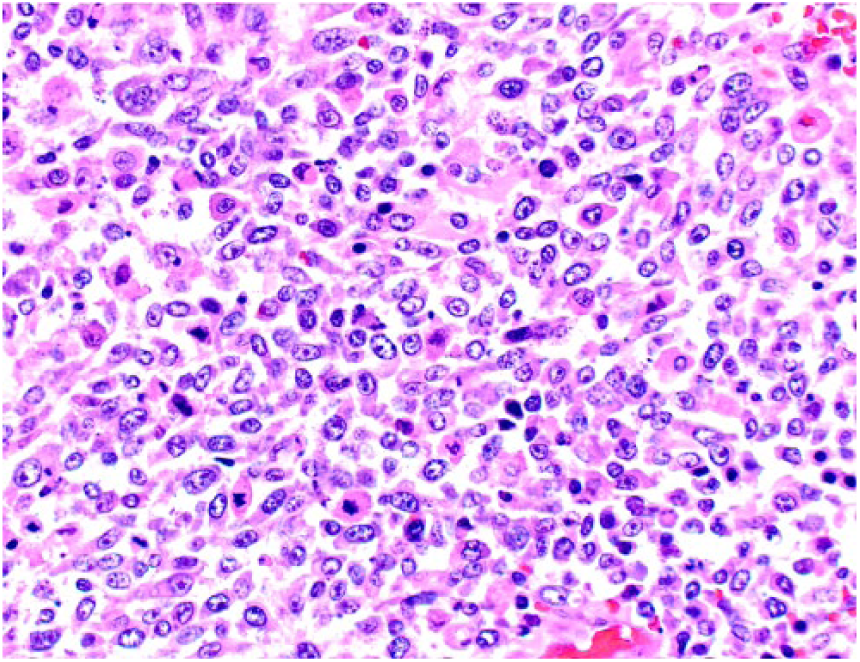
Sheets of neoplastic cells exhibiting epithelioid morphology are occasionally observed within the fibroblastic osteosarcoma. H&E.
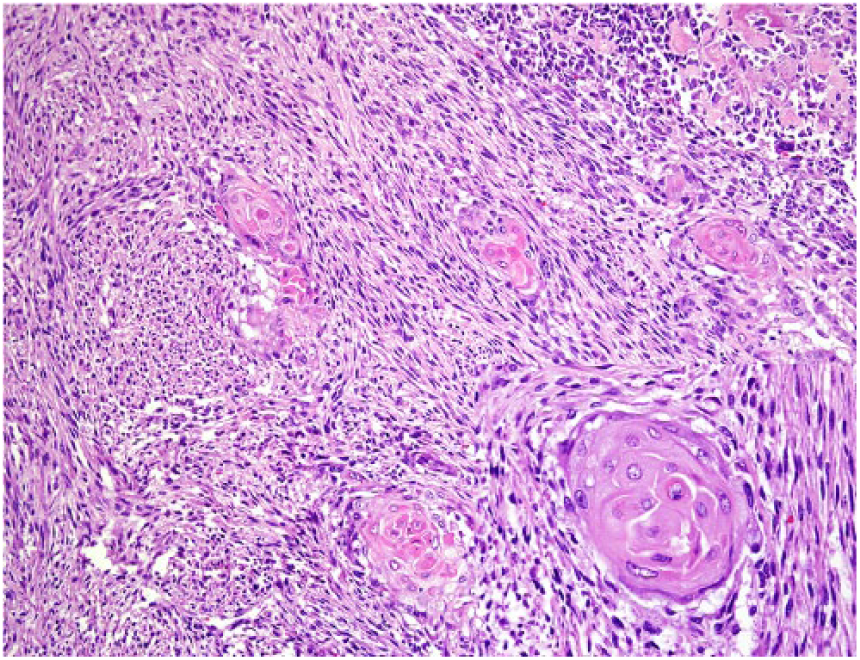
Multiple, well-demarcated clusters of squamous differentiation among neoplastic spindle cells are distributed throughout the fibroblastic osteosarcoma. H&E.
Tissue sections were stained immunohistochemically for osteocalcin (mouse monoclonal, 1:250 dilution at 32 min, Abcam, Cambridge, UK), osteonectin (goat polyclonal, 1:500 dilution at 60 min, R&D Systems, Minneapolis, MN), vimentin (mouse monoclonal, 1:3,000 dilution at 60 min, BioGenex, San Ramon, CA), pancytokeratin Lu-5 (PCK Lu-5; mouse monoclonal, 1:100 dilution at 90 min, Biocare Medical, Pacheco, CA), cytokeratin 5 and 6 (CK 5-6; mouse monoclonal, ready-to-use dilution at 60 min, Cell Marque, Rocklin, CA), high molecular weight cytokeratin (HMW CK; mouse monoclonal, 1:100 dilution at 90 min, Cell Marque), low molecular weight cytokeratin (LMW CK; mouse monoclonal, 1:100 dilution at 90 min, Cell Marque), and factor VIII−related antigen (rabbit polyclonal, RTU at 60 min, Cell Marque). Neoplastic spindle cells (Fig. 5), epithelioid cells (Fig. 6), and areas of squamous differentiation (Fig. 7) expressed diffuse cytoplasmic immunostaining for osteocalcin and osteonectin. Neoplastic spindle cells and epithelioid cells were also immunopositive for vimentin. Epithelioid cells (Fig. 8) expressed occasional cytoplasmic immunostaining for PCK Lu-5, and areas of squamous differentiation were immunoreactive for PCK Lu-5 (Fig. 9) and HMW CK (Fig. 10); these areas were faintly immunoreactive for CK 5-6 (Fig. 11) and immunonegative for LMW CK. Areas of squamous differentiation were not located within blood or lymphatic vessels, as no immunoreactivity for factor VIII−related antigen was observed around these areas.
Neoplastic spindle cells exhibit cytoplasmic immunostaining for osteocalcin. Inset: spindle cells are also immunoreactive for osteonectin.
Epithelioid osteoblasts exhibit cytoplasmic immuno-staining for osteocalcin.
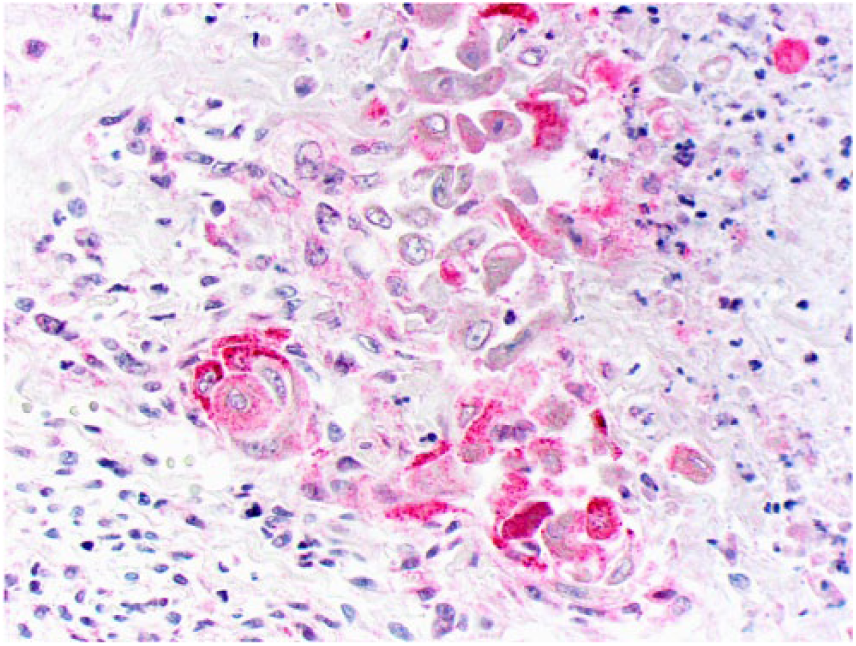
Clusters of squamous differentiation exhibit cytoplasmic immunostaining for osteocalcin.
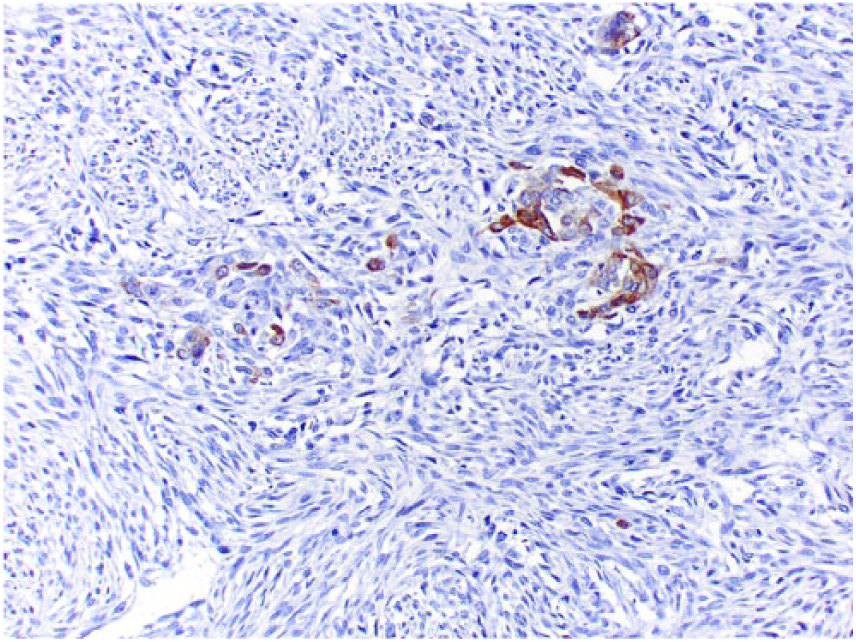
Scattered epithelioid osteoblasts exhibit cytoplasmic immunostaining for pancytokeratin Lu-5.
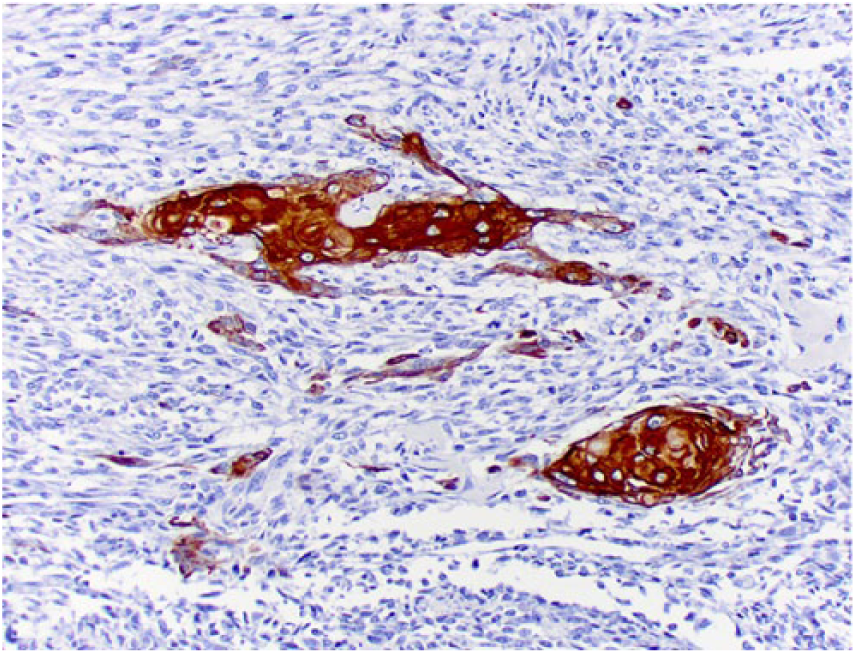
Clusters of squamous differentiation exhibit cytoplasmic immunostaining for pancytokeratin Lu-5.
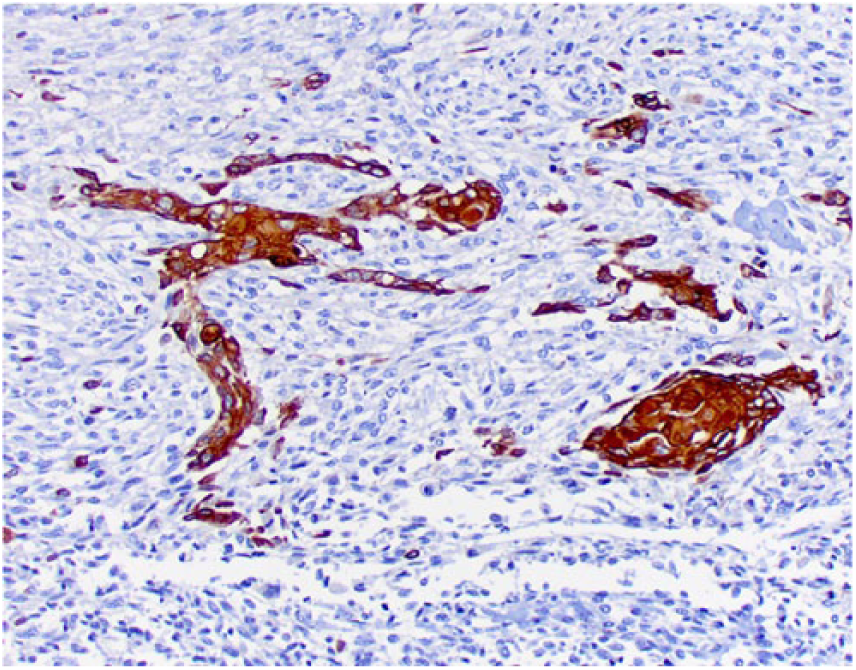
Clusters of squamous differentiation exhibit cytoplasmic immunostaining for high molecular weight cytokeratin.
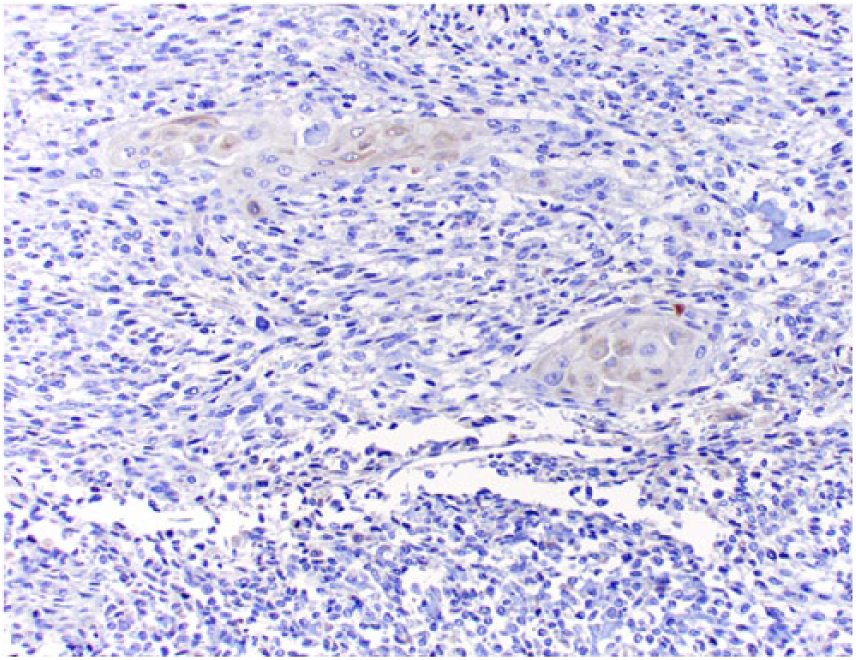
Clusters of squamous differentiation exhibit faint cytoplasmic immunostaining for cytokeratin 5 and 6.
The diagnosis of fibroblastic osteosarcoma was based on tumor location, gross appearance, and the typical histopathologic features of neoplastic cells, as well as on the presence of multiple areas of mineralized and non-mineralized osteoid matrix scattered throughout the tumor.1,2,4,9,13–15 Although tumor morphology typically satisfies a diagnosis of osteosarcoma, immunohistochemical expression of osteocalcin, osteonectin, and vimentin by neoplastic cells was used for diagnostic support in our case.14,15
The majority of neoplastic cells in our case were elongate, consistent with a diagnosis of fibroblastic osteosarcoma. 14 However, epithelioid cells and well-defined areas of squamous differentiation with keratin pearls were present throughout the tumor. Osteosarcoma with epithelioid differentiation has been rarely described in humans and in the skull of dogs, but squamous differentiation has only been reported in human tumors.8,10 In our case, epithelioid cells expressed PCK Lu-5, and areas of squamous differentiation were immunopositive for PCK Lu-5, HMW CK, and CK 5-6. PCK expression in human osteosarcoma has been documented in cases of osteoblastic, chondroblastic, and fibroblastic osteosarcoma, in which staining is more evident in areas of epithelioid and squamous differentiation.8,11 Such tumors are clinically and pathologically similar to conventional osteosarcomas, but appear to affect older individuals and be more resistant to chemotherapy. 11 CK expression in canine osteosarcoma has also been described in a variety of tumors, including osteoblastic, fibroblastic, telangiectatic, poorly differentiated, and epithelioid subtypes. 10 Given the low frequency of these features in canine osteosarcomas, it remains unknown whether these tumors have any difference in behavior when compared to typical osteosarcomas.
The presence of epithelioid cells and areas of glandular or squamous differentiation within osteosarcomas poses a diagnostic challenge for the pathologist, because these features may mimic a carcinoma metastatic to bone.3,8,11 Based on these previous reports in human medicine and on the clinical and pathologic evidence drawn from our case, the diagnosis is most likely of an osteosarcoma with epithelioid and squamous differentiation rather than a metastatic carcinoma within an osteosarcoma. A detailed examination of the medical history, a thorough autopsy, and the presence of osteoid are key features that should favor a diagnosis of osteosarcoma over a metastatic carcinoma. 11 Additionally, the fact that LMW CK is expressed by simple and glandular epithelium but not squamous epithelium and the absence of LMW CK expression in our case also supports our diagnosis. 12 Lastly, immunohistochemical staining for factor VIII–related antigen revealed that the areas of squamous differentiation were not located within blood or lymphatic vessels, giving further support to squamous differentiation rather than a metastatic carcinoma in our case. The faint and inconsistent expression of CK 5-6 and the absence of abdominal, thoracic, or pericardial lesions sufficed to rule out a metastatic epithelial mesothelioma. 12
CK expression without morphologic evidence of epithelial differentiation may indicate anomalous expression related to CK expression by mesenchymal cells or to technical artifacts that result in false-positive CK staining. 8 The presence of epithelioid cells and areas of squamous differentiation that were consistently immunoreactive for a variety of CK subtypes on IHC provide substantial evidence of true epithelial differentiation and not anomalous or aberrant CK expression by mesenchymal neoplastic cells. 8 This is a puzzling tumor subtype with a widely unknown biology. The most accepted hypothesis to explain this biphenotypic nature of osteosarcomas in humans is that epithelial differentiation arises from uncommitted multipotential stem cells present within the tumor, which may acquire epithelial morphology following disorderly differentiation. 7 Other interpretations include epithelioid or squamous foci within osteosarcomas derived from cell rests displaced from other tissues, 2 different and unrelated tumors occurring simultaneously in the same tissue (collision tumor), or the possibility of a primary carcinosarcoma of the bone.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
