Abstract
Identification of freeze-thaw artifact in fish can help to determine whether they have been harvested within the appropriate season and monitor adherence to fishing regulations. Recognition of freeze-specific changes will also prevent potential misinterpretation due to decomposition, disease, injury, or species variation. An initial survey using black rockfish (Sebastes melanops) identified which tissues reliably exhibit freeze artifact. Tissues were exposed to different treatments: immediate formalin fixation; refrigeration or storage at room temperature for 24, 48, or 72 hours; or freezing for 1, 8, or 28 days. Three fish underwent a combination of treatments. Tissue changes in each treatment group were compared macroscopically and microscopically. Macroscopic changes in frozen-thawed and never-frozen fish overlapped somewhat; however, microscopic findings of skeletal myocyte cavitation, lens liquefaction, and brain tissue fractures were consistent findings only in frozen-thawed tissues. A validation study was then done to establish the accuracy of microscopic analysis. Brain and paired ocular and skeletal muscle samples from 61 steelhead trout (Oncorhynchus mykiss) were fixed in formalin either fresh or after being frozen for 4 weeks. Weighted kappa values showed both high observer accuracy and interobserver agreement in the identification of freeze-thaw status. Based on these findings, microscopic changes in the skeletal muscle, eye, and brain are considered consistent and easily identifiable indicators of a previous freeze-thaw cycle and should not be confused with a pathologic process.
Freezing and thawing alters the gross and histologic appearance of tissues. Identification of freeze-specific change is often only necessary to avoid misdiagnosis; however, in forensic casework, it may be a pivotal point in a suspicious death investigation. Perpetrators might freeze bodies to hide them or obfuscate the time of death. Freezing may be a cause of death or occur naturally with exposure to the elements.12,13,19,30 Determining whether a body has been frozen may help to corroborate or refute a suspect’s statements and, ultimately, determine whether the law has been broken. In veterinary forensic investigations involving illegal fishing activity, poachers may attempt to disguise the fact that fish have been harvested out of season by freezing the fish for storage and then thawing them during the legal fishing season to sell as fresh-caught or vice versa. In these cases, the question presented to the pathologist is simple: has this fish ever been frozen?
Illegal fishing and overfishing contribute to the destabilization of marine environments, with far-reaching ecological and economic effects.8,29 Illegal fishing activity that violates the United States federal law can result in severe penalties including fines, loss of licensing, forfeiture of boats, and jail time. Undercover operations have targeted illegal harvesting, importation of protected or potentially invasive species, and fraud. Notable recent cases in the United States include Operation Bahamarama (Florida) and Operation Fishing for Funds (Michigan) which have resulted in convictions and fines as high as US$1,000,000. Money and forfeited property are taken as restitution and are often used to help fund conservation efforts.25,26
The casework prompting this study originated from small operation fishing violations along the West Coast of the United States. In this area, Pacific salmon populations have been severely reduced or extirpated through a combination of habitat loss, pollution, and overfishing. Illegal activity in this instance involved salmon being sold outside of the legal fishing season. The suspect claimed that fishing had occurred in season with the carcasses being held frozen until the outdoor market commenced. The investigator wanted to know if freeze-thaw status could be verified through examination of the gutted carcasses.
Freezing and thawing of soft tissues damages individual cells and disrupts the larger tissue architecture, but there is a lack of clear understanding of the mechanism and the factors that influence severity. Cell wall pore formation and rupture are thought to be the result of fluid shifts that occur during the freezing and thawing process. As extracellular water freezes, fluid flows out of the cell into the extracellular space. 10 This fluid movement increases the intracellular solute concentration, resulting in cell shrinkage and disruption of cellular junctions. 9 When the tissue thaws, fluid moves back into the cells. The subsequent volume change can rupture the weakened cell walls. These collective effects are thought to be the driving force of change rather than physical damage due to ice crystals.10,19 Freeze-related changes documented in mammalian tissue include cell shrinkage, basement membrane separation, transudate accumulation, hemolysis, and endothelial and epithelial cell sloughing. Fractures or cleft formation along tissue planes appear most notably in the brain, liver, and heart.1,15,19
Freeze artifact in boney fish has been infrequently and incompletely documented, with most studies focusing on freezing as it relates to commercial food storage.1 –4,6,11,14,16 Frozen storage of food fish causes macroscopic changes in muscle color, texture, and weight due to fluid loss, ice crystal formation, and bacterial growth.4,9 These changes are somewhat subjective, require comparison to the tissue before storage, and may differ with species and seasonality. 4 Microscopic changes are more consistent and specific. In studies on fishery-harvested fish and fish of unspecified origin, the presence of irregularly shaped spaces within skeletal myocytes (either clear or pale eosinophilic) was used to accurately differentiate fresh from fresh-frozen muscle.3,4,11,14,16,20 Cross-sectional changes are described as having “breakdown processes in the central part and structurally intact periphery” or “vacuoles,” with the changes being somewhat dependent on the fat and protein compositions of the different species examined and the rapidity and method of freezing.3,4,20 In tissues other than muscle, freeze artifact in fish has been described minimally or not at all, with, to the authors’ knowledge, no description of freeze artifact in fish brains or in eyes of any vertebrate.
This study comprises 2 parts. First, a pilot study was done to detect and further characterize gross and microscopic freeze artifact in a variety of fish tissues and to compare the findings to those of never frozen tissues preserved fresh and at various stages of decomposition. Based on those findings and likely tissue availability in market-prepared carcasses, skeletal muscle, eye, and brain were identified as the best freeze-thaw indicators. A second study was a validation study supporting the reliability of these freeze-thaw indicators. This study was a blinded analysis of a larger cohort of steelhead trout; a species that may be encountered in this type of casework.
Materials and Methods
Pilot Study
Donated, filleted carcasses of legally harvested adult black rockfish (n = 14) from the Oregon central coast (at Newport, OR, USA) were line caught (jigged with a spinning reel and rod) and killed immediately after being caught via manually applied blunt force trauma (cranial concussion). Black rockfish (hereafter rockfish) were chosen based on availability and were not harvested for the purposes of this project. While on the boat, the fish were held in plastic containers at air temperature. Air temperatures taken from land on that day ranged from 70 to 80°F/21-26°C. Sea temperature in the collection area during this time of year averages 56°F/13.3°C (ncei.noaa.gov/access/coastal-water-temperature_guide). The fish were filleted and collected dockside within 5 hours of being caught. They did not have direct contact with ice at any time, though they were externally rinsed with fresh water immediately prior to cleaning. The cleaning method consisted of lateral body wall muscle layer (fillet) removal with preservation of the coelomic cavity and internal organs, head/gills, skeletal elements, lumbar and some body wall muscling, and overlying skin and fins.
To assess as many potential freeze-thaw scenarios as possible, fish were assigned randomly to a variety of fresh or frozen methods of preparation (Supplemental Table S1). Tissues from 3 of the rockfish were fixed in 10% neutral buffered formalin immediately (Fish 1–3; designated as time 0). Tissues saved from Fish 1 and Fish 2 included only the heads, attached gills and fragments of the anterior kidneys. Tissues saved from Fish 3 consisted of only the internal organs which were removed and fixed en bloc. The remaining 11 rockfish were transported for approximately 5 hours in a cooler with bagged ice and then either stored whole in a 40°F/4.4°C refrigerator (Fish 4–6) for 24, 48, and 72 hours, respectively; left at room temperature at approximately 68°F/20°C (Fish 7–9) for 24, 48, and 72 hours, respectively; or stored in a −8°F/−22°C, freezer (Fish 11–14) for 1 to 28 days (see Supplemental Table S1). The freezer was a walk-in freezer with a regular defrost cycle, having approximately 7 temperature peaks of 3°F/−16°C and 4 peaks of 20°F/−6.6°C per 24 hours.
All frozen tissues were trimmed and fixed in 10% neutral buffered formalin (hereafter, formalin) as soon as they had completely thawed. Thawing occurred at room temperature except for Fish 13, which was thawed in the 40°F/4.4°C refrigerator. Thawing at room temperature took 5 to 8 hours. The heads were partially split via a dorsal midline cut through the skull and then formalin-fixed whole. This method facilitated intact retrieval of the brain, which becomes soft to partially liquefied following freezing or decomposition.
Tissue sets consisted of eye, brain, skin, skeletal muscle, stomach, pyloric ceca, gill, heart, spleen, liver, gonad, anterior kidney, and intestine. After formalin fixation, the eye, gill, and skin were decalcified over a 24-hour period using a rapid decalcifier (Leica Surgipath Decalcifier II). All tissues were routinely processed and stained with hematoxylin and eosin. One sample of each tissue was examined.
Validation Study
Sixty-one steelhead trout consisting of 26 whole bodies and 35 heads were obtained fresh during a routine cull of summer (winter-spawning) adults. The fish were collected at a hatchery approximately 150 river miles from the ocean. All fish were killed on site via cranial concussion. The heads were bagged individually immediately after euthanasia and refrigerated overnight prior to transportation and sampling. The bodies were collected immediately after euthanasia. The bodies and bagged heads were placed in a cooler with blue ice for 45-minute transport and were sampled immediately upon arrival at the lab. Thirty-four fish were female and 27 fish were male. Eyes, skeletal (lateral and levator operculi) muscle from the head or epaxial musculus lateralis from the body wall, and brain were sampled. From each individual fish, one eye and a section of muscle were retrieved fresh and fixed in formalin. Because of the small size, the brain was sampled whole; half of the whole samples (assigned randomly) were immediately fixed in formalin and the other half were preserved within the skull and frozen. The remainder of the head including the contralateral eye from all fish and an approximately 5 cm wide transverse section of body wall from whole fish (a steak cut) were each wrapped in 3 layers of plastic wrap and an outer layer of aluminum foil, placed in a Ziploc bag and stored in the previously described walk-in freezer for 4 weeks.
After 4 weeks, tissues were removed from the wrappings and allowed to thaw in a single layer at 40°F/4.4°C overnight. Further sampling and preparation, including longitudinal splitting of the skull prior to formalin fixation, was as described for the rockfish. One section each of eye and muscle and 2 hemisections of brain were processed as described for the rockfish. Slides were randomized, unmarked as to treatment group, and reviewed by 2 ACVP (American College of Veterinary Pathology) board-certified veterinary anatomic pathologists (RAK and TCV). Muscle was scored for the presence or absence of myocyte cavitations, relative numbers of cavitations observed in cross-section, and the presence or absence of hemolysis. Cavitations in less than 25% of myocytes was scored as +, cavitations in 25% to 75% of myocytes was scored as ++, and cavitations in greater than 75% of myocytes were scored as +++. Brain was examined for the presence or absence of tissue fractures and hemolysis. Sagittal sections of eyes were evaluated for lens liquefaction, hemolysis, and myocyte (ocular muscle) cavitations. Myocyte cavitations were scored as for the head and body wall sections. Based on the combination of features, an opinion was given for each individual tissue as to whether it had been frozen, never frozen, or could not be determined.
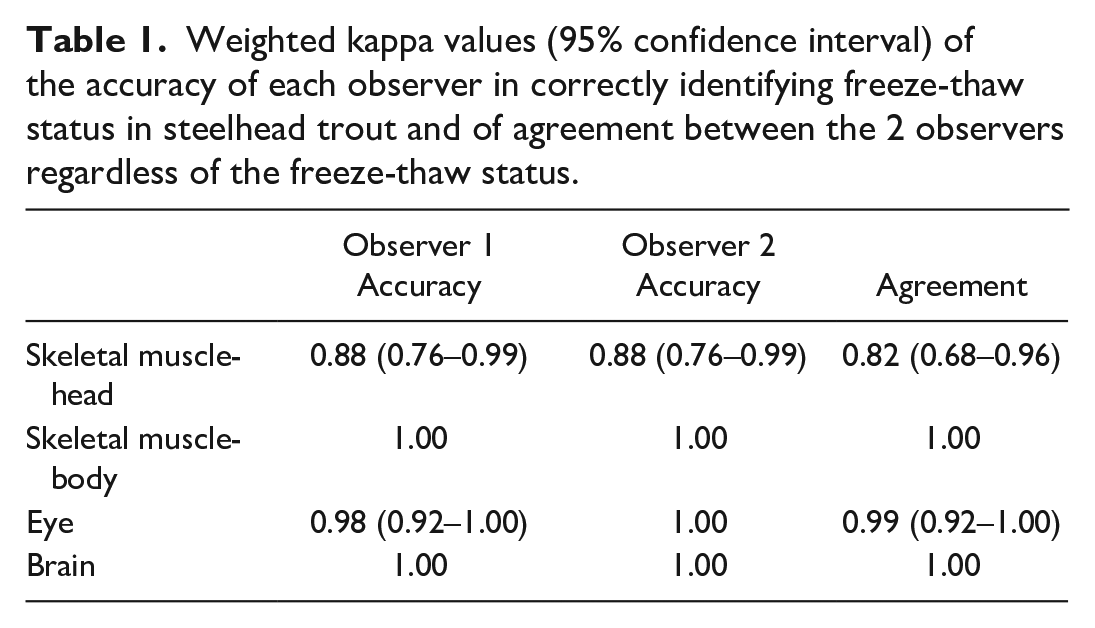
Statistical analysis was performed in R version 4.0.3. 17 Weighted kappa values were used to assess the accuracy of freeze-thaw determination for each tissue type and interobserver agreement (Table 1).
Weighted kappa values (95% confidence interval) of the accuracy of each observer in correctly identifying freeze-thaw status in steelhead trout and of agreement between the 2 observers regardless of the freeze-thaw status.
Results
Pilot Study
Macroscopic changes associated with freezing
Frozen-thawed fish had centrally cloudy lenses (Fig. 1). The skin surface had a “ground glass” appearance almost entirely devoid of mucus, even after 48 hours at room temperature (Fish 14).
Macroscopic changes associated with decomposition
All never-frozen fish held at room temperature had clear corneas at time 0 (Fig. 2), which became progressively cloudier at 48 and 72 hours (Fig. 3). A film of mucus developed on the skin surface and increased in volume over 72 hours. Over the 72-hour time period (Fish 9), tissues became increasingly malodorous, the muscle and skin developed a yellow tinge, and internal organs softened. By 72 hours, the ribs and skeletal muscles had started to separate, a large amount of watery, reddish brown, intracoelomic fluid accumulated; and solid organs partially liquefied.

Frozen-thawed, room temperature and fresh eyes, adult black rockfish.
Overlapping macroscopic findings of frozen-thawed and decomposed fish
Gills from both frozen-thawed and decomposed fish had pale, thickened filaments. Fish that were frozen and thawed multiple times (Fish 12) or allowed to decompose after being frozen (Fish 13 and Fish 14) had ocular cloudiness, though the affected areas differed, with cloudy corneas obscuring the lenses in decomposed fish and clear corneas with cloudy lenses in frozen-thawed fish.
Microscopic changes associated with freezing
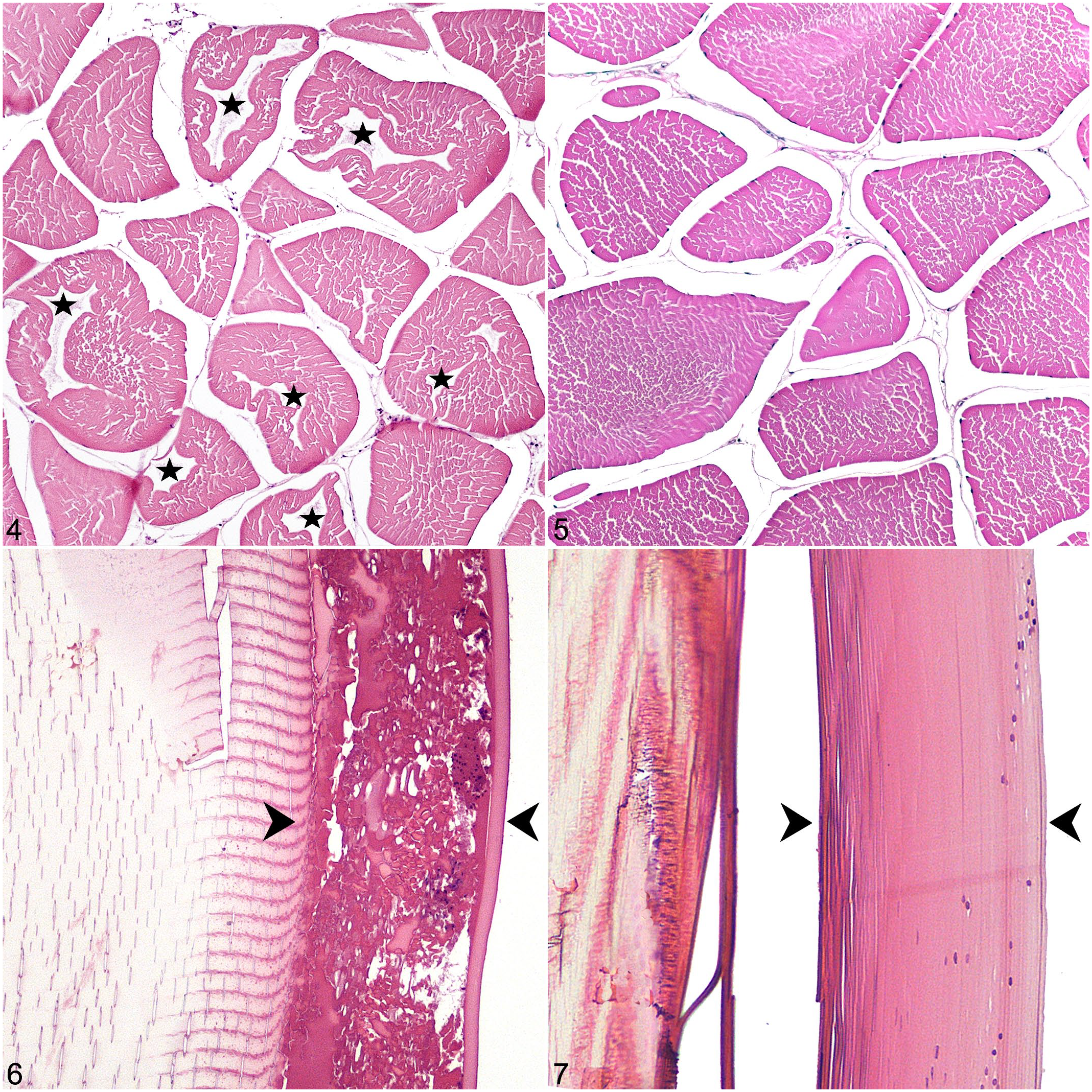
A summary of the histologic features of each treatment type is in Supplemental Table S2. Skeletal myocytes had central cavitated areas best visualized on cross-section. These cavitations had defined, undulant borders around variable amounts of colorless space and/or pale eosinophilic, lacy material (Fig. 4). Severely affected myocytes were partially collapsed around this central area. Myocyte cavitations were in 100% of the tissues that had been frozen and none of the never-frozen tissues (Fig. 5). In fish frozen for more than a week, these cavitations appeared to be more numerous; however, too few fish were examined to make a statistical inference. Cavitated myocytes were occasionally present in other tissues, including the peri-ocular muscle (in 83% of the eyes) and branchial arch. In general, however, muscles in these areas were scant and often included few or no myocyte cross-sections.
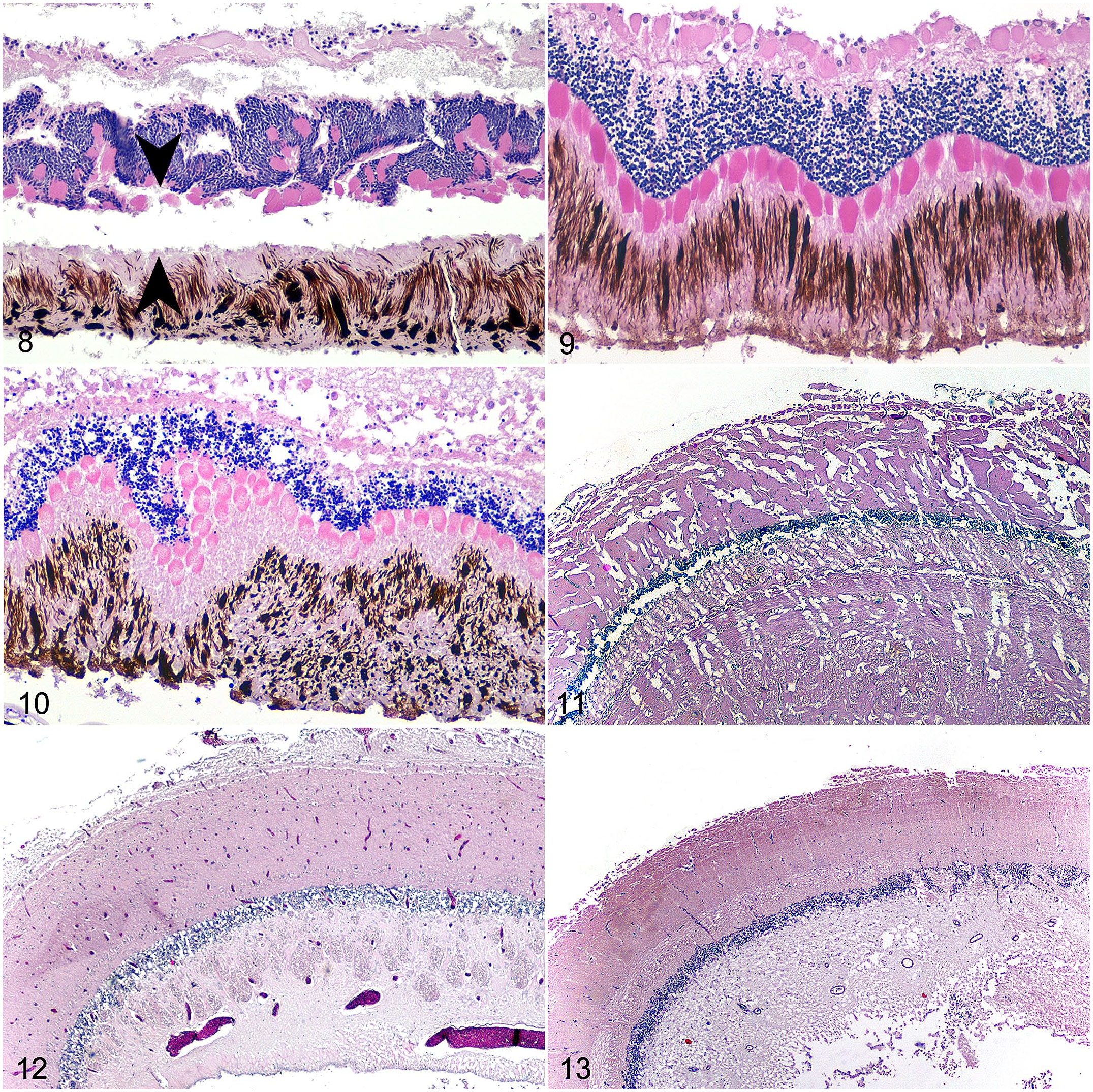
In all frozen-thawed eyes, the lens had a peripheral accumulation of homogenous to globular eosinophilic material either subjacent or external to the lens sheath (Fig. 6). Lens changes were in 100% of the frozen samples and none of the never-frozen samples (Fig. 7). In the retina, rods and cones in approximately 50% of the frozen-thawed samples were rounded, disordered and multifocally to diffusely separated (described hereafter as retinal clefting) from the retinal pigment epithelium (Fig. 8). The pigment epithelium had a jumbled appearance with pigment swirled throughout the layer instead of in an orderly, basal orientation. In 100% of frozen-thawed brains, there were linear to crisscrossing fractures throughout the white and gray matter (Fig. 9).
Comparison of frozen-thawed and never-frozen tissues, adult black rockfish. Hematoxylin eosin.
In the ventricles of the heart, trabecular spaces were widened with a slight disarray of cardiomyocytes and vacuole-like separations were visible in cross-section that were similar to, but less pronounced than, the skeletal musculature. These cavitary areas were in 50% of the frozen hearts. The liver was variably affected by the freeze artifact. In liver frozen for 4 weeks, sinusoidal spaces were diffusely widened with intra-sinusoidal accumulation of pale basophilic, lacy to finely vacuolar material. In liver frozen for only 24 hours, the sinusoidal changes were multifocal and less pronounced, occurring more often in pericholangial regions. In the muscular walls of the stomach and intestines, there was multifocal fissuring with smooth myocyte and collagen fiber separation. Changes were most pronounced after 4 weeks of freezing and in the (thicker) wall of the stomach.
Microscopic changes associated with decomposition and autolysis
Variable skeletal myofiber separation and angular myocyte fragmentation through the sarcoplasm without cell collapse were presumed to be processing artifact (Fig. 5). There were also occasional sections with multifocal, mild, sarcoplasmic fragmentation, and loss of cross-striations, possibly related to capture.
In the eyes, lens fiber fragmentation occurred with increasing time at room temperature (Fish 8 and Fish 9). Superficially, fragmentation was angular to squared. Deeper portions of the lens were circumferentially fragmented, resulting in an onion ring-like appearance (Fig. 10). Rounding of the rods and cones occurred with autolysis but lacked the disarray and separation associated with freeze-thaw (Fig. 11).
Brain autolysis was relatively mild for up to 48 hours at room temperature (Fig. 12). Changes consisted of multifocal areas of intravascular hemolysis, an overall increase in tissue pallor, and widening of spaces around neurons and blood vessels (Fig. 13). At 72 hours, there was partial dissolution of the telencephalon, giving the tissue a granular appearance. Cellular details, including nuclei, were not visible. The caudal portions of the brain were comparatively well-preserved, maintaining distinct granular layers in the optic lobe and cerebellum, a relatively compact neuropil, and occasional intact erythrocytes.
Comparison of frozen-thawed and never-frozen tissues, adult black rockfish. Hematoxylin eosin.
The gill epithelium began to slough as early as 24 hours at room temperature and 48 hours of refrigeration. In the decomposed tissues, hemolysis and branchial epithelial sloughing worsened over 48 and 72 hours. By 72 hours, there was widespread pallor and separation of the gill arch stroma.
In the heart, there was tissue pallor, dissolution of sarcomeres, and multifocal to diffuse reticulo-endothelial cell loss. At 72 hours, there was also myocardial gas bubble formation. Autolysis of the liver in some samples obscured the sinusoidal spaces. By 48 hours at room temperature, hepatocellular and biliary epithelial detail and sinusoidal spaces were lost. Intravascular lumina were filled with wispy eosinophilic material, and there was diffuse hemolysis.
Decomposition and autolysis in the stomach and intestine consisted of mild epithelial sloughing with good preservation of sloughed cells at time 0. By 48 hours at room temperature, there was transmural loss of differential staining and wide-spread hemolysis. By 72 hours at room temperature, the intestinal tract was diffusely pale and eosinophilic with loss of the mucosa and mural cellular detail. The gastric mucosa was multifocally absent with loss of differential staining, hemolysis, and severe transmural bacterial overgrowth.
Overlapping microscopic changes of frozen-thawed and decomposed fish
The most common overlapping change was intra- and extravascular hemolysis, characterized by the loss of erythrocyte cytoplasm with or without preservation of nuclei. Hemolysis was present to some degree in all tissues except those fixed at time 0, in which there was no hemolysis. Hemolysis was generally less diffuse in sections with a larger volume of blood, such as within the heart chambers and large-caliber blood vessels. The extent of the hemolysis varied with the length of time, becoming diffuse by 72 hours at room temperature.
The cutaneous and branchial epithelium only remained in those tissues fixed at time 0. Within as few as 24 hours of refrigeration and in all frozen tissues, the epithelium was largely sloughed.
Other than mild internal parasitism, there were no signs of disease. There were also no signs of chronic injury. There were rare foci of acute hemorrhage, likely related to capture.
Validation Study
To validate the pilot study, a larger sampling of skeletal muscle, eyes, and brain from steelhead trout were formalin-fixed either fresh or after being frozen for 4 weeks. They were then evaluated blindly to determine whether features identified as freeze-thaw artifact in the pilot study could be reliably used to identify previously frozen tissue. Amongst the sections of brain and skeletal muscle from the body wall, there was 100% agreement and accuracy as to whether the tissues had been frozen. Accuracy was slightly less when only skeletal muscle from the head was considered (weighted kappa value of 0.88 for both observers; Table 1). In tissues falsely determined to be frozen, there were only rare myocytes that appeared to be cavitated. Fracturing of the brain affected all frozen-thawed and no fresh sections. Hemolysis occurred in 26/28 of the frozen brains and 0/29 of the fresh brains. In 3 of the samples (2 frozen and 1 fresh), hemolysis was indeterminate due to a lack of erythrocytes.
Peripheral liquefaction affected all frozen lenses (n = 60) and no fresh lenses (n = 59). In cases where the lens was completely lost during processing (from 1 frozen and 2 fresh eyes), a determination of fresh versus frozen was correctly made based on a combination of retinal disarray, hemolysis, and peri-ocular myocyte cavitations. Inaccuracy in ocular assessment only occurred in one eye with a partial lens and no retina due to sectioning and processing artifacts. One observer marked it as “undetermined” and the other incorrectly assessed it as fresh. In the peri-ocular myocytes, cavitations were not consistently visible in frozen samples due to a paucity of muscle and lack of myocyte cross-sections. There were, however, no false positives based on myocyte features. Retinal disarray is 100% correlated with frozen status.
As in the rockfish, there were no signs of disease in any of the examined tissues. There was occasional, acute, skeletal myocyte necrosis and rare, acute, cerebral hemorrhage attributed to capture and euthanasia.
Discussion
The most specific and easily identifiable freeze-thaw artifacts in fish were fissuring of the brain, partial liquefaction of the lens, and cavitations in myocytes. These changes should not be confused with disease or antemortem injury. In skeletal muscle, our findings agree with studies showing the development of distinct myocyte cavitary changes after being frozen.3,11,16 Myocytes from other vertebrates, including species of Aves and Mammalia, have similar cavitations from freezing (personal observation), though they appear to be less pronounced and less numerous. To the authors’ knowledge, published studies of mammalian freeze artifact have not included skeletal muscle. This is perhaps because the frozen artifact is easily identified in other organs such as liver, making examination of muscles unnecessary.1,2,19 Other than the brain, structural changes in the solid organs of fish were either not present or not as distinct as in mammals. Therefore, coelomic viscera does not seem to be a useful indicator of freeze-thaw in fish.
In prior studies, when only skeletal muscle was examined, there was high specificity and sensitivity of freeze-thaw identification even when the evaluation was being done by veterinarians unfamiliar with the histologic changes of freezing. 3 Similarly, in this study, only rare false positive and false negative assessments of skeletal muscle occurred, and only in samples taken from the head. One reason for the relative difficulty in assessing this anatomical region may be the comparative lack of cranial muscling in fish, which makes capture of adequate myocyte cross-sections more difficult.
The influence of muscle type, both individual and interspecific, was also considered. There are 2 types of skeletal muscle: red (type 1/slow twitch) and white (type 2/fast). Muscles along the dorsal-caudal aspect of the head of boney fish (analogous to the temporal region in mammals) consist of levator muscles, which, in zebrafish (Danio rerio), are a mix of white and red muscle. 7 In contrast, the body wall and epaxial muscles of salmonids are made up of white muscle with only very small foci of red muscle superficially at the lateral line and dorsal midline. 21 Whether or not freeze-artifact differs between these 2 muscle types is uncertain; however, if freeze-artifact is less prominent in red muscle, it is unlikely to affect interpretation in full-thickness body wall sections. It is therefore recommended that the body wall and epaxial muscles be preferentially sampled. Sections should be deep and wide enough to avoid sampling only around the lateral line or dorsal spinous processes. If only the head is available, analyzing skeletal muscle in conjunction with the eye and brain will still allow for accurate identification of freeze artifact.
Muscle composition of fish also varies between species and is often described in terms of fat content. Species variation as a response to different environments (fresh vs. salt and warm vs. cold water) could make a difference in the development of freeze artifact. Fish inhabiting arctic waters have adaptations to hinder freezing and their tissues may respond differently to cold storage. 28 Thus far, freeze-thaw myocyte changes have been observed in studies done on a mixture of warm water fatty, semi-fatty, and lean fish species.3,11 Two types of cold-water fish, a lean saltwater and a fatty anadromous species, were evaluated in this study with similar results. Given the results of this and prior studies, species differences may not preclude the development of freeze artifact; however, if casework is required, comparative studies on a wider range of species may be of value.
In the eye, partial lens liquefaction was a consistent and easily identifiable change related to freezing. The microscopic features are reminiscent of Morgagnian globules but lack features indicating a vital reaction such as bladder cells, capsular epithelial hyperplasia, mineralization, and hypermaturity.
Widespread hemolysis should raise suspicion of frozen-thawed tissue but, when taken alone, is not diagnostic of freeze-thaw. There are many other causes of hemolysis, including exposure to fresh water, decomposition, hemolytic disease, and toxins.5,23 Branchial tissue is also not a reliable indicator of freeze-thaw. The gill has a limited ability to respond to stimuli, and it is important not to overinterpret changes in this tissue. Epithelial lifting or sloughing, for example, is a common finding caused by a variety of stimuli, including fixation and histologic processing, antemortem toxin exposure, and decomposition. 27
The gross appearance of a fish carcass can raise suspicion of freeze-thaw, though changes often overlap with decomposition. Fish with translucent lenses and pale, thickened gill filaments in the absence of other signs of decomposition, such as odor, internal liquefaction, and tissue discoloration, should prompt the collection of appropriate samples for histologic examination.
Also noted grossly was the lack of epidermal mucous accumulation on a frozen-thawed fish allowed to decompose at room temperature. Although only one fish was examined under these conditions, this striking difference as compared to the decomposed never-frozen fish may warrant further investigation. In life, the surface film of fish is a mixture of mucin, immune molecules, dead epithelial cells, and a commensal microbiome. 18 Freezing of tissue could affect the mucous character by altering any of those components.15,24 A comparison of the microbial communities of frozen and never-frozen fish may provide further information about freeze-thaw related microbiome changes.
It should be noted that the sample size of this pilot study was limited to only a few individuals. A more thorough analysis of decomposition-related changes in fish tissue was not considered to be within the scope of this research, but additional information can be found in existing literature.6,27
Regarding exposure-related nuances of freeze-thaw artifact, it has been observed that severity increases with longer time spent frozen. 19 This may be because freezer temperature fluctuations can cause partial to complete thawing and refreezing that has a cumulative impact on freeze artifact. 4 While in the pilot study, more myocytes appeared to be affected in fish frozen for longer periods of time, only a very small subset was compared in this way, and inferences based on statistical analysis cannot be made.
Different freezing and preservation methods may also influence the freeze-thaw artifact. Tissues frozen more slowly (at the higher temperatures of most home freezers) have more time for the cellular shrinkage that results in fluid-shift related damage. Tissues frozen more rapidly (at the lower temperatures recommended for commercial freezers) develop intracellular ice crystals.10,19 Tissues flash-frozen in liquid nitrogen have shown preservation of erythrocytes and little to no freeze artifact in eye, gill, liver, intestine, pancreas, and immature gonad. 22 In the legal cases prompting this study, flash-freezing was not a method used, nor were the suspects part of large-scale commercial operations. Until further research can be done, this highlights the need for a thorough history before ruling out freeze-thaw artifact, particularly when fisheries are involved. In this study, an automatic defrosting (frost-free) freezer was used because of its popularity for home use and availability to the authors. Frost-free refrigerators cyclically warm evaporator coils to prevent frost build-up which causes mild interior temperature fluctuations. These fluctuations, theoretically, do not occur in a manual defrost freezer, though opening and closing the door during home use may negate the temperature stability. A study comparing tissues stored in manual defrost, automatic defrost, and ultra-low freezers is in progress.
In summary, the authors have identified several microscopic features allowing for the identification of freeze-thaw artifact in fish. Macroscopic features may heighten the suspicion of a previous freeze-thaw, but microscopic examination allows for the most accurate conclusions. The tissues with the most consistent and identifiable changes (body wall muscle, eye, and brain) are also likely to be available in market-prepared fish. Whether or not the goal is to determine freeze-thaw status, it is necessary to recognize freeze artifact as such and not as a pathologic process. It is hoped that the information provided in this paper will aid pathologists in the investigation of wildlife crimes and in the interpretation of non-lesions in fish.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221120012 – Supplemental material for Identification of freeze-thaw artifact in fresh and decomposed black rockfish (Sebastes melanops) and steelhead trout (Oncorhynchus mykiss)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221120012 for Identification of freeze-thaw artifact in fresh and decomposed black rockfish (Sebastes melanops) and steelhead trout (Oncorhynchus mykiss) by Rebecca Kagan and Tabitha C. Viner in Veterinary Pathology
Footnotes
Acknowledgements
We thank Colleen Wilson HT, the Oregon State University Fisheries and Wildlife Club, and the Oregon Department of Fish & Wildlife Cole Rivers Hatchery for their help with sample collection and preparation. The findings and opinions contained herein are those of the authors and do not necessarily reflect the views of the US Fish and Wildlife Service.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
