Abstract
Expression of histamine, serotonin, and KIT was evaluated in 61 archived feline mast cell tumors (MCTs) from the skin (n = 29), spleen (n = 17), and gastrointestinal (GI) tract (n = 15) using immunohistochemistry. Twenty-eight percent of cutaneous MCTs, 18% of splenic MCTs, and 53% of GI MCTs displayed histamine immunoreactivity. Serotonin immunoreactivity was detected in 3 GI and 1 cutaneous MCT. Sixty-nine percent of cutaneous MCTs, 35% of splenic MCTs, and 33% of GI MCTs were positive for KIT. Expression of these biogenic amines and KIT was less common than expected. Results of this study suggest heterogeneity in feline MCTs based on anatomic location. Further studies are needed to explain the significance of these differences.
Mast cell tumors (MCTs) are common in cats, arising primarily in the skin, spleen, and gastrointestinal tract. In each of these locations, they exhibit a different clinical presentation, cellular morphology, and biologic behavior.1,3,6 To better understand differences in feline MCTs based on anatomic location, we describe in this study the expression of histamine, serotonin, and KIT in cutaneous, splenic, and gastrointestinal (GI) MCTs using immunohistochemistry.
Archived biopsy specimens from cats with histologically confirmed MCTs were identified at the University of Georgia College of Veterinary Medicine and Athens Veterinary Diagnostic Laboratory, University of California–Davis School of Veterinary Medicine, Cornell University College of Veterinary Medicine, Louisiana State University School of Veterinary Medicine, and Purdue University School of Veterinary Medicine. Inclusion criteria were histologic diagnosis of MCT of the skin, spleen, or intestine and adequate tissue for evaluation. Samples from patients with multifocal MCT with no identifiable primary location were excluded. All samples had been fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Hematoxylin and eosin–stained sections of each tumor were reviewed by a single pathologist (EWH) to confirm the diagnosis and that samples were adequate for immunohistochemistry (IHC). Any tumor with a questionable diagnosis was stained with Giemsa, and those without a conclusive diagnosis were excluded.
Staining for histamine, serotonin, and KIT was performed on deparaffinized 4-μm sections with a DakoCytomation autostainer (DakoCytomation, Carpinteria, CA). Sections were treated with hydrogen peroxide (3%) for 5 minutes for inhibition of endogenous peroxidase. Antigen retrieval was not performed for histamine and serotonin. For KIT, antigen retrieval was performed by heating sections in a commercial pH 6.0 citrate buffer (BioGenex, San Ramon, CA). Nonspecific binding was blocked using commercial buffered casein (BioGenex) for 5 minutes. The primary antibodies used in this study were polyclonal rabbit antihuman histamine (ab43870, 1:500; Abcam, Cambridge, MA; 1:500), polyclonal rabbit antihuman serotonin (ab10385, 1:400; Abcam), and polyclonal rabbit antihuman CD117/c-KIT (1:300 or 1:400; Dako, Glostrup, Denmark).8,9 Secondary biotinylated goat antirabbit antibodies (Vector Laboratories, Burlingame, CA), streptavidin conjugated to horseradish peroxidase, and diaminobenzidine (DAB) as the chromogen substrate were used to visualize bound primary antibodies. Samples were counterstained with Gills II hematoxylin. The positive control for the histamine antibody was skin from a cat with a hypersensitivity reaction containing large numbers of mast cells. Positive controls for the serotonin antibody were human appendix and normal feline small intestine (specifically the enterochromaffin cells); enterochromaffin cells served as internal positive controls for GI MCTs. Positive controls for the KIT antibody were a KIT+ canine cutaneous MCT and a KIT+ feline cutaneous MCT. Negative controls for all antibodies used negative control rabbit IgG (Biocare Medical, Concord, CA) in place of primary antibody.
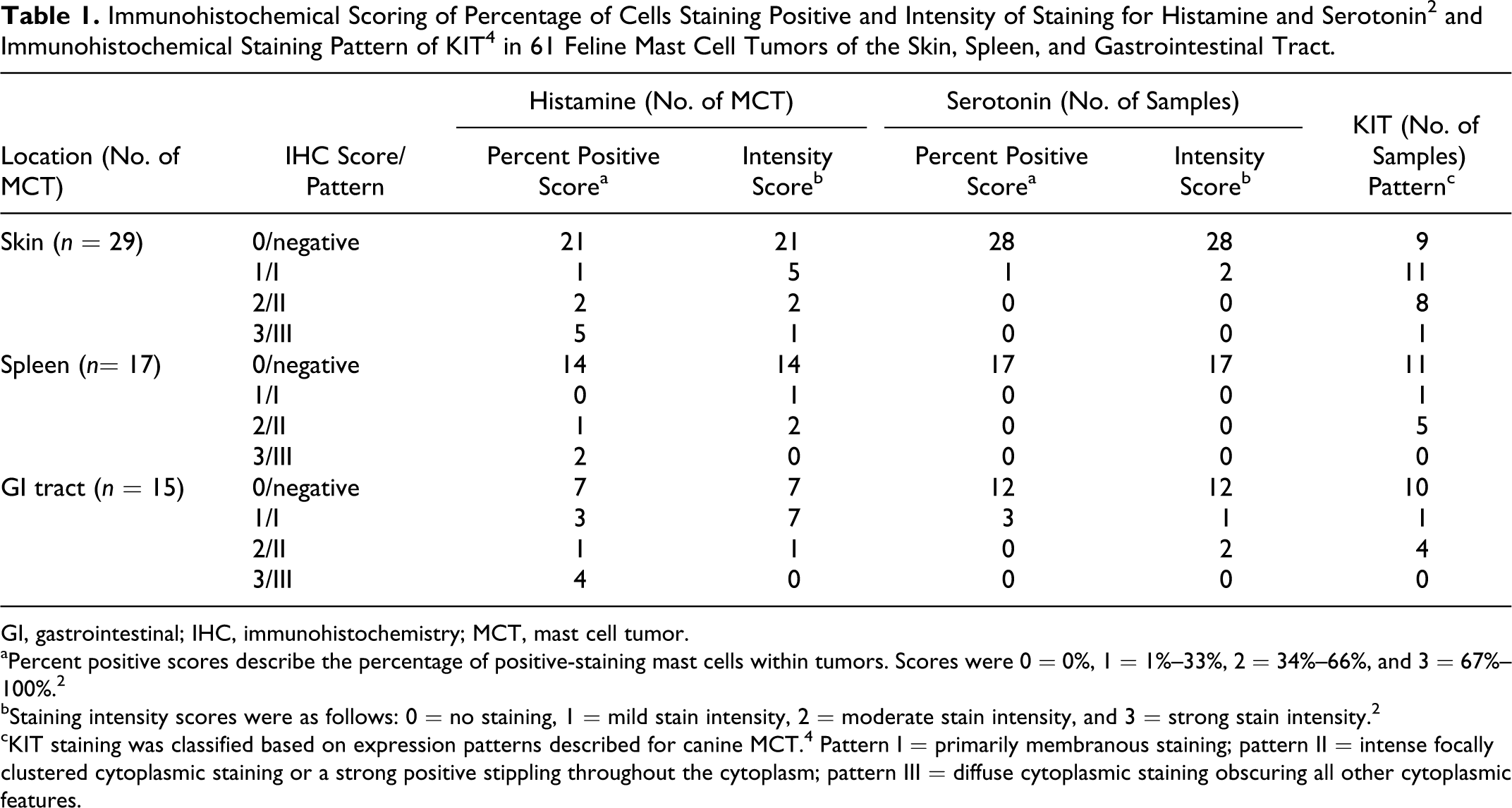
Slides were evaluated by the primary investigator (CLM) and a single pathologist (EWH). For histamine and serotonin, a staining classification system based on the percentage of positive cells and the intensity of the staining was used (modified from Doré et al 2 ; Table 1). For KIT, staining was classified using patterns described for canine MCTs (Table 1). 4
GI, gastrointestinal; IHC, immunohistochemistry; MCT, mast cell tumor.
aPercent positive scores describe the percentage of positive-staining mast cells within tumors. Scores were 0 = 0%, 1 = 1%–33%, 2 = 34%–66%, and 3 = 67%–100%. 2
bStaining intensity scores were as follows: 0 = no staining, 1 = mild stain intensity, 2 = moderate stain intensity, and 3 = strong stain intensity. 2
cKIT staining was classified based on expression patterns described for canine MCT. 4 Pattern I = primarily membranous staining; pattern II = intense focally clustered cytoplasmic staining or a strong positive stippling throughout the cytoplasm; pattern III = diffuse cytoplasmic staining obscuring all other cytoplasmic features.
Seventy samples were identified. Tissues were obtained between 1985 and 2009. Nine tumors were excluded; the primary site could not be determined for 5, and 4 could not be confirmed as MCTs. Sixty-one MCTs were evaluated: 29 cutaneous, 17 splenic, and 15 GI. None of the samples evaluated were consistent with sclerosing GI MCTs.
Scoring of IHC for histamine and serotonin and KIT staining patterns is presented in Table 1, with examples shown in Figs. 1 to 6. Eight cutaneous, 8 GI, and 3 splenic MCTs expressed histamine. Three GI and 1 cutaneous MCT expressed serotonin. Normal and hyperplastic mast cells (MCs) in feline skin did not stain for serotonin. Twenty cutaneous, 6 splenic, and 5 GI MCTs expressed KIT. The majority of KIT-positive tumors exhibited pattern I or II staining; however, most positive splenic and GI MCTs exhibited pattern II staining. Normal feline cutaneous MC had mild membranous KIT staining.
On the basis of the theory that MCTs develop from dysregulation of resident tissue MC, we expected differential expression of histamine and serotonin based on tumor site; however, our data failed to confirm this suspicion. The biochemical composition of feline MC and MCT granules is poorly understood. It has been suggested that serotonin may be the predominant biogenic amine in the MC granules of cats. 7 The present study failed to support this contention, as normal and hyperplastic MCs in the feline hypersensitivity reaction tissue did not stain for serotonin, and only 4 MCTs had positive staining for serotonin. In addition, only one-third of the tumors were positive for histamine. It is possible that formalin fixation hindered our ability to detect serotonin and histamine; through cross-linking of proteins, aldehyde solutions potentially block binding and mask detection of amines. 5 Therefore, use of antigen retrieval may have improved our limits of detection. However, when optimizing the protocol, we found that these techniques resulted in less effective amine staining. In addition, histamine staining was noted in the similarly fixed control tissue, and serotonin staining was noted in enterochromaffin cells of the GI MCTs, suggesting fixation was unlikely a problem. A final consideration is reliability of the chosen antibodies. As all antibodies exhibited strong staining in feline positive controls, we believe it is unlikely that different antibodies would have increased detection of histamine or serotonin.
At 51%, the percentage of KIT-positive MCTs in this study was low. A higher percentage of cutaneous MCTs were positive, but at 69%, this was still lower than previously reported.9,10 Reasons for the lower percentage are unclear. The antibody used was the same across studies and was independently validated in our laboratory using positive and negative controls. One explanation could be excessive tissue fixation hindering antibody binding, as these tissues were collected at multiple institutions, and fixation techniques were potentially different. In addition, to our knowledge, this is the only report clearly including visceral MCTs, and it is possible that KIT is differentially expressed based on anatomic location. Finally, it is also possible that this protein is less prevalent in feline MCTs than previously reported.8,9
In conclusion, results of this study suggest that histamine and serotonin are uncommonly expressed in feline MCTs, and further study of the biochemical composition of normal feline MCs and MCTs from different locations is indicated. KIT expression was lower than expected, and further studies are needed to evaluate KIT expression in normal feline MCs and MCTs. This descriptive study did not attempt to correlate expression of KIT with prognosis. Further evaluation of c-KIT mutations in feline MCTs and correlation with tumor behavior are indicated.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funded by the Georgia CaRES (Cancer Research, Education, and Service) for Pets Fund.