Abstract
Amoebiasis is one of the most common protozoal diseases of reptiles, but amoebic myositis has not been reported in any animal species. An 11-year-old, male common water monitor lizard (Varanus salvator) was found dead with several subacute ulcerated skin wounds. Gross examination revealed multiple discrete to coalescing, white-yellow to gray, caseous foci scattered in the skeletal muscles and liver. The mucosa of small intestine was thickened, red, and contained many variably sized, dark red ulcers, with depressed and hemorrhagic centers. Histopathologic examination revealed severe necrotizing and granulomatous myositis, hepatitis, and enteritis accompanied by large numbers of intralesional, 10-20-μm diameter, periodic acid-Schiff-positive, amoeboid protozoa. Gene sequence analysis of a 136-bp region of the 18S ribosomal RNA amplified by polymerase chain reaction revealed 98–100% similarity with Entamoeba invadens. Aside from intestinal and hepatic involvement, no other internal organs were affected. The muscular infection by E. invadens likely resulted from a combination of direct invasion of trophozoites via skin wounds and hematogenous spread.
Amoebiasis associated with ulcerative enteritis and hepatitis caused by Entamoeba invadens is one of the most common and serious protozoal diseases in lizards, snakes, and some other reptiles. 2,12 E. invadens has a direct life cycle with no intermediate host. 5 The life cycle contains 2 morphologically distinct stages: the labile trophozoite, or mobile form, which inhabits the host, and the resistant cyst protected by a cell wall that allows its survival under adverse external environmental conditions. 5 The cyst is the main infectious form, which releases the motile trophozoite after oral ingestion by the host. 7 The most common presentation of invasive amoebiasis is enteritis via fecal-oral transmission, with further development of hepatitis through hematogenous dissemination via the portal vein. 6 The trophozoite first adheres to the intestinal mucus and epithelial cells by a Gal/GalNAc-specific lectin. 9 Following this intimate contact, pore-forming polypeptides called amoebapores are released by the trophozoite. 3 Proteolytic enzymes secreted by the parasite disrupt the intestinal mucus and epithelial barrier and facilitate tissue penetration. 3 Trophozoites secondarily enter the mesenteric circulation and invade the liver or other organs, causing hepatic abscesses or other extra-intestinal lesions. However, amoebic skeletal myositis has not yet been described. 11,13,15 We report here an unusual infection caused by E. invadens in a common water monitor (Varanus salvator) resulting from a combination of direct invasion via skin wounds and hematogenous spread.
An 11-year-old, male common water monitor lizard (Varanus salvator) showed signs of anorexia and occasional regurgitation for 7 days prior to being found dead beside the water pool in its new exhibition area. Prior to death, this animal was housed with another male common water monitor in a 120 × 60 × 60-cm tank with the temperature maintained at 22–28°C. The tank was cleaned and the water was changed daily. During this period, the lizard developed several nonhealing ulcerative skin wounds scattered over the abdomen, medial aspect of hind limbs, foot pads, and tail. These wounds were attributed to aggression by the enclosure mate. Six months prior to its death, the lizard escaped its enclosure and occupied a tortoise exhibit for 3 days.
A thorough necropsy was performed immediately following discovery. Gross examination revealed a total of seven 0.5–4-cm unhealed linear ulcerative skin wounds scattered over the abdomen, tail, and medial aspect of both hind limbs. After removing the skin, there were multiple, 1 × 0.5 × 1-cm to 8 × 2.3 × 2-cm, discrete to coalescing, white-yellow to gray, caseous foci randomly distributed in the superficial and deeper epaxial, intercostal, thigh, and tail muscles (Fig. 1). The liver showed approximately 30% enlargement in size, with multiple irregular, discrete to coalescing, friable, white-yellow foci occupying more than 70% of the liver parenchyma (Fig. 2). The lumen of small intestine was distended by gas and contained dark brown watery content; the mucosa was thickened, reddened, and contained many variably sized, dark red ulcers with depressed and hemorrhagic centers. No gross lesions were found in other visceral organs. Samples of the lungs, heart, liver, kidney, stomach, small intestine, large intestine, and epaxial, intercostal, thigh, and tail muscles were fixed in 10% neutral buffered formalin for further examination. The tissues were embedded in paraffin, sectioned at 5 µm in thickness, and stained with HE. Some selected sections were also subjected to periodic acid–Schiff (PAS), acid-fast, and Gram staining.
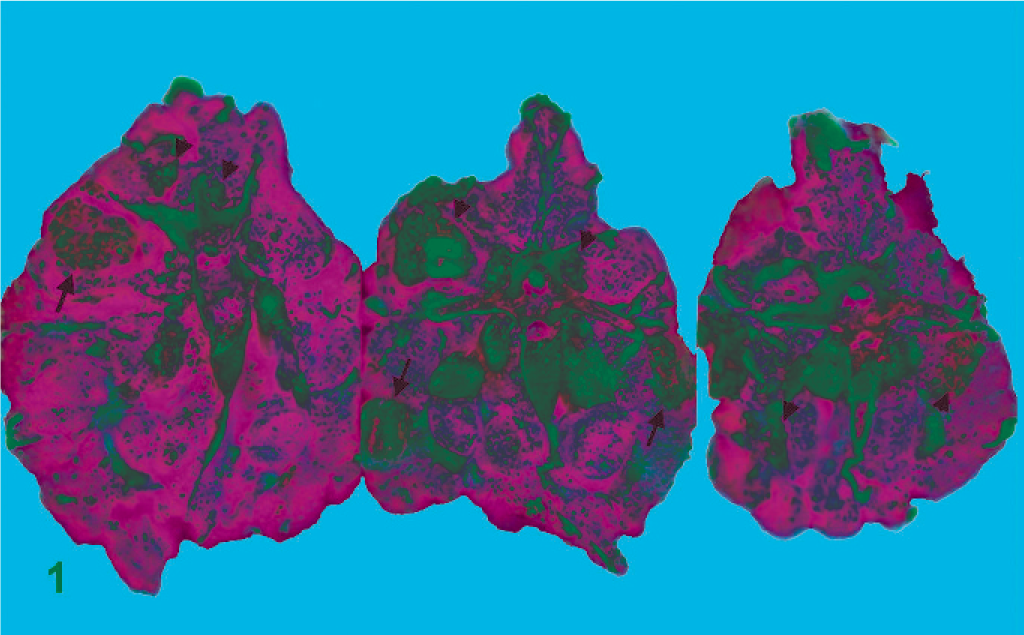
Tail muscles; common water monitor lizard. Transverse sections of tail show scattered necrotic foci in the superficial (arrows) and deeper (arrowheads) muscles.
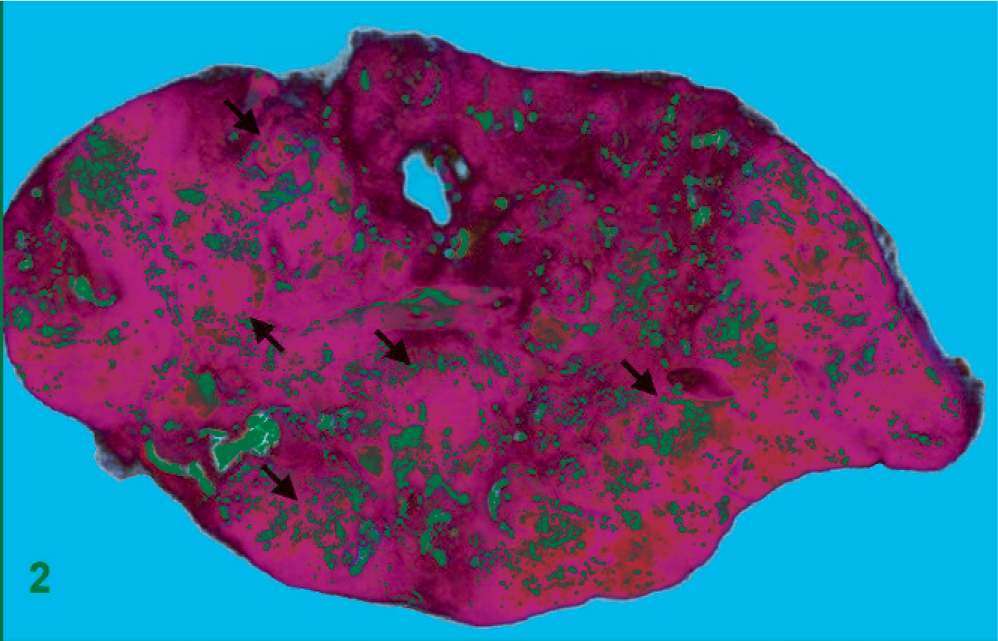
Liver; common water monitor lizard. Section of liver with discrete to coalescing, pale yellow foci occupying approximately 70% of the liver parenchyma (arrows).
Histologically, the affected skeletal muscle had severe, multifocal to coalescing, necrotizing and granulomatous myositis accompanied by varying numbers of intralesional PAS-positive, round to ovoid amoebic trophozoites (Fig. 3). Occasionally, amoebic trophozoites could be detected in the lumens of blood vessels at periphery of the affected regions (Fig. 3). The lesions were characterized by marked myofiber degeneration and necrosis and nodular infiltrates composed of large numbers of lymphocytes, macrophages, foreign-body and Langhans giant cells, and occasional heterophils with varying amounts of cellular debris and fibrosis. Some of the more superficially located affected muscles were adjacent to the cutaneous wounds; however, no direct extension was noted between the more deeply located affected muscles and the skin wounds. Many of the muscle lesions noted were distant from overlying skin wounds. The trophozoites had a size of 10–20 µm in diameter, vacuolated cytoplasm, a thick eosinophilic wall, and a single centrally to eccentrically located, round to ovoid, 2–3-µm nucleus containing chromatin plaques with a small indistinct endosome. The morphologic features are consistent with those of the genus Entamoeba. The liver also contained multiple, discrete to coalescing, variably sized necrotic foci containing amoebic trophozoites similar to those seen in the muscles. Trophozoites were most often located periphery of the necrotic areas. Occasionally, trophozoites could also be found within the lumens of portal veins (Fig. 4). Along with hepatocellular necrosis, there was infiltration of a variable number of heterophils, lymphocytes, plasma cells, macrophages, and foreign-body and Langhans giant cells, and mild to moderate fibrosis in the portal triads. The remaining hepatocytes showed marked cytoplasmic vacuolation. The affected intestinal tract contained areas of transmural, ulcerative, granulomatous enteritis. The ulcers were flask-shaped with congestion and hemorrhage and covered by a thick fibrinonecrotic exudate mixed with a varying number of heterophils, macrophages, and foreign-body and Langhans giant cells. The submucosa contained hemorrhage, edema, and an infiltration of inflammatory cells. Similar amoebic trophozoites were found within the edematous submucosa and in the areas surrounding blood vessels in the submucosa and muscularis. Secondary bacterial infection of various muscles, intestine, and liver was ruled out by the negative results of acid-fast and Gram staining. No lesions were noted in other organs.

Skeletal muscle; common water monitor lizard. The normal architecture of the affected muscle (M) is effaced by multiple coalescing necrotic areas infiltrated by large numbers of macrophages, multinucleated giant cells, lymphocytes, and plasma cells with many intralesional protozoan trophozoites (arrowheads). Inset: Higher magnification of an amoebic trophozoite present within the lumen of a blood vessel in the necrotic region (arrow). HE. Bar = 50 µm.
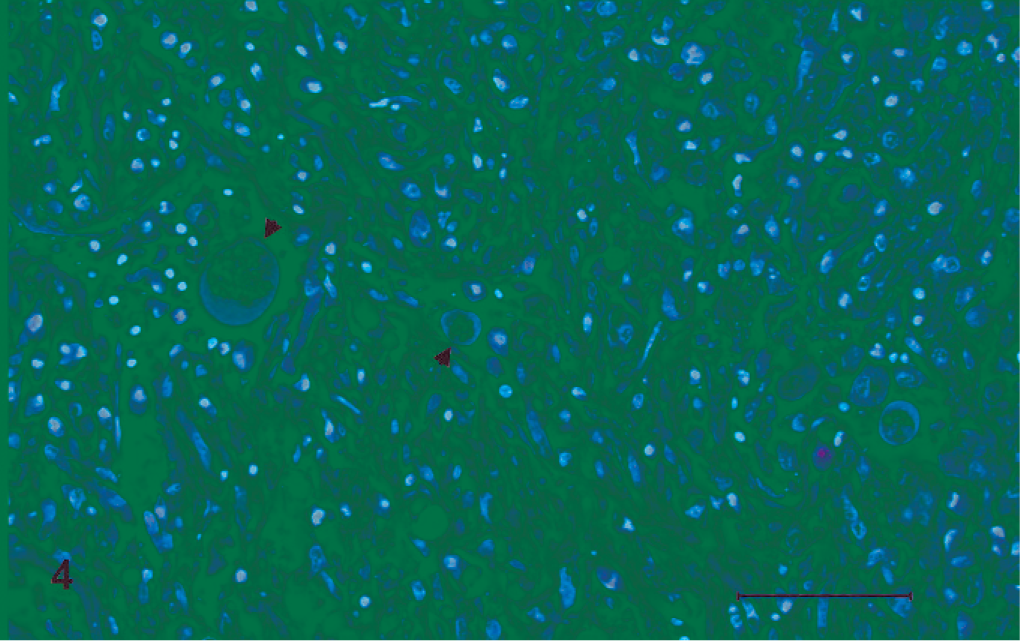
Liver; common water monitor lizard. In the affected area seen grossly, there is extensive necrosis, with some E. invadens present within the portal vein (arrowheads). HE. Bar = 50 µm.
In order to identify the species of Entamoeba, DNA was extracted from eight 5-µm serial sections of paraffin-embedded muscle or liver tissue by DNease Blood & Tissue Kit (Qiagen, Chatsworth, CA, USA) and amplified by polymerase chain reaction (PCR) using the primer set and method as described previously followed by gene sequence analysis. 14 For PCR amplification, the primer set used targeted a 136-bp region of 18S rRNA from various Entamoeba species. The 50 µl of reaction mixture was composed of 14 µl of Go Taq Flexi DNA Polymerase Mix (Promega, Madison, WI, USA), 0.5 µM of each forward and reverse primer, 0.2 mM of each dNTP, 1 µl of template DNA, and 32 µl of distilled water. Because no E. invadens–confirmed positive tissue sample was available, no positive PCR control was included. The negative PCR control contained 1 µl of distilled water instead of template DNA. The PCR amplification was performed using PTC-100 programmable thermal controller (MJ Research, Watertown, MA, USA) started with denaturation at 94°C for 2 minutes, followed by 40 cycles of heat denaturation at 94°C for 30 seconds, primer annealing at 60°C for 30 seconds, and DNA extension at 72°C for 1 minute, and ended with a final extension at 72°C for 5 minutes. Fifteen microliters of each PCR product was electrophoresed in a 2% Tris acetate-EDTA-agarose gel and stained with ethidium-bromide. The amplification product of the predicted size was sequenced in both directions with the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, CA, USA) according to the manufacturer's instructions. A sequence of 136-bp in the primer-binding regions was clearly readable in both directions. When submitted to Basic Local Alignment Search Tool (BLAST, www.ncbi.nlm.gov/blast) against GenBank databases, it showed 98–100% similarity with the gene of E. invadens (accession number AF149905, AY190083, AY769863). Other Entamoeba species showed similarity of less than 87% in this genome region.
To our knowledge, muscular amoebiasis has not been reported in animals. Human cases of amoebiasis with lesion development in the skeletal musculature have also rarely been reported, including one case with a psoas muscle abscess and one case with an unusual amoebic abscess localized in the right hip. 11,15 Hematogenous or lymphatic dissemination and direct spread from perforation of intestinal lesions or rupture of hepatic abscesses into the abdominal cavity were the presumed causes for the unusual muscular involvement in humans. Similarly, parasitemia as a result of hematogenous or lymphatic spread from gastrointestinal tract has been postulated as one of the possible modes of infection for the extremely rare cases of primary cutaneous amoebiasis in human. 13 In the present case, the superficial muscular invasion by amoeba might have resulted from direct invasion through skin wounds by virulent trophozoite spread from contaminated stool. The deeper muscular invasion might develop from 3 possible mechanisms: direct invasion of E. invadens via relatively deeper skin wounds; migration of peritoneal floating trophozoites released from ruptured intestinal and/or liver lesions; or hematogenous or lymphatic spread via systemic amoebiasis secondary to intestinal infection. In the present case, although both small intestine and liver were severely affected, no evidence of intestinal perforation, rupture of hepatic lesions, and peritoneal involvement was revealed grossly. A series of 0.5-cm dissections of the epaxial and tail muscles were made and carefully examined grossly, and no direct connection between the skin wounds and those more deeply located muscular lesions was revealed. In addition, muscular lesions were also found in areas with no detectable wounds in the overlying skin. Amoebic trophozoites could be found in the lumens of portal veins and blood vessels present at the periphery of the affected muscle. These findings strongly suggest that the intestine was the primary site of infection with further spread to the liver via the portal system and that the muscular involvement was possibly the result of a combination of hematogenous spread and direct infection through traumatized skin by trophozoites excreted from the digestive tract.
Fecal-oral route is the primary mode of transmission for E. invadens and accounts for the high incidence in a confined and unsanitary environment. In areas of enzootic infection, a variety of conditions, including overcrowding and inadequate and contaminated water supplies, favor direct fecal-oral transmission of amoeba from one animal to another. 7 It is known that the ingested mature quadrinucleate cysts develop into trophozoites in the intestine, invade the mucosa of gastrointestinal tract and the small blood vessels of submucosa, and then gain access to the superior mesentery and portal system to reach liver. 6 Alternatively, extension via the biliary system is also a possible route for the transportation of amoeba from the intestine to the liver. 6 In the present case, morphologic evidence of blood vessel invasion by amoeba in the liver with no pathologic changes in the biliary system is consistent with a hematogenous spread via portal system to the liver.
Chelonians and crocodilians are considered to be symptomless carriers of E. invadens and may serve as a reservoir for infection in snakes and lizards in captivity. 4 In the intestine of turtles, amoebic protozoa take nourishment from ingested plants to form cysts and complete their life cycle without being pathogenic to the host. 10 The mixing of chelonians and crocodilians with snakes and lizards in the same exhibit or the recirculation of water through separate exhibits can result in outbreaks of amoebiasis in susceptible reptile species. 7 Cockroaches, house flies, and other arthropods may mechanically transfer contaminated feces via their legs or mouthparts to food sources. 8 Chelonians could also be the primary source of amoeba in the present case as the affected lizard was exposed to tortoises for 3 days. Moreover, in experimental infections in snake, E. invadens caused disease at 25°C but not at 10°C or 30°C. 1 This common water monitor lizard was housed in a tank within the temperature range for the optimal growth of amoebae.
Clinically, metronidazole, an antiprotozoan drug, has been used to treat reptilian amoebiasis at a dose of 275 mg/kg body weight by given orally via stomach tube. 2 Metronidazole is the most active against the trophozoites but dose not eliminate all cysts from the gut lumen; however, at high doses it can induce hepatotoxicity with neurologic signs. To prevent amoebic infections in reptiles, strict hygienic measures should be applied among reptilian terraria. Special attention should be paid to avoid cross-contamination between chelonian and lizard housing. 7 Animal caretakers should be aware of the correct sanitation procedures.
In summary, the severe necrotizing amoebic granulomatous enteritis and hepatitis are the primary cause for the death of this lizard. Infection might have been acquired through mature quadrinucleate cysts in chelonian feces-contaminated water and equipment. The widespread muscular amoebiasis possibly resulted from both direct invasion via skin wounds by environmental fresh trophozoites and hematogenous spread.
