Abstract
Scrapie is a naturally occurring fatal neurodegenerative disease of sheep and goats. Susceptibility to the disease is partly dependent upon the genetic makeup of the host. In a previous study it was shown that sheep intracerebrally inoculated with US scrapie inoculum (No. 13–7) developed terminal disease within an average of 19 months. We have since produced an inoculum, No. x124 from pooled brains of US-origin sheep scrapie, that results in incubations nearly threefold shorter. The present study documents clinicopathologic findings and the distribution of abnormal prion proteins (PrPSc) by immunohistochemical (IHC) and Western blot (WB) techniques, in tissues of sheep inoculated with No. x124. All inoculated sheep developed clinical disease and were euthanatized within an average of 7.7 months postinoculation (MPI). Sheep that had valine/valine or alamine/valine at codon 136 of prion protein (PRNP) gene developed the disease faster and were euthanatized at an average of 4.3 and 5.6 MPI, respectively. Also, the inoculum was able to induce disease in a short time (7 MPI) in a sheep that was relatively resistant (QR at codon 171) to scrapie. This indicates that inoculum No. x124 appears to induce scrapie in shorter time than inoculum No. 13–7, especially in sheep homozygous or heterozygous for valine at codon 136.
Introduction
Scrapie belongs to a family of diseases that are collectively known as transmissible spongiform encephalopathies (TSEs) of animals and humans. It is a naturally occurring disease of sheep and goats. Infection leads to accumulations of an abnormal form of prion protein (PrPSc) in tissues of the central nervous system (CNS) and other organs of the affected animals. Susceptibility to scrapie is partially dependent on genetic composition of the host, and in sheep, polymorphisms of the PRNP gene at codons 136 (valine [V] or alanine [A]), 154 (histidine [H] or arginine [R]), and 171 (glutamine [Q], arginine [R], or histidine [H]) are the dominant determinants.1, 9 When Suffolk sheep (QQ at codon 171) were intracerebrally inoculated with a US scrapie inoculum (No. 13-7), scrapie was induced within an average of 19 months.6 However, since then it has been shown that another US scrapie isolate, No. x124, from the University of Idaho, Caine Veterinary Center, induced scrapie much faster (<12 months) even when given to sheep orally.3
The objective of this study was to make a comparison of the incubation periods and clinicopathologic findings for inocula No. x124 and those described earlier for No. 13-7,6 when sheep are inoculated intracerebrally.
Material and Methods
Experimental animals
For this study, 14 four-month-old Cheviot lambs (all castrated males) of various PRNP genotypes (at codons 136, 154, and 171) were obtained from a scrapie-free flock from western Iowa. The lambs were divided by genotypes into 4 groups (A–D) and inoculated by the intracerebral route. Group A (n = 3) were AA/RR/QQ; Group B (n = 5) were AV/RR/QQ; Group C (n = 5) were VV/RR/QQ, and Group D (n = 1) was AV/RR/QR (Table 1).
Survival periods, histology, immunohistochemistry, and Western blot findings in tissues of 14 Cheviot sheep that developed clinical scrapie after inoculation with US scrapie agent (inoculum No. x124) by intracerebral route. ∗
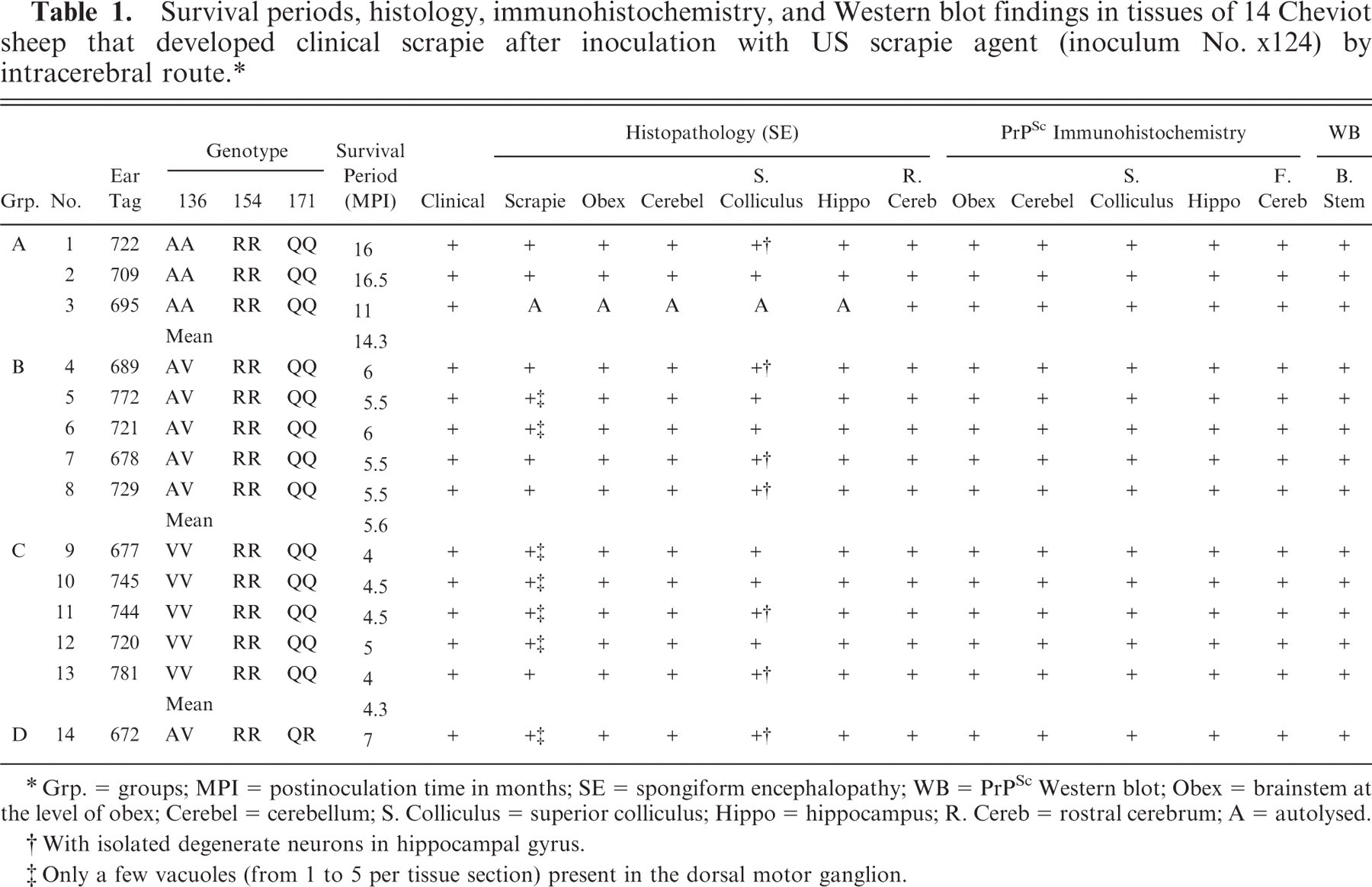
Grp. = groups; MPI = postinoculation time in months; SE = spongiform encephalopathy; WB = PrPSc Western blot; Obex = brainstem at the level of obex; Cerebel = cerebellum; S. Colliculus = superior colliculus; Hippo = hippocampus; R. Cereb = rostral cerebrum; A = autolysed.
With isolated degenerate neurons in hippocampal gyrus.
Only a few vacuoles (from 1 to 5 per tissue section) present in the dorsal motor ganglion.
Housing
Inoculated animals were initially housed in a biosafety level-2 containment facility (2 per pen) and 2 weeks postinoculation were moved to outside pens at the National Animal Disease Center (NADC), Ames, Iowa. The sheep were fed pelleted growth and maintenance rations that contained no ruminant protein, and clean water was available ad libitum.
Inoculum
Inoculum No. x124 was prepared from a pool of 7 scrapie-affected sheep brains from 5 flocks3 obtained from the Caine Veterinary Teaching Center, Idaho. Five brains were from AA/RR/QQ and one each was from AV/RR/QQ and VV/RR/QQ at codon 136, 154, and 171 of PRNP gene, respectively. All were positive by immunohistochemical and Western blot analysis.3 The infected brains were homogenized by ultrasonication at the final concentration of 20% (wt/vol) in phosphate-buffered saline (PBS). This stock was diluted to 10% (wt/vol) in PBS at the time of inoculation. Each animal was inoculated with 1 ml of this inoculum.
Inoculation procedure
The procedure for intracerebral inoculation of lambs has been described previously.7 Briefly, the animals were sedated with xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 1-mm hole was trephined through the calvarium. The inoculum (1 ml of 10% wt/vol brain suspension) was injected into the midbrain via a 22-gauge 9-cm long needle while withdrawing the needle from the brain. The skin incision was closed with a single suture.
Necropsy, samples, and laboratory procedures
Except for one sheep (No. 695, Table 1), all others were euthanatized when they developed terminal clinical signs of scrapie. They were examined at necropsy and 2 sets of tissue samples were collected. One set of tissues included representative sections of liver, kidney, spleen, skin, striated muscles (including heart, tongue, diaphragm, masseter), tonsils (pharyngeal, palatine), thyroid gland, turbinates, lung, intestines (ileum), lymph nodes (retropharyngeal, mesenteric), pituitary gland, Gasserian ganglion, brain (hemisections of cerebral cortex, cerebellum, superior colliculi, and brainstem including obex), and eye. These tissues were fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 5 μm, and stained with HE for light microscopy. The second set of tissues was frozen at −20°C.
All paraffin-embedded tissues were also stained by an automated immunohistologic (IHC) method for detection of PrPSc as described previously.6 Briefly, following deparaffinization and rehydration, tissue sections were autoclaved for 30 minutes in an antigen retrieval solution (Dako Target Retrieval Solution, Dako Corp., Carpinteria, CA) and stained with an indirect avidin biotin system (Basic Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems, Inc., Tucson, AZ) designed for an automated immunostainer (NexES IHC module, Ventana Medical Systems). The primary antibody was a cocktail of 2 monoclonal antibodies, F89/160.1.511 and F99/97.6.1,10 each used at a concentration of 5 μg/ml, and incubation was carried out at 37°C for 32 minutes. The secondary antibody was biotinylated anti-mouse (Biotinylated anti-mouse IgG [made in horse], Vector Laboratories, Burlingame, CA), diluted 1:200 and incubated for 8 minutes at 37°C.
One half of the brain (cut longitudinally) was frozen for Western blot (WB) as described previously.6 Briefly, the modification consisted of the use of P4 as primary antibody and a secondary biotinylated anti-mouse antibody (dilution 1:10,000; Biotinylated anti-mouse IgG, Amersham Biosciences, Piscataway, NJ), followed by a streptavidin horseradish-peroxidase conjugate (dilution 1:10,000; Streptavidin horseradish-peroxidase conjugate, Amersham Biosciences). The immunoblot was developed with an ECL Plus chemiluminescence solution (ECL Plus, Amersham Biosciences) before being exposed to film or an electronic imaging system.
Results
All inoculated animals developed clinical signs of scrapie prior to euthanasia. Initial clinical signs of the disease consisted of a progressive decrease in appetite and associated weight loss. Later, the affected sheep developed fine head tremors, listlessness, progressive problems with locomotion, and in terminal stages, sternal recumbency. None of the affected sheep exhibited obvious pruritus or loss of wool. At necropsy the animals were thin with scant body fat. None appeared emaciated, and other significant lesions were not seen.
The postinoculation survival periods for the individual animals varied with genotypes and are shown in Table 1 and Fig. 1. As a group, the shortest postinoculation survival time was Group C (VV/RR/QQ; 4.3 MPI). This was followed by Group B (AV/RR/QQ; 5.6 MPI); Group D (AV/RR/QR; 7 MPI); and Group A (AA/RR/QQ; 14.3 MPI) (Table 1).
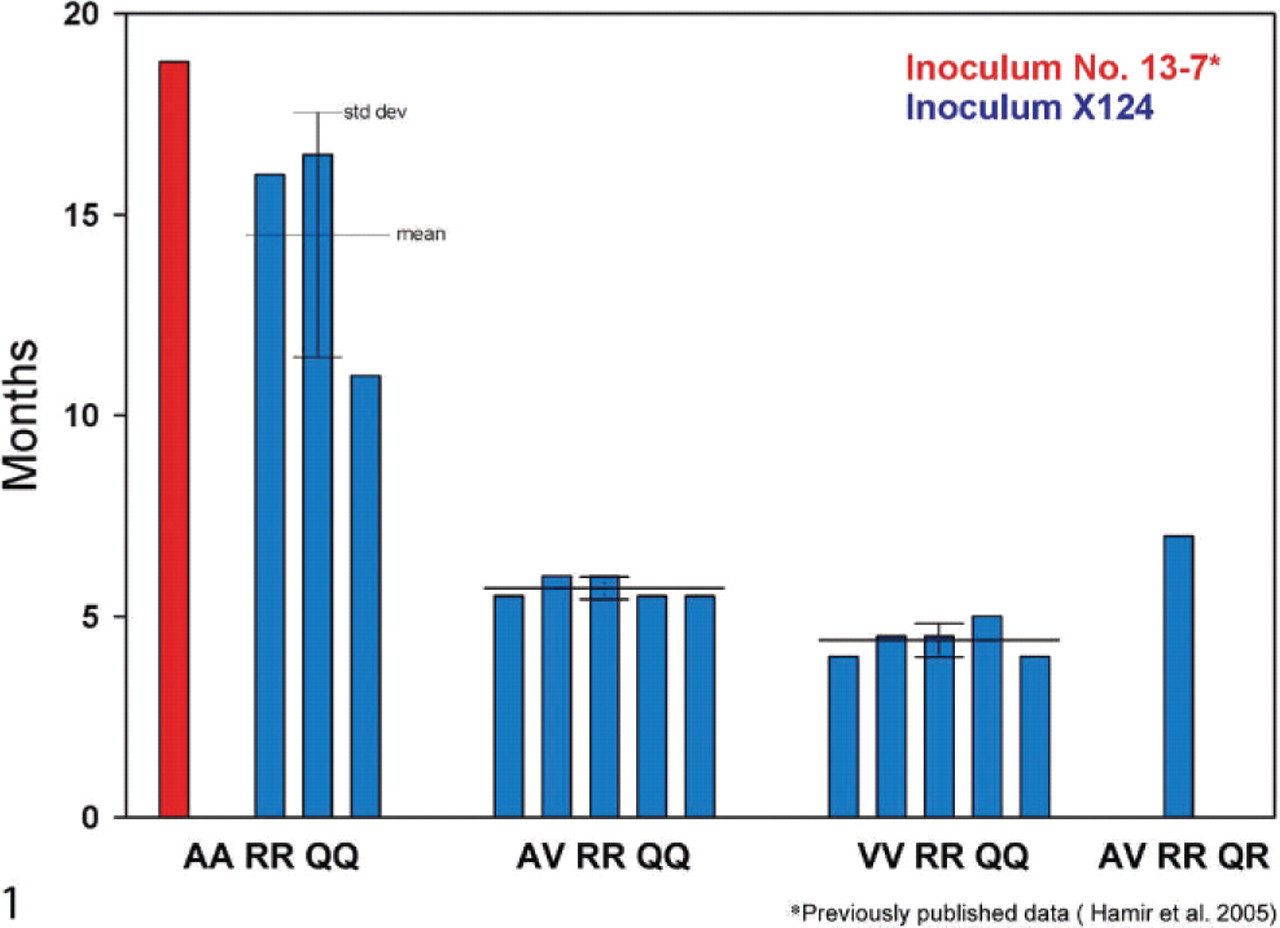
Comparison of survival periods and genotypes of sheep inoculated with 2 US scrapie agents (inoculum No. 13-7 and No. x124) by intracerebral route.
The distribution of histologic lesions in the brain and the results of IHC and WB are given in Tables 1 and 2. Microscopic lesions of spongiform encephalopathy (SE) were present in all sheep except No. 695, which was extensively autolysed (Table 1). However, even in this sheep, PrPSc was readily demonstrated in brain tissue by IHC and WB techniques (Table 1). In better preserved specimens, the SE lesions were generally most severe in the cerebellum and obex (Figs. 2–6). However, the sheep in Groups C (VV/RR/QQ) and D (AV/RR/QR) had only a few characteristic vacuoles that were observed in either the neuronal cytoplasm or in the neuropil of dorsal motor nucleus of the vagus nerve (Fig. 8). In the cerebellum, the vacuoles were present in the molecular, Purkinje cell and in the granular layers (Fig. 5). Both the cerebral cortex (Fig. 2) and the colliculus (Fig. 4) had diffuse vacuoles in neuropil. In the hippocampus, the vacuoles were in the neuropil surrounding the hippocampal gyrus (Fig. 3). In 7/14 sheep there were isolated degenerate neurons in the hippocampal gyrus (Table 1).
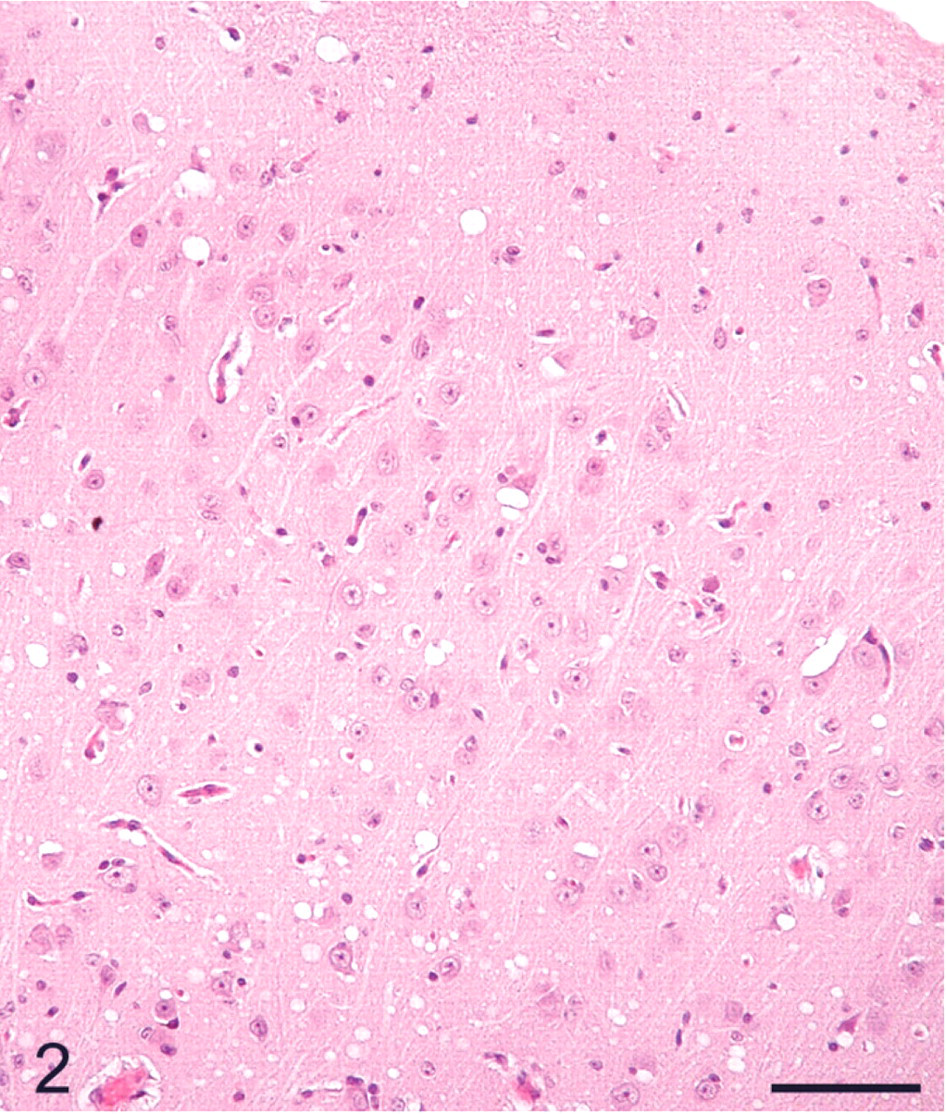
Cerebral cortex; sheep No. 709. Diffuse vacuoles in the neuropil. HE. Bar = 80 μm.
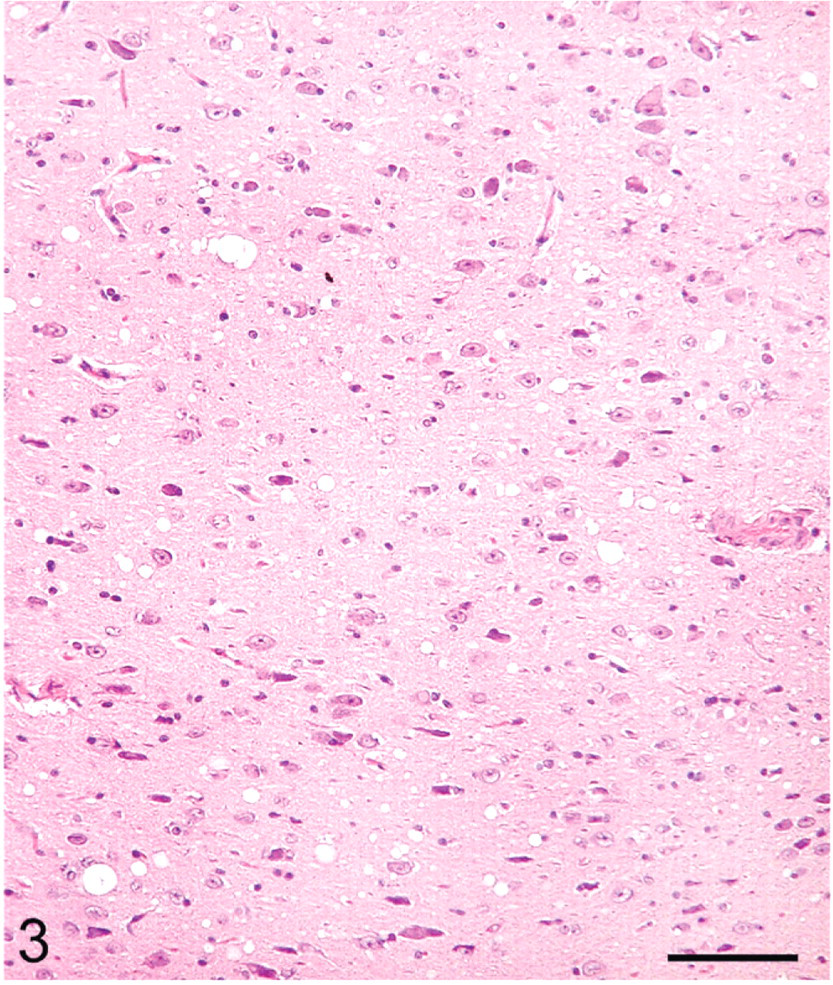
Hippocampus; sheep No. 709. Multifocal vacuoles in the hippocampal gyrus. HE. Bar = 80 μm.
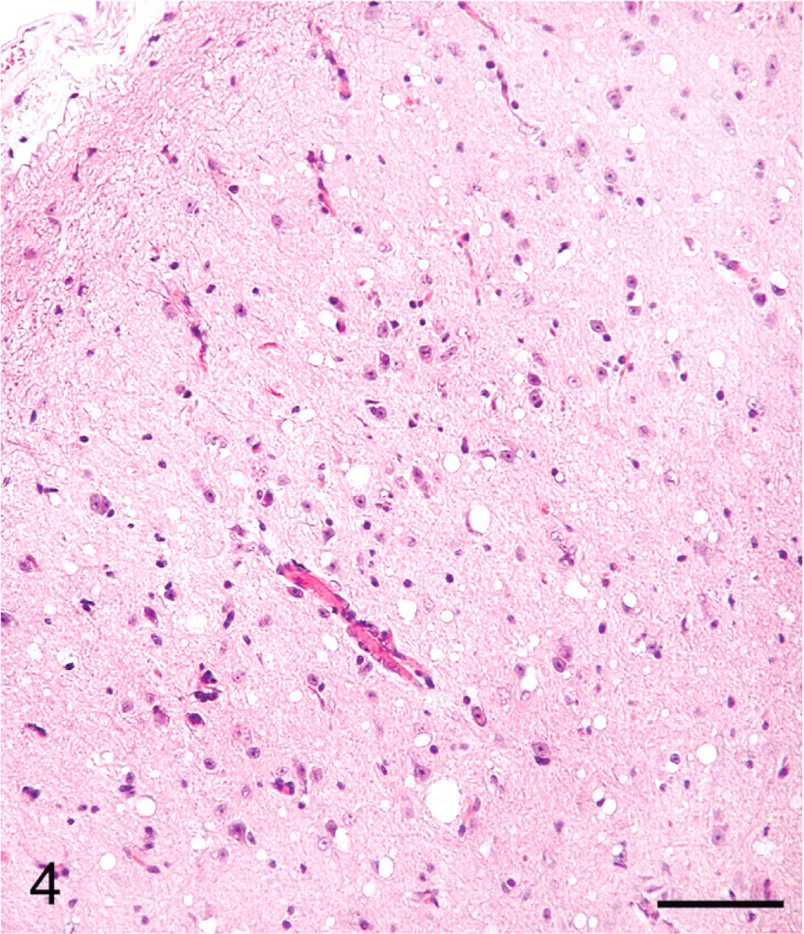
Colliculus; sheep No. 709. Multifocal vacuoles in the neuropil. HE. Bar = 80 μm.
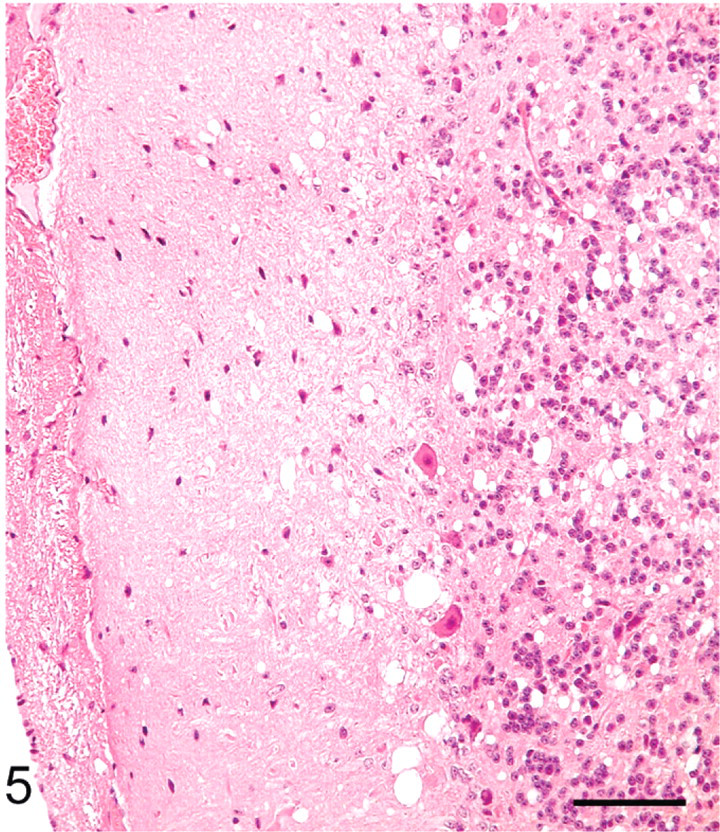
Cerebellum; sheep No. 709. Multifocal vacuoles in the molecular area. HE. Bar = 80 μm.
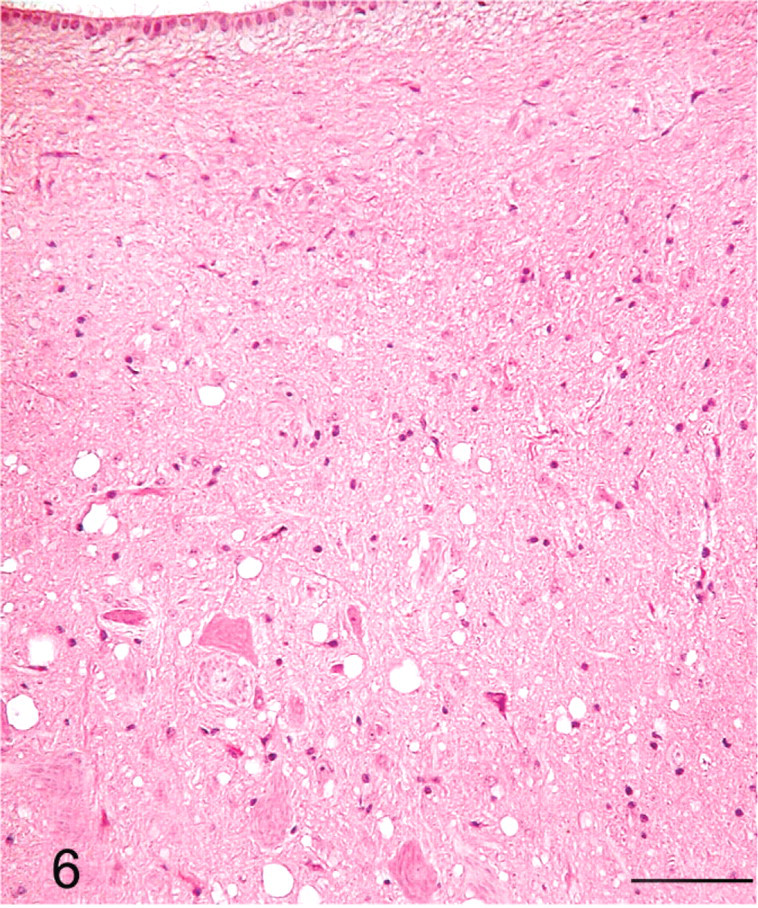
Brainstem, dorsal motor nucleus of vagus; sheep No. 709. Extensive SE lesions. HE. Bar = 80 μm.
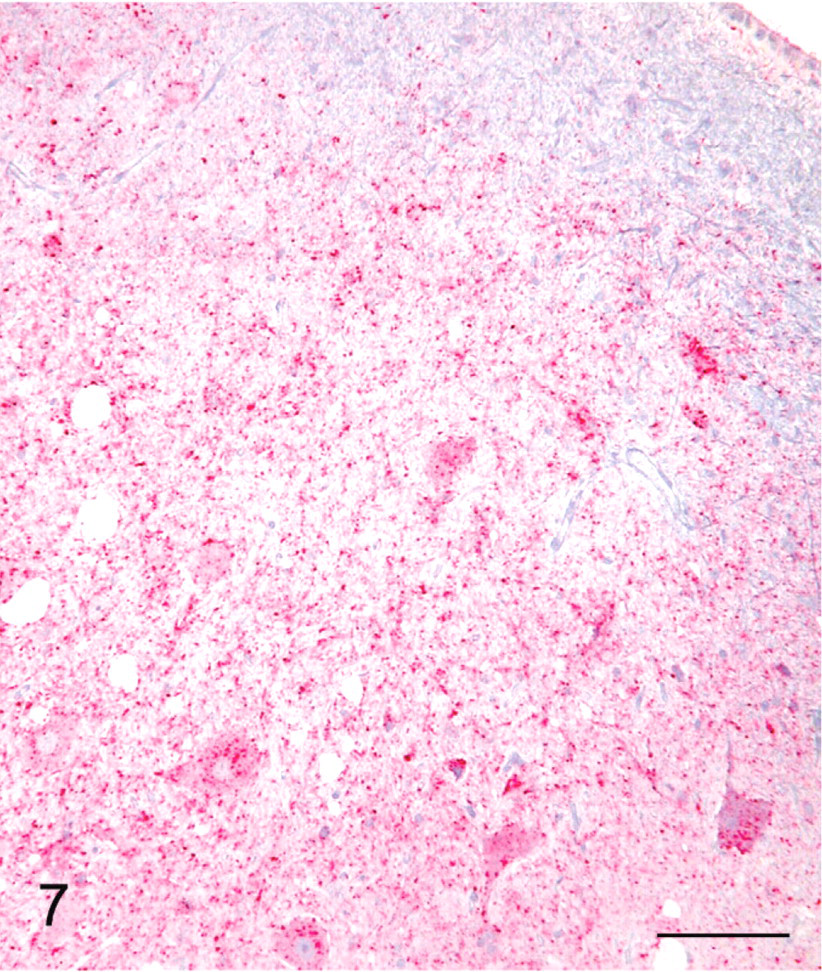
Brainstem, dorsal motor nucleus of vagus; sheep No. 709. Extensive SE lesions showing diffuse labeling with PrPSc. Stained for PrPSc labeling (red) by IHC (immuno-alkaline phosphatase) and counterstained with hematoxylin. Bar = 80 μm.
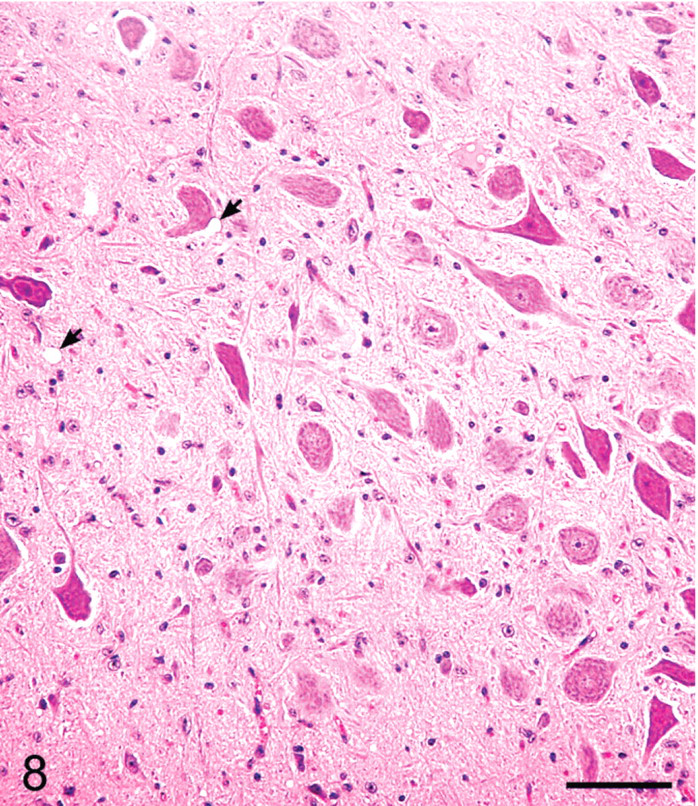
Brainstem, dorsal motor nucleus of vagus; sheep No. 720. Minimal SE lesions. Two small vacuoles are shown by arrows. HE. Bar = 80 μm.
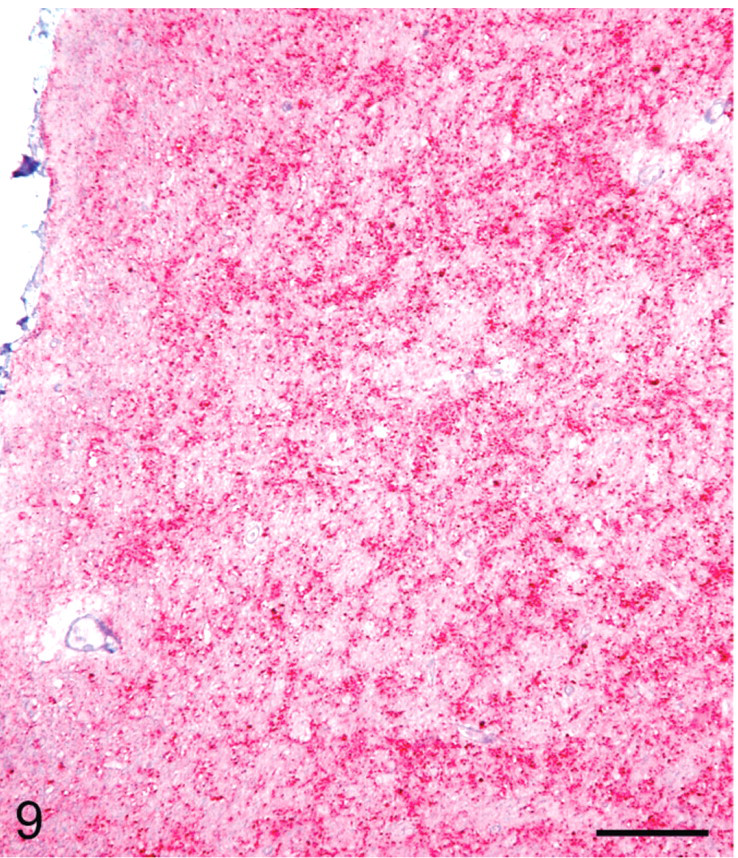
IHC labeling of PrPSc in the lymphoreticular system (LRS) and CNS tissues was present in 14/14 scrapie-affected sheep (Table 1). The intensity and distribution of PrPSc labeling in the CNS were similar in all sheep. The intensity of PrPSc labeling was greatest in midbrain (level of superior colliculus) with slightly less labeling present in brainstem (area of obex, Figs. 7, 9), cerebellum, hippocampus and rostral cerebrum. In substantial areas of brainstem, midbrain, and cerebellar folia and basal nuclei, the PrPSc accumulations tended to coalesce to the extent of confluence. The pattern of PrPSc accretion in the hippocampus and cerebral cortex was typified by intense perineuronal labeling and multifocal coarse granular accumulations in the neuropil. The plexiform layers of retina contained confluent accumulations, whereas receptor, nuclear, and ganglion cell layers had multifocal punctuate PrPSc labeling. PrPSc labeling in Gasserian ganglia was relatively less intense, punctuate in character, and confined to neuronal perikarya. Moderately intense punctuate labeling was present in the pars nervosa of the pituitary glands examined.
Brainstem, dorsal motor nucleus of vagus; sheep No. 720. Minimal SE lesions showing diffuse labeling with PrPSc. Stained for PrPSc labeling (red) by IHC (immuno-alkaline phosphatase) and counterstained with hematoxylin. Bar = 80 μm.
Localization of PrPSc in the peripheral nervous system was limited to a moderate amount of punctate labeling of the perikaryon of several neuron groupings in the myenteric plexus of 4/5 Group C (VV/RR/QQ) sheep, a finding not present in the other sheep groups. Multifocal punctuate PrPSc labeling of adrenal cortical and medullary cells (1/5 Group C sheep) was noted.
All or some of the LRS tissues collected from 13/13 QQ sheep contained PrPSc labeling in lymphocyte follicles, predominately in germinal centers with occasional and lesser labeling in paracortical areas. PrPSc was identified in all of the lymphoid organs collected from 2/2 Group A sheep (AA/RR/QQ) and 5/5 Group C sheep (VV/RR/QQ), whereas 2–4 of the lymphoid organs collected from Group B (AV/RR/QQ) and Group D (AV/RR/QR) were PrPSc-negative (Table 2). Submucosal lymphocyte follicles in the third eyelid labeled for PrPSc in 3/4 Group C sheep, but were PrPSc-negative in 5/5 Group B and 1/1 Group D sheep; third eyelid tissue samples were not collected from Group A sheep. Submucosal lymphocyte follicles in the ileum (Peyer's patches) from Group B sheep were PrPSc-negative, whereas PrPSc-labeled Peyer's patches were seen in the remaining 8/8 sheep from which samples of ileum were collected. PrPSc labeling was present in the spleen of 13/13 sheep, localized to peri-arteriolar lymphoid sheaths (Table 2).
Immunohistochemical findings in tissues of 14 Cheviot sheep that developed clinical scrapie after inoculation with US scrapie agent (inoculum No. xl24) by intracerebral route. ∗
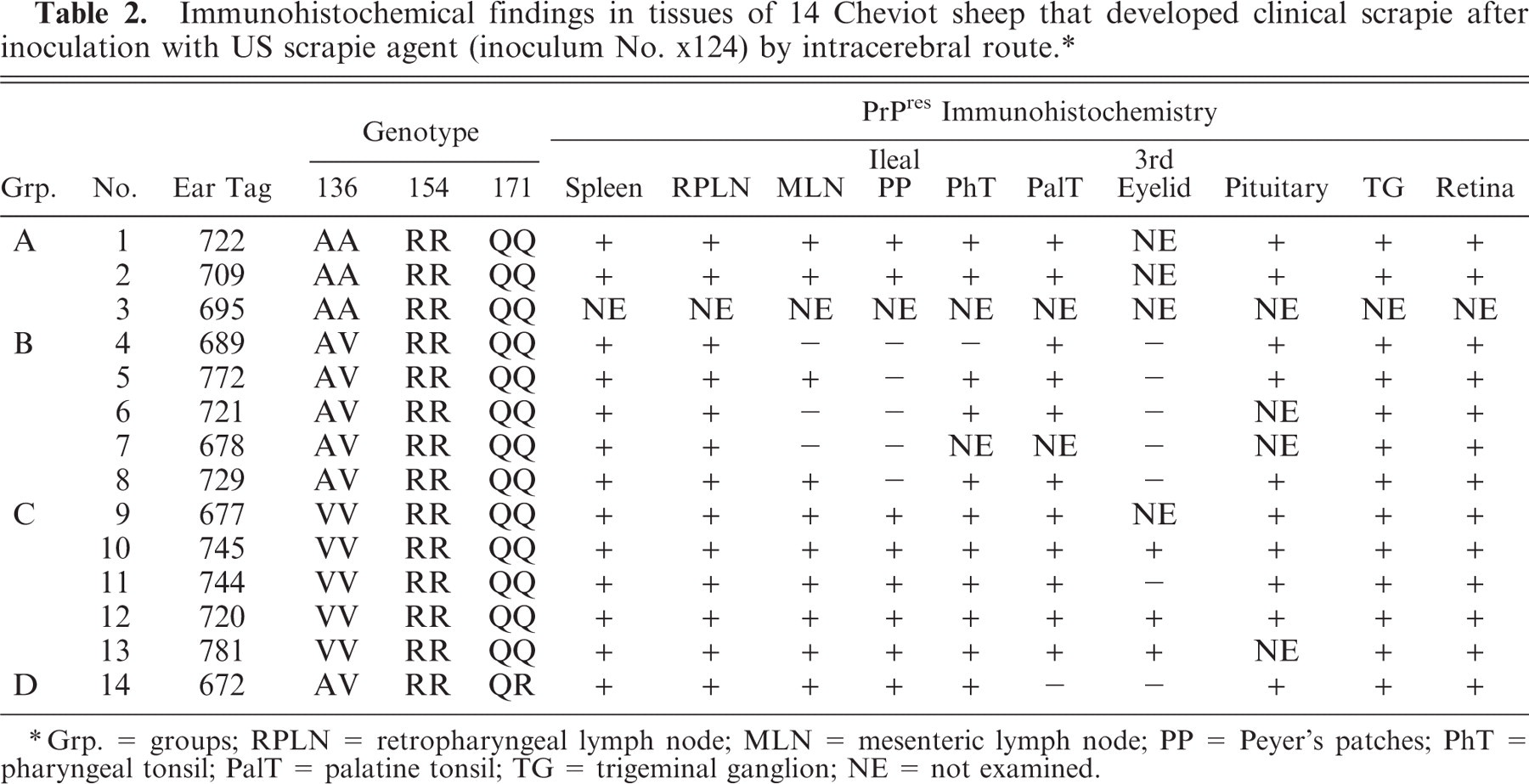
Grp. = groups; RPLN = retropharyngeal lymph node; MLN = mesenteric lymph node; PP = Peyer's patches; PhT = pharyngeal tonsil; PalT = palatine tonsil; TG = trigeminal ganglion; NE = not examined.
IHC-stained tissue sections of non-CNS and nonlymphoid tissues, including striated muscles (heart, diaphragm, and masseter muscle), did not reveal PrPSc labeling.
Western blot analyses of brainstem samples from these animals revealed all sheep inoculated with the scrapie agent were positive for the presence of PrPSc as detected by monoclonal antibody P4 (Fig. 10). The PrPSc isoforms from all scrapie-positive brainstems had a similar glycosylation pattern and distribution as well as similar electrophoretic mobility (Fig. 9).
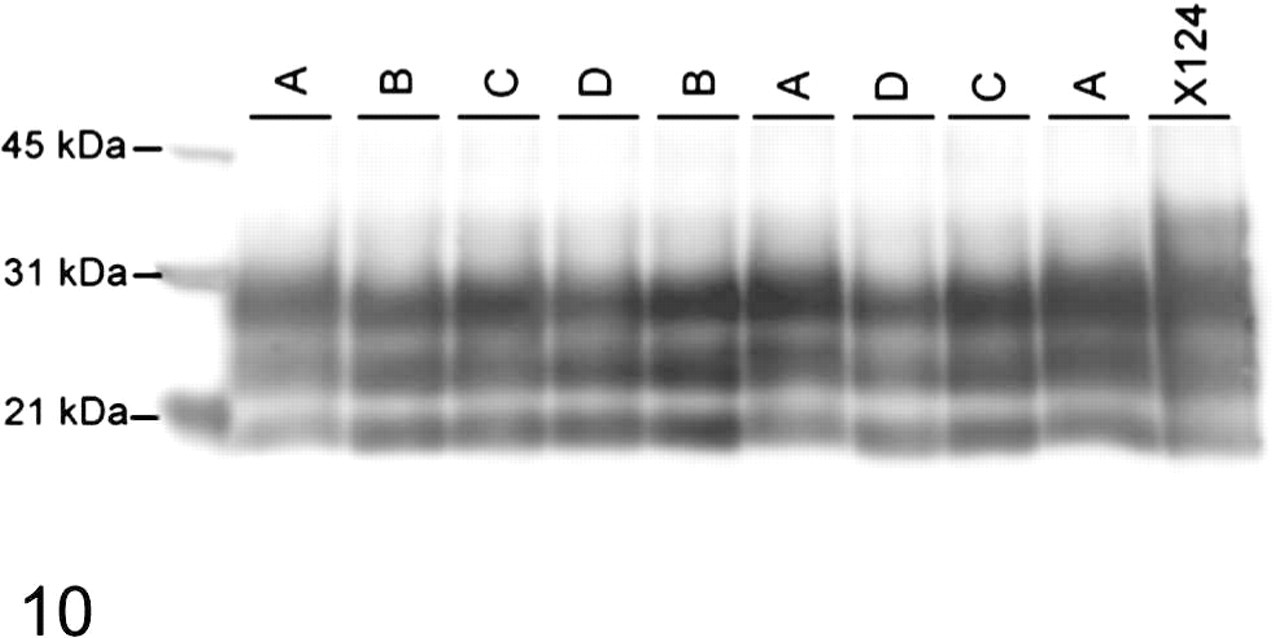
Western blot comparison of PrPSc in brainstems of individual sheep representing 4 different groups (based on genotype). Samples are as follows: Group A, AA/RR/QQ; Group B, AV/RR/QQ; Group C, VV/RR/QQ; Group D, AV/RR/QR. Samples were loaded at 0.1 mg per lane and probed with monoclonal antibody P4. Molecular weight markers in kDa are indicated on the left side of the blot.
Discussion
This study utilized the intracerebral route of inoculation to provide the most efficient means of testing the susceptibility of sheep to the scrapie agent (No. x124) and to provide a comparison with similarly designed previous studies.4, 6 In general, the intracerebral route of inoculation of TSE agents is the fastest means of inducing disease with these agents in susceptible hosts. In AA/RR/QQ sheep using a pooled scrapie inoculum (No. 13-7), similar intracerebral inoculations resulted in an average incubation time of 18.8 MPI,6 and when the same inoculum was intracerebrally inoculated in VV/RR/QQ lambs, the mean incubation time was 17.8 MPI (R. A. Kunkle et al., unpublished). However, in the present study, the sheep in Groups A and C with similar PRNP genotypes (AA/RR/QQ and VV/RR/QQ) had shorter survival times—14.3 and 4.3 MPI, respectively. Also, the sheep in Group B with AV/RR/QQ revealed a relatively short survival time of 5.6 MPI. These are very short survival times and are comparable with those obtained in New Zealand-derived Cheviot sheep inoculated subcutaneously with scrapie inoculum No. SSBP/1.8 From the PRNP genotypes, it appears that valine at codon 136 has a major influence on the incubation period of the disease in this breed of sheep. These data are consistent with previously reported short incubation times for the same inoculum delivered orally in sheep with valine at codon 136.3
Current programs for prevention and control of prion diseases in sheep are based on selective breeding for sheep carrying resistant genotypes for breeding. Sheep with 171 QR have been highly resistant to currently circulating strains of scrapie, and prior to 1996 no cases of scrapie had been found in sheep with this genotype.12 However, in the present study, the survival time for the Group D (AV/RR/QR) animal was half that of sheep in Group A (AA/RR/QQ), which were expected to be much more susceptible to the disease.
In recent years, sheep producers in the USA and Europe have been encouraged to eliminate codon 171QQ homozygous sheep in favor of QR or RR homozygous animals as a means of controlling scrapie in their flocks. However, the results of the present study indicate that the 171QR sheep in Group D succumbed to the disease in half the time of that of 171QQ sheep in Group A. Since there was only one sheep in Group D, this part of the study needs to be repeated with larger number of codon 171QR as well as in RR sheep.
Although all sheep in this study had severe and terminal clinical signs of scrapie, the neuropathologic changes of spongiform encephalopathy observed in dorsal motor nucleus of vagus were minimal in Groups B, C, and D in either 136VV or AV sheep. Compared with previous studies of intracerebral inoculations of inoculum (No. x124) into Suffolk sheep,4 the morphologic lesions seen in the obex region of the brainstem in the present study were relatively mild. Otherwise, the histopathologic lesions, IHC, and WB findings were comparable and generally similar to the previous findings.4, 6
There are reportedly more than 20 different strains of sheep scrapie identified in the UK.2 Strain typing studies have not been done in this country to determine the number of strains of scrapie that are circulating in US sheep. Recently, inoculum No. 13-7 has been propagated into 4 consecutive generations of Suffolks in an attempt to stabilize it, so that it can be used as comparison to other scrapie isolates.5 From the results of this and previous studies, it would appear that there are at least 2 different strains/isolates in pooled inocula of Nos. x124 and 13-7 that can be identified on the basis of survival times and neuropathologic findings in experimental sheep.6, 8
In conclusion, when compared with previous studies with inoculum 13-7,6, 8 inoculum x124 showed reduced survival times in all sheep with different PRNP genotypes, and the reduced survival time was most prominent in sheep with homozygous or heterozygous valine at PRNP codon 136, suggesting a major effect of this codon on the survival times in Cheviots inoculated with scrapie. It is also concluded from the results of this study that sheep with QR at codon 171 may be as susceptible to scrapie (inoculum x124) as those with QQ at this codon.
Footnotes
Acknowledgements
We thank Dr. M. Kehrli for constructive comments and for his enthusiastic support, Dr. K. O'Rourke, Pullman, Washington, for prion antibodies for IHC, the late Dr. L. Nusz and Drs. J. Laufer and T. Anspach for clinical assistance, and Mr. J. Fosse for the photomicrographs. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Joseph Lesan, Deb Clouser, Kevin Hassall, Sharla Van Roekel, Ginny Montgomery, Micky Fenneman, and the TSE animal caretakers.
This study was carried out under the guidelines of the institutional Animal Care and Use Committee at National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
