Abstract
C-KIT is the cellular homolog of the feline sarcoma viral oncogene v-KIT, which encodes the tyrosine kinase receptor protein KIT. Mutations and varied expression of this gene have been demonstrated within multiple neoplasms in people and domestic animals. The purpose of this study was to determine if KIT protein is expressed in feline soft tissue fibrosarcomas (ST FSA) using immunohistochemistry (IHC). The computer database at the Oklahoma Animal Disease Diagnostic Laboratory was searched from January 1, 2006, to December 31, 2007, for any domestic cat with an ST FSA. Routinely stained slides from 46 feline ST FSAs were reviewed and graded based on the scale outlined by Kuntz et al. Immunohistochemistry for KIT protein was performed on one representative section from each cat. There were a total of 12/46 (26%) cats that were immunoreactive for KIT. Immunoreactivity was detected in greater than 80% of the neoplastic cells in 4/46 (9%) cats. Immunoreactivity was detected in less than 10% of the neoplastic cells in 8/46 (17%) cats. Immunoreactivity was characterized by evenly distributed cytoplasmic stippling within the neoplastic spindle-shaped cells and/or multinucleated giant cells. Based on these results, KIT immunoreactivity can be detected within feline ST FSAs using IHC. The results of this study also indicate that KIT immunoreactivity in feline ST FSA does not correlate with the histologic grade (P = .141, X 2 = 2.166), survivability (P = .241, X 2 = 1.373), or whether the neoplasm was a spontaneous or an injection site FSA (P = .074, X 2 = 3.184).
Soft tissue fibrosarcoma (ST FSA) is one of the most common neoplasms in cats. 3 The cause of this neoplasm may be viral, injection site associated, or spontaneous. 2,9,19 Regardless of the origin, this neoplasm tends to be locally aggressive with low metastatic potential. The neoplasm recurs in 30 to 60% of cats following surgical resection. 7,12
C-KIT is a gene that encodes a membrane-associated tyrosine kinase growth factor receptor (CD 117). 35 The c-KIT gene is expressed by many cell types, including mast cells and interstitial cells of Cajal (ICC). 10,27 The normal function of this growth factor receptor has been extensively studied, and it is thought to be associated with growth and differentiation of immature cells. 29,35 KIT protein has been shown to be expressed by neoplastic cells as well; most notably, gastrointestinal stromal tumors (GIST) in people and mast cell tumors (MCT) in dogs. 8,10,25
Neoplastic transformation of fibroblasts as a consequence of Feline Sarcoma Virus (FeSV) infection is induced by expression of the viral oncogene v-KIT. 2,13,26 This gene is the product of random incorporation of the cellular proto-oncogene c-KIT into the viral genome. 2 Because of the relationship between v-KIT and c-KIT, c-KIT expression may also play a role in the formation of ST FSA in cats, independent of cause.
The purpose of this study was to document if KIT is expressed in ST FSA using immunohistochemistry (IHC). Additional goals of this study were to document the pattern of KIT immunoreactivity and determine if there was a correlation between KIT immunoreactivity and histologic grade of the neoplasm, survival time, and/or injection site sarcoma (ISS).
Materials and Methods
Case selection
The Universal Veterinary Information System laboratory database (UVIS version 3.1.8, Ross Group; Dayton, OH) at the Oklahoma Animal Disease Diagnostic Laboratory was searched for any soft tissue biopsy specimens from a domestic cat submitted between January 1, 2006, and December 31, 2007, with the diagnosis of fibrosarcoma. Routinely HE-stained slides from each cat were reviewed. For a cat to be included in the study, the diagnosis of ST FSA had to be confirmed by 2 board-certified anatomic pathologists. Slides with a diagnosis other than FSA were not included.
Data collection
The pathology reports from all cats included in the study were reviewed, and the following information was collected: age, gender, location of the neoplasm, and recent history of an injection at the site. If any of the above data were not included in the report, the referring veterinarian was contacted for that information. Referring veterinarians were also contacted to determine if the cats in the study were alive or deceased as of June 2008. The date of recurrence of the neoplasm, if any, was recorded and the disease-free interval was calculated. The number of days the cat survived after surgery was also recorded when available.
Tumor grading
Slides from each cat were reviewed blindly and were graded independently by 2 board-certified anatomic pathologists. Each neoplasm was graded as low, intermediate, or high grade based on the criteria outlined previously. 22 Neoplasms in the study were considered to be ISS if the UVIS pathology report included histologic features consistent with ISS (anaplasia, giant cells, inflammatory cell infiltration, and/or adjuvant) and either of the 2 following criteria were met: neoplasm was located at site commonly used for vaccinations or history of injection within the last year at the site of the neoplasm. 6
c-KIT IHC
IHC was performed on one 4–5 µm thick section from each feline ST FSAs using a horseradish peroxidase-streptavidin method. 27 Rabbit polyclonal c-KIT antibody (Neomarkers, Fremont, CA) at a 1 : 200 dilution in Da Vinci Green diluent (Biocare Medical, Concorde, CA) was used as the primary antibody. The slides were counterstained with Mayer's hematoxylin.
A well-differentiated canine mast cell tumor (2006 and 2007 cases) and a feline mastocytoma (2006 cases) were used as positive controls. An additional slide from each cat without the primary antibody was used as a negative control.
IHC interpretation
The slides were blindly evaluated by 2 board-certified anatomic pathologists for immunoreactivity. The results were scored as follows, based on the approximate number of cells within the neoplasm that were immunoreactive: greater than 80% of cells were immunoreactive; 80–10% of the cells were immunoreactive; less than 10% of the cells were immunoreactive; or negative (no immunoreactivity was noted in the section). Distribution of immunoreactivity was also noted and recorded as KIT-staining pattern I, II, or III as previously described for mast cell tumors in dogs. 33 The morphology of immunoreactive cells was also recorded for each cat.
Statistics
The total number of feline ST FSAs with immunoreactivity for KIT was calculated for each grade. For statistical purposes, the number of high and intermediate histologic grade ST FSAs with immunoreactivity were combined and compared with the number of low histologic grade ST FSAs with immunoreactivity using Excel (Microsoft, Redmond, WA). A chi-square and P value were also generated using Excel. Survival was defined as either alive or deceased as of June 2008 for the purposes of chi-square analysis. An unpaired Student's t-test was used to compare the mean survival for each of the categories of immunoreactivity using Excel. Only those cats with survival data available from the referring veterinarian were included in the Student's t-test. Finally, the histologic grades were evaluated for a statistical difference in disease-free interval using a single factor analysis of variance (ANOVA) test using Excel. In all statistical analyses, a P value <.01 was considered significant.
Results
Study population
A total of 46 cats met the inclusion criteria. Cats ranged in age from 4 years to 17 years, with an average age of 10.6 years. There were 23 male and 23 female cats. The breeds of cats within the study group were as follows: 30 domestic short-haired cats, 1 domestic medium-haired cat, 6 domestic long-haired cats, 2 Maine coons, 2 Persians, 1 Siamese, 1 Siamese cross, 1 Tonkinese, and 2 cats for which the breed was not reported.
Included in the study were 17 high, 18 intermediate, and 11 low histologic grade ST FSAs. In this study group, the most common locations of ST FSAs were the trunk (26%, 12/46), limb (24%, 11/46), and shoulder (17%, 8/46). Other locations included the head (9%, 4/46), hip (7%, 3/46), and one case each on the abdomen, ear, flank, neck, and oral cavity. There were 3 cats for which the location of the neoplasm was not reported.
KIT immunoreactivity
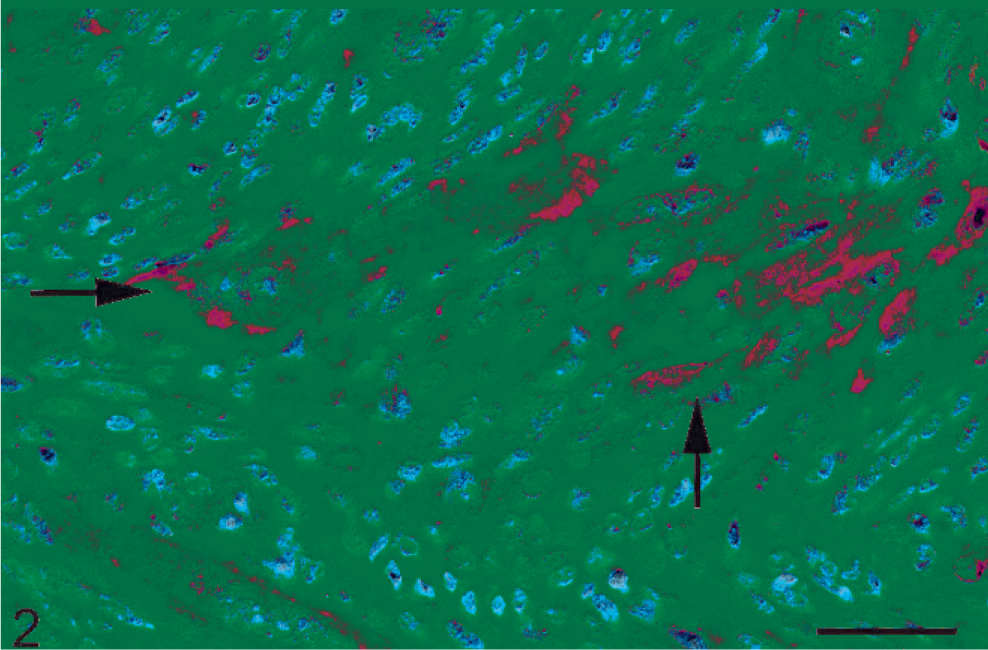
Approximately 9% (4/46) of the ST FSAs had KIT immunoreactivity within over 80% of the neoplastic cells (Fig. 1). Seventeen percent (8/46) of the ST FSAs had KIT immunoreactivity in less than 10% of the cells (Fig. 2). Immunoreactivity was not seen in the remaining 34 ST FSAs (74%). The immunoreactivity was characterized by evenly distributed cytoplasmic stippling within the neoplastic spindle-shaped cells and/or multinucleated giant cells, which is consistent with a type-II staining pattern. 33

Soft tissue fibrosarcoma; cat. KIT immunoreactivity is noted as intense stippling throughout the cytoplasm of neoplastic spindle cells, consistent with KIT staining pattern II (arrows). Streptavidin-horseradish peroxidase for KIT counterstained with Mayer's hematoxylin. Bar = 100 µm.
Soft tissue fibrosarcoma; cat. KIT immunoreactivity is noted in the cytoplasm of multinucleated giant cells within the neoplasm, consistent with KIT staining pattern II (arrows). Streptavidin-horseradish peroxidase for KIT counterstained with Mayer's hematoxylin. Bar = 100 µm.
Correlation of KIT immunoreactivity to histologic grade
There was no significant difference between KIT immunoreactivity within the high and intermediate histologic grade ST FSAs compared with low histologic grade ST FSAs, independent of whether the neoplasms with less than 10% immunoreactivity were included in the statistical analysis (Table 1).
Statistical correlation between ST FSAs with and without immunoreactivity for histologic grade, survivability, and whether the neoplasm was considered an ISS. Statistical analysis was performed comparing cases without immunoreactivity (n = 34) to those with greater than 80% immunoreactivity (n = 4). Statistical analysis was also used to compare cases without immunoreactivity to all cases with immunoreactivity (n = 12). Seventeen cases of ISS were included in this study. A P value <.01 was considered statistically significant.∗

∗HI = greater than 80% immunoreactivity; LI = less than 10% immunoreactivity; ISS = injection site sarcoma.
Correlation of KIT immunoreactivity to disease-free interval and survivability
Of the 46 cats included, only 33 had follow-up information regarding local recurrence. Of these 33 neoplasms, 8/12 high histologic grade, 8/13 intermediate histologic grade, and 1/8 low histologic grade ST FSAs had local recurrence. The mean disease-free interval was 154 days for high-grade, 203 days for intermediate-grade, and 306 days for low-grade ST FSAs. There were no significant differences in disease-free interval between the histologic grades (P = .300).
Information on survivability was available on 30 cats. The mean days of survival was 284 days for high histologic grade, 303 days for intermediate histologic grade, and 267 days for low histologic grade ST FSAs. No significant difference was found between ST FSAs with KIT immunoreactivity and those without, with respect to whether the cat was alive or dead as of June 2008 (Table 1). Comparison of mean survival time in days showed no significant difference between those cats with no immunoreactivity, those with less than 10% immunoreactivity, and those with greater than 80% immunoreactivity (Table 2).
Student's t-test results comparing mean survival in days among feline soft tissue fibrosarcomas grouped by immunoreactivity to KIT.∗

∗HI = greater than 80% immunoreactivity (n = 4); N = no immunoreactivity (n = 34); LI = less than 10% immunoreactivity (n = 8); AI = all immunoreactivity (n = 12).
Correlation of KIT immunoreactivity to diagnosis of ISS
Of the 46 cats included in this study, 17 ST FSAs were considered to be ISS. No significant difference in KIT immunoreactivity was found between ISS and the other ST FSAs in this study (Table 1).
Discussion
KIT immunoreactivity is frequently used to confirm the diagnosis of GIST in people and dogs and is also used as a tool for prognostication of MCT in dogs. 8,20,33 KIT immunoreactivity has been demonstrated in feline MCT, mammary gland adenoma and carcinoma, GIST, and granulosa cell tumors. 27 The results of this study indicate that ST FSA should be added to the list of feline neoplastic tissues with known KIT immunoreactivity. As the list of KIT immunoreactive cells increases, interpretation of KIT IHC in soft tissue neoplasms as a diagnostic tool for determining cell of origin must be made with increasing caution.
ST FSA in cats is a locally invasive neoplasm, which is usually treated with aggressive surgical excision with or without adjunctive therapy such as radiation or chemotherapy. 7,31 The decision of whether to use an adjunctive therapy is often based on histologic grading of the neoplasm. 7 The most widely used grading scale is that described by Kuntz et al. for soft tissue sarcomas in dogs. 22 This scale categorizes the neoplasms as low, intermediate, or high grade based on specific histologic features including mitotic rate, degree of cellular differentiation, and the presence of necrosis and/or hemorrhage. The results of this study show no correlation between KIT immunoreactivity and the histologic grade of ST FSA.
No significant correlation was found between KIT immunoreactivity and survivability. This finding indicates that factors other than KIT expression contribute to the biologic behavior of this neoplasm. Of interest, the cats with high histologic grade FSA had a high mean survivability (284 days) though the average disease-free interval was short in this group (154 days). This may be a reflection of aggressive, repeated surgical resection in cases of high-grade neoplasms. The mean disease-free interval in cats with low histologic grade ST FSAs was high (306 days) though the mean survivability was lower (267 days). Two of the cats with low-grade neoplasms were euthanatized for disease unrelated to the neoplasm soon after diagnosis with an ST FSA, which likely skewed the average survivability for this group. When examining disease-free interval between the histologic grades, no significant difference was found. This finding is in stark contrast to previous studies, which demonstrated prognostic value associated with histologic grading. 4,5,30,31 While the results of the present study are intriguing, it is difficult to draw any conclusions from that information as this was not the focus of the present study and confounding variables are almost certainly present.
Distribution of KIT protein within the immunoreactive cells correlates with prognosis in canine MCT. 33 Membranous and focal cytoplasmic localization of KIT (KIT staining patterns I and II) correlates with a favorable prognosis, while diffuse cytoplasmic localization correlates with a poor prognosis. 33 In the ST FSAs in this study, immunoreactivity in all positive neoplasms was characterized by evenly distributed cytoplasmic staining in both spindle cells and/or multinucleated giant cells (KIT staining pattern II). Though distribution of immunoreactivity appears to have a prognostic value in canine MCT, the same correlation was not found within the feline ST FSAs included in this study.
The final goal of this study was to determine if expression of KIT protein in ISS was different than that associated with other ST FSAs. Several studies have been published detailing common histologic features associated with ISS, including presence of inflammation, anaplasia, giant cells, and the presence of adjuvant within the neoplasm. 1,6,9 In addition, ISS tend to show increased frequency of local recurrence as well as increased likelihood of distant metastasis when compared with other ST FSAs, despite histologic grade. 14,31 There was no significant difference between KIT immunoreactivity, in terms of percentage of immunoreactive cells or pattern, in ISS and the other cases of ST FSA in this study. Therefore, it is unlikely that increased expression of KIT relates to the increased aggressive biologic behavior of ISS.
Of interest, mutations in c-KIT have been associated with higher grade MCT in dogs. 34,36 Mutations in c-KIT have also been demonstrated in GIST in people. 15,16 These mutations allow autophosphorylation of the receptor, independent of ligand interaction, and result in unregulated activation. 16,23,28 Tyrosine kinase inhibitors are a targeted therapy that is being successfully utilized for GIST in people and is in clinical trials for canine MCT. 11,18,21,24,32 In one case report, a case of feline MCT was treated successfully with a tyrosine kinase inhibitor (imatinib mesylate) after identification of a c-KIT mutation from neoplastic mast cells. 17 The possibility remains that mutations in c-KIT may have some role in the behavior of a subset of feline ST FSAs that express a mutated KIT protein. Further work is required to determine if these mutations exist in feline ST FSA, and if targeted therapies, such as tyrosine kinase inhibitors, may be beneficial in the treatment of this neoplasm.
In summary, KIT expression has been demonstrated in feline ST FSAs with IHC. Based on this finding, KIT immunoreactivity should not be considered specific for MCTs when IHC is used to differentiate between feline soft tissue sarcomas. In the cases of ST FSA included in this study, KIT immunoreactivity did not correlate with histologic grade, survivability, or a diagnosis of ISS. These results suggest that factors other than expression of KIT play a role in neoplastic transformation and proliferation in feline soft tissue fibrosarcoma.
Footnotes
Acknowledgements
We thank Darlene Giracello for assistance with the sample preparation and KIT IHC. This study was completed through the Oklahoma State University's (OSU) Veterinary Research Scholars Program. This program is supported by competitive grants from the National Institutes of Health, the Merck-Merial Veterinary Scholars Program, the Morris Animal Foundation, and by the Center for Veterinary Health Sciences at OSU.
