Abstract
The c-kit receptor is responsible for transmission of promigration signals to melanocytes; its downregulation may be involved in malignant progression of human melanocytic neoplasms. Expression of this receptor has not been examined in normal or neoplastic melanocytes from dogs. In this study, 14 benign dermal and 61 malignant mucosal melanocytic tumors were examined for c-kit (KIT) expression. Sites of the mucosal melanomas were gingiva (not further specified; n = 30), buccal gingiva (n = 6), soft palate (n = 4), hard palate (n = 5), tongue (n = 7), lip (n = 6), and conjunctiva (n = 3). Melan A was expressed in all 14 dermal melanocytomas and in 59 of 61 (96.7%) tumors from oral or conjunctival mucosa, confirming melanocytic origin. C-kit receptor expression was strong and diffuse throughout the cytoplasm in all 14 dermal melanocytomas and was identified in basilar mucosal melanocytes over submucosal neoplasms (27 of 61, 44.3%), junctional (neoplastic) melanocytes (17 of 61, 27.9%), and, less commonly, neoplastic melanocytes of the subepithelial tumors (6 of 61, 9.8%). KIT expression anywhere within the resected melanomas correlated with significantly longer survival. These results suggest that c-kit receptor expression may be altered in canine melanomas and may have potential as a prognostic indicator for mucosal melanomas.
The pathobiology of mucocutaneous melanomas in humans differs from that of cutaneous counterparts. 1,6,9 Similarly, canine mucosal melanomas are often more aggressive than most cutaneous melanocytic tumors. 4 Identification of a prognostic marker in canine mucosal melanomas would help with treatment decisions and facilitate targeted therapy. For example, use of the c-kit inhibitor toceranib phosphate (Palladia, Pfizer Animal Health, New York, NY) has improved outcomes in canine mast cell tumors that express KIT.
In humans, the c-kit gene, which encodes a tyrosine kinase receptor (KIT, CD117), may be expressed, overexpressed, and/or mutated in mucocutaneous melanomas. 2,6,9 The most frequently mutated exons include 11, 13, 17, and 18, but mutations were not detected in cutaneous melanocytomas. 1 The goal for the current study was to characterize expression of KIT in canine cutaneous melanocytomas and mucosal melanomas to understand the potential role of c-kit in tumorigenesis.
Materials and Methods
Cases of oral or conjunctival melanoma—submitted from January 2008 through June 2010 by the small animal hospital of the University of Tennessee College of Veterinary Medicine or from the referral biopsy service—were retrieved from the college’s pathobiology archives. All hematoxylin and eosin–stained sections were reviewed by a pathologist (S.J.N.) to confirm the diagnosis. Melan A—an immunohistochemical marker within the tyrosinase pathway, positive in 95% of canine oral melanomas 4 —was used to confirm cell of origin.
Signalment for each patient and size and location of the tumor were determined by review of submission forms and histopathology trim sheets. Twelve cases from the college had complete medical records available. For the remaining 48 dogs, surveys were sent to the referring veterinarians to obtain information on treatment, recurrence, survival data, and so on. Thirty-two were returned (66.7%), and in the remaining 16, an additional phone call to the clinic provided the necessary information. Survival time was recorded as days from diagnosis to the time of death or euthanasia, when known, with the assumption that death or euthanasia was a result of melanoma. Survival was recorded as the 15th of the month in which dogs were reported dead/euthanized, unless the specific date was recorded. For dogs that were lost to follow-up, survival time was calculated to the last date of observation by the veterinarian. Of 60 dogs, 15 (25%) were lost to follow-up; 6 survived zero days after diagnosis; and 1 case was diagnosed at necropsy. Dogs with unknown mortality were eliminated from the survival analysis; dogs lost to follow-up or alive at the end of the study were right-censored.
One representative 5-μm tissue section from each case was placed on charged glass slides for immunohistochemical detection of Melan A, S100, and c-kit (CD117). Slides were air-dried and heated at 60°C for 15 minutes before being deparaffinized with xylene and rehydrated through graded alcohols to deionized water. Following antigen retrieval (Table 1), all slides were soaked in Tris-buffered saline (pH 7.6) for 10 minutes, incubated in 3% hydrogen peroxide for 5 minutes, and processed using an automated stainer (Dako, model S3400). A 10-minute alkaline phosphatase block (Dako, Dual Endogenous Enzyme Block) and a 5-minute nonserum protein block (Dako) were used on Melan A and c-kit slides, respectively. A 5-minute 3% hydrogen peroxide block, followed by a 5-minute nonserum protein block, was used for the S100 slides. Last, slides were rinsed, counterstained with Harris hematoxylin, dehydrated, and coverslipped. For negative control slides, Universal Negative Control+ rabbit and mouse serum (Dako) was substituted for primary antibodies. Paraffin-embedded oral melanoma, cutaneous mast cell tumor, and peripheral nerve were used as positive controls for Melan A, c-kit, and S100, respectively. Immunohistochemical expression of c-kit was also evaluated in 14 archived canine gingival samples (nonneoplastic with variable pigmentation) and 14 dermal melanocytomas.
Immunohistochemistry Techniques

Statistical analysis was performed using SAS 9.2. The method of Kaplan-Meier (PROC LIFETEST) was performed to determine the effect of KIT expression and its location on survival—with location being anywhere in the section, in basilar melanocytes, in junctional melanocytes, in the submucosal portion of the mass, or in any of the subset of oral melanomas (excluding conjunctival melanomas). Data were right-censored on the basis of the last day that the subject was known to be alive; subjects were removed from analysis when no follow-up information was available. Values of P < .05 were considered significant.
Results
Sixty-one mucosal melanomas were evaluated from 60 dogs. Of the 61 tumors, 58 (95%) were oral, and 3 (4.9%) were conjunctival. Of the oral melanomas, 7 (10.3%) were lingual; 45 (79.3%) were gingival (6 in buccal gingiva; 5, hard palate gingiva; 4, soft palate gingiva; remainder, not further specified); and 6 (10.3%) were in labial mucosa. Twenty-six (44.8%) oral melanomas were well pigmented; 22 (37.9%) were poorly pigmented; and 10 (17.2%) were amelanotic. The most-pigmented oral masses were typically in the mucosa of the lip or hard palate. The 3 conjunctival melanomas were classified as amelanotic, poorly pigmented, or well pigmented.
All 14 dermal melanocytomas had strong cytoplasmic expression of Melan A. Melan A was also strongly expressed in the cytoplasm of 59 of 61 mucosal melanomas, including in the neoplastic melanocytes, in junctional melanocytes, and in basilar melanocytes within the overlying mucosa. To confirm melanocytic origin, 6 pleomorphic tumors were evaluated immunohistochemically for S100 protein expression. One tumor that was negative for Melan A had strong diffuse immunoreactivity for S100 protein. Another tumor that did not express Melan A or S100 protein had junctional activity, a histologic characteristic of melanocytic tumors, justifying its inclusion in the study.
C-kit expression was assessed for all 61 melanomas. Submucosal neoplastic melanocytes had moderate cytoplasmic expression in 6 tumors (9.8%). In 3 of these, no KIT expression was detected in basilar melanocytes of the overlying epithelium or in junctional neoplastic melanocytes (Fig. 1). KIT expression was most common in basilar melanocytes of the overlying mucosal epithelium, where it was recorded in 27 of 61 cases (44.3%) (Fig. 2). Of 61 cases, 17 (27.9%) had KIT expression in junctional melanocytes (Fig. 3). Thirty melanomas (49.2%) had KIT expression in at least 1 of these regions. Of the 6 labial melanomas, 2 had submucosal immunoreactivity; 2 had junctional immunoreactivity; 3 had basilar melanocyte immunoreactivity; and 4 had KIT immunoreactivity somewhere in the section. Of the remaining oral melanomas, 24 of 52 (48.1%) had KIT expression; 4 had submucosal reactivity (none of which were amelanotic); 23 had basilar melanocyte reactivity; and 15 had junctional melanocyte reactivity. Junctional melanocyte KIT expression was always accompanied by basilar melanocyte reactivity, but junctional or basilar melanocyte expression was detected in cases without reactivity in the submucosal neoplasm (Fig. 2) or in addition to reactivity in the submucosal neoplasm (Fig. 3). Of the 3 conjunctival melanomas, only 1 (an amelanotic variant) had basilar melanocyte KIT expression; submucosal neoplastic cells did not express KIT.
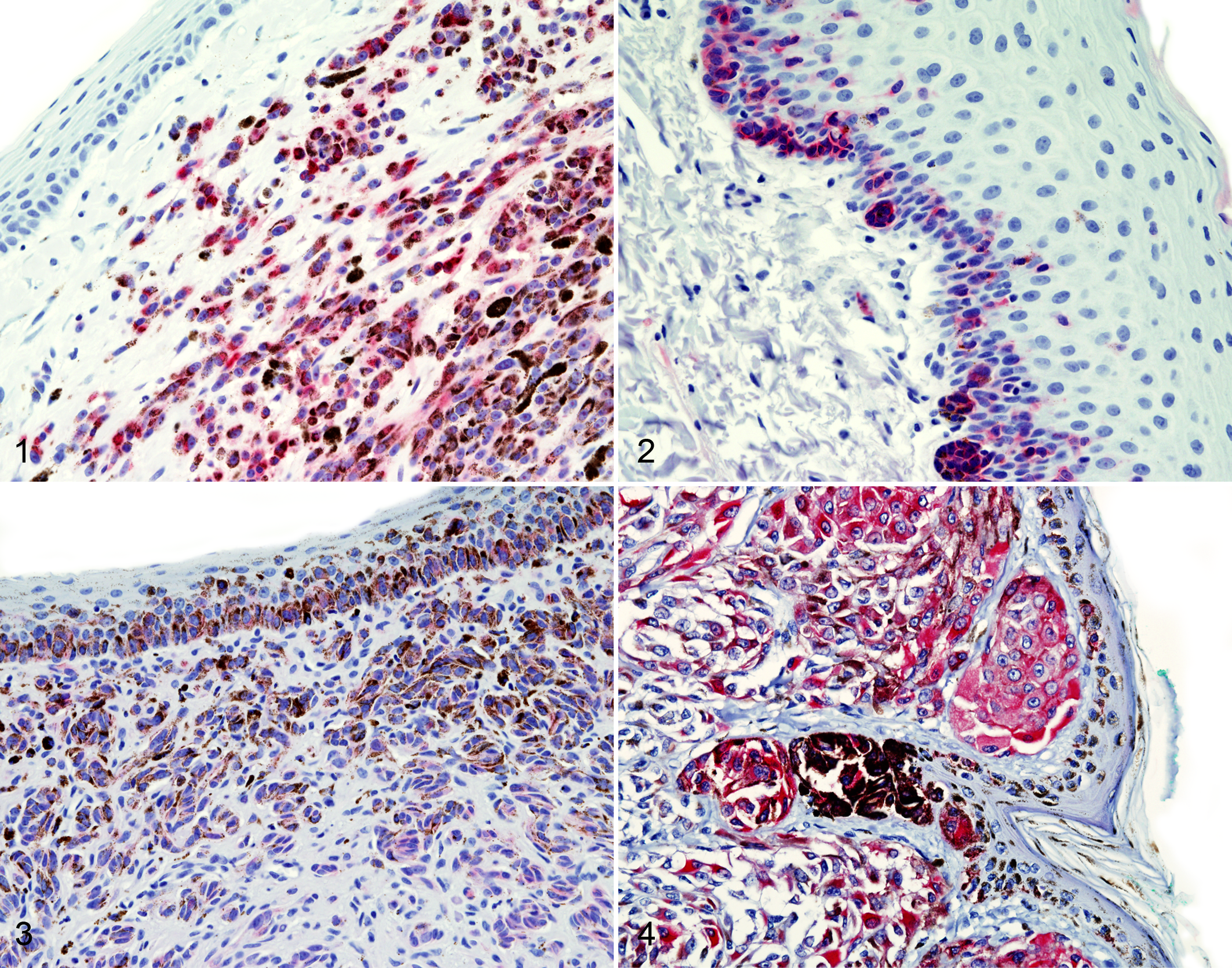
Oral melanoma; dog No. 1. Scattered strong cytoplasmic c-kit expression in neoplastic melanocytes. Vulcan Fast Red.
C-kit was not expressed in any of the 14 variably pigmented normal gingival samples. However, c-kit expression was strong and diffuse in all 14 dermal melanocytomas, including in junctional melanocytes and in basilar melanocytes of the epidermis (Fig. 4).
Statistical analysis for effect of breed, sex, age, and tumor site and size on survival was similar to that published for larger studies of canine melanoma. 4,7 suggesting that our study population was appropriate. Dogs with melanomas of the labial mucosa survived longer (310 days) than those with oral melanomas at other sites (123 days), although the difference was not statistically significant.
Survival was significantly associated with the presence of KIT (Table 2) anywhere in the section of a melanoma (submucosal neoplasm, junctional melanocytes, or basal layer melanocyte; Fig. 5), anywhere in any oral melanoma section (excluding conjunctival sites), and within basilar melanocytes overlying any melanoma. Expression of KIT in junctional melanocytes and in the submucosal neoplasm itself did not correlate significantly with survival.
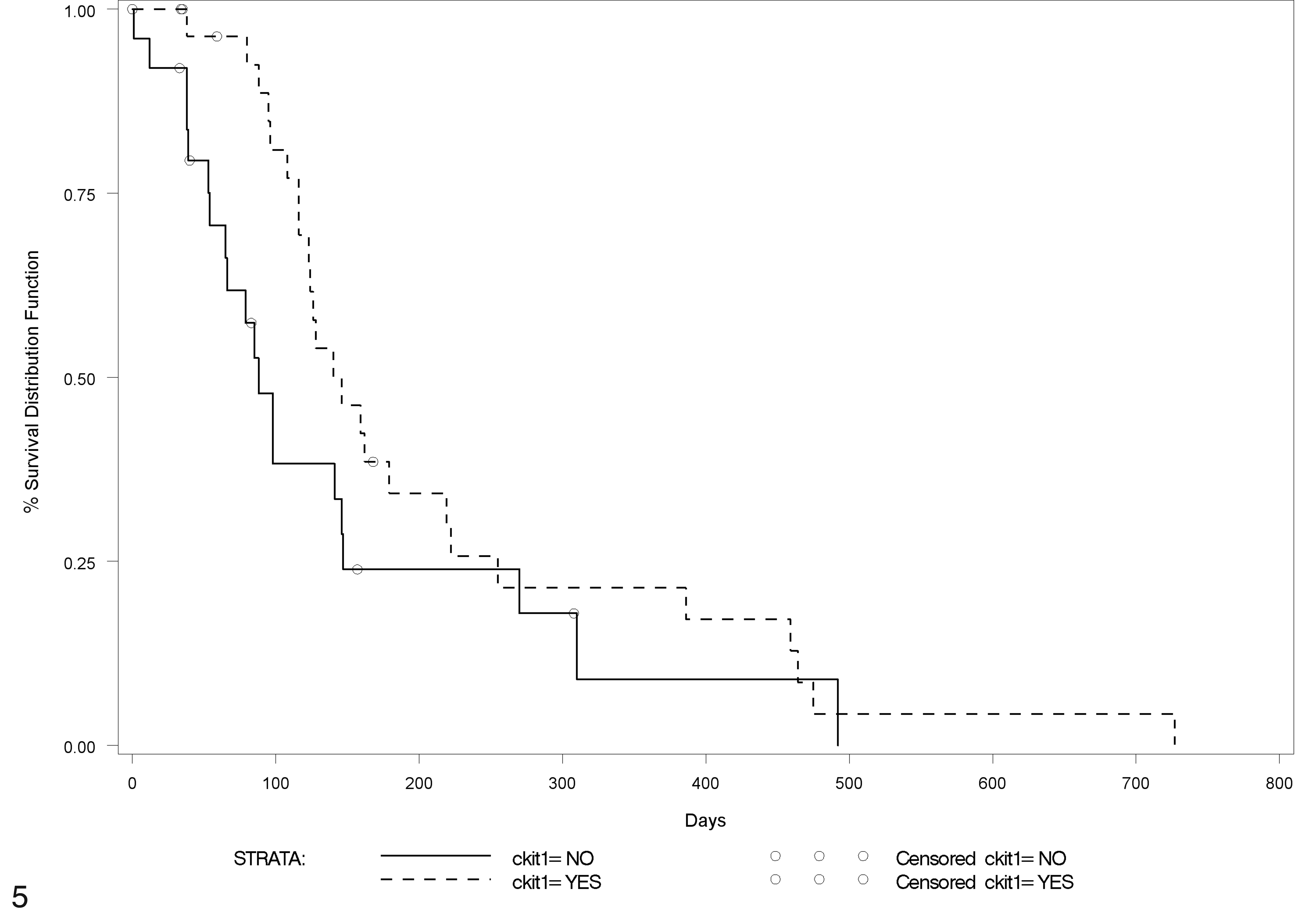
Survival curve for dogs with mucosal melanoma. Percentage survival is represented by the y axis. Median survival for dogs with KIT expression anywhere in the melanoma was 146 days; for dogs without KIT expression in the melanoma, 88 days (P = .01).
KIT Localization and Survival in Dogs With Melanoma
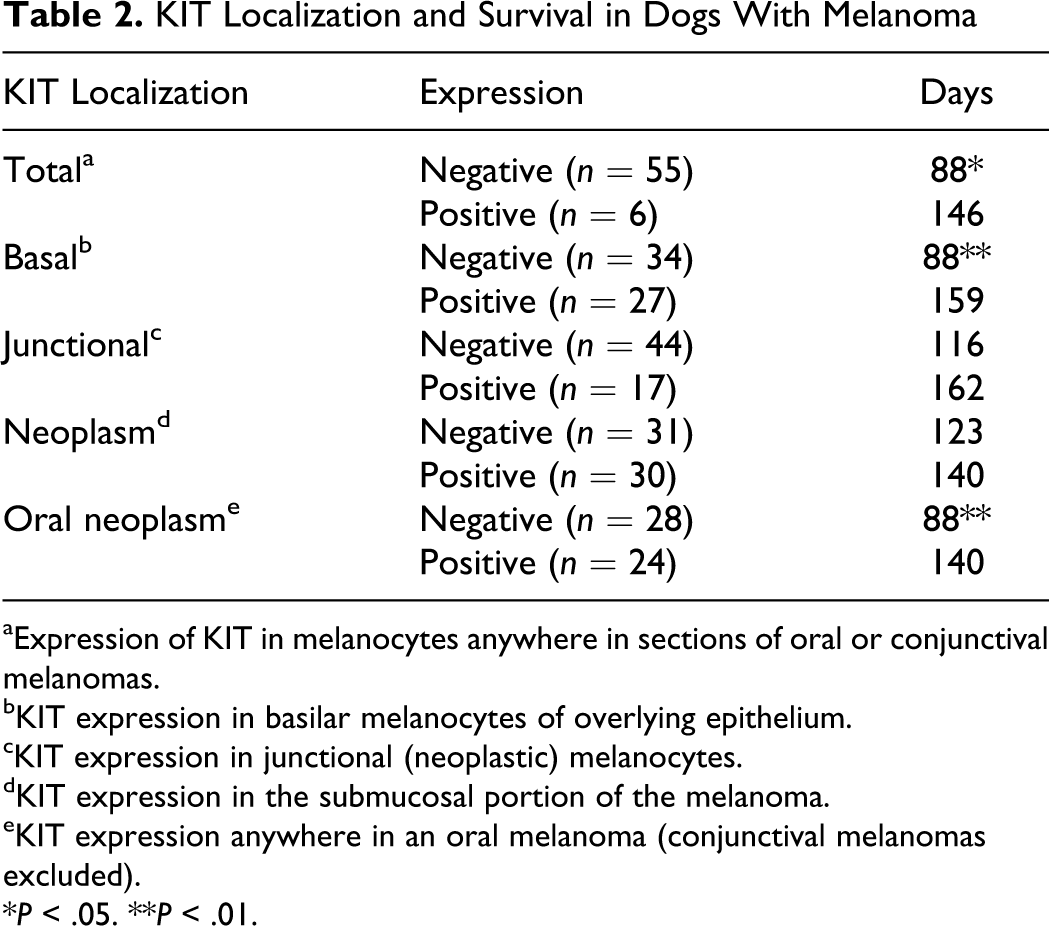
aExpression of KIT in melanocytes anywhere in sections of oral or conjunctival melanomas.
bKIT expression in basilar melanocytes of overlying epithelium.
cKIT expression in junctional (neoplastic) melanocytes.
dKIT expression in the submucosal portion of the melanoma.
eKIT expression anywhere in an oral melanoma (conjunctival melanomas excluded).
*P < .05. **P < .01.
Discussion
Melanoma is considered the most common malignant oral tumor in the dog, with an estimated prevalence of 12.7 per 10 000 tumor cases or 6% of all canine tumors. 4 The average age of dogs with oral melanoma is 10.4 years (range, 1–17 years). 4,7 The average age of dogs in our study was similar. Male dogs were overrepresented in some studies, 4 but our group had slightly more females (51.7%) than males. The gingiva and lip are the most commonly reported sites for oral melanoma in dogs, 4 accounting for 87.9% (51 of 58) of the tumors in our study. In our case series, 17% were amelanotic.
The mean postdiagnosis survival for dogs with oral melanoma is 3 to 10 months, 7 similar to that for dogs without KIT expression in our series. However, dogs with KIT expression in their tumor survived 50% longer.
Conjunctival melanomas are rare in dogs; the predilection site is the nictitating membrane. 3 The 3 conjunctival melanomas in this study (1 heavily pigmented, 1 lightly pigmented, and 1 amelanotic) all expressed Melan A. Only 1 tumor (amelanotic) expressed KIT, which was limited to basilar melanocytes.
KIT was expressed in submucosal neoplastic melanocytes in 9.8% of the melanomas in our study. However, KIT was expressed in basilar (nonneoplastic) melanocytes, in junctional (neoplastic) melanocytes, or in both cell populations in 27, 17, or 17 of the 61 melanomas, respectively. Junctional melanocyte KIT expression was always accompanied by expression in basilar melanocytes. Because KIT was not expressed in normal canine gingival melanocytes, it may be upregulated in proliferative lesions. The strong diffuse KIT expression in basilar melanocytes, junctional melanocytes, and dermal melanocytes in all 14 dermal melanocytomas could suggest a role for KIT in neoplastic transformation. Additionally, KIT expression was much more common in basilar and junctional melanocytes than in the invasive submucosal melanocytic masses; hence, KIT may be downregulated or lost with invasive progression.
The meaning of c-kit expression in human melanocytic neoplasms is unclear because increased expression has been seen in benign and malignant tumors. 2 Although c-kit is expressed in normal human melanocytes, 5 it is not expressed immunohistochemically in normal canine epidermal or gingival melanocytes. Human benign melanocytic nevi and in situ melanomas express c-kit, but c-kit expression is downregulated in dysplastic nevi, invasive melanomas, and metastatic melanomas. 2,8 Additionally, c-kit is expressed in junctional regions of benign compound nevi and in superficially spreading melanomas, but expression is lost in the vertical growth phase of invasive melanomas. 2 Apparently, c-kit expression can decrease during melanoma progression. 6,9
Differences in c-kit expression between benign and malignant melanocytic neoplasms were similar to those described in human tumors. C-kit expression was diffuse and strong in all dermal melanocytomas, but in malignant melanocytic tumors, c-kit expression was most common in basilar melanocytes (44% of tumors), less common in junctional melanocytes (27%), and least common in submucosal masses (9.8%). Junctional melanocyte c-kit expression was always accompanied by basilar melanocyte immunoreactivity, but the reverse was not true. Thus, canine melanomas may have a downregulation or loss of c-kit expression with increasing invasiveness, as in human melanomas. The complete lack of c-kit expression in melanocytes of the variably pigmented nonneoplastic gingival mucosal samples could indicate that c-kit expression is upregulated during melanocyte proliferation. Whether the mucosal melanocytes, especially those in the overlying basal layer, that expressed c-kit were neoplastic is not clear. However, melanocytes at the periphery of histologic sections rarely expressed c-kit, suggesting a possible location effect on those melanocytes in the epithelium overlying a melanoma. Survival was significantly prolonged by the presence of c-kit expression in canine melanomas. Thus, KIT status of canine melanomas could have prognostic value.
The differential expression of KIT between benign dermal and malignant mucosal canine melanocytic neoplasms could be the result of site-specific differences. Epidermal keratinocytes have membrane-type kit ligands, which are essential for the upregulation of c-kit expression in the epidermis and epidermal melanocytes. 2 In normal human skin, basilar melanocytes in the epidermis and hair follicle epithelium have strong c-kit expression. 9 In contrast, dermal melanocytes have much lower c-kit immunoreactivity than do dermoepidermal junction or epidermal melanocytes. 2 Therefore, c-kit loss in dermal melanocytes and dermal nevi might result from the dermal milieu downregulating c-kit expression or because dermal melanocytes have a different growth regulatory system. 9 Additionally, dermal melanocytes may be independent of KIT ligands for survival and proliferation. 2
Differential c-kit expression is also seen in human cutaneous acral lentiginous melanoma, in which basilar epidermal melanoma cells have strong immunoreactivity; yet, melanoma cells in the superficial epidermis or those growing vertically in the dermis do not express c-kit. 9 The c-kit-positive cells could represent an early stage of tumor progression. 9 We postulate a similar theory for the location-specific differential expression of c-kit in our canine melanocytic neoplasms.
Although c-kit expression may promote human melanocytic proliferation, in studies of ligand-independent activation of the receptor tyrosine kinase KIT in mice, constitutive signaling from the c-kit receptor exclusively promoted migration of melanocytes in vitro and in vivo and actually slowed cell cycle progression. Hence, c-kit may be linked to invasiveness in melanocytic neoplasms rather than to proliferation. 6
In this study, dogs with KIT expression anywhere in representative sections of their melanomas had significantly longer survival than did those without KIT expression. Fewer submucosal melanocytes expressed KIT than did the melanocytes in the overlying epithelium or those at sites of junctional activity; thus, c-kit may contribute to progression of melanomas. Additionally, c-kit may be downregulated as invasive growth progresses. If so, its absence in diagnostic samples could correlate with behavior. In conclusion, c-kit expression correlated with survival in dogs with mucosal melanomas, and immunoreactivity decreased with increasing distance from the overlying mucosa.
Footnotes
Acknowledgements
The Center of Excellence in Livestock Diseases and Human Health, University of Tennessee, supported J. M. Jankovsky in the center’s summer student research program. We thank the departments of pathobiology and small animal clinical sciences for additional financial support, A. Vasington for graphic support, M. Bailey for editorial assistance, and D. Durtschi for immunohistochemistry.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
