Abstract
Despite the profound impact that skeletal muscle disorders may pose for the daily activities of wild terrestrial and marine mammals, such conditions have been rarely described in cetaceans. In this study, the authors aimed to determine the nature and prevalence of skeletal muscle lesions in small and large odontocetes and mysticetes (n = 153) from 19 different species. A macroscopic evaluation of the epaxial muscle mass and a histologic examination of the longissimus dorsi muscle were performed in all cases. The only macroscopically evident change was variable degrees of atrophy of the epaxial muscles (longissimus dorsi, multifidus, spinalis) in emaciated specimens. The histopathological study revealed single or combined morphological changes in 91.5% of the cases. These changes included the following: degenerative lesions (75.2%), muscle atrophy (37.9%), chronic myopathic changes (25.5%), parasitic infestation (9.2%), and myositis (1.9%). The skeletal muscle is easily sampled during a necropsy and provides essential microscopic information that reflects both local and systemic conditions. Thus, skeletal muscle should be systematically sampled, processed, and examined in all stranded cetaceans.
Keywords
Skeletal muscle disorders have been rarely described in cetaceans. These disorders include granulomatous mucormycosis in a killer whale (Orcinus orca); 1 sarcocystosis in several cetacean species (beluga whale, Delphinapterus leucas; Atlantic white-sided dolphin, Lagenorhynchus acutus; long-finned pilot whales, Globicephala melaena; 9,17,28 striped dolphin, Stenella coeruleoalba; 15 sperm whale, Physeter macrocephalus; and northern right whale dolphin, Lissodelphis borealis); 11 and bacterial necrotizing fasciitis and myositis in the captive common bottlenose dolphin (Tursiops truncatus). 97
The different cetacean species exhibit a wide range of body masses. 26,55 However, the distribution of the skeletal muscle is similar in all the species and provides similar tail-stroke power for swimming. The longissimus dorsi is part of the epaxial musculature, which lies along both sides of the vertebral column, and is involved primarily in cetacean locomotion and has been shown to be 1 of the 2 muscles that power the dolphin’s upstroke. 44,63 The impact of skeletal muscle disorders may range from subclinical or non-life-threatening situations (eg, mild intra or interspecific interactions) to grave localized injuries (eg, those caused by ship collision) or disseminated disease processes (eg, infectious myositis), which could impede locomotion and lead to death. The presence of complex polysaccharide inclusions, 78 myopathic changes related to capture myopathy, and ship-strike-associated muscle lesions have been previously reported in the populations studied here. 42,43,76 Age-related histological findings, including variations in the number and the size of different types of myofibers, have also been described in this group of animals. 75 In addition, the presence of sarcoplasmic masses within the skeletal muscle of a stranded pygmy sperm whale (Kogia breviceps) have been previously published. 74
Given the scarcity of reports on the muscle pathology of these species and the wide range of muscle lesions encountered in the stranded cetaceans of our study, this article aimed to determine the nature and prevalence of skeletal muscle lesions in small and large odontocetes and mysticetes stranded on the Canary Islands, Spain.
Material and Methods
Stranding Epidemiologic Data and Necropsy Examination
Animals included in this study were stranded cetaceans on the Canary Islands, except for 5 animals from other locations. The specimens included in this study are summarized in Table 1 (individual case details are given in Table S1 in the supplemental material).
Muscle Pathology in Stranded Cetaceans (1996–2013): Number of Cases by Species in the Different Etiological Categories.
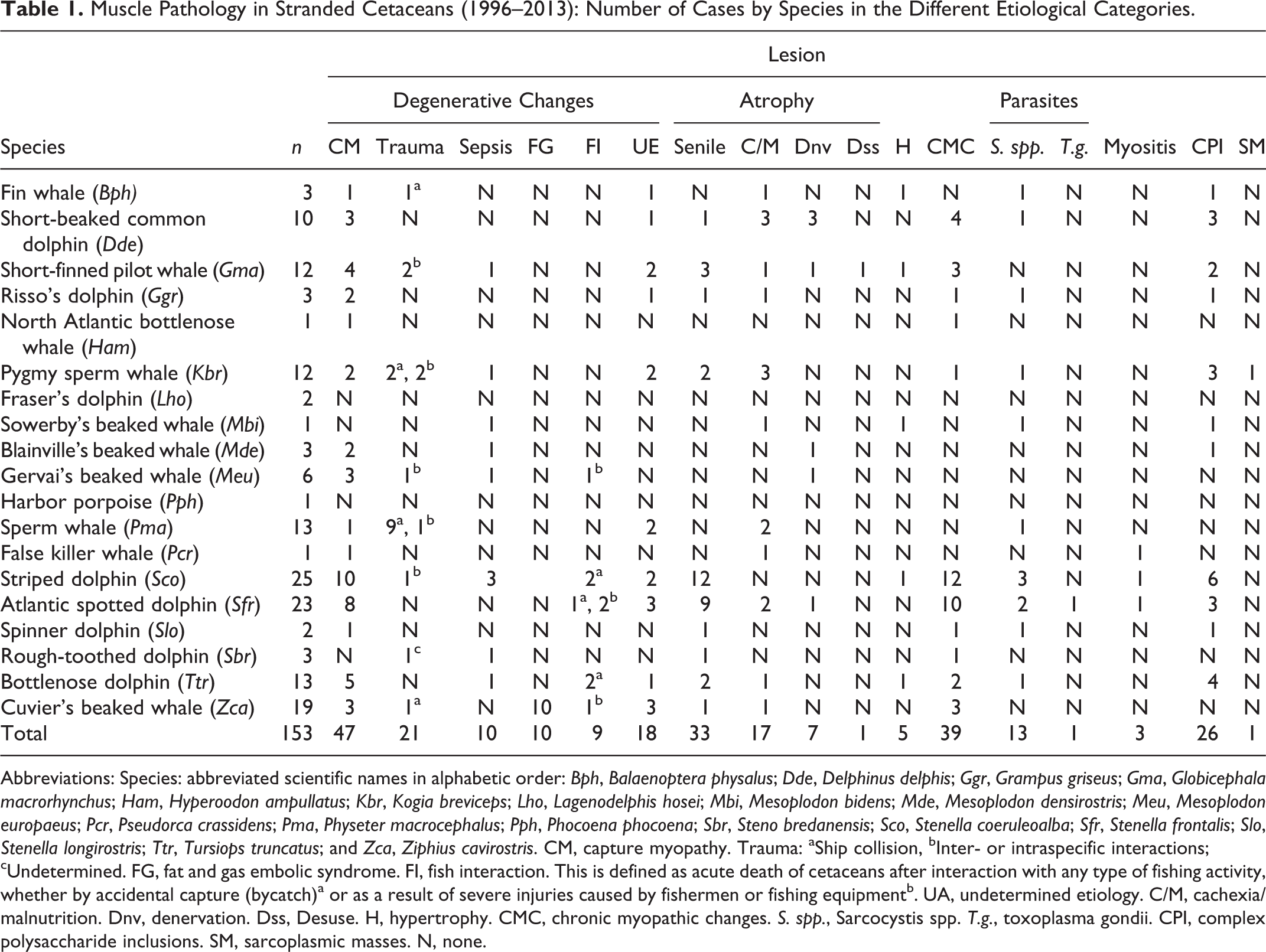
Abbreviations: Species: abbreviated scientific names in alphabetic order: Bph, Balaenoptera physalus; Dde, Delphinus delphis; Ggr, Grampus griseus; Gma, Globicephala macrorhynchus; Ham, Hyperoodon ampullatus; Kbr, Kogia breviceps; Lho, Lagenodelphis hosei; Mbi, Mesoplodon bidens; Mde, Mesoplodon densirostris; Meu, Mesoplodon europaeus; Pcr, Pseudorca crassidens; Pma, Physeter macrocephalus; Pph, Phocoena phocoena; Sbr, Steno bredanensis; Sco, Stenella coeruleoalba; Sfr, Stenella frontalis; Slo, Stenella longirostris; Ttr, Tursiops truncatus; and Zca, Ziphius cavirostris. CM, capture myopathy. Trauma: aShip collision, bInter- or intraspecific interactions; cUndetermined. FG, fat and gas embolic syndrome. FI, fish interaction. This is defined as acute death of cetaceans after interaction with any type of fishing activity, whether by accidental capture (bycatch)a or as a result of severe injuries caused by fishermen or fishing equipmentb. UA, undetermined etiology. C/M, cachexia/malnutrition. Dnv, denervation. Dss, Desuse. H, hypertrophy. CMC, chronic myopathic changes. S. spp., Sarcocystis spp. T.g., toxoplasma gondii. CPI, complex polysaccharide inclusions. SM, sarcoplasmic masses. N, none.
Skeletal muscle samples from stranded odontocetes and mysticetes (n = 153), collected over a 14-year period (1996 to 2013), were examined. The animals were of both sexes and ranged in age from neonatal to mature adults, as determined by biological and morphometric parameters. The skeletal muscle samples were collected during necropsies, following standard protocols. 53 Necropsies were performed either on site (the beach or coast) (17.6%), in facilities built specifically for this purpose (33.3%), or when possible, in the necropsy room at the Veterinary College of the University of Las Palmas of Gran Canaria (Spain) (29.4%). Large cetaceans necropsies were also frequently performed at the rubbish dump of the corresponding island (19.6%). Permission for the management of the stranded dead cetaceans was issued by the environmental department of the Canary Islands government.
The preservation status of the carcasses was established according to Geraci and Lounsbury. 35 The body condition of each animal was estimated morphologically based on the prominence of the spinous and transverse vertebral processes, the mass of the epaxial musculature (longissimus dorsi, multifidus, spinalis), and the absence or limited presence of subcutaneous fat deposits. 51 These estimates were made taking into account the species and age of the animals. Using these parameters, we classified their body condition as good, moderate, poor or emaciated.
The muscle examination included a macroscopic evaluation of the epaxial and hypaxial muscle mass based on transverse and longitudinal sections throughout the entire muscle body that were approximately 1–2 cm apart.
Histopathology and histochemistry
All skeletal muscle samples were taken from the region of the longissimus dorsi muscle immediately lateral to the dorsal fin as previously described. 62,84 Samples from other skeletal muscle tissue that displayed grossly visible alterations were also collected for histopathology.
The samples were mounted on a tongue depressor, fixed at both ends by pins with the myofibers oriented lengthwise, and immersed in 4% neutral-buffered formalin for 24–48 hours. Transverse and longitudinal muscle sections were routinely processed, embedded in paraffin, serially sectioned and stained with hematoxylin and eosin (HE), periodic acid-Schiff (PAS) with and without amylase digestion, phosphotungstic acid-hematoxylin (PTAH), osmium tetroxide (OsO4), and von Kossa staining as previously described. 2,4,6,34,52
Histological assessments of the longissimus dorsi muscle samples were obtained from all animals and were performed in a blind manner by 3 veterinary pathologists (ES, AEM, PH).
Diagnostic criteria for degenerative changes included segmental hypercontraction (characterized by a hyperacidophilic sarcoplasm and endomysial edema) and/or degeneration/necrosis (with a hyaline, floccular, granular, and/or discoid appearance), contraction band necrosis, presence of globules in the sarcolemmal tube and/or under the basal lamina, an inflammatory response, and fiber regeneration. Lesions were confirmed as ante mortem when accompanied by histochemical demonstration of a loss of myofibril striations (affected fibers failed to show the normal dark-blue coloration after PTAH staining and stain pale blue, yellow, or pink instead), and myoglobin leakage and intrafibrillar fibrinogen immunolabeling. The degree of skeletal myodegeneration was subjectively judged as follows: mild, when scattered individual fibers distributed randomly throughout the section were detected; moderate, when multiple foci of affected fibers were distributed randomly through the section; or severe, when multiple, large, widespread, often coalescing areas of myodegeneration were detected.
Evaluation criteria for myofiber atrophy/hypertrophy included an assessment of muscle fiber size and morphology. Atrophic fibers usually displayed decreased sizes and angular shapes, while hypertrophic fibers tended to have increased sizes and more rounded contours along with internal disorganized myofibrils. Determination of the fiber types involved and any alterations in the distribution of the fibers required immunohistochemical fiber typing in each case. This analysis required the previous knowledge of the normal histology of the specimens in relation to their different diving behaviors and their different ages. Variations in fiber type composition among marine mammals have been proposed to result from differences in routine dive duration and swimming speed, as well as from age. 16,75,77
Intramyofiber inclusions/deposits were identified with the HE and PAS stains. PAS-positive, diastase-resistant inclusions within skeletal muscles are consistent with deposits of abnormal glycogen, often referred to as a complex polysaccharide. 85 Intramyofiber vacuolations diagnoses were based on the presence of various, small, clear vacuoles within the sarcoplasm of the myofibers as shown in HE-stained sections.
Chronic myopathic changes (CMCs) were determined based on excessive fiber size variation, increases in the number of internal nuclei, and intramyofiber pigment deposits, which were identified by the presence of large amounts of juxtanuclear brownish pigmentation, which is a pattern of PAS staining that has been show to be compatible with lipofuscinosis.
Ring fibers were identified as cytoarchitectural alterations consisting of peripheral rings around maloriented myofilaments.
Myositis was determined based on the presence of focal/multifocal areas of inflammatory infiltrates (mainly macrophages and polymorphonuclear leucocytes).
Intramuscular parasites were identified by a combination of HE, PAS, and PTAH stains.
Immunohistochemistry
Fast and slow myosin heavy chain (MHC) isoforms, indicating type II and type I fibers, respectively, were detected using monoclonal mouse antihuman myosin heavy-chain isoform type I (slow twitch) (skeletal, slow, M8421) and type II (fast twitch) (skeletal, fast, M4276) antibodies (Sigma Co, St. Louis, MO, USA), and myoglobin and fibrinogen were detected using polyclonal rabbit antihuman myoglobin (A0324) and fibrinogen (A0080) (Dako, Glostrup, Denmark). Antibodies were visualized using the avidin-biotin-peroxidase method (Vector Laboratories, Burlingame, CA, USA) as previously described. 42 In addition, polyclonal anti–Toxoplasma gondii (Dako, Glostrup, Denmark) diluted 1:200 with a previous pronase treatment (5 min) was used to confirm histopathological diagnoses of toxoplasmosis. Tissue sections in which the primary antibodies were replaced by phosphate buffered saline or nonimmune serum were used as negative controls to confirm the specificity of the tests. 65 Goat and human tissues were used as positive controls.
Electron microscopy
For ultrastructural studies, skeletal muscle samples were fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.2). The specimens were postfixed in 1% osmium tetroxide in 0.2% veronal buffer, gradually dehydrated in an alcohol series, and embedded in EMbed 812 epoxy resin (Electron Microscopic Science, Hatfield, PA, USA). Thin sections were stained with uranyl acetate and lead citrate. The sections were examined with a ZEISS EM-912 transmission electron microscope (Carl Zeiss, Oberkochen, Germany).
Myofiber sizes comparisons
Myofiber size measurements were performed in a selected group of animals from our study (n = 39). For morphometric analysis, 10 randomly selected fields/images per slide (200X magnification) of a muscle section that was immunohistochemically stained for antifast myosin were captured using an Altra20 digital camera (2 MegaPixel CMOS color camera for light microscopy, Olympus Soft Imaging Solutions GmbH, Münster, Germany). The cross-sectional area (CSA) and the lesser diameter (the greatest distance between the opposite sides of the narrowest aspect of the fiber) of all the fibers in a field with red-stained fast-twitch muscle fibers (type II) and blue-stained slow-twitch muscle fibers (type I), were measured. The relative number of type I and type II fibers was assessed by counting each fiber type from the 10 randomly selected fields of each animal. The percentage of each specific fiber type was calculated as the specific fiber count divided by the total fiber count. The muscle morphology data (CSA, lesser diameter and number of fiber types) was calculated using the digital software CellA (Imaging Solutions). The mean values were obtained using the Soft Imaging System for Life Science Microscopy.
Interpretation/analysis
The combination of the histopathologic findings and data regarding the age, diving profile, type of stranding, and the cause of death for each case (hereafter referred as the necropsy report) 5 were used to establish an etiological diagnosis of muscle pathology in the current study. In addition, fiber size measurements were taken into consideration for the diagnosis of myofiber atrophy in selected cases, including 25 animals with a morphological diagnosis of focal/multifocal or generalized myofiber atrophy and 14 specimens with no myofiber size changes.
Results
A varied and often compound set of morphophysiological and pathological changes were microscopically identified in 140 of the 153 (91.5%) animals (these data are summarized in Table 1, while full descriptions are available in Table S1 in the supplemental material). The percentage of animals with 1, 2, 3, 4, or 5 histopathologic features were 49.3%, 19.3%, 17.9%, 7.1%, and 2.1%, respectively. Some lesions tended to occur together: chronic myopathic changes were frequently present in animals with senile or denervation muscle atrophy (82.9%), while 69.2% and 46.15% of the animals with complex polysaccharide inclusions displayed at least 1 of the muscle atrophies described in our study or had degenerative changes associated to stranding stress syndrome, respectively.
The only macroscopically visible muscle change among the animals evaluated was atrophy, which was more evident along the epaxial musculature than in the muscles of the thoracic wall. Several degrees of atrophy, ranging from mild to a profound reduction in muscle mass with marked prominence of the adjacent axial osseous relieves (ribs, transverse and spinous vertebral apophyses) were observed in 41 of the 153 (Supplemental Table S2) animals. Generalized, focal, or multifocal myofiber atrophy was observed in 58 of the 153 (37.9%) animals, although only cases of emaciation corresponded with generalized myofiber atrophy when examined by light microscope.
Myofiber Atrophy/Hypertrophy
The histologic features (fiber size and morphology, and distribution pattern), based on the available epidemiological data, were combined to classify myofiber atrophies as senile atrophy, cachexia/inanition atrophy, denervation atrophy, and disuse atrophy.
Senile atrophy represented 56.9% of the cases of atrophy and was detected in 33 animals (old adults) belonging to 10 different species. Most of the animals presented a moderate-good overall body condition. However, histologically, all of the animals in this group were morphologically diagnosed with generalized muscle atrophy of type II fibers, while type I fibers were enlarged compared with those of juvenile and young adult specimens. No remarkable morphological changes were observed within the atrophic myofibers. Cytoplasmic accumulation of PAS-positive pigment, indicating lipofuscin, within myofibers alone or in combination with CMCs was observed in 54.4% and 33.3% of the cases, respectively.
Cachexia/inanition atrophy represented 29.3% of the cases of atrophy. Similar to the patterns observed with senile atrophy, all the animals in this group showed generalized myofiber atrophy at the microscopic level. However, a severe wasting of the epaxial musculature was only determined in this category based on gross examination, from which 14 of the 17 (82.4%) animals showed evidence of a consumptive pathological process. In 4 cases, foreign bodies lodged in the mouth, tongue or stomachs in the affected specimens were responsible for their condition. Histologically, generalized muscle atrophy, primarily of type II fibers, was noted. In addition, atrophied myofibers showed evidence of variable degrees of flattening and angular shape changes (Fig. 1). Occasional irregular shapes were also noticed.
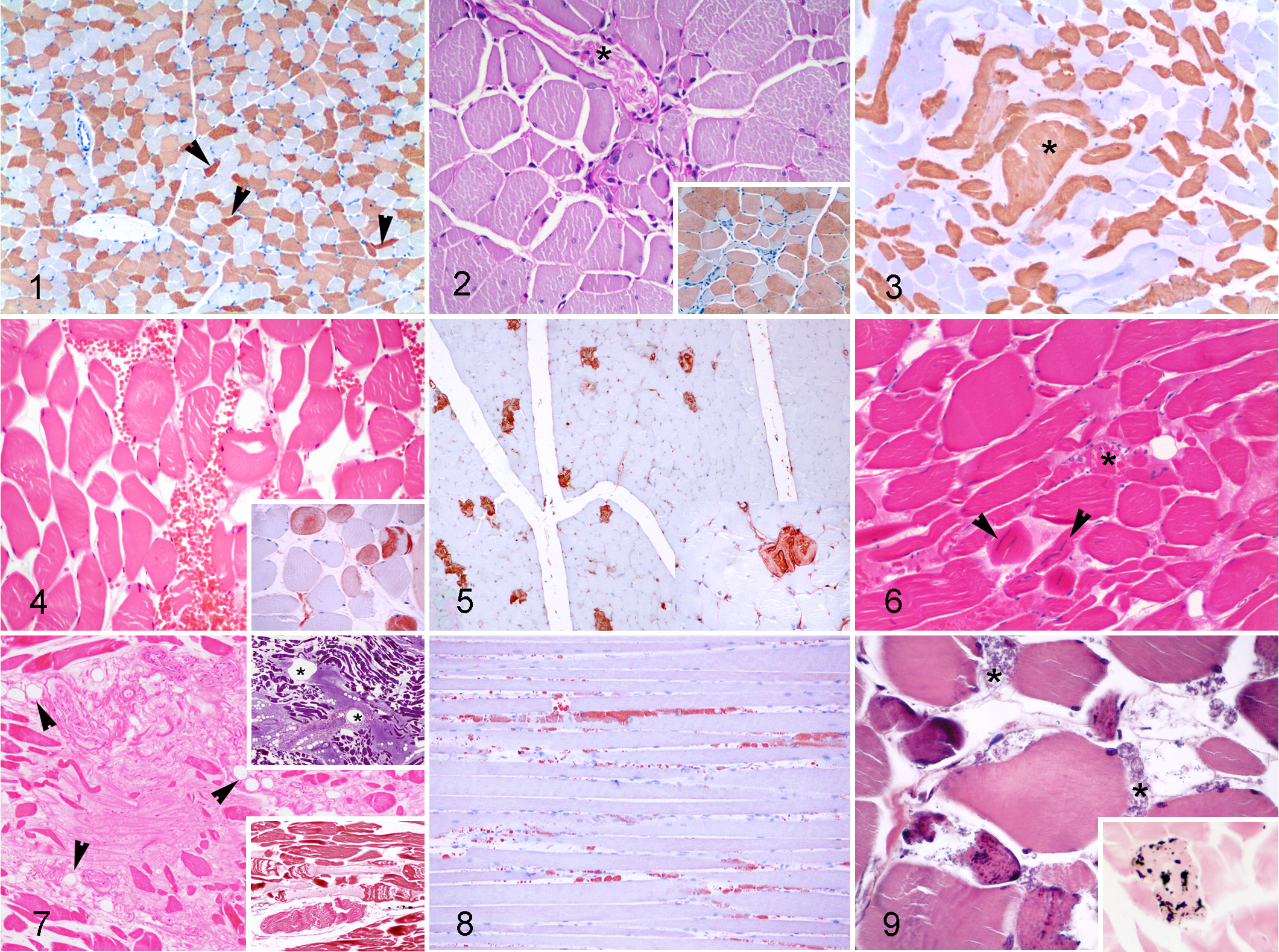
Denervation atrophy represented 16.7% of the atrophies diagnosed. The morphologic diagnosis in this group was multifocal myofiber atrophy, frequently adjacent to an intramuscular nerve (Fig. 2). The affected fibers were of both types, and fiber type grouping (the clustering of a specific fiber type instead a random distribution) was common (Fig. 2, inset). Focal concentration of myofiber nuclei was a specific feature of this type of muscle atrophy. More than half of the specimens in this group were considered senile based on biological and morphometrical parameters. Intramuscular nerves in this group showed an increase in perineurial and epineurial connective tissue and decreased myelin, as shown by osmium tetroxide postfixation staining, when compared to other cases.
Disuse atrophy was diagnosed in 1 specimen; an adult female short-finned pilot whale (Globicephala macrorhynchus) that had severe lateral column deviation (scoliosis). Histologically, there were diffuse atrophic fibers of both types, as well as a slight enlargement of the endomysial fibrous stroma.
Presumed compensatory hypertrophy was observed in 5 specimens with myofiber atrophy regardless of the etiology (Fig. 3). Hypertrophied myofibers usually showed longitudinal fiber splitting (muscle fiber divisions).
Degenerative Muscle Changes
Degenerative muscle changes (DMCs) were the most frequently identified lesions (115/153; 75.2%).
Capture myopathy in active-stranded animals, which died shortly after the stranding or after a period of rehabilitation, was the most commonly identified underlying disease associated with acute skeletal rhabdomyolysis in our study (40.9%). 42,43
Physical trauma was the second most frequent etiological diagnosis (18.3%) associated with active degenerative muscle lesions in our study. Hematomas and hemorrhages were frequently observed upon gross examination of traumatized muscle areas, which correlated with histological diagnoses of hemorrhages and acute monophasic myonecrosis affecting both types of myofibers (Fig. 4). Affected myofibers often displayed a hyaline and/or a floccular appearance, contraction band necrosis, fragmentation of myofibers and discoid degeneration. Discoid degeneration was exclusively observed in animals in which sharp trauma from ship strikes was the cause of death.
Systemic infection or septicemia was the third most common cause of myonecrosis (8.7%) in our study and was found in 8 different species (Table 2). The related histological muscle findings ranged from scattered acute single-myocyte degeneration and necrosis (Fig. 5) to, more frequently, diffuse and polyphasic myocyte degeneration and necrosis (Fig. 6). Regenerating myofibers were commonly encountered along with acute degenerative changes. An inflammatory reaction, mainly composed of reactive and phagocytizing macrophages, was frequently observed within the affected areas.
Muscle Degenerative Changes Associated With Systemic Infection or Septicemia.
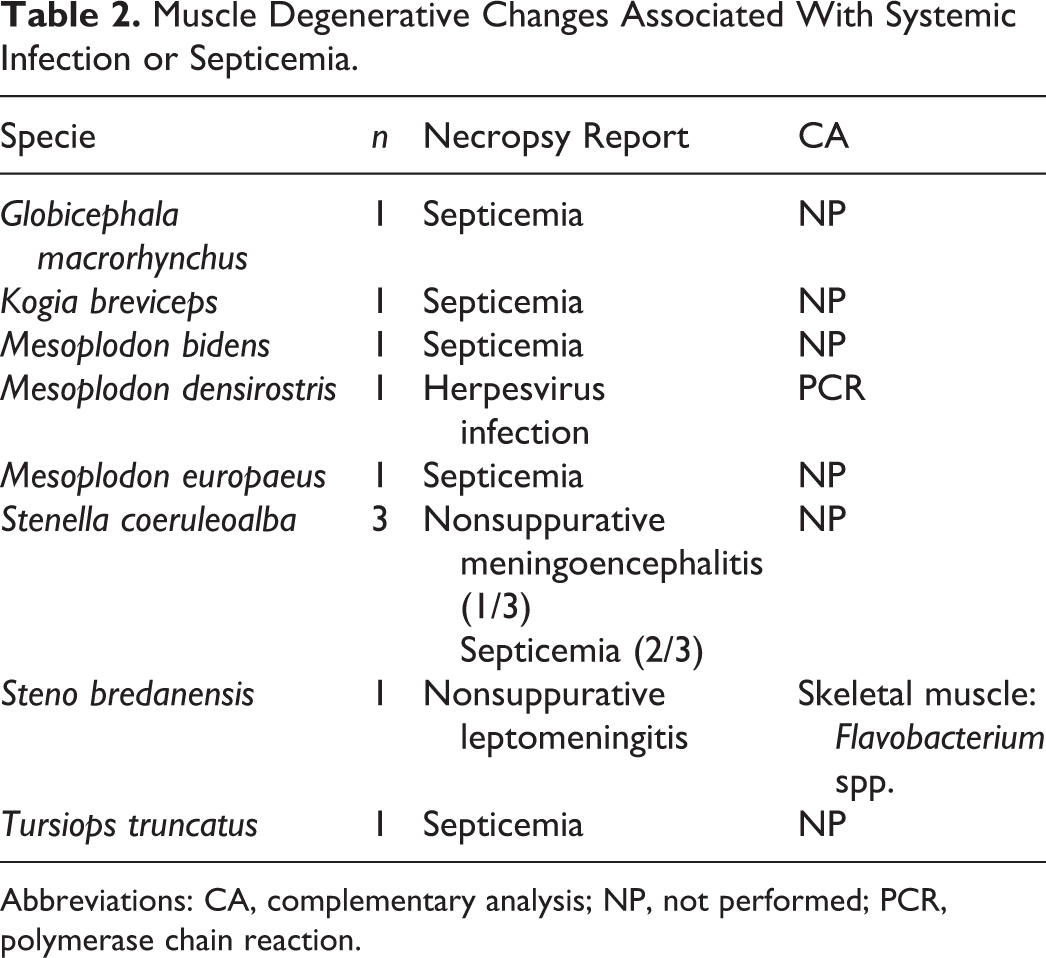
Abbreviations: CA, complementary analysis; NP, not performed; PCR, polymerase chain reaction.
Other causes associated with DMCs in the present study included fat and gas embolism syndrome (8.7%) and interactions with fishing activities (7.8%).
Those cases with an etiological diagnosis of fat and gas embolism as the cause of stranding/death not only displayed degenerative changes in their muscle fibers (acute monophasic myonecrosis) but also in the interstitial connective tissue and intramuscular nerves. Fibers in advanced stages of degeneration frequently appeared as hyaline-filled sarcolemmal sheaths, and the interstitial connective tissue was replaced by similarly large amounts of amorphous hyaline material (Fig. 7). Intraneural and intermyofiber edema was a consistent finding in this group of animals.
An interaction with fishing activities was considered to be responsible for DMCs in 9 individuals, including 5 different species. In these cases, entanglement (55.6%) and/or a lethal interaction with any other fishing gear (44.4%) was determined as the cause of death. Entanglement caused milder and more discrete histopathological muscle lesions than any other fishing-related trauma. Moreover, altered sarcolemmal permeability, evidenced by large amounts of myoglobin-positive globules between the unaffected segments of the myofibers, was a common finding with both of these causes (Fig. 8).
Degenerative muscle lesions of undetermined etiology represented 15.6% of the cases and were found in 18 specimens of 10 different species. Despite the heterogeneity of this group, some common features were shared among some animals. Notably, 3 animals in this group showed acute degenerative changes that primarily affected type I fibers, which were similar to those observed in capture myopathy (Fig. 9). Calcification (von Kossa-positive) of a necrotic myofiber was noted in 1 of these 3 individuals (the only detected case of calcification in the present study) (Fig. 9, inset). Interestingly, 9 of the 18 muscle samples in this group showed additional histological features compatible with muscle atrophy. In the remaining 6 animals, a cause for the DMCs was not apparent even thought the changes were severe in some cases.
Degenerative muscle lesions typically showed immunohistochemical depletion of the sarcoplasmic myoglobin coupled with intra and extracellular globular accumulation of myoglobin and sarcoplasmic fibrinogen deposition (all of these strongly support an antemortem degeneration).
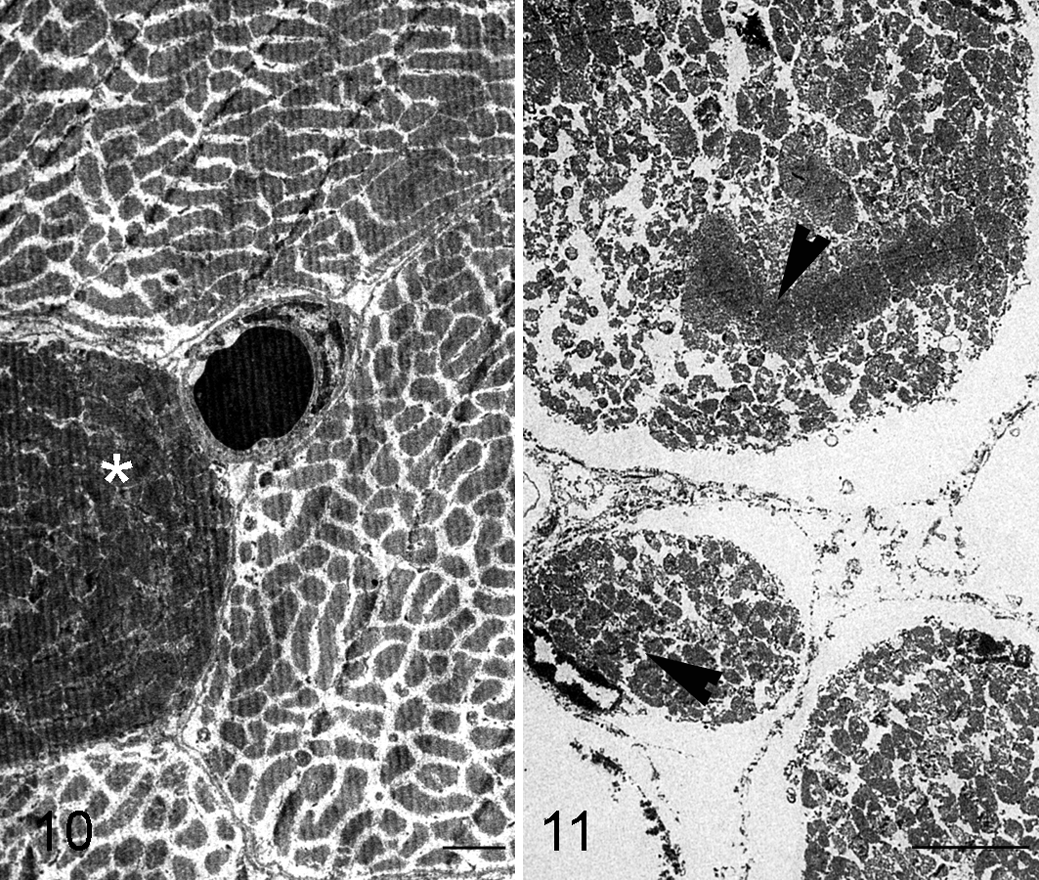
Some of these changes were confirmed by the electron microscopy study. The ultrastructural changes depended on the stage of the degeneration process, and included the formation of contraction band necrosis, fragmentation of myofibrils, mitochondrial swelling, and the disappearance of the myofibrillar structure in necrotic fibers (Figs. 10, 11).
Chronic Myopathic Changes
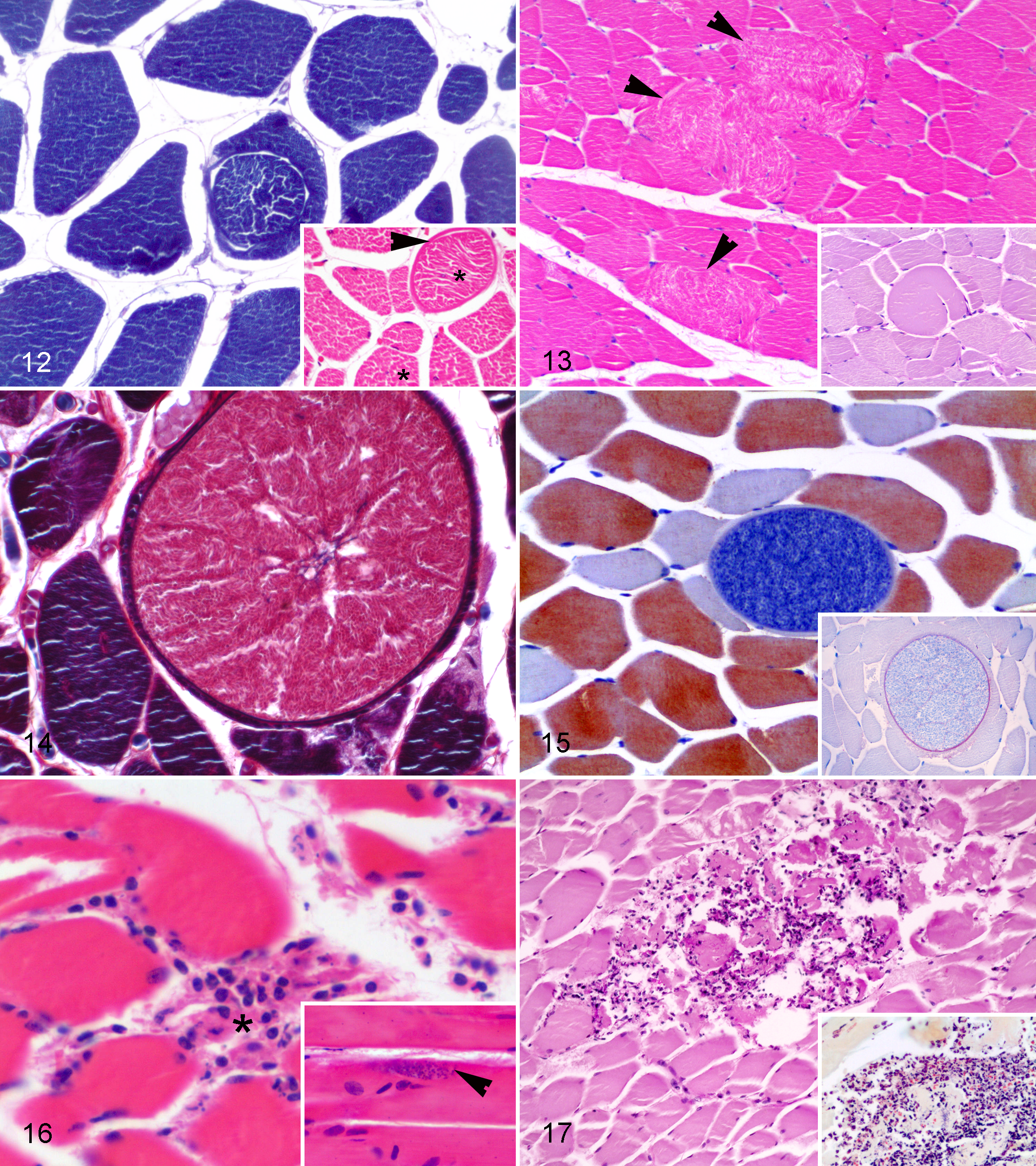
Chronic myopathic changes, detected in 39 of the 153 (25.5%) animals, included ring fibers (Fig. 12), increased fiber size variation (Fig. 13), longitudinal splitting in hypertrophied fibers (Fig. 13, inset), and the presence of juxtanuclear lipofuscin.
Complex Polysaccharide Inclusions
Complex polysaccharide inclusions, as previously reported, 78 were observed in 26 specimens representing 11 different species. An association was established with histological skeletal muscle lesions that was compatible with rhabdomyolysis, and generalized or multifocal muscular atrophy in 20 and 12 of the 26 animals, respectively.
Parasitic Infestation
Parasitic infestation of skeletal muscle was observed in 14 of the 153 (9.2%) animals, including Sarcocystis spp. (8.8%) and Toxoplasma gondii (0.7%).
Sarcocystis spp. were detected in at least 1 specimen of 10 different species: fin whale (Balaenoptera physalus), short-beaked common dolphin (Delphinus delphis), Risso’s dolphin (Grampus griseus), pygmy sperm whale, Sowerby’s beaked whale (Mesoplodon bidens), sperm whale, striped dolphin, Atlantic spotted dolphin (Stenella frontalis), spinner dolphin (Stenella longirostris), and bottlenose dolphin. Two species were represented by more than 1 individual: the striped dolphin (n = 3) and the Atlantic spotted dolphin (n = 2). Histologically, the protozoal cysts appeared round to oval and were located within myofibers. There was no detectable inflammation in any case (Figs. 14–15). Cysts contained metrocytes or bradyzoites and were surrounded by a fibrinogen-positive wall (Fig. 15, inset). Cysts varied in size, ranging from 43.5 µm × 28.3 µm to 113 µm × 121.7 µm.
Muscle parasitization by T. gondii was only detected in an adult Atlantic spotted dolphin. The protozoal cysts were intracellular, affecting the myofibers and intramuscular nerves. They were typically ovoid-shaped and approximately 8.6 µm × 8.6 µm size (Fig. 16). They stained positive with PAS and showed positive immunolabeling with a specific monoclonal antibody. Moderate interstitial lymphohistiocytic myositis was associated with these zoites.
Myositis
Myositis was detected in 3 of the 153 (1.9%) animals. All of these cases showed multifocal pyogranulomatous inflammation associated with a systemic inflammatory process of bacterial origin (Fig. 17). Nocardia farcinica were isolated from the lung tissue of an adult striped dolphin and Staphylococcus spp from the pulmonary tissue of an Atlantic spotted dolphin. In the remaining case, a juvenile false killer whale (Pseudorca crassidens), the etiologic agent was not determined, although a penetrating wound was observed in the adjacent skin and subcutis suggested a secondary bacterial infection.
Sarcoplasmic Masses
The presence of sarcoplasmic masses within skeletal myocytes in an adult pygmy sperm whale has been previously reported. 74 Other histopathologic muscle changes in that case included a high number of internal nuclei, variations in fiber size and shape, and a predominance of type I fibers compared to that in the control group.
Discussion
Very little information regarding muscle pathology in marine mammals was available at the time of this study. Our results indicate that muscle lesions are more common in cetaceans (92.2%) than in other domestic and wild species based on the small amount of published data in terrestrial mammals. 81,86 In a postmortem study in equids, 65% of the analyzed specimens showed muscle lesions of 1 or more type at necropsy, 86 excluding the polysaccharide storage myopathy, which was previously reported on the same population (44.1%). 88 In equids, myonecrosis was the most common finding (17%) followed by chronic myopathic changes (15.7%), generalized muscle atrophy (13.1%), intramyofiber protozoa (8.3%), denervation atrophy (6.1%), and neoplasia (1.3%), as well as other conditions. 86 In wild animals, a high incidence of muscle lesions have been previously described in the pichi (Zaedyus pichiy) (76.5%). These lesions were mainly due to rhabdomyolysis (66.9%), Sarcocystis parasitization (10.3%), and inflammation (6.2%). 81
Determining the cause of muscle injury based exclusively on gross and/or microscopic findings is seldom possible. Additional tests and life histories are often required. However, this is particularly difficult in free-ranging or stranded cetaceans, where their life history is frequently lacking and only necropsy data are available. In our study, the muscle condition classification and establishment of a correlation with an etiological diagnosis for the stranding/death were deemed necessary in each case. This allowed us to better understand the underlying pathogenic mechanisms of muscle pathology, as well as the pathological significance and the repercussions of muscle lesions in these marine mammals.
Degenerative Muscle Changes
Degenerative muscle change was the most common finding in our study (75.2%), as was the case in the 2 previous terrestrial species. 81,86 However, rhabdomyolysis is multifactorial in origin, and possible causes include sepsis, muscle compression, seizures, trauma, hypothermia, and severe exertion. 33
In our study, degenerative muscle lesions were observed in association with a variety of disorders, including capture myopathy syndrome, 42,43 trauma, septicemia, gas- and fat-embolism syndrome and interactions with fishery activities.
Active muscle degeneration was mainly associated with a stressful situation in 47 of the active-stranded cetaceans (40.9%), as has been previously reported. 42 In addition to the degenerative lesions observed in the skeletal muscles, this group of animals also displayed hemodynamic lesions indicative of multiorgan vascular shock, degenerative muscle lesions affecting cardiac muscles and myoglobinuric nephrosis typical of capture myopathy (CM). 42 Terrestrial and marine wildlife experience CM, often with fatal consequences, after human capture/rescue interactions, as described previously, 42,80 and, in the case of active-stranded cetaceans, stress-associated myopathy contributes to the comparatively low success rate of rehabilitation. 68,96
Degenerative muscle changes related to traumatic processes (18.3%) were mainly attributed to inter/intraspecific interactions and to ship collisions, 76 representing 33.3% and 61.9% of the cases, respectively. The first category refers to animals with gross lesions that varied from superficial parallel skin marks, due to inter/intraspecific interaction, to severe traumatic lesions represented by fractured ribs and subcutaneous and pulmonary hemorrhages. 5 The second category includes animals that suffered severe trauma that resulted in peracute or acute death associated with a collision with a ship. The cause of trauma was not determined in the remaining 4.7% of the traumatized cetaceans with skeletal muscle degeneration in our study. Interstitial edema, hemorrhages and severe, diffuse, acute monophasic myonecrosis were the main histopathological findings in this group of animals, although the major and more distinguishable features were discoid degeneration or fragmentation of the myofibers. This type of myodegeneration was observed predominantly in the cetaceans in which the sharp trauma was deep enough to cause evisceration or bone fractures. 76 An acute stress response was mainly, although not exclusively, implicated in the pathogenesis of DMCs in this physically traumatized group of animals. 71,72 The typical stress response is activated after a wounding or painful trauma. 18,37 Although physiological stress has some benefits, an extreme or prolonged response with the continued release of catecholamines and sustained, elevated blood cortisol and aldosterone levels are potentially damaging to the heart and the skeletal muscle. 10,20 In addition to stress, rhabdomyolysis is a recognized complication of traumatic injury, 25,45 in which ischemia, reperfusion and/or vascular damage are the main pathophysiological mechanisms underlying muscle damage. 92
Degenerative muscle changes related to infectious processes (8.7%) were characterized by scattered single-cell necrosis in our study. Myodegenerative changes associated with a systemic infection or septicemia have been previously reported in humans and animals 33,41 and have been attributed to direct muscle invasion by the pathogens, direct damage to muscle fibers by endotoxins, and/or the increased levels of inflammatory mediators, such as tumor necrosis factor alpha (TNF-α). 54 Acute, multifocal degenerative changes were also present in muscle samples from animals where gas- and fat-embolism syndrome was the cause of stranding/death in our study. Animals with gas- and fat-embolism syndrome included 10 Cuvier’s beaked whales (Ziphius cavirostris) that had evidence of lesions consistent with a severe decompression process associated with the use of sonar during military maneuvers. 29,30 Diffuse, mild to moderate acute monophasic myonecrosis (hyaline degeneration) was a common feature in this group of animals, although additional pathological findings included the “disintegration” of interstitial connective tissue and related structures (intramuscular nerves, adipose tissue, and/or intrafusal muscle fibers) and their replacement by large amount of amorphous hyaline material, which was assumed to be degraded material. A previous association between arterial gas embolism, decompression sickness, and rhabdomyolysis (as suggested by elevated levels of plasma creatine kinase) has been reported. 64,73,79 Although the mechanisms leading to the release of muscle enzymes after arterial gas embolism remain unclear, it has been suggested that gas emboli entering the systemic circulation and reaching the skeletal muscle is the most plausible explanation for the muscle hypoxic and ischemic damage that occurs in the muscle. Decompression sickness is the result of the supersaturation of body tissues with nitrogen gas and the subsequent release of nitrogen bubbles to the bloodstream. The amount of gas dissolved in specific tissues depends on the dive depth and duration, the descent and ascent rates, the lipid content of the tissue, and time at the surface between successive dives. 31 Tissues with a high lipid content can take up a larger amount of nitrogen because of its higher solubility in lipids than in any other biological tissue. 36 A variable fatty component in the interstitial connective tissue has been observed in the deep-diving cetaceans that were examined in this study (mainly pygmy sperm whales, short-finned pilot whales, sperm whales, and the Ziphiidae family) compared with that of other species. 77 The disruption of interstitial fat depots may result in the release of nitrogen gas bubbles from supersaturated tissues and could explain the acute muscle degenerative lesions observed in this group of animals when suffering from decompression sickness.
An interaction with fishing activities (ie, accidental capture [bycatch], and/or severe injuries directly caused by fishermen or fishing equipment) was the etiological diagnosis in 9 cetaceans with a morphological diagnosis of skeletal muscle degeneration (7.8%). In our study, although no distinctive degenerative findings were detected in this group of animals, the leakage of myoglobin strongly supports myofiber damage. Rhabdomyolysis can be defined as the disruption of skeletal muscle leading to leakage of intracellular muscle constituents, especially myoglobin, into the extracellular fluid. 45,50 It has been proposed that tuna purse-seine and net fishery operations could elicit short-term responses in dolphins, including severe muscle damage, resulting in a condition similar to CM, and hyperthermia. 14,94 Capture myopathy is a multifactorial, pathophysiological syndrome associated with stress, excessive muscular activity, heat, trauma, prolonged muscle compression, restraint and being transported. In by-caught animals, some of these mechanisms could contribute to the muscle damage. Despite the fact that there is currently not full descriptions of skeletal muscle disorders in net-caught cetaceans, signs ranging from minor temporary effects (eg, muscle stiffness) to massive muscle damage and death have been sporadically described in some individual cases. 66 In addition, net-caught cetaceans probably share common behaviors, such as extreme and desperate struggles to escape, which often causes severe trauma and eventual death by asphyxiation. Related to this, near-drowning in humans has been shown to induce rhabdomyolysis, and the potential causes include hypothermia, physical exercise and tissue hypoxia. 8
The cause of myonecrosis was not determined in 18 individuals, although CM syndrome was suspected in 3 cases. Despite the fact that these cases were found dead-stranded, the fresh state of the carcasses along with multiple cutaneous erosions, ulcerations and/or abrasions on the head of all or some of these animals, strongly suggested an active stranding in all cases. Histological features of muscle atrophy were observed in 50% of the animals in which the cause of myonecrosis was not apparent.
One exception for myonecrosis in cetaceans from our study, when compared with myonecrosis in other species, is that post necrotic calcification (dystrophic mineralization) was infrequent. Myofiber mineralization is considered a nonspecific sequela of myofiber necrosis, in which diverse circumstances are involved. 91 Since overt mineralization was rarely observed within the injured skeletal muscle from the stranded cetaceans in our study, the causes of this difference require further investigation.
Stress associated with capture and handling and endotoxic injury from bacterial infection were the main underlying causes of rhabdomyolysis in wild pichis and equids, respectively. 81,86,90 However, although rhabdomyolysis may be caused by a wide range of mechanisms, it is always characterized by a loss of muscle integrity and subsequent leakage of intracellular enzymes and electrolytes into the bloodstream, which can be life-threatening. 83 Therefore, regardless of the etiology and depending on the severity of the lesions, rhabdomyolysis can adversely affect the health status of the affected cetaceans.
Muscular Atrophy/Hypertrophy
Generalized muscle atrophy was related to senescence in 56.9% of the cases with a morphological diagnosis of muscle atrophy. This group of animals was considered “senile” according to the available epidemiological data. Loss of skeletal muscle mass can occur during aging (sarcopenia) in humans and animals, including cetaceans, as we have previously reported, 75 although the many potential cellular and molecular mechanisms that underlie sarcopenia are unclear. Oxidative stress, inflammation, endocrine disorders, malnutrition, and inactivity have all been proposed as responsible cofactors for muscular senescence in humans and animals. 59
Generalized muscle atrophy was detected in 17 specimens in association with an etiological diagnosis of cachexia/inanition in our study, which represented 29.3% of the atrophy-related causes. The term atrophy implies a reduction in either the diameter of the muscle as a whole or in the diameter of a myofiber. 91 Both conditions were observed in this group of animals, in which type II fibers were preferentially affected. Histological features of the muscles of this group of animals were similar to previous descriptions in sheep and human beings that were associated with consumptive disease and inanition. 46,70
Cachexia is a metabolic condition associated with an underlying illness and inflammation, 27 which cannot be completely reversed by nutritional interventions. Limited success has been shown in preserving fat but not muscle mass in these cases. 57 In a similar way, inanition atrophy is due to reduced caloric intake and results in a general reduction in muscle fiber size, although recovery is typically relatively quickly when nutrition is restored. An identical histological pattern was detected in all the specimens in this group regardless of the cause of the atrophy, which was either inanition (foreign bodies that prevented feeding, such as a long and thick cable wrapped around the tongue of a fin whale, or a strand of plastic wrapped around the body of the mandible in a sperm whale) or chronic systemic diseases. 5
Generalized muscle atrophy related to inactivity (disuse) was recognized in only 1 animal in the present study. This case displayed external signs of scoliosis, a condition previously described in cetaceans. 7,19 It was not possible to determine the cause of this malformation, although it could be 1 of the several causative factors previously associated in documented cases of vertebral column malformation in cetaceans, including stress or exertion, spondylodiscitis or spondylo-osteomyelitis, trauma, or congenital malformations. The etiopathogenic mechanisms underlying the atrophy of both fiber types in this case might be related to abnormal swimming patterns and/or starvation due to impaired feeding.
Although starvation, cachexia, and sarcopenia can be defined as distinct clinical syndromes, a certain degree of overlap might have occurred in animals from the present study, as has been previously suggested. 70 Regardless of the etiology, muscle atrophy has important implications for the biomechanical performance and health status because a reduction in fiber size is related to lower force production. 93
Scattered fiber necrosis has been described as an additional histological change in cachexia, 95 aggravating the wasting process. Multifocal muscle atrophy associated to denervation atrophy was detected in 7 specimens, representing the 16.7% of the atrophy-related etiologies, in which identical histological features (group atrophy and fiber type grouping with no fiber type predisposition) to previous reports were observed. 23,89 Involvement of both types of fibers was confirmed by IHC (fiber typing) in 6/7 animals. Although determination of fiber types is necessary to confirm denervation atrophy, 24 small group atrophy and fiber type grouping are virtually pathognomonic of denervation atrophy following reinnervation. 56,89
More than half of the specimens in this group were considered senile (based on morpho-pathological findings) and myofiber atrophy might have been partially attributable to aging, as has been previously proposed. 69 Denervation atrophy of skeletal muscle is a consequence of damage to lower motor neurons and results in focal areas characterized by fibers with reduced size, although selective atrophy of a specific fiber type does not generally occur. 23 Fundamental physiological and metabolic skeletal muscle changes have been described as occurring after damage to the nerve supply. 32
As found in our study, fiber hypertrophy often accompanies fiber atrophy, which contributes to increased variation in fiber size. 91 Pathologically hypertrophic fibers have decreased oxygen diffusion from the interstitial capillaries to the internal regions. This leads to fiber splitting to ensure an adequate oxygen supply. Hypertrophied fibers could also undergo segmental necrosis if overloading occurs. 91 Type II fiber atrophy together with enlarged type I fibers, resulting in similar sized fiber types, have been described in the aged animals from our study. This likely represents an adaptive mechanism related to the diving abilities of cetaceans. 75
Chronic Myopathic Changes
Chronic myopathic changes were the third most common finding (25.5%) in our study and preferentially involved specimens with concomitant age-related muscle changes, such as senile or denervation atrophy. 75 Some of the CMCs, in particular, increased myofiber size variation and the number of internal nuclei, have been defined as nonspecific findings that are associated with a variety of neuromuscular disorders. 23 However, they have also been shown to be related to aging in equids 86 and humans, 49 an association that is also apparent in the cetaceans examined here. In addition, the presence of lipofuscin and lipofuscin-like granules has been observed in a variety of pathologic and physiologic conditions, including aging. 47
Chronic myopathic changes are nonspecific changes that are mainly related to advanced age, which could variably affect the fiber integrity and viability. However, the functional significance of some of these changes are currently unknown. 91
Complex Polysaccharide Inclusions
The presence of complex polysaccharide inclusions, which has previously been reported, 78 was the fourth most common myopathic change identified (22.6%) in our study. An interesting finding was that 69.2% of the animals with CPI displayed other muscle atrophies described in our study, as follows: senile atrophy (50%), denervation atrophy (27.7%), cachexia/inanition (16.6%), and disuse atrophy (5.5%). All of the animals affected by senile atrophy in our study were older adults, which explains why some of them also had complex polysaccharide depositions; a condition previously reported to be associated with aging in other species. 39,87 Muscular atrophy and neuromuscular disorders have also been reported in horses with polysaccharide storage myopathy. 58,91 In addition, 46.15% of the animals with CPI displayed degenerative changes associated to the stranding stress syndrome. This association introduces the intriguing possibility that a polysaccharide storage disease affecting myocytes might predispose some cetaceans to capture-related myopathy, which has been reported to occur in exertional rhabdomyolysis in horses. 68,78
Parasitic Infestation
Skeletal muscle parasitism was observed in 9.2% of the animals in our study. Intrasarcoplasmic protozoan cysts of Sarcocystis spp were observed in 13 specimens from 10 different species. This represents the first description of this type of infestation in 8 species: Atlantic spotted dolphin, short-beaked common dolphin, common bottlenose dolphin, spinner dolphin, pygmy sperm whale, fin whale, Sowerby’s beaked whale, and Risso’s dolphin. Our results suggest a lower prevalence in these species compared to the relatively high presence of muscle sarcocystosis found in stranded Atlantic white-sided dolphins (39%). 28 No inflammatory infiltrate was detected in any of the muscle tissue samples displaying variable Sarcocystis parasitization, suggesting that this protozoan is largely an incidental finding with non apparent pathogenic relevance. Although the life cycle of Sarcocystis spp in marine mammals is unknown, it has been suggested that dolphins are probably intermediate hosts for this parasite. 28
Tissue cysts containing bradyzoites of T. gondii were only observed in the skeletal muscle of 1 specimen in our study, an adult male Atlantic spotted dolphin. To our knowledge, this is the first description of skeletal muscle toxoplasmosis in a cetacean species. Two major presentations for T. gondii infection have been reported in different cetacean species: localized encephalitis, and systemic infection, 12,21,22,48,60,67 including in the Atlantic spotted dolphin. 5 T. gondii is able to infect a broad spectrum of cell types, although neurons and muscle cells are considered the preferred cell type for parasite persistence during infection, which in turn affects their transmission to new hosts. 82 A major route of transmission of T. gondii is the ingestion of contaminated food from chronically infected intermediate hosts, which in turns relies on the development of a bradyzoite stage. These can survive within muscle tissue cysts for at least 31 days. 38 The life cycle of T. gondii in marine mammals remains unsolved. Although it is a matter of speculation at this time, the role of skeletal muscle toxoplasmosis in Toxoplasma transmission is conceivable.
Myositis
Myositis was uncommon in the current study and was primarily related to bacterial infections. In 2 of the 3 specimens with myositis from this group, bacterial myositis was related to a systemic infection due to Nocardia farcinica and Staphylococcus spp, 5 while no microbiological analysis was performed in the remaining case. It is possible that several opportunistic bacteria might have enter through the wound contributing to the development of the lesions.
Several cases of pyo- and polymyositis have been reported in humans associated with Nocardia septicemia. 3,40,61 However, muscle involvement in bacterial infections is relatively uncommon and may result from contiguous sites of infection, penetrating trauma, vascular insufficiency, or hematogenous dissemination. 13 Associations between the route of infection and the infecting organism have been recognized. For instance, in human beings, acute bacterial infections of skeletal muscle secondary to hematogenous spread are mostly associated with Staphylococcus aureus, whereas infections as a result of penetrating wounds are often polymicrobial. 13
Depending on the etiological agents responsible for the infection and on the severity of the lesions, myositis in cetaceans can have different outcomes, with some fatal cases having been reported. 97
Sarcoplasmic Masses
The histopathological findings observed in the muscle of a pygmy sperm whale were comparable to those classically described as typical of myotonic dystrophy in humans and other animals. 74 The pathogenic relevance of this myopathy in the present case remains uncertain, although it might have contributed to the increase in the reported cases of primary muscle disorders in wild animals.
Conclusion
Our results provide evidence for a wide spectrum of muscle changes in wild cetaceans, which include lesions associated with systemic disorders (eg, generalized atrophy in consumptive processes, or bacterial myositis), incidental findings (eg, Sarcocystis spp parasitization), and degenerative processes of variable severity that might involve other organs (eg, myodegeneration and necrosis with subsequent myoglobinuric tubulonephrosis) and indicate a grave prognosis, aggravate the underlying disease processes, or have a negative impact on rehabilitation attempts. 42,43
Skeletal muscle is easily sampled during necropsy examinations and provides essential microscopic information on both local and systemic pathological conditions. Accordingly, skeletal muscle should be systematically sampled, processed and examined in all stranded cetaceans.
In conclusion, muscle lesions have been shown to be common among free ranging cetaceans. Because myopathic disorders can adversely affect their locomotion, 86 these conditions are highly significant for cetaceans, which strictly depend on effective skeletal muscle function for survival and the performance of specific daily activities in their aquatic environment.
Footnotes
Acknowledgements
The authors thank the members of the different stranding networks: Canary Islands (the SECAC and the Canarias Conservación), Almeria, and United Kingdom.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was possible thanks to the partial funding by National Project CGL2012-39681 (Subprograma BOS); Regional Project SolSub C200801000288, and ProID 20100091; Technical Assistant Contract by Canary Islands Government delegation (TEC0002955); and precompetitive project ULPGC2013-21.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
