Abstract
Osteosarcoma (OSA) is a malignant heterogeneous primary bone tumor responsible for up to 90% of all primary bone tumors in dogs. In this study, osteocalcin (OC) and osteonectin (ON) immunoreactivity was evaluated in 23 canine OSAs, 4 chondrosarcomas, 4 fibrosarcomas, 2 hemangiosarcomas, and 4 histiocytic sarcomas. The effects of three different decalcification agents (ethylenediaminetetraetic acid [EDTA], formic acid and hydrochloric acid [HCl]) on the immunoreactivity for OC and ON was also assessed. Immunoreactivity to OC was present in 19/23 (83%) cases of OSA and all cases of chondrosarcoma. In three OSAs the extracellular matrix showed immunoreactivity to OC. None of the fibrosarcomas, histiocytic sarcomas or hemangiosarcomas showed immunoreactivity to OC. The sensitivity and specificity for OC in canine OSA in this study was 83% and 71% respectively. For ON, 100% of both OSAs (23/23) and non-OSAs (14/14) showed cytoplasmic immunoreactivity to this antibody, giving a sensitivity of 100% but a complete lack of specificity. There were no significant differences in immunoreactivity for OC and ON between the different decalcification agents used. In conclusion, OC showed high sensitivity for identifying OSA but it failed to distinguish between OSA and chondrosarcoma, and the osteoid produced by neoplastic cells in most cases did not show immunoreactivity to OC. These factors may limit the practical utility of OC in the diagnosis of OSA in dogs when chondrosarcoma is a differential diagnosis. ON showed no specificity in detecting OSA and has little practical application for the diagnosis of OSA in dogs.
Keywords
Osteosarcoma (OSA) is a heterogeneous malignant primary bone tumor responsible for up to 90% of all primary bone tumors in dogs, with the remaining 10% including chondrosarcoma, fibrosarcoma and hemangiosarcoma. 4,15,19,21,29 The histological diagnosis of primary OSA can be challenging. Because the different cell types that form OSAs originate from a common pluripotential mesenchymal precursor cell, OSAs may show marked histological diversity, making definitive diagnosis difficult. 7,28 Also, to make a definitive histological diagnosis of OSA, tumor osteoid must be present among neoplastic cells, which is not always the case in all sections, especially those prepared from small core biopsies. 6,9 Difficulties may also arise in differentiating osteoid from either collagenous fibrous tissue or fibrin in areas of hemorrhage. Finally, the samples may contain only reactive bone and be nondiagnostic. 6,28 This histological heterogeneity frequently results in confusion amongst pathologists in the diagnosis of OSA and has prompted the study of different cellular markers for the immunohistochemical (IHC) characterization of the phenotype of the proliferating cells in cases of suspected OSA in humans. 2,3,6 –8,11,30
During intramembranous bone formation, bone morphogenetic proteins (BMP-2, -4 and -7) instruct mesenchymal cells to differentiate into osteoblasts that produce and secrete osteocalcin (OC) and osteopontin, as well as other extracellular matrix proteins such as osteonectin (ON). 14 OC is a low-molecular weight protein that binds hydroxyapatite crystals in bone matrix and is considered a marker for late osteoblastic differentiation, while ON is an extracellular matrix-associated glycoprotein that is mainly expressed in tissue undergoing growth, remodeling or repair. 3,5,20,27
IHC studies investigating OC and ON expression in both normal bone and OSAs in people have demonstrated that the immunoreactivity of both markers is primarily cytoplasmic, with more variable and sporadic expression by extracellular osteoid matrix. Human studies report good sensitivity (>64%) and excellent specificity (100%) for OC in the identification of neoplastic osteoblasts in a histologically diverse range of OSAs, making OC a potentially useful marker for OSA diagnosis in equivocal cases. 3,5 –8,11,27 However, while ON shows excellent sensitivity (>90%) in the detection of neoplastic osteoblasts in humans, it is of limited use for the diagnosis of OSA because of its low specificity (<55%) for osteoblast detection and its tendency to show cytoplasmic immunoreactivity in cells from a wide range of tumors, including fibrosarcomas, chondrosarcomas and carcinomas. 2,5,6
In dogs, only one report describes the use of immunohistochemistry for OC and ON, among many other markers, on a single case of a multipotential OSA with various mesenchymal differentiations; however, detailed methods were not provided. 13
The primary aim of this study was to evaluate the use of commercially available OC and ON antibodies in the diagnosis of canine OSA by immunohistochemistry. An additional aim was to assess the effect of three commonly used decalcification agents on the immunoreactivity for OC and ON.
Materials and Methods
Thirty-seven formalin-fixed paraffin-embedded sarcoma specimens were selected from the archives of the Pathobiology Section, Institute of Veterinary, Animal and Biomedical Sciences (IVABS), Massey University, Palmerston North, and the New Zealand Veterinary Pathology (NZVP) laboratory, Palmerston North. Cases were selected based on the histological diagnosis of sarcoma and availability of representative tissue for IHC labeling. Cases were reviewed by two authors (ASWM and KED), and a diagnosis of OSA was reached when there was unequivocal histological evidence of neoplastic mesenchymal cells producing osteoid. Histologic subtypes of OSA were defined according to the World Health Organization (WHO) criteria as previously applied to dogs. 10,16,17,26 The 37 cases retrieved were diagnosed between 1997-2013 and consisted of OSA (n = 23), chondrosarcoma (n = 4), fibrosarcoma (n = 4), hemangiosarcoma (n = 2), and histiocytic sarcoma (n = 4). All OSAs and chondrosarcomas were located within bone. Two of four fibrosarcomas were located within the maxilla, one was located in the subcutaneous tissue while the other was located within abdominal wall. One histiocytic sarcoma affected the tibia, while the remainder were extraosseous, located within the liver, abdomen and brain. One hemangiosarcoma was located in the humerus and the other was located within the spleen. Clinical data and histological diagnoses from the cases used are summarized in Supplemental Table S1.
Data concerning the type of decalcifying solution used and duration of decalcification were unknown in all cases except numbers 22 and 23. These two cases were used to assess different decalcification methods. Tumor and affected bone were placed in 10% formalin for a minimum of 48 hours. Subsequently, several parallel bone slabs 3-5 mm thick were sectioned using a fine toothed band saw, and then decalcified using either 15% EDTA (pH 7.4) for approximately 90-100 days, 10% formic acid for approximately 60-70 days, or 10% hydrochloric acid (HCl) for approximately 10 days.
Sections 5 µm thick were cut and mounted on advanced adhesive microscope slides (HD Scientific, Wetherill Park, Australia). Sections were then deparaffinized in xylene and hydrated in graded alcohols. Optimization of IHC protocols was performed using sections of non-neoplastic bone (positive control), OSA (case control), dog liver (negative control), and bone without primary antibody (negative control), to determine the optimal antigen retrieval method and primary antibody dilution. For OC, the optimal antigen retrieval method was determined to be citrate buffer (CB), 10 mM pH 6.0 at 95°C for 35 minutes; and for ON, CB, 10 mM pH 6.0 at 105°C for 10 minutes. For both methods, a benchtop pressure cooker (Decloaking Chamber, Biocare Medical, Concord, CA, USA) was used. After washing three times in running water, the slides were placed into the Sequenza apparatus (ThermoFisher Scientific, Dreieich, Germany) for immunolabeling. IHC labeling was accomplished using the avidin-biotin-peroxidase complex (ABC) procedure (Vectastain® Elite ABC Kit; Vector Laboratories, Burlingame, CA, USA) on paraffin sections. The primary antibodies used were: mouse monoclonal antiosteocalcin antibody (clone: OCG3) (Abcam®, Cambridge, England) and rabbit polyclonal SPARC antibody (ON) (Abbiotec, San Diego, CA, USA). The OC antibody was diluted 1:100, and ON 1:400, and tissue sections were incubated with the primary antibody at 4°C overnight. Sections were then incubated with biotinylated secondary antibody for 30 minutes at room temperature, and endogenous peroxidases were then quenched using 3% H2O2 diluted in methanol for 10 minutes. Finally, sections were incubated with ABC for 30 minutes at room temperature and visualization was accomplished using 3,3’-diaminobenzidine (Vector® DAB Substrate, Burlingame, CA, USA) for 10 minutes, also at room temperature. Gill’s II hematoxylin stain was used as a counterstain.
Immunoreactivity was evaluated using a semi quantitative 4-grade scale (− = no immunoreactivity; + = mild immunoreactivity; ++ = moderate immunoreactivity; +++ = strong immunoreactivity) as previously described. 12 A score of +/- was given when only low numbers of focal or random cells showed immunoreactivity for any of the antibodies. The type of positive cell was determined and the distribution of labeling (nuclear, cell membrane, cytoplasmic, extracellular matrix) recorded. This grading system was also used to compare the effect of decalcification method on the immunoreactivity for OC and ON.
Results
The full results for each case are summarized in Supplemental Table S1. The 23 cases of OSAs comprised histological subtypes (classified according to the WHO classification 26 ) of osteoblastic OSA (n = 16), fibroblastic OSA (n = 3), chondroblastic OSA (n = 2), telangiectatic OSA (n = 1), and giant cell rich OSA (n = 1). The remaining 14 sarcomas were histologically reconfirmed as fibrosarcoma (n = 4), histiocytic sarcoma (n = 4), hemangiosarcoma (n = 2), and chondrosarcoma (n = 4).
Effect of Decalcification Method on Immunoreactivity for Osteocalcin and Osteonectin
The effect of decalcification on the immunoreactivity for OC and ON was evaluated in two cases (case numbers 22 and 23). In both cases there was a slight reduction in immunoreactivity in samples decalcified with 10% HCl for both antibodies and for ON when 10% formic acid was used compared to 15% EDTA (Table 1).
Results of the Effect of Decalcification on the Immunoreactivity for Osteocalcin and Osteonectin in Canine Osteosarcoma Bone Tissue Sections.
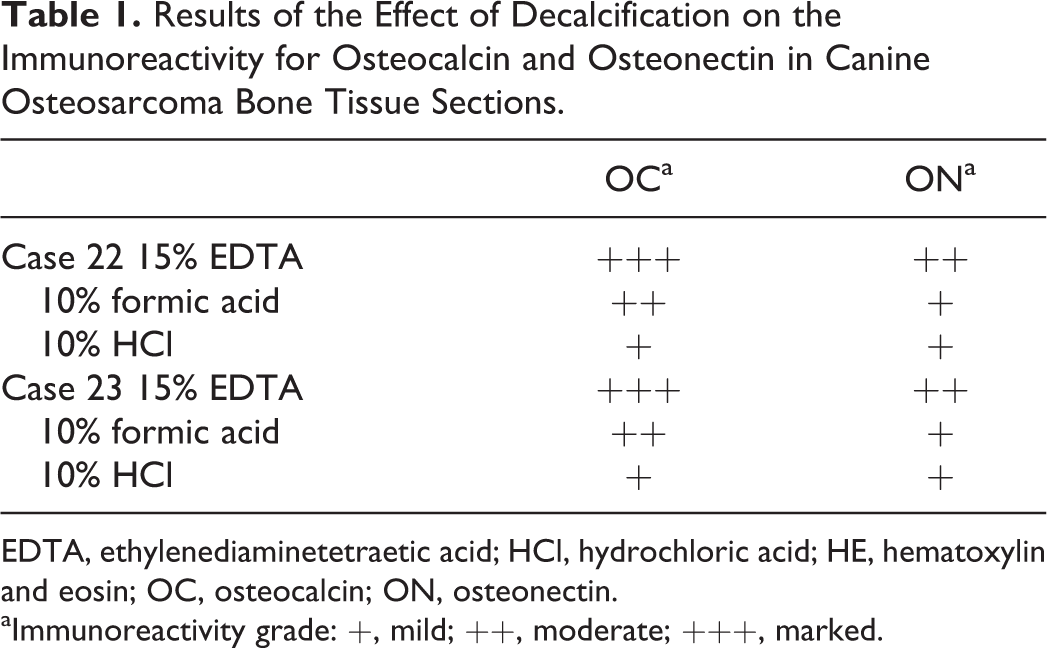
EDTA, ethylenediaminetetraetic acid; HCl, hydrochloric acid; HE, hematoxylin and eosin; OC, osteocalcin; ON, osteonectin.
aImmunoreactivity grade: +, mild; ++, moderate; +++, marked.
Immunoreactivity of Osteosarcoma for Osteocalcin and Osteonectin Antibodies
The results of OC and ON immunohistochemistry are summarized in Table 2. Of the 23 OSAs, 19/23 (83%) showed cytoplasmic immunoreactivity for OC. Of the 16 osteoblastic OSAs, 13/16 (81%) were immunoreactive for OC (Fig. 1b). Based on the amount of osteoid produced by the neoplastic cells, 7/16 (44%) osteoblastic OSAs were classified as moderately productive and 9/16 (56%) as productive (large amounts of osteoid produced by neoplastic cells). In the moderately productive osteoblastic OSAs, immunoreactivity for OC was mild (+) in 3/7 (43%), moderate (++) in 2/7 (28%), and absent in two cases (case numbers 3 and 18). Of the 9 productive osteoblastic OSAs, immunoreactivity for OC was mild (+) in 4/9 (44%), moderate (++) in 4/9 (44%) (Fig. 1b), and negative in one case (case number 16). One chondroblastic OSA showed moderate (++) immunoreactivity for OC (Fig. 2b); the other was negative. All fibroblastic OSAs (3/3, 100%) were immunoreactive for OC; this was mild (+) in two cases and moderate (++) in one case (Fig. 3b). The single telangiectatic OSA (Fig. 4b) and giant cell rich type OSA both demonstrated moderate (++) immunoreactivity for OC.
Summary of Immunohistochemical Results for the Different Canine Osteosarcoma Subtypes and Tumors Other Than Osteosarcoma.
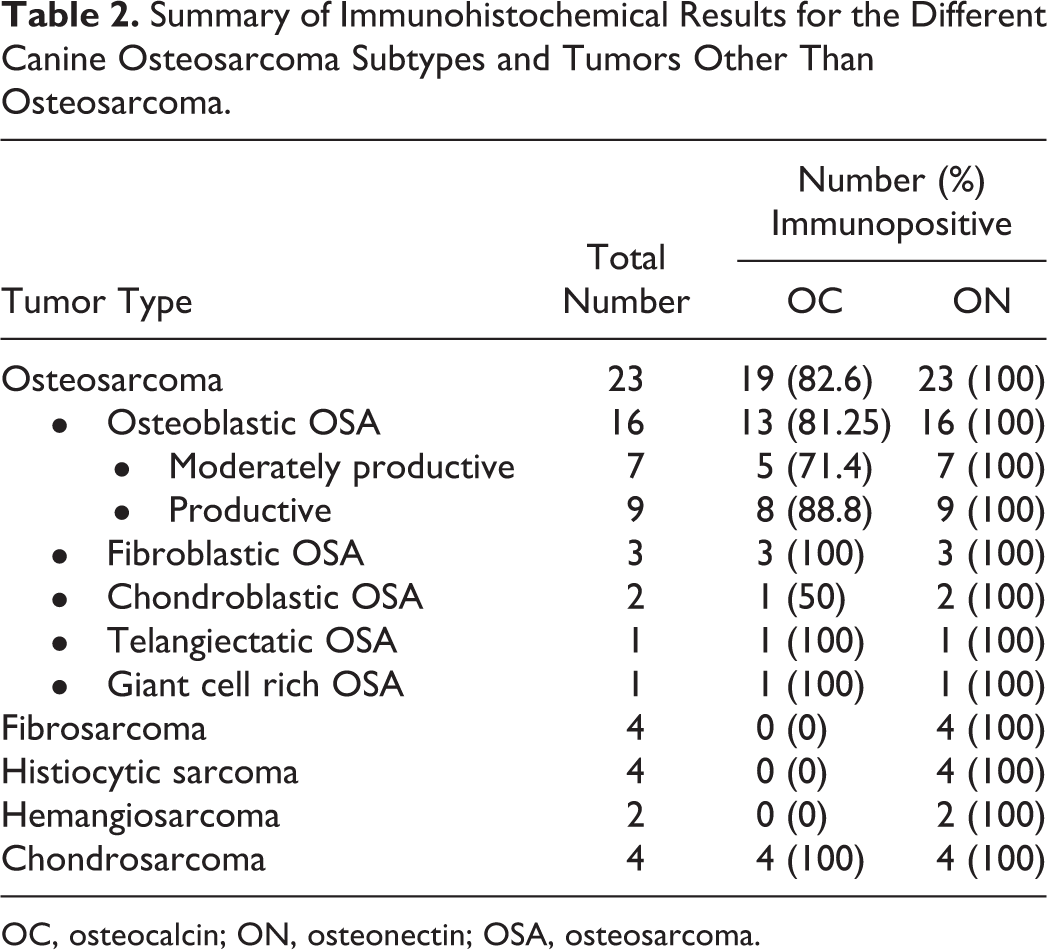
OC, osteocalcin; ON, osteonectin; OSA, osteosarcoma.
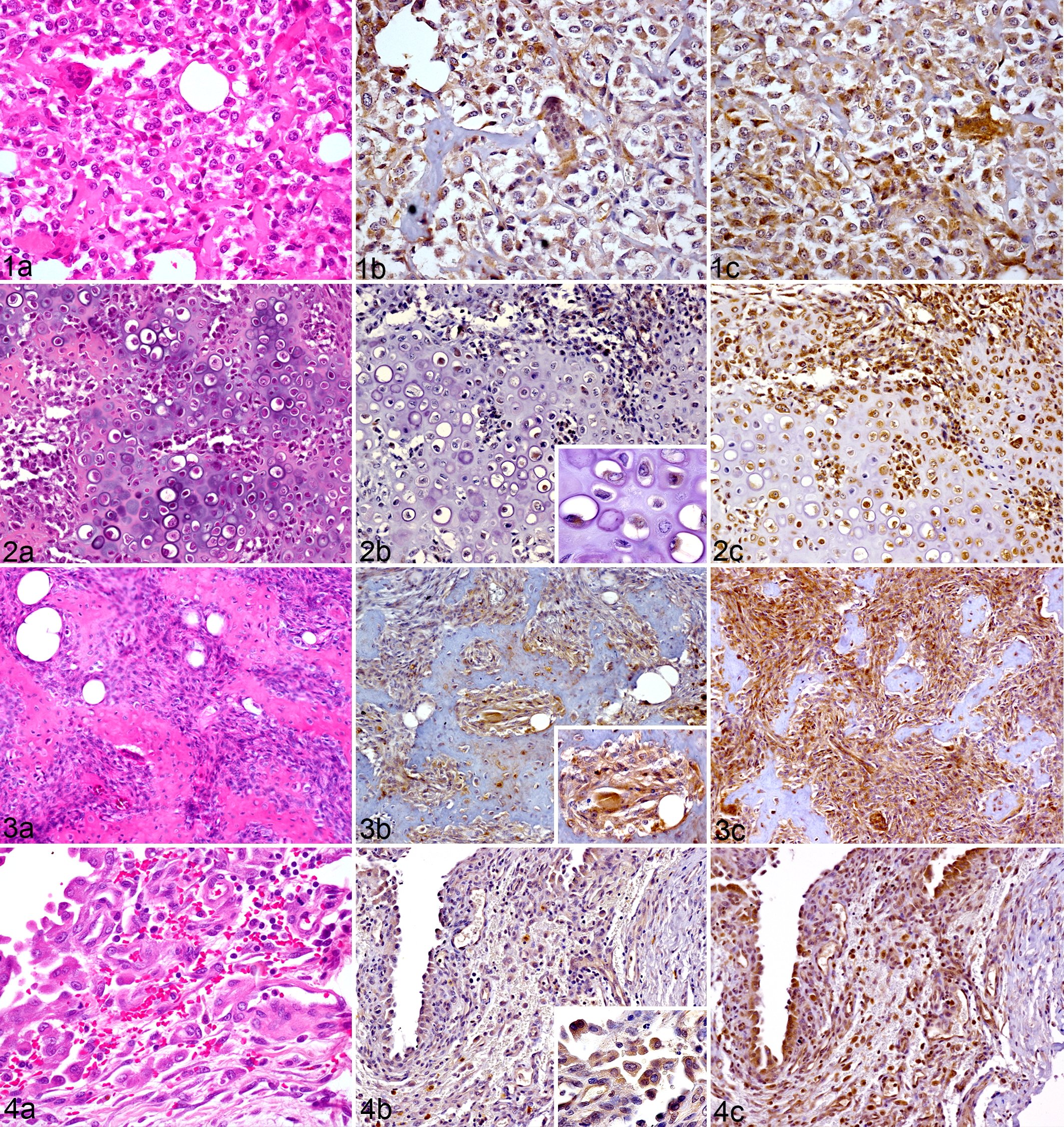
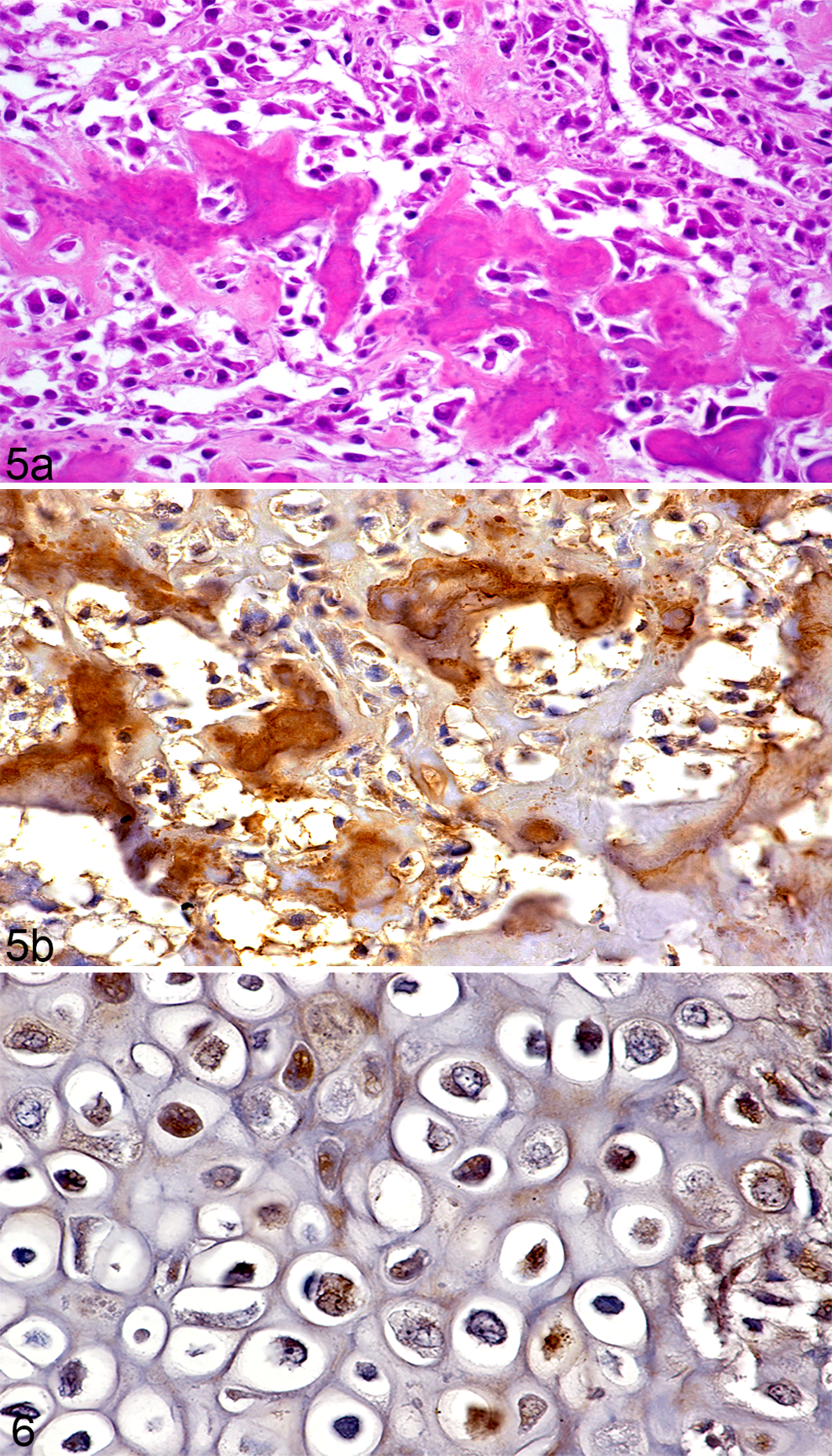
In all cases of OSA evaluated, immunoreactivity for OC was present in the cytoplasm of neoplastic cells in addition to the cytoplasm of non-neoplastic osteocytes, osteoclasts, and reactive periosteal mesenchymal cells. In 3/23 (13%) OSAs (case numbers 19, 21, and 23), the osteoid surrounded by neoplastic osteoblasts showed marked immunoreactivity for OC (Fig. 5b). In the remaining OSAs (20/23, 87%) the extracellular matrix (osteoid or chondroid) was immunonegative for OC.
All OSAs (23/23, 100%), demonstrated cytoplasmic immunoreactivity for ON. Similar to OC, immunoreactivity for ON was present in the cytoplasm of all OSA subtypes (osteoblastic, chondroblastic, fibroblastic, telangiectatic, and giant cell type) (Figs. 1c, 2c, 3c, and 4c). However, the variety of ON immunoreactive cell types was much greater than with OC and included non-neoplastic osteocytes, osteoclasts, endothelial cells, pericytes, fibroblasts, chondroblasts, and skeletal myocytes. In all cases, the osteoid showed no immunoreactivity to ON.
The dog liver was negative for OC, but hepatocytes were immunoreactive for ON. Sections without primary antibody were also repeatedly negative for OC and ON.
Immunoreactivity of Fibrosarcoma, Hemangiosarcoma, Chondrosarcoma, and Histiocytic Sarcoma for Osteocalcin and Osteonectin
Other (non-OSA) mesenchymal tumors included in this study showed variable cytoplasmic immunoreactivity for OC (Table 2). Immunoreactivity for OC was mild-to-moderate in the cytoplasm of neoplastic chondrocytes of all (4/4, 100%) chondrosarcomas (Fig. 6). In addition, in 3/4 chondrosarcomas the chondroid matrix demonstrated immunoreactivity for OC. In 2/4 (50%) fibrosarcomas there were only focal or scattered OC immunoreactive spindloid cells, but the majority of the neoplastic spindle cells were negative; hence both cases were considered equivocal (+/-). Similarly, in 1/4 (25%) histiocytic sarcomas, neoplastic cells near foci of dystrophic calcification demonstrated marked immunoreactivity for OC antibody but most neoplastic cells were immunonegative for OC. Neoplastic cells in the two hemangiosarcomas were immunonegative for OC.
Cytoplasmic immunoreactivity for ON was present in all cases of non-OSA tumors. Neoplastic cells were immunoreactive for ON along with non-neoplastic epithelial cells, endothelial cells and skeletal myocytes. In case number 31, the cytoplasm of neurons also showed immunoreactivity for ON.
Discussion
The results of the present study suggest that in dogs immunohistochemistry for OC may be a useful adjunctive test to assist in the diagnosis of OSA, but immunohistochemistry for ON is of little value due to its poor specificity. To our knowledge this is the first study that has evaluated the immunoreactivity of OC and ON in canine OSAs and other bone and soft tissue sarcomas.
Previous studies in people have provided a considerable amount of information regarding OC and ON immunoreactivity in cases of OSA, and other tumors from various locations. 2,3,5 –7,11,27,30 Reported OC sensitivity and specificity for osteoblastic detection in bone-forming tumors in people is 70% and 100%, respectively, while for ON the reported sensitivity is 90% and specificity 54%. 5,6,8 Immunoreactivity for OC in human OSA is typically cytoplasmic within osteoblasts, but immunolabeling of the extracellular osteoid matrix is also often present, and can further aid diagnosis. 5 –7,11 Fanburg et al 5 evaluated 106 tumors in humans, including multiple different OSA subtypes, benign bone tumors, non-bone-forming sarcomas, carcinoma, melanoma, and lymphoma, and found that all osteoblastic OSAs, as well as a proportion of fibroblastic (6/8 cases) and chondroblastic (4/7 cases) OSAs were immunopositive for OC. 5 Similar results have also been described in other studies. 3,6,11 Immunoexpression for OC may be higher in low-grade OSAs compared to poorly differentiated OSAs. 7 As OC appears in the later stages of bone development, and is considered a late marker for osteoblastic differentiation, it is not unexpected that OC expression will be greater in well-differentiated OSAs such as osteoblastic OSAs. 3,6,7,25
In our study, 83% of canine OSAs were immunoreactive for OC, with immunoreactivity present in the cytoplasm of neoplastic cells. Immunopositive OSA subtypes included osteoblastic, fibroblastic, chondroblastic, telangiectatic, and giant cell type OSAs. As in the human studies, there was slight variation between moderately productive and productive OSAs, with greater immunoreactivity (moderate/++) in cases of productive osteoblastic OSAs than in either moderately productive or fibroblastic OSAs (mild/+ immunoreactivity). Other OSA subtypes were also immunoreactive for OC except for one case of chondroblastic OSA, which did not show immunoreactivity. All 3 cases of fibroblastic OSAs were positive in our study. This is a very interesting finding since this type of OSAs can be easily misdiagnosed especially when only small biopsy samples are available. Similar findings have been described in human OSA. 3,5,6,11 The lack of OC labeling in four OSAs in our study may be due to the poorer differentiation of these tumors and the loss of mature differentiation of the neoplastic osteoblasts, which is a hallmark of malignancy. Lower immunoreactivity has also been reported in metastatic cells of OSA in people and this is believed to be due to either the presence of less differentiated anaplastic cells, or suppression of the cells’ ability to express OC in the altered tumor microenvironment. 3 Furthermore, neoplastic proliferating cells in OSAs are very diverse, reflecting a pluripotential mesenchymal cell origin, so lack of expression of certain markers or the expression of aberrant proteins is not unexpected. 6,7,11,22,25 Finally, data concerning the type of decalcifying solution used and duration of decalcification were unknown in the cases where no OC immunoreactivity was seen. Lack of immunoreactivity could therefore be due to prolonged or delayed fixation, or over-decalcification, factors that may artifactually reduce detectable immunoreactivity in tissue sections. 5,11
The specificity of OC immunonegativity in non-OSA sarcomas in this study was 71%. The two cases of fibrosarcoma contained only rare focal or random OC immunoreactive cells with the vast majority of neoplastic cells showing no immunoreactivity, and were therefore considered negative for OC. As these two cases originated from the maxillary bone, the scattered OC immunopositive cells present were believed to be reactive osteoblastic-type cells from the maxillary bone rather than neoplastic cells. The rare focal positive cells in one case of histiocytic sarcoma were considered immunonegative because these cells were only those surrounding foci of dystrophic mineralization, and cells away from these foci were all negative. A false-positive “diffusion” effect, due to the ability of OC to bind calcium and hydroxyapatite resulting in marked immunoreactivity in the areas where calcium is present, has been previously described in humans, and it is likely that this process also occurred here. 5 The immunoreactivity of neoplastic cells for OC in all of the chondrosarcomas in this study is not surprising since these cells share a common origin with osteoblasts. This limits the value of OC immunohistochemistry in supporting a diagnosis of OSA, particularly since chondrosarcoma is the second most common primary bone tumor in dogs and can be difficult to distinguish histologically from OSA in small biopsy samples. As the prognosis for chondrosarcoma is better than for OSA in dogs, reliably distinguishing these two tumors is prognostically important. 28
Another possible explanation for the OC immunoreactivity of chondrosarcomas in this study is that all paraffin-embedded sections were obtained from archived samples. OSAs are heterogeneous tumors and the type of proliferating cell and matrix produced can vary greatly throughout the tumor. It could be that in those cases there were other areas (not evaluated) of osteoid production by neoplastic cells. If the cases diagnosed as chondrosarcoma were actually chondroblastic OSAs then the specificity of OC would have been much higher.
The tumor matrix (osteoid or chondroid) was immunonegative for OC in most cases. In the three osteoblastic OSAs cases where the osteoid was immunoreactive, that matrix was more basophilic with HE than the nonimmunoreactive matrix. This type of HE staining pattern is associated with a less mature bone matrix and is due to the presence of increased noncollagenous proteins, such as proteoglycans and bone-specific vitamin K-dependent proteins (OC). 24 The components of the ground substance vary according to the tissue and it could be that in these cases the immunoreactivity was due to the presence of a higher concentration of OC in the matrix. The lack of immunoreactivity for most of the tumoral osteoid is a disadvantage, especially when it is important to differentiate osteoid from collagen or fibrin when confirming a diagnosis of OSA. Various studies of human OSA describe positive immunoreactivity of the neoplastic osteoid, allowing a better specificity for OC in OSA diagnosis. A novel antibody generated specifically against canine OC may increase the usefulness of this protein for the IHC detection of OC in the osteoid of canine OSA.
Previous studies of human bone tumors and tumors (sarcomas and nonsarcomas) in other body locations have concluded that ON antibody lacks specificity in the identification of cells with an osteoblastic phenotype. 2,5 –7,25 Our results showed similar findings, with all cases showing moderate to marked immunoreactivity for ON; not only cells from the osteoblastic, fibroblastic, chondroblastic, telangiectatic, and giant cell type OSAs but also the neoplastic cells in fibrosarcomas, histiocytic sarcomas, hemangiosarcomas, and chondrosarcomas. A wide range of non-neoplastic cells, including endothelial cells, pericytes, reactive fibroblasts, skeletal myocytes, neurons, tunica media of blood vessels, hepatocytes, and epithelial cells also showed variable immunoreactivity with ON. Due to this low specificity of ON, and its immunoreactivity in multiple neoplastic and non-neoplastic cells, ON cannot be considered a useful marker for osteoblastic differentiation and is of little use in OSA diagnosis in dogs.
When immunoreactivity for OC and ON was compared, the lowest immunoreactivity was observed in the samples that were decalcified using 10% HCl and the strongest immunoreactivity was seen when 15% EDTA was used as the decalcifying agent. Although immunoreactivity was reduced in samples decalcified with 10% HCl compared with the other agents, immunoreactivity was still easily detected. This is important because in many laboratories, 10% HCl is regularly used as a decalcification agent. This study therefore showed that immunoreactivity for commercially available OC and ON antibodies was not lost even when HCl was used or if samples were decalcified for a long period. While many authors do not recommend the use of strong acids such as HCl, studies that evaluated immunoreactivity in various tissues decalcified with HCl did not find a significant deleterious effect on the immunoreactivity of different cellular antigens; 1,18,23 our results support this finding.
Conclusion
Osteocalcin showed high sensitivity and moderate specificity for osteoblastic identification in dogs suggesting it may be a useful adjunct in the diagnosis of canine OSA in small biopsy specimens where the histological diagnosis is equivocal. However, OC IHC failed to reliably distinguish between OSA and chondrosarcoma, two common sarcomas of bone, and did not allow reliable identification of osteoid. Osteonectin did not differentiate between a variety of sarcomas of bone and appears to have little practical application in OSA diagnosis in dogs.
Footnotes
Acknowledgements
We would like to thank Matthew Perrott, Adrienne French, Evelyn Lupton, and Saritha Gils for the technical support and the Institute of Veterinary, Animal and Biomedical Science (IVABS) postgraduate fund for financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
