Abstract
Scrapie, a transmissible spongiform encephalopathy of sheep and goats, exists in most small ruminant-producing countries of the world. A novel form of this disease was recently recognized and is known by various names, including Nor98, Nor98-like, and atypical scrapie. Differing from classic scrapie in epidemiology, histopathology, and biochemical characteristics, atypical scrapie cases have been identified throughout Europe and in the United States. Enhanced scrapie surveillance efforts recently identified 3 cases of atypical scrapie in Canada.
Scrapie is a naturally occurring transmissible spongiform encephalopathy of sheep and goats that has been recognized in Canada since 1938. 15 Slowly progressive and uniformly fatal, affected sheep may develop ataxia, hypermetria, anxiety, tremors, intractable pruritus, and a loss of body condition, although clinical signs are variable and not necessarily overt. Although a suspicion of scrapie can be based on clinical signs, the demonstration of pathologic prion protein associated with scrapie (PrPSc) in target tissues is required for the confirmation of disease. Typically, accumulations of PrPSc are most abundant in the dorsal motor nucleus of the vagal nerve at the level of the obex of the medulla but may also be found in other regions of the central nervous system and peripheral lymphoid tissues, including tonsils and lymph nodes. Histologic lesions are generally restricted to the central nervous system and include vacuolation of neurons and neuropil, particularly at the obex.
The classic form of sheep scrapie is characterized by a broadly consistent disease phenotype, with variations in clinical signs and neuropathology being attributed to prion strain differences, with host genotype heavily influencing disease susceptibility. Multiple strains of classic scrapie have been discerned through strain-specific neuropathology in rodent models 5 or through differences in the prion protein biochemical properties. 4 Polymorphisms in the host-encoded prion gene (PRNP) are major determinants of susceptibility to classic scrapie, with variations at codons 136, 154, and 171 conveying variable degrees of resistance. 1 The existence of genetic resistance has led to the implementation of breeding programs in many countries aimed at reducing national flock susceptibility.
In 2003, a novel and distinct scrapie phenotype was described in Norway 3 and has become known as Nor98, Nor98-like, nonclassic, or atypical scrapie. The epidemiology, neuropathology, and biochemical properties of atypical scrapie differ from those of classic scrapie, and diagnostic discrimination requires Western blot or immunohistochemistry (IHC) of target tissues. Atypical scrapie-affected sheep are generally older (5 to >10 years of age), whereas classic scrapie tends to cause disease in 3–5-year-old animals. 9 Clinical signs can be vague or absent with atypical scrapie, may include ataxia and emaciation, but notably do not involve the pruritus and wool loss observed with classic scrapie. 2 Of relevance to national sheep-breeding programs, cases of atypical scrapie are frequently found in animals with genetic resistance to classic scrapie. The neuropathology and neuroanatomical distribution of PrPSc differs widely between atypical and classic scrapie, 11,13 and PrPSc has not yet been detected in lymphoid tissues of atypical cases, as it most often is in classic scrapie. 2 The biochemical properties of atypical scrapie and its passage into mice 7 identify it as a unique strain of scrapie.
Summary of diagnostic results from 3 cases of atypical scrapie in Canada.*
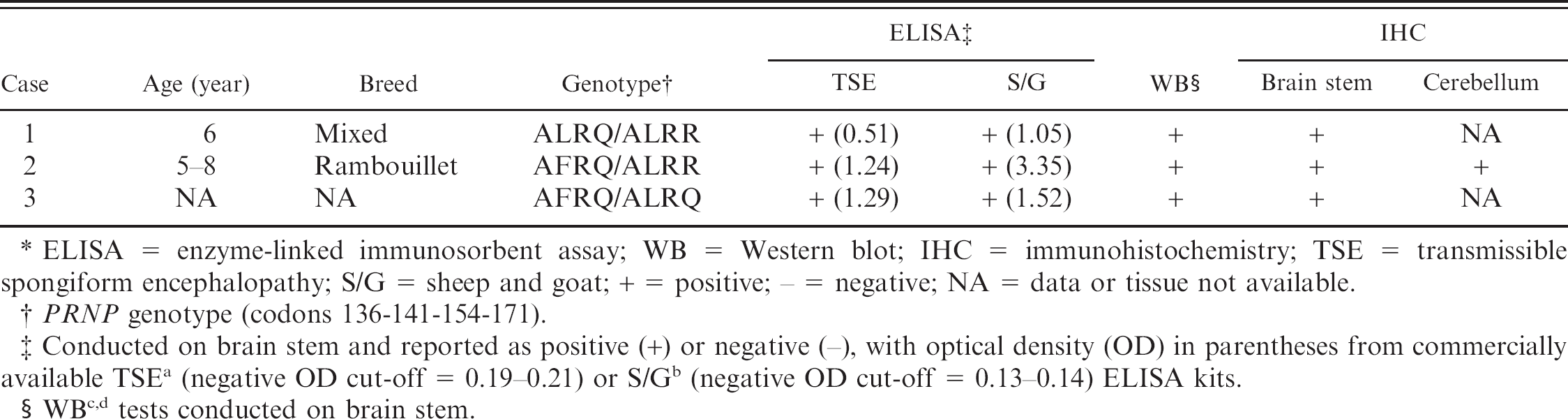
ELISA = enzyme-linked immunosorbent assay; WB = Western blot; IHC = immunohistochemistry; TSE = transmissible spongiform encephalopathy; S/G = sheep and goat; + = positive; − = negative; NA = data or tissue not available.
PRNP genotype (codons 136-141-154-171).
Conducted on brain stem and reported as positive (+) or negative (-), with optical density (OD) in parentheses from commercially available TSE a (negative OD cut-off = 0.19–0.21) or S/G b (negative OD cut-off = 0.13–0.14) ELISA kits.
WB c,d tests conducted on brain stem.
The development of high-throughput testing methods for transmissible spongiform encephalopathies, the possibility that bovine spongiform encephalopathy could enter the sheep population, and the need to determine the prevalence of scrapie for the purpose of control or eradication, have led to large-scale surveillance efforts in Europe and North America. Enhanced scrapie surveillance throughout Europe has demonstrated that atypical scrapie represents an increasingly larger proportion of scrapie cases in many countries. Recently, the first 6 cases of atypical scrapie detected in the United States were reported. 8 The Canadian National Scrapie Surveillance Program was initiated in 2005 and involves the active testing of sheep more than 12 months of age, which are slaughtered at federal and provincial abattoirs, or are identified as fallen stock from rendering companies, sales barns, cull ewe feedlots, or farms. In the current study, the first 3 cases of atypical scrapie detected in Canadian sheep are presented and discussed.
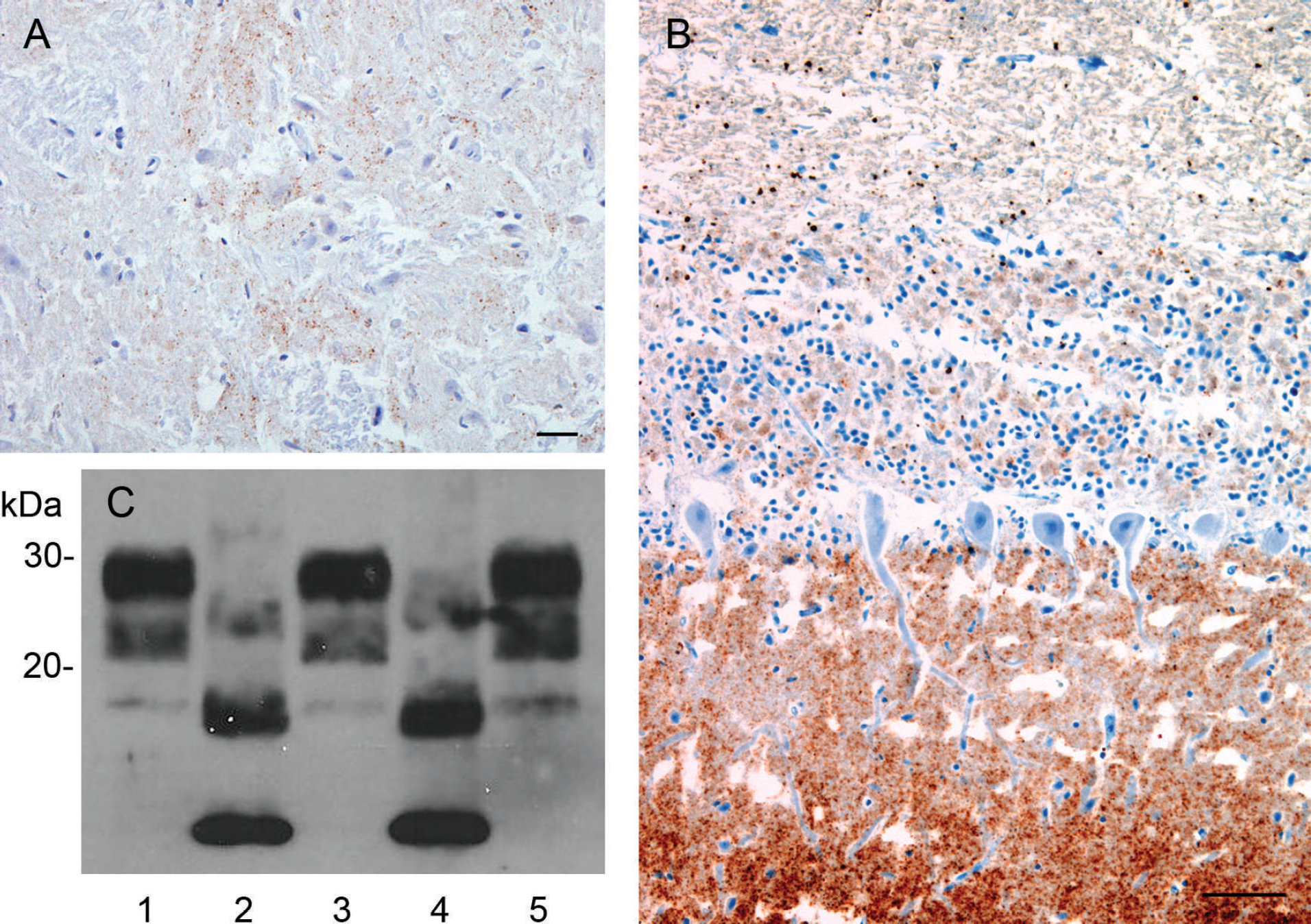
The initial case involved a clinically normal 6-year-old, mixed breed (Dorset x Suffolk) ewe that was euthanized after participation in a research project at a Canadian academic institution. The ewe had been obtained from a farm with no history of scrapie, and frozen brainstem tissue was submitted for routine scrapie surveillance testing. Positive results were obtained from 2 independent enzyme-linked immunosorbent assays a,b (ELISAs; Table 1). The molecular profile of the isolate was investigated by Western blot, c,d and an atypical scrapie banding pattern emerged, with the distinct low molecular weight band (<15 kDa) distinguishing it from classic scrapie isolates. Immunohistochemical analysis by using monoclonal antibody (mAb) 2G11, e as previously described, 16 revealed fine granular neuropil staining in the spinal tract of the trigeminal nerve (Fig. 1A) and globular white matter staining in adjacent corticospinal tracts; PrPSc staining was not observed in the dorsal motor nucleus of the vagus (DMNV), consistent with the staining distribution characteristic of atypical scrapie. The PRNP genotype of the affected animal was ALRQ/ALRR (136 AA, 141 LL, 154 RR, 171 QR), and, after quarantine, the 440 animal flock of origin was genotyped. Rectal biopsies were performed on all animals to screen for the presence of classic scrapie; however, no cases were detected. Intracerebral inoculation of brain stem homogenate from this case into tg338 mice (transgenic for ovine VRQ PrP) 6 caused clinical disease at a mean (± standard error of the mean) of 265 ± 6.5 days postinoculation. Transmission of this atypical scrapie isolate into mice was confirmed by IHC and Western blot, which demonstrated conservation of the prominent low molecular weight band (<15 kDa; data not shown). The distribution pattern of PrPSc immunostaining observed in these mice was consistent with the previous finding that atypical scrapie isolates tend to cause intense staining in the thalamus, corpus callosum, and cerebral cortex. 7
In the second case, a 5–8-year-old clinically normal Rambouillet ewe was culled from a commercial flock of approximately 360 animals in which it had spent its entire life and had had multiple lambings. Other than several breeding rams, no other animals had been introduced into the flock in more than 10 years. A portion of brainstem, including a fragment of cerebellum, was submitted for routine scrapie surveillance and was found to be positive on first and repeated testing by ELISA. a,b Confirmatory IHC by using either mAb 2G11 e or a 1:1 mixture of F89 f and F99 f mAbs revealed intense immunostaining in the molecular layer and, to a lesser extent, in the granular layer of the cerebellum (Fig. 1B). Scant, predominantly globular, or fine granular staining was scattered throughout the white matter of the cerebellum and neuropil of the medulla oblongata, but no staining was observed in the DMNV. Analysis of the sample by parallel Western blot tests c,d demonstrated the presence of a low molecular weight band (<15 kDa), consistent with atypical scrapie, and the PRNP genotype of this animal was determined to be AFRQ/ALRR (136 AA, 141 FL, 154 RR, 171 QR). The entire source flock was quarantined and genotyped, and rectal biopsies were performed on animals with QQ at codon 171 (71 animals). Classic scrapie was not detected in any of these sheep through IHC of rectal biopsy specimens.
Prion protein associated with scrapie (PrPSc) immunohistochemistry.
The third case was detected by routine ELISA surveillance testing and confirmed by IHC and Western blot c,d (Fig. 1C) as above. This was a clinically normal animal culled from a 650 animal commercial flock primarily composed of Rambouillet sheep with smaller numbers of Merino, Suffolk, and Dorper animals. Records of the age and breed of this particular animal were not available. Repeated ELISAs a,b were positive, and Western blot c,d demonstrated a characteristic atypical banding pattern. Immunohistochemistry revealed the diffuse presence of fine granular and globular PrPSc throughout the obex, with increased regional intensity in the spinal-tract nucleus of the trigeminal nerve and a paucity of staining in the DMNV. The PRNP genotype of this animal was AFRQ/ALRQ (136 AA, 141 FL, 154 RR, 171 QQ). Screening of the quarantined source flock for classic scrapie detected no positive cases by using the above-mentioned strategy of genotype-directed rectal biopsy testing by IHC.
Similarities exist among the 3 cases described and between other cases of atypical scrapie reported in Europe and North America. Two of the animals were known to be more than 5 years of age (case 3, age unknown), and all 3 were clinically healthy before sampling, although the presence of subtle neurologic deficits cannot be ruled out. These attributes are consistent with findings in other countries in which atypical scrapie is more frequently diagnosed in sheep older than 5 years of age, and clinical signs can be vague and are not always present. 2 The ELISA testing initially detected each Canadian case, and the identification of atypical scrapie was confirmed by the findings of a characteristic low molecular weight (<15 kDa) band on Western blot and the distinctive distribution pattern of PrPSc by IHC.
Two animals were sampled at slaughter facilities (cases 2 and 3), and 1 animal was sampled at a diagnostic pathology service (case 1). In all cases, frozen brainstem tissue was received and displayed moderate-to-marked autolysis when confirmatory testing was performed. Autolysis precluded detailed interpretation of histologic lesions and confounded identification of gross neuroanatomical sites in some instances. The presence of cerebellum in case 2 facilitated a rapid IHC diagnosis, because of the abundant staining observed in this region compared with weak staining in the brainstem and no staining in the DMNV. This is an important diagnostic feature that differentiates atypical from classic scrapie, in which staining in the DMNV at the level of the obex is always present. Concentrations of PrPSc in the medulla are generally lower in atypical scrapie, and a sensitive diagnostic screening test is thus required if medulla is to be the only tissue tested. A potential means to enhance detection of atypical cases would be to include testing of cerebellar samples in addition to brain stem.
Variations in sheep PRNP genotype at codons 136, 154, and 171 greatly influence susceptibility to classic scrapie and may have some influence over susceptibility to atypical scrapie. Although conferring moderate-to-marked resistance to classic scrapie, the AHQ and ARR alleles are often associated with atypical scrapie. 10,14 In addition, a leucine (L) to phenylalanine (F) polymorphism at codon 141 is more frequent in atypical cases. 12,14 Two of the Canadian atypical cases are considered genetically resistant to classic scrapie (ARR/ARQ), whereas 1 case is classified as having little genetic resistance (ARQ/ARQ); 2 cases carry an F at position 141. The PRNP genotypes of all 3 Canadian atypical cases have been associated with atypical scrapie in other countries. Perhaps not unexpectedly, the PRNP genotypic distribution profiles of the 3 source flocks were found to mirror that of the Canadian national flock (GeneNovaS: Genotyping statistics. Available at https://genenovas.ca/public/stats_pub.html. Accessed on December 23, 2009).
Although atypical scrapie can be experimentally transmitted to sheep 16 and ovinized transgenic mice, 7 epidemiologic evidence suggests that natural transmission within a flock occurs rarely if at all. Only a single animal per flock is usually detected, with rare exceptions being larger flocks that contain 1–3 additional atypical scrapie—positive animals. This contrasts with classic scrapie in which transmission within and between flocks has been well characterized. Interestingly, for reasons that are unclear, atypical scrapie and classic scrapie are rarely found within the same flock, 2 and no evidence of coexistence was found in the 3 flocks investigated in the current study. Although the origin of atypical scrapie is still not clear, mounting epidemiologic and biochemical evidence suggests it may arise sporadically, etiologically similar to sporadic Creutzfeldt-Jakob disease in humans. 2 As with classic scrapie, there is no current evidence that atypical scrapie is transmissible to humans, although a theoretical risk cannot be ruled out.
The classic form of scrapie has been extensively studied, with surveillance and breeding programs currently aimed at reducing disease prevalence. The existence of atypical scrapie in the Canadian sheep flock is not unexpected, and additional cases will undoubtedly arise during continued scrapie surveillance efforts. Rapid identification and differentiation of these cases is necessary to understand the national prevalence of atypical scrapie and complement the Canadian initiative to control classic scrapie.
Acknowledgements The authors thank the support staff in the Transmissible Spongiform Encephalopathy Unit and the Animal Care Facility at the Ottawa Laboratory Fallowfield for their outstanding technical assistance. This work was supported by the Canadian Food Inspection Agency. Disclaimer: Mention of trade names and commercial products is solely for the purpose of providing information and does not necessarily imply endorsement of their use by the Canadian Food Inspection Agency.
Footnotes
a.
TeSeE™ ELISA, Bio-Rad Laboratories, Hercules, CA.
b.
TeSeE™ Sheep/Goat ELISA, Bio-Rad Laboratories, Hercules, CA.
c.
TeSeE™ Western immunoblotting kit, Bio-Rad Laboratories, Hercules, CA.
d.
USDA-Pullman Western blot protocol by using mAb L42, U.S. Department of Agriculture, Pullman, WA.
e.
Institut Pourquier, France.
f.
VMRD Inc., Pullman, WA.
