Abstract
Metastatic carcinoma of urogenital origin is a common cause of mortality in free-ranging California sea lions (Zalophus californianus). The etiology of this cancer is likely multifactorial, with viral infection, genetic factors, and exposure to environmental organochlorine contaminants possible contributing factors. In this study, expression of estrogen receptor α (ER α), progesterone receptor (PR), p53, and Ki67 were evaluated by immunohistochemistry in 12 sea lions with metastatic carcinoma, genital epithelial dysplasia, and intraepithelial neoplasia; 4 with genital epithelial dysplasia and intraepithelial neoplasia without metastases; and 6 control animals. Dysplastic and neoplastic lesions were identified in multiple areas of the cervix, vagina, penis, prepuce, and urethra in affected animals, suggesting multicentric development. Lesions were graded according to degree of epithelial dysplasia and infiltration and lesions of different grades were evaluated separately. Estrogen receptor expression was lower in intraepithelial lesions compared with normal genital epithelium, and expression in metastatic lesions was completely absent. There was progesterone receptor expression in neoplastic cells in intraepithelial lesions of all grades and in metastases, with no significant difference between lesion grades or between control and affected epithelium. Ki67 index and p53 expression increased with lesion grade and were higher in lesions than normal epithelium. Metastatic tumors exhibited highly variable morphology; however, proliferation index, ER α, PR, and p53 expression were similar in tumors with different patterns of growth. These results suggest that endogenous hormones, environmental contaminants that interact with steroid hormone receptors, and alterations in p53 may play a role in urogenital carcinogenesis in California sea lions.
Keywords
Introduction
Disseminated carcinoma of urogenital origin is a common cause of mortality in stranded adult California sea lions, whereas other types of tumors are much less common. 19,26 In one investigation, 66 of 370 (18%) subadult and adult sea lions necropsied after stranding along the central California coast over a 25-year period were diagnosed with metastatic carcinoma. 19 Both infiltrative tumors and intraepithelial neoplasia can be found in the cervix and vagina of affected females and penis and prepuce of affected males. 26 Metastases are found throughout pelvic and abdominal lymph nodes as well as abdominal and thoracic viscera and often can exhibit varying histologic patterns both within and between affected individuals. 19 Intraepithelial lesions, ranging from mild epithelial dysplasia to carcinoma in situ, have been noted in the genital tract of sea lions with metastatic carcinoma and in some sea lions dying from other causes without metastatic lesions (K. M. Colegrove and L. J. Lowenstine, personal observation). 26
Genital carcinomas and intraepithelial lesions in sea lions bear remarkable similarity to cervical neoplasia in women, including similarities in sites of distant metastases. 25 In women, it is widely accepted that cervical intraepithelial neoplasia represents a precursor lesion to invasive carcinoma, and there is a continuum between mild dysplastic lesions and invasive carcinoma. 33 Human papillomaviruses (HPV) are important for the development of many cases of cervical cancer; however, infection is not always sufficient to induce neoplasia. 43 Other factors such as cigarette smoking, endogenous hormones, oral contraceptive use, nutrition, and polymorphisms in chemical metabolizing genes are also thought to play a role in cervical carcinogenesis. 2,4,13,35,41
Several potential factors involved in genital carcinogenesis in sea lions have been previously investigated. 1,3,20,50 Intranuclear inclusions have been noted in some neoplastic cells, and herpes viral particles have been identified via transmission electron microscopy.19,26 A gammaherpesvirus in the genus Rhadinovirus, Otarine herpesvirus-1 (OtHV-1), has been identified in genital tumors and metastatic genital tumors and in one study was amplified via polymerase chain reaction (PCR) in 100% of those tumors examined. 19,22,26 More recently, OtHV-1 DNA was also amplified from lower genital tract tissue of male and female sea lions both with and without metastatic carcinoma but not in any other type of tumor. 6 Sea lions with genital carcinoma have been found to have a high internal relatedness factor, a measure of inbreeding determined through examination of polymorphic microsatellite DNA markers. 1 Additionally, the MHC class II locus Zaca-DRB.A has been shown to be strongly associated with an increased risk of urogenital cancer. 3 Bacterial infection of the lower genital tract is common in free-ranging sea lions, with β-hemolytic Streptococcus being statistically associated with carcinoma in female sea lions. 20 California sea lions are also exposed to organochlorines, such as polychlorinated biphenyls (PCBs) and dichloro-diphenyl-trichloroethane (DDT), in utero, through lactational transfer, and via contaminated food sources. 14,18,21 Some organochlorines are estrogenic and can act as endocrine disruptors as well as tumor initiators and tumor promoters. 23 Sea lions with genital carcinoma have higher blubber burdens of PCBs and DDT than sea lions without cancer. 50 Given these factors, it is highly likely that, as with human cervical cancer, genital carcinogenesis in sea lions has a multifactorial etiology.
Estrogen (ER) and progesterone receptors (PR) mediate the effects of endogenous hormones in target cells of the reproductive system. Some endocrine-disrupting effects of organochlorines also depend on chemical interaction with steroid hormone receptors causing transcriptional activation of hormone-responsive genes. Activation of ER and PR in hormone-sensitive tissues has been associated with exposure to PCBs and DDT. 9,39,48 Although the association between endocrine disrupting environmental contaminants and breast cancer is inconclusive, a recent study suggested that interaction between organochlorines and ER α led to upregulation of ER α and increased cell proliferation in hormone-responsive breast cancer in women. 5,32 The relationship among endogenous hormones, exposure to endocrine-disrupting organochlorines, and tumor formation in the hormone-sensitive sea lion genital tract is not currently known. Previous investigations revealed that both ER α and PR are expressed throughout epithelia and stroma in the unaffected sea lion reproductive tract, including the cervix, vagina, penis, and prepuce (K. M. Colegrove, unpublished data).
The tumor-suppressor gene p53 is involved in many types of cancer through regulation of cell proliferation, apoptosis, response to DNA damage, and genome stability. Cancer can develop through mutations or inactivation of p53 via interaction with viral oncogenes. 45 Specific chemically induced mutations have also been identified. 10 The potential involvement of steroid hormone receptors and p53 in the pathogenesis of sea lion genital carcinoma has not been previously examined. Additionally, cell proliferation has not been investigated in sea lion genital carcinomas. The objective of this investigation was to compare Ki67 index, ER α, PR, and p53 immunohistochemical expression among normal, dysplastic, and neoplastic sea lion genital epithelium and in metastatic tumors with different histologic patterns.
Materials and Methods
Animals and case selection
Twelve California sea lions (7 females and 5 males) with metastatic carcinoma, genital epithelial dysplasia, and intraepithelial neoplasia, 4 (2 females and 2 males) with genital epithelial dysplasia and/or intraepithelial neoplasia but no evidence of metastases, and 6 (3 females and 3 males) unaffected control animals were included in this investigation. All sea lions were adults that had stranded live on the central California coast and died or were euthanized during rehabilitation at The Marine Mammal Center, Sausalito, California. Clinical signs consistent with metastatic carcinoma have been previously reported. 19 Cause of clinical disease and/or death in sea lions with intraepithelial lesions but no metastases included trauma (N = 3) and pneumonia (N = 1). Causes of clinical disease and/or death in control sea lions included domoic acid toxicity (N = 2), trauma (N = 1), leptospirosis (N = 1), and pneumonia (N = 1). Animals selected as controls had no significant reproductive tract lesions based on gross and histologic examination. All sea lions included in this investigation received full necropsies completed within 12 hours of death or euthanasia, and the entire reproductive tract was examined grossly and submitted for histologic examination.
Histopathology and grading
Tissue samples from all major organs including the entire reproductive tract were fixed in 10% neutral buffered formalin and submitted to the Pathology Service, Veterinary Medical Teaching Hospital, University of California, Davis, California, for evaluation. After fixation, representative samples were taken from all major tissues and lesions noted on gross examination. In females, the following sections were routinely taken from the urogenital tract: midsagittal sections through the right and left ovary; transverse sections from each uterine horn, cranial vagina, middle vagina at the hymeneal fold, and urinary bladder; and longitudinal sections through the endocervix, ectocervix, and fornix. In males, sections included cross-sections of the glans penis, shaft of the penis with distal urethra, and prostate gland with urethra; and transverse sections of the prepuce and urinary bladder. Formalin-fixed samples were processed routinely for paraffin embedding, sectioned at 5 µm, and stained with HE for light microscopic examination. Epithelium within affected cervix, vagina, penis, prepuce, and urethra was graded according to degree of epithelial dysplasia, approximating established human grading schemes for cervical, vaginal, and penile neoplasia. 25 Lesions were classified as low grade intraepithelial lesions (LGIL) (Score 1) if there was mild dysplasia and proliferation was confined to the lower third of the epithelium. These lesions were similar to cervical intraepithelial neoplasia (CIN) 1 and vaginal intraepithelial neoplasia (VIN) 1 lesions in women and penile low-grade squamous intraepithelial lesions in men. 25,51 Lesions were given a classification of high-grade intraepithelial lesions (HGIL) (Score 2) if there was moderate to marked dysplasia and proliferation of atypical parabasal cells that involved between one third to the entire thickness of the epithelium. These lesions were similar to CIN 2, CIN 3, VIN 2, and VIN 3 lesions in women and high-grade squamous intraepithelial lesions in men. 25,51 Lesions were classified as invasive (Score 3) if there was distinct invasion beyond the epithelial basal layer into the underlying stroma, distinct from metastatic foci. 39 Metastatic tumors (Score 4) were classified as solid squamous, cribriform, adenosquamous, comedone, or basaloid. Solid squamous tumors had neoplastic cells arranged in sheets and lobules. Neoplastic cells had prominent intraepithelial bridges and variable evidence of keratinization. Cribriform lesions were composed of neoplastic cells lining small fenestrations often containing debris or mucous. Comedone lesions were composed of solid nodular tumor foci containing central regions of necrotic debris. Adenosquamous lesions contained both glandular and squamous components. Basaloid lesions were characterized by nests, cords, and ribbons of small oval cells with hyperchromatic nuclei, which resembled basal cell carcinoma of the skin.
Immunohistochemistry
Immunohistochemistry was performed on serial sections of select urogenital tissues and metastatic tumors from all cases. Animals without evidence of neoplasia or dysplasia were considered control animals. Affected and unaffected portions of reproductive epithelium were examined from animals with evidence of intraepithelial lesions or invasive cancers. After deparaffiniation, endogenous peroxidase activity was blocked by incubating sections in 0.2% H2O2 in methanol for 30 minutes. Antigen retrieval was accomplished for ER α and PR by heating sections to 95°C for 30 minutes in Citrate buffer (pH = 6.2; Dako Cytomation, Carpinteria, CA) in a rice steamer. For p53, sections were microwaved for 20 minutes in Citrate buffer (pH = 6.0; Dako Cytomation). Sections were incubated in 3% normal goat serum for 30 minutes. Serial sections were incubated with a monoclonal antibody to human ER α (1 : 125; 1D5, Immunotech, Marseille, Cedex 9, France), a monoclonal antibody to human PR (1 : 200;10A9, Immunotech), and a polyclonal antibody to p53 (1:200; CM1, Biocare Medical, Concord, CA) overnight at 4°C in a moist chamber. For Ki67, sections were incubated with the primary monoclonal antibody (1 : 75; MIB-1, Dako Cytomation). Slides were incubated with a biotinylated antimouse link reagent (Biocare Medical) for 10 minutes and then incubated with streptavidin horseradish peroxidase (Biocare Medical) for 10 minutes. Positive staining was visualized using 3-amino-9-ethylcarbazole (AEC) chromogen (Zymed Labs, San Francisco, CA). After all steps, sections were rinsed in phosphate buffered saline (PBS) spiked with polyoxyethylenesorbitan monolaurate (TWEEN 20, Sigma-Aldrich, Inc., St. Louis, MO). Sections of canine and sea lion uterus known to be positive for ER α and PR were included in each procedure as positive controls. Negative controls were sections incubated with omission of the primary antibody.
Immunostained sections representative of the range of genital and metastatic lesions present in each case were evaluated by a single person (KMC) without prior knowledge of the animal from which the tissue was sampled. In any given tissue, genital lesions given a separate neoplasia grade via light microscopy were evaluated separately for each immunhistochemical stain. The stroma underlying lesions of differing grades was also evaluated for ER α and PR. Three of the most well-preserved metastatic foci were evaluated for each animal. Representative areas from each tissue or lesion grade were divided into 5 regions of approximately equal surface area, and randomly selected areas within those regions were evaluated. For each region, 100 cells were evaluated for a grand total of 500 cells evaluated. Expression of ER α and PR was scored using a semiquantitative grading system. 12 For each tissue and lesion grade, a proportional score (PS) and an intensity score (IS) were established. The PS corresponded to the percentage of 500 cell nuclei that stained positive, where 0 = zero, 1 = <1%, 2 = 1–9%, 3 = 10–32%, 4 = 33–65%, and 5 = >% 66 positively stained nuclei. The IS reflected the subjectively evaluated intensity of positive, brown–red nuclear staining, where 0 = no staining, 1 = mild, 2 = moderate, and 3 = strong staining. A total score (TS) between 0 and 8 was then calculated by the following formula: TS = PS + IS. For p53 expression, the percentage of positively stained nuclei out of the 500 evaluated cells was determined. Tumor and intraepithelial lesions were considered positive if >10% of cells exhibited positive staining. 49 Ki67 index was calculated as a percentage of positively stained nuclei out of the 500 total evaluated cells.
Statistical analysis
Total ER and PR scores from different lesion grades in the cervix, vagina, penis, and prepuce were compared using the nonparametric Kruskal-Wallis test. If a significant difference was found, the Mann-Whitney test was used to determine which groups were significantly different from each other. The Ki67 index and p53 data were tested for equality of variances using Levene's test for equality of variances and for normality using the Kolmogorov-Smirnov test, with P < .05 set as the level of significance. Variances between groups were significantly different, and most data sets were not normally distributed; therefore, the Kruskal-Wallis and Mann-Whitney tests were also used to test for differences between groups. For all analyses, a P value of <.05 was considered significant. Statistical analysis could not be completed on groups with fewer than 3 samples using these procedures. Statistical calculations were performed using Medcalc statistical software, Version 9.1.0.1 1993 (Medcalc, Mariakerke, Belgium).
Results
Gross necropsy findings
On gross necropsy examination of sea lions with metastatic carcinoma, multiple tan neoplastic masses were noted in lymph nodes and abdominal and thoracic viscera, similar to previously described cases. 26 Seven animals (Nos. 3, 5, 10–14) had marked hydronephrosis and hydroureter caused by compression of the ureters by neoplastic masses infiltrating the sublumbar and renal lymph nodes. Four of 7 females (Nos. 1, 4–6) and 4 of 5 males (Nos. 11–14) with metastatic carcinoma had lesions noted in the reproductive tract during necropsy or during sectioning, after formalin fixation. In females, there were tan firm nodules or subtly raised, irregular plaques in the vaginal epithelium, and the cervix was mildly thickened (Fig. 1). In males, well-demarcated, pale tan, irregular plaques were noted along the epithelium of the penis and prepuce (Fig. 2). The transition zone between the penis and prepuce was affected in 3 males with metastatic carcinoma and 1 male with intraepithelial lesions only (Nos. 11–14). Often, subtle lesions became more appreciable after formalin fixation. Three females (Nos. 3, 5, and 6) had well-demarcated, circumferential bands of yellow to brown endometrium in 1 uterine horn, corresponding to the site of previous attachment of the zonary placenta.

Cervix and vagina; sea lion No. 4. Coalescing, raised, tan neoplastic masses expand the vaginal epithelium. Formalin fixed. Scale = 1.0 cm.
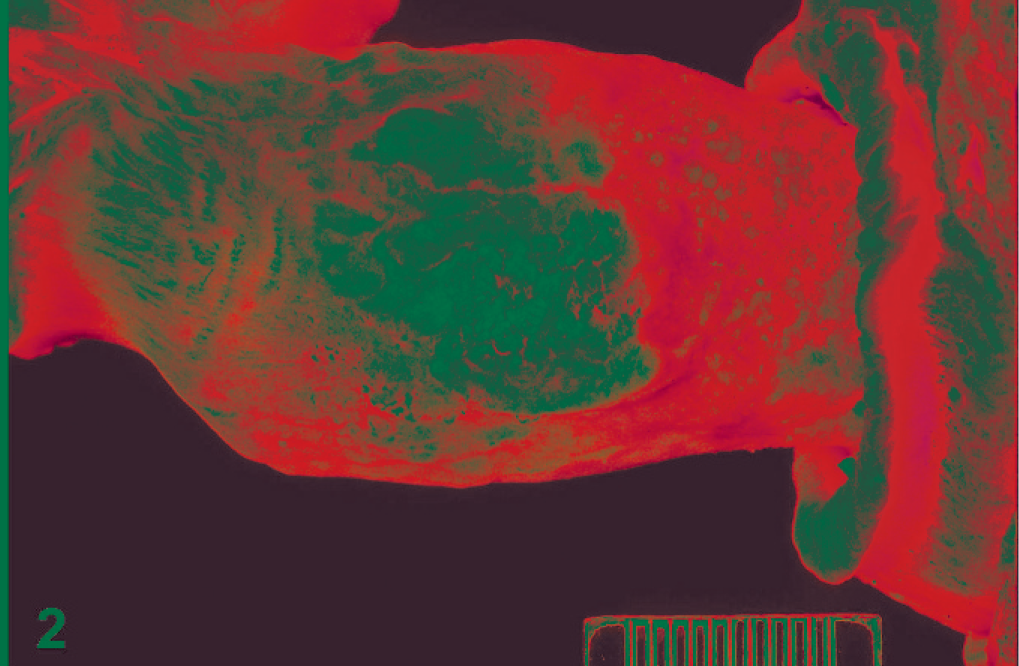
Penis; sea lion No. 11. A raised, irregular area of thickened penile mucosa. Formalin fixed. Scale = 1.0 cm.
Histopathologic findings
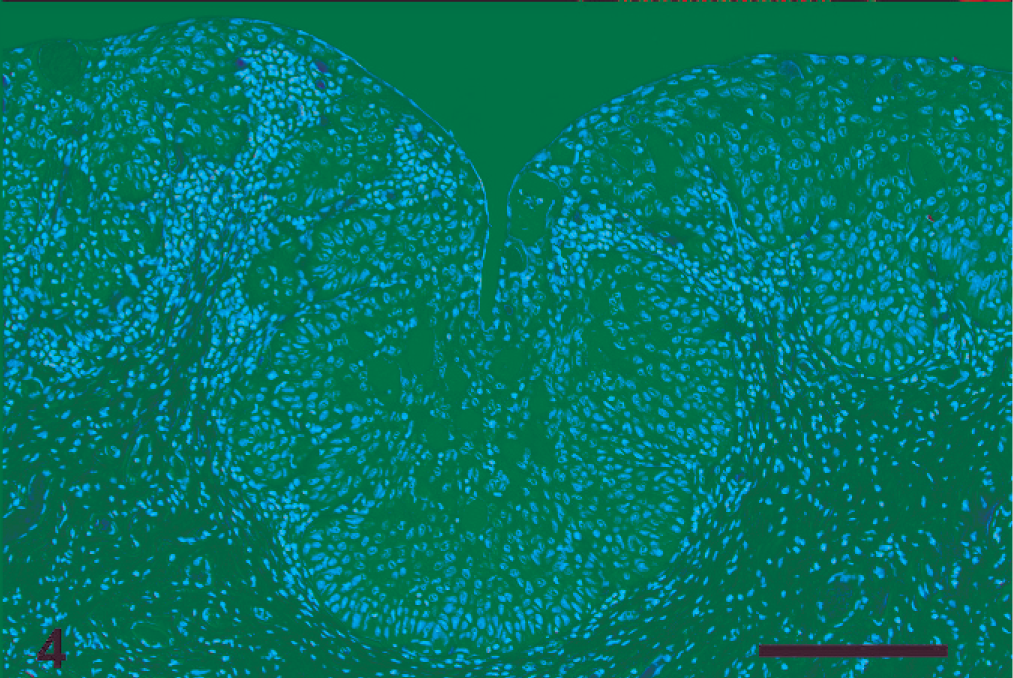
Intraepithelial lesions, with morphological features similar to those previously described, were noted in the middle vagina (N = 7), external cervix (N = 6), cranial vagina (N = 5), internal cervix (N = 4), and distal vagina (N = 3) in females with metastatic cancer (Table 1). 26 The penis (N = 5) and prepuce were similarly affected (N = 5) in males with metastatic carcinoma (Table 2). In some cases, there was a gradual transition between unaffected genital epithelium and LGIL; however, in other cases, HGIL occurred directly adjacent to normal epithelium (Fig. 3). Intraepithelial lesions occurred in multiple, often noncontiguous, locations along the reproductive tract in both males and females. Intraepithelial lesions in animals without evidence of metastasis were morphologically identical to lesions in those with metastatic carcinoma. Intranuclear inclusions, similar in morphology to the inclusions associated with OtHV-1 infection, were noted in genital lesions in the vagina of 1 female (No. 1) and the penis of 2 males (Nos. 10 and 14). In 3 females, there was cribriform change within intraepithelial lesions (Nos. 3, 4, and 7) in the external cervix and cranial and middle vagina (Fig. 4). In males, histologic evidence of invasion was rare; however, invasion was noted in varying locations in 6 of 7 females. In females and males with metastatic carcinoma, metastatic foci were found in many tissues, with 1 female having 12 separate metastates. In females, metastatic tumor morphology was highly variable both between and within affected individuals, and individual tumors often exhibited regionally different morphologic features. In males, the majority of metastatic foci had a solid squamous and comedone morphology. Tumors with an adenosquamous and basaloid morphology were less common than tumors with a solid squamous, comedone, and cribriform morphology.
Histopathologic findings in 7 adult female California sea lions with metastatic urogenital carcinoma (Nos. 1–7) and 2 adult females with genital epithelial dysplasia and/or intraepithelial neoplasia but no metastases (Nos. 8 and 9). Score 0 = not affected; 1 = LGIL; 2 = HGIL; 3 = invasive carcinoma; 4 = metastatic carcinoma.
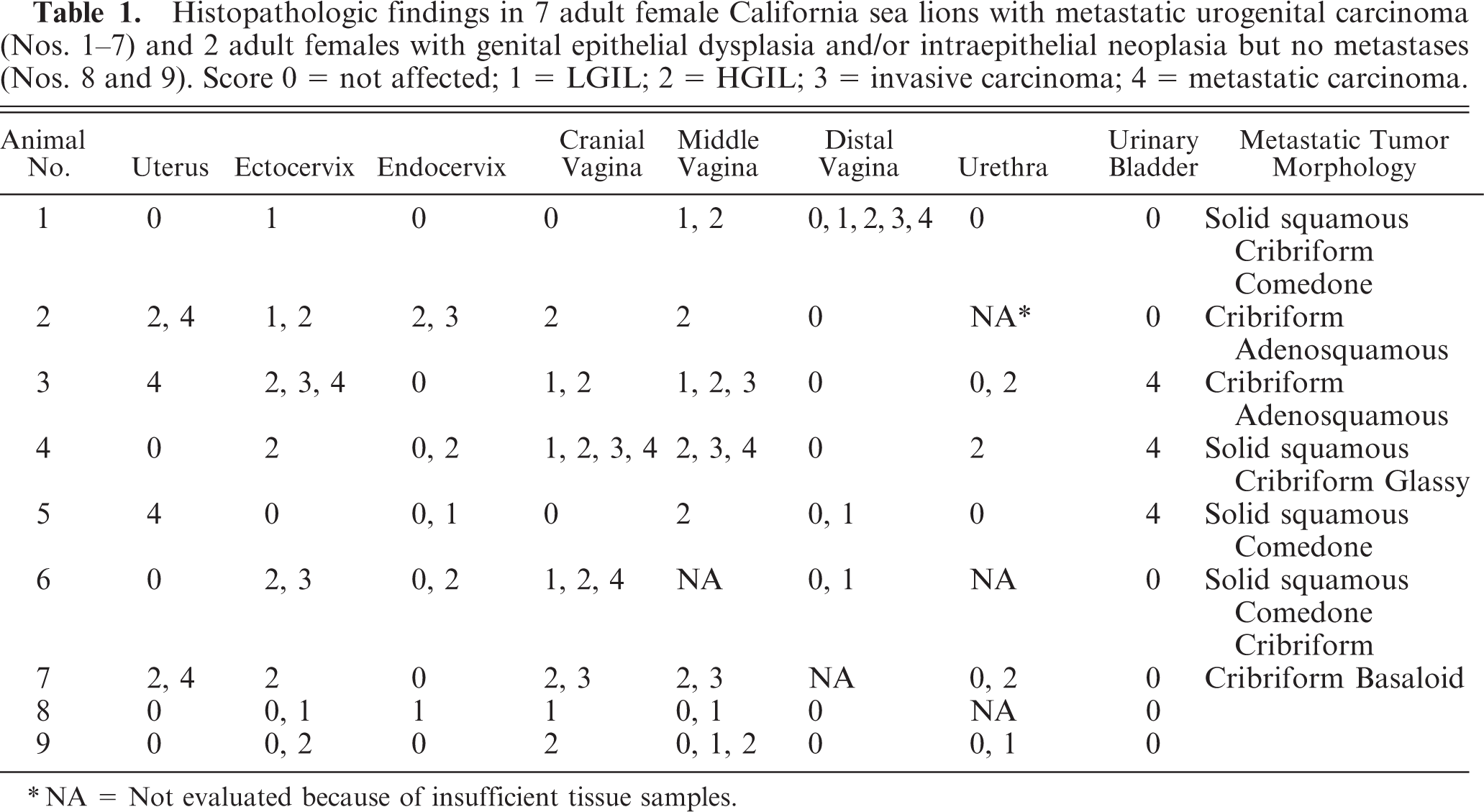
∗NA = Not evaluated because of insufficient tissue samples.
Histopathologic findings in 5 adult male California sea lions with metastatic urogenital carcinoma (Nos. 10–14) and 2 adult males with genital epithelial dysplasia and/or intraepithelial neoplasia but no metastases (Nos. 15 and 16). Score 0 = not affected; 1 = LGIL; 2 = HGIL; 3 = invasive carcinoma; 4 = metastatic carcinoma.

2NA = Not evaluated because of insufficient tissue samples

Middle vagina; sea lion No. 8. Transition zone (arrow) between normal epithelium on the left and LGIL on the right. HE. Bar = 100 µm.
Cranial vagina; sea lion No. 3. HGIL with a cribriform pattern. There is dysplasia throughout the entire thickness of the epithelium and proliferation of atypical basal cells. HE. Bar = 50 µm.
Primary neoplastic lesions were not noted in the urinary bladder in any sea lion examined. However, intraepithelial lesions identical to those noted in the genital epithelia were noted in the penile urethra in 2 males and in the urethra adjacent to the middle vagina in 4 females (Fig. 5). In 1 male (No. 13), urethral lesions were noted from the distal tip of the glans penis to the penis shaft approximately 10.0 cm from the distal tip in serial sections. At the distal end of the urethra, dysplastic epithelium was adjacent to transitional epithelium that exhibited moderate squamous metaplasia. Along the shaft of the penis, the penile epithelium overlying the urethra was unaffected. Serial sections of the urethra were not examined in other cases; therefore, the extent of the urethra lesions in other affected animals is not known.

Urethra; sea lion No. 3. Transition between normal urethral epithelium on the left and HGIL on the right. HE. Bar = 100 µm.
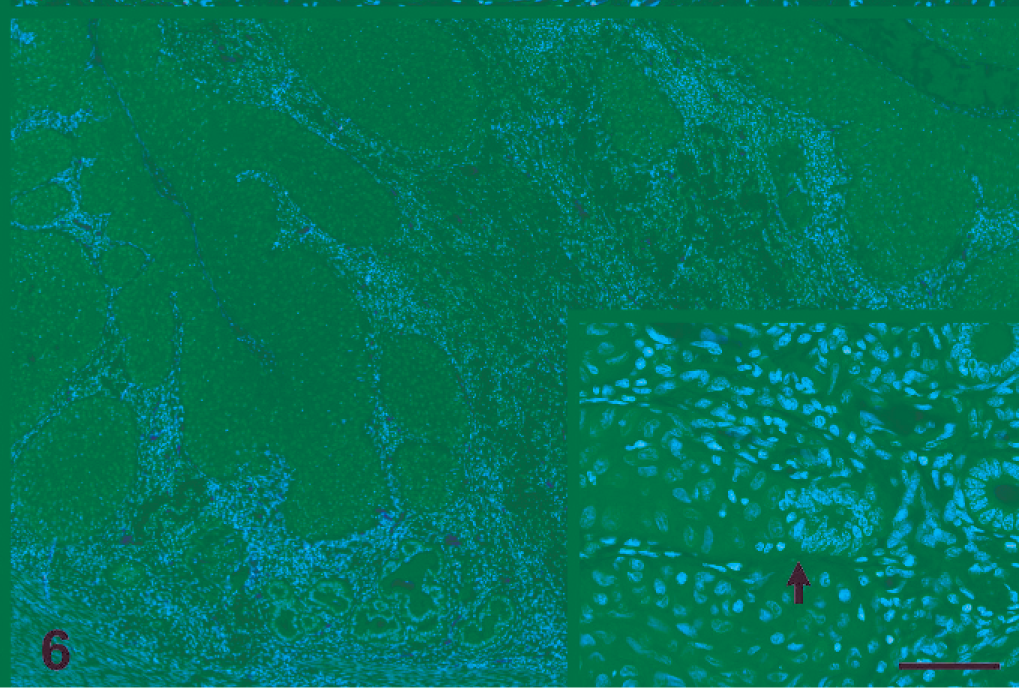
In 2 sea lions (Nos. 2 and 7), a distinct lesion was noted in the uterus. In addition to intravascular metastases, the surface and large portions of the glandular epithelium were replaced by multiple layers of dysplastic, nonkeratinized squamous epithelium morphologically similar to the cervical and vaginal intraepithelial lesions (Fig. 6). In a few areas, normal uterine gland epithelium transitioned into dysplastic squamous epithelium (Fig. 6, inset).
Uterus; sea lion No. 7. The endometrial surface and glandular epithelium contain multiple layers of dysplastic, nonkeratinized squamous epithelium morphologically similar to HGIL commonly found in the cervix and vagina. Inset provides a higher magnification of an area of apparent transition between normal glandular epithelium and neoplastic cells (arrow). HE. Bar = 25 µm.
Immunohistochemistry
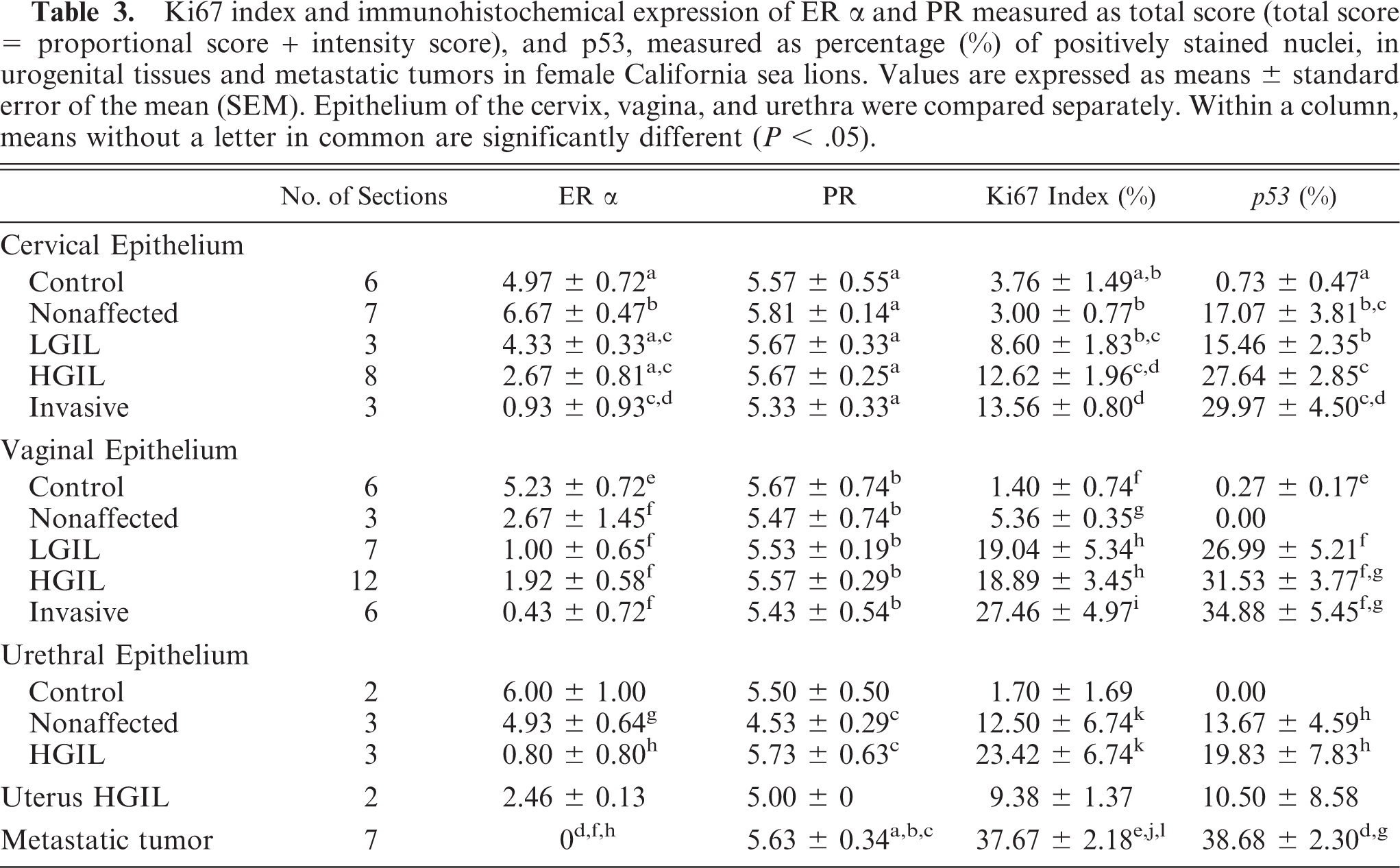
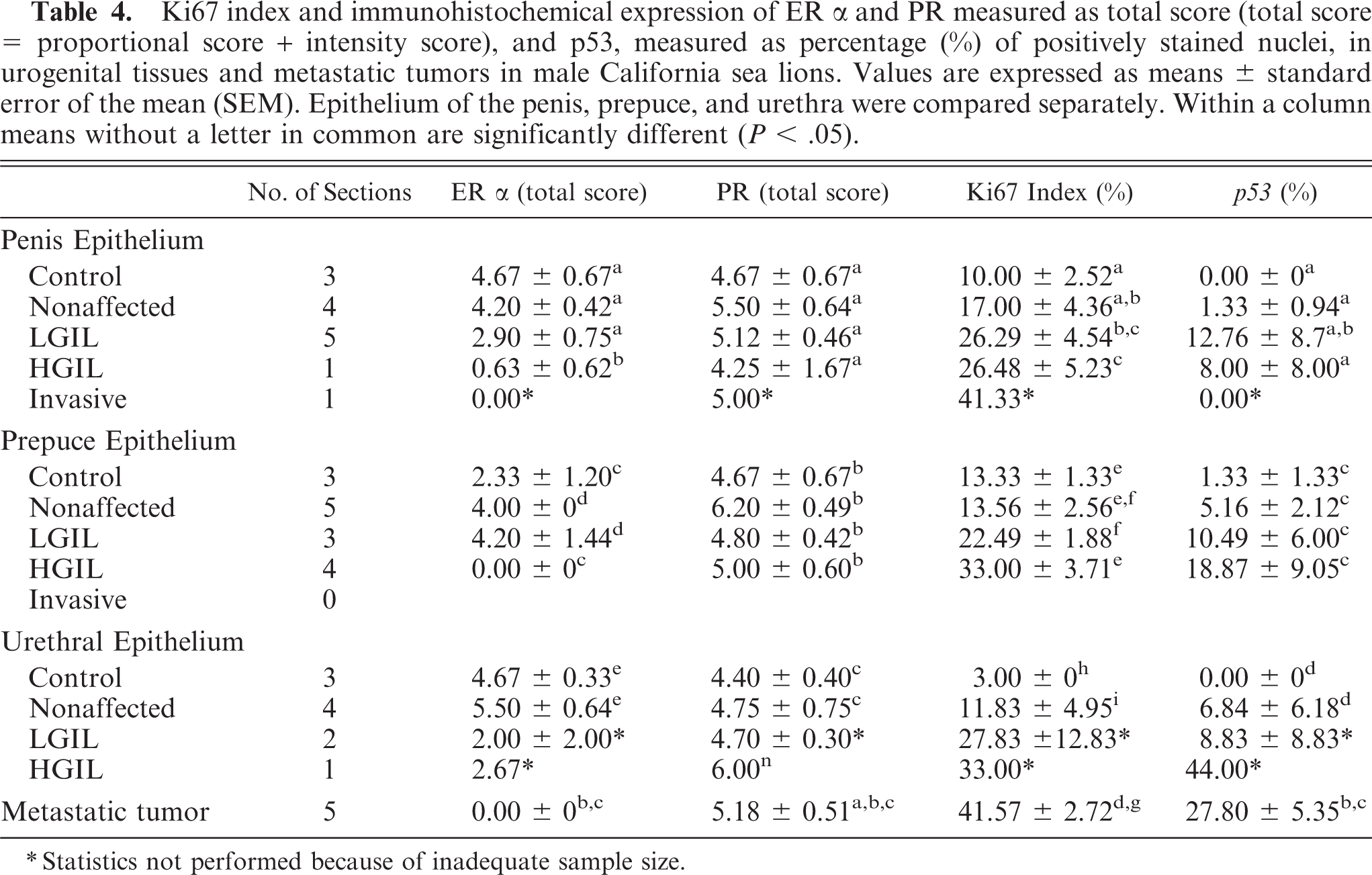
Dark red, positive nuclear immunoreactivity for both ER α and PR was noted in control and unaffected genital epithelium in both females and males (Tables 3, 4). Estrogen receptor expression was significantly lower in intraepithelial lesions compared with control and/or unaffected epithelium (Fig. 7), and expression was completely absent in all metastatic tumors examined (Fig. 8). Intraepithelial lesions were entirely negative for ER α in 1 female with metastatic carcinoma (No. 3) and 1 female and 1 male without metastatic lesions (Nos. 9 and 15). When present, ER α immunostaining was often confined to the basal or parabasal portions of the epithelium (Fig. 7). In contrast, PR receptor expression was common in all lesion grades and in metastatic tumors. Positively stained nuclei were scattered throughout the entire thickness of the epithelium in intraepithelial lesions in both genital and urethral epithelium (Fig. 9) and in infiltrative and metastatic lesions (Fig. 10). Although the intensity of PR staining often was lower in lesions compared with normal or unaffected epithelium, a higher percentage of cells exhibited positive staining. There was no statistically significant difference in PR immunohistochemical scores between different grades of intraepithelial lesions or between metastatic tumors, intraepithelial lesions, and unaffected control epithelium. A similar pattern of ER α and PR expression was noted in genital intraepithelial lesions in sea lions without distant metastases, including absence of ER α staining in HGIL (data not shown). Sample numbers were insufficient for complete statistical assessment. Metastatic tumors with an adenosquamous morphology had significantly higher PR scores than solid squamous tumors (P = .012); however, tumors with other morphologic patterns did not have significantly different scores. In metastatic foci, the intensity of PR immunostaining was often higher at the periphery of the nodules. Within individual animals, hormone receptor immunohistochemical scores in lesions of the same grade were similar regardless of location in the urogenital tract. Strong intranuclear expression of both ER α and PR was noted in stromal cells in the submucosa underlying control, nonaffected, and affected genital epithelium, even in the absence of positive ER α expression in overlying epithelial cells (Fig. 8). Positive staining was most frequent in nuclei of connective tissue and smooth muscle cells and less frequent in endothelial cells. Stromal ER α scores ranged from 3.97 to 4.80 in the cervix, 5.50 to 6.50 in the vagina, 4.33 to 6.00 in the penis, and 3.31 to 5.40 in the prepuce. Stromal PR scores ranged from 6.33 to 6.48 in the cervix, 5.43 to 6.33 in the vagina, 4.25 to 6.00 in the penis, and 3.67 to 6.12 in the prepuce. There was no statistically significant difference in stromal ER α or PR scores between control tissue, nonaffected tissue, or genital lesions of different grades. Unaffected urethral epithelium exhibited strong ER α and PR expression; however, ER α expression decreased significantly in affected urothelium. Sections of urinary bladder transitional epithelium were negative for ER α in 100% of the sea lions. There was occasional weak to moderate PR immunostaining in the urinary bladder of 7 sea lions with intraepithelial and metastatic neoplasia (Nos. 2–4, 8, 9, 11, and 15) and in the 3 control males (score range: 2–5).
Ki67 index and immunohistochemical expression of ER α and PR measured as total score (total score = proportional score + intensity score), and p53, measured as percentage (%) of positively stained nuclei, in urogenital tissues and metastatic tumors in female California sea lions. Values are expressed as means ± standard error of the mean (SEM). Epithelium of the cervix, vagina, and urethra were compared separately. Within a column, means without a letter in common are significantly different (P < .05).
Ki67 index and immunohistochemical expression of ER α and PR measured as total score (total score = proportional score + intensity score), and p53, measured as percentage (%) of positively stained nuclei, in urogenital tissues and metastatic tumors in male California sea lions. Values are expressed as means ± standard error of the mean (SEM). Epithelium of the penis, prepuce, and urethra were compared separately. Within a column means without a letter in common are significantly different (P < .05).
∗Statistics not performed because of inadequate sample size.

Ectocervix; sea lion No. 7. Immunohistochemistry for ER α illustrating positive nuclear staining throughout normal epithelium on the left. Immunostaining is confined to the basal layer in the area of LGIL on the right. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.
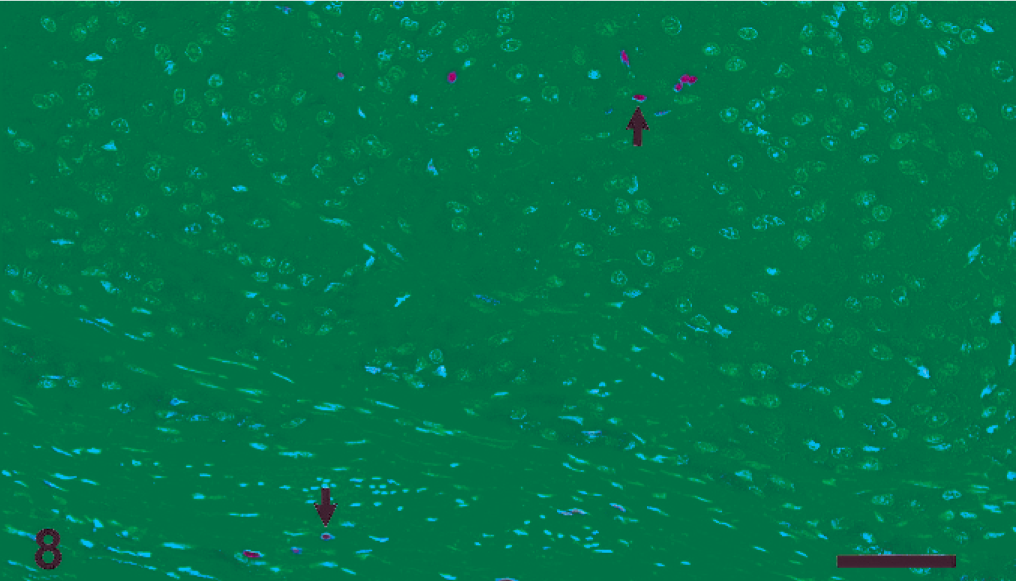
Cranial vagina; sea lion No. 7. Positive immunohistochemical expression of ER α in stromal fibrovascular cell nuclei (arrows) within and adjacent to invasive urogenital carcinoma. Note the lack of nuclear staining in neoplastic cells. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.

Urethra; sea lion No. 3. Immunohistochemical staining with PR antibody in urethral epithelium in a HGIL-grade lesion. There is nuclear staining of many cells throughout the epithelium including within multinucleated epithelial cells (arrows). Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.

Ectocervix; sea lion No. 5. Positive immunohistochemical staining for PR in neoplastic cell nuclei (arrows) within an intravascular tumor embolus near an area of invasion and metastasis. Immunostaining is more frequent at the periphery of the tumor. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.
In general, Ki67 index increased with increasing lesion grade and was significantly higher in HGIL lesions compared with normal and/or unaffected epithelium of the cervix and vagina in females with and without metastasis. In control and unaffected epithelium, positively labeled cells were most common in the parabasal layer; however, positively labeled cells extended into the intermediate layers in LGIL (Fig. 11) and throughout the entire thickness of the epithelium in some HGIL. In males, the Ki67 index in normal and unaffected epithelium was higher than in females, and statistical significance between normal and unaffected male genital epithelium and different lesion grades was more variable. In all tissues, however, Ki67 index was significantly higher in metastatic foci than in normal unaffected epithelium and intraepithelial lesions (Fig. 12). In preputial lesions, Ki67 index was significantly higher in HGIL compared with LGIL. Ki67 index was higher in HGIL compared with normal epithelium and LGIL in sea lions without metastasis (data not shown); however, sample size was insufficient for statistical analyses. Statistically significant differences in Ki67 index were not noted between metastatic tumors with different morphologic patterns.

Penis; sea lion No. 10. Immunohistochemistry for Ki67 illustrating frequent positively labeled nuclei in proliferating epithelial cells in the intermediate layers of a LGIL lesion. Streptavidin-biotin horseradish peroxidase method. Bar = 100 µm.

Lymph node, metastatic carcinoma; sea lion No. 12. Numerous neoplastic cells, including cells undergoing mitosis (arrows), exhibit nuclear expression of Ki67, indicating a high Ki67 index in the tumor. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.
The percentage of p53 positively stained epithelial cells similarly increased with lesion grade in most tissues; however, immunohistochemical staining was highly variable. Positive immunostaining for p53 (>10% positively stained nuclei) was noted within intraepithelial lesions in 9 of 12 animals (75%). The 3 negative animals were males. In 11 of 12 sea lions (92%), metastatic tumors were positive for p53 (range: 14.50–50.00% positively stained nuclei). In intraepithelial lesions, nuclei in the basal, parabasal, and intermediate layers more commonly expressed p53, with less frequent expression in cells in the superficial layers (Fig. 13). In metastatic tumors, p53 immunolabelled neoplastic cells were multifocally distributed throughout the tumor (Fig. 14). Staining was significantly reduced in areas of necrosis. In both intraepithelial and metastatic lesions, positively immunolabelled cells exhibited granular nuclear staining. One animal (No. 1) had positive p53 immunolabelling of nuclei containing inclusions. There was little p53 expression in the genital tract of control animals; however, p53 expression was commonly noted in unaffected epithelium of sea lions with concurrent genital lesions. In sea lions with intraepithelial lesions but no evidence of metastasis, p53 expression was similar, including positive immunoexpression in nonaffected epithelium adjacent to genital lesions. Positive immunostaining was noted in both females and in 1 of 2 males with intraepithelial lesions but no metastases; however, samples sizes were insufficient for statistical analysis. Statistically significant differences in percentage of p53 positive cells were not noted between metastatic tumors exhibiting different morphologic patterns.

Distal vagina; sea lion No. 1. Immunohistochemistry for p53 illustrating positive epithelial nuclear staining (arrows) in LGIL. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.
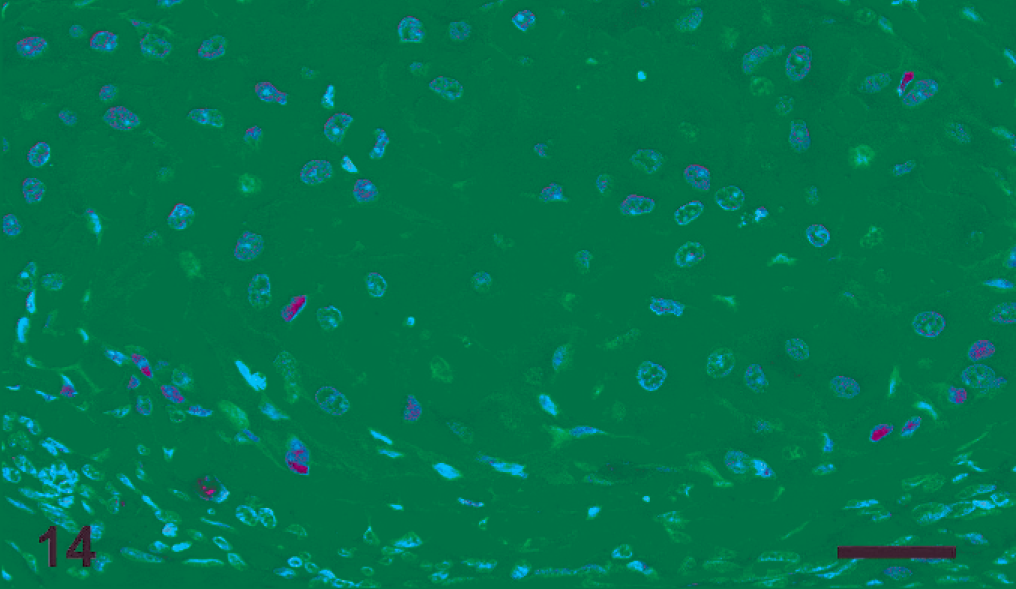
Ovary, metastatic carcinoma; sea lion No. 3. There is frequent immunoexpression of p53 in neoplastic cell nuclei. Streptavidin-biotin horseradish peroxidase method. Bar = 50 µm.
Discussion
The results of this study and in previous immunohistochemical investigations in unaffected sea lions (K. M. Colegrove, unpublished data) demonstrate that both ER α and PR are expressed throughout the genital epithelium and underlying stroma in both males and females. Intriguingly, the loss of ER α expression with continued PR immunohistochemical expression in intraepithelial lesions with high-growth fraction suggests the potential for hormone-responsive growth and mirrors results of similar studies of cervical cancer in women. In most studies in women, epithelial cells in high-grade cervical neoplastic lesions exhibit PR immunohistochemical expression, but expression of ER α is low or completely negative. 24,31 Hormone cofactors are hypothesized to play a role in cervical carcinogenesis in women. Increased 16α-hydroxylation has been shown in HPV-infected cervical epithelial cells, and estrogen increases transcription of oncogenic HPV-16. 2 Additionally, progesterone can increase transcription of the E6 and E7 genes of HPV-16 and increase viral mRNA. 8,30 The continued growth of transformed cervical cells, however, is thought to become hormone independent. 13 Although papillomavirus infection has not been associated with sea lion genital carcinomas, and the oncogenic potential of the sea lion gammaherpesvirus, OtHV-1, has yet to be determined, investigation into a possible interaction between hormones, viral infection, and carcinogenesis is needed. 6,26 Proliferation of sea lion neoplastic cells in response to progestins could occur, similar to the suspected role of progesterone in promoting tumor growth in feline mammary carcinoma. 28,29 Additionally, the strong expression of stromal ER α and PR in urogenital lesions is of interest, given the experimental evidence that stromal hormone receptors can mediate epithelial hormone responses even in the absence of epithelial cell receptors. 11
Some organochlorine compounds, such as some congeners and metabolites of PCBs and DDT, are estrogenic and have endocrine disrupting potential. 38,48 Although the effects of these compounds in sea lions are unknown, there is mounting experimental evidence in other species of potential deleterious effects in hormone responsive tissues, some of which may occur with exposure during development. 27,34 Mink exposed to organochlorines exhibit reproductive abnormalities and upregulation of hormone receptors. 34 Exposure to the synthetic estrogen diethylstilbestrol (DES) during development has been shown to cause upregulation of ER α and developmental abnormalities in the penis of rodents. 17 In a recent study, neonatal mice exposed to estrogenic PCB metabolites developed cervicovaginal carcinomas with age and morphology of the tumors was related to the specific mixture given. 27 Similar to the vaginal adenocarcinomas in women developmentally exposed to DES, exposure to the estrogenic organochlorines during development could prime the sea lion reproductive tract to respond abnormally to endogenous hormones. 17 Given the distinct pattern of hormone receptor expression in sea lion genital carcinomas, further evaluation of the potential relationship between organochlorine exposure, hormones, and tumor development is needed.
Urethral lesions have not been previously described in cases of sea lion genital carcinomas. In both male and female sea lions, the distal end of the urethra is in close apposition to the penile and middle vaginal epithelium, respectively. Exposure rate to potential transforming agents, such as oncogenic viruses, genitourinary bacteria, or genotoxic chemicals, is likely similar in the distal end of the urethra and genital epithelium. Given the extent of the urethra lesions in sea lion No. 13, however, other factors may be involved. Unaffected distal urethral epithelium in both males and females normally expressed ER α and PR, and stromal cells surrounding the urethra in the penis and vagina also exhibited strong hormone receptor expression. If hormone-related factors do play a role in tumor development, then the distal urethral epithelium may respond similarly as the adjacent genital epithelium. In contrast, ER α expression was absent and PR receptor expression very weak in the urinary bladder, where there was no evidence of neoplastic transformation. Given the involvement of the urethra in 31% of the cases in this study, we propose that metastatic carcinomas in sea lions should more accurately be considered urogenital in origin. Although genital and metastatic lesions in other affected sea lions have not exhibited positive immunostaining for the urothelial marker uroplakin (L. J. Lowenstine, unpublished data), uretheral lesions have not been evaluated, and further investigation is warranted.
This study further confirms that sea lion metastatic carcinomas arise most commonly from the lower genital tract of both males and females. Neoplastic lesions can often be noted with careful examination of the entire lower genital tract during gross necropsy and formalin fixation can enhance lesion detection. By taking multiple samples throughout the length of the lower genital tract, progression of intraepithelial neoplasia to invasive carcinoma can often be detected in affected females. More rigorous sampling of affected males may be needed to increase detection of neoplastic cell invasion in the prepuce and penis. The multifocal, often discontinuous distribution of intraepithelial lesions is highly suggestive of multicentric development. Multicentric dysplastic foci could develop synchronously in different areas of the genital tract as a result of a common initiating event or could develop metachronously. As is hypothesized in human cervical cancer, development of metachronous lesions could occur via independent transformation events or through intraepithelial spread of a single dysplastic transformed cell clone. 46
Although sample numbers are low, the histologic findings in this study suggest that the cervix, cranial, and middle vagina may have approximately equal susceptibility to neoplastic transformation in female sea lions. In women, the cervical transformation zone, where glandular epithelium of the endocervix transforms into squamous epithelium, is highly susceptible to HPV and estrogen-induced neoplastic transformation. 16,37 Accordingly, cervical intraepithelial neoplasia is approximately 10 times more common than vaginal or vulvar lesions. 46 The transformation zone is hypothesized to contain multipotent stem cells that can give rise to glandular or squamous epithelium. 15 The normal female sea lion genital tract has morphologic features that could account for similar regional susceptibilities, including a transition between pseudostratified columnar epithelium with pseudoglands and stratified squamous epithelium in both the ectocervix and the middle vagina. Although presence of a distinct transformational zone in sea lions is unconfirmed, transition zones in the cervix and middle vagina could contain stem cells with an increased susceptibility to neoplastic transformation. These distinctive morphologic features could also account for the variable histologic patterns noted in metastatic tumors in female sea lions. In women, both squamous cell carcinomas and adenocarcinomas can arise in the cervix, and metastases can also exhibit varying morphologies depending on degree of differentiation. 25 In contrast, the penis and preputial epithelia are morphologically similar, and the metastatic tumors in male sea lions had less diverse morphologies.
It was extremely difficult to differentiate between potential primary endometrial lesions and metastases. Although the diffuse involvement of the endometrial surface in some cases suggests a primary lesion, apparent transition between normal glandular epithelium and neoplastic epithelial cells was only rarely detected. Protrusion of metastatic foci into normal endometrial glands could have mimicked this apparent transition. Similar endometrial lesions were noted in a previous study and the authors suggested that the glandular morphology of metastatic tumors may be a result of either primary endometrial or cervical tumors. 26 In this study, however, metastatic tumors having adenosquamous morphology were not consistently noted in animals with uterine lesions. Squamous metaplasia of endometrial glands and endometrial gland hyperplasia are rarely diagnosed in free-ranging stranded sea lions. To our knowledge, primary endometrial carcinoma, without lower genital tract involvement, has never been reported. It appears that secondary involvement of the uterus in metastatic genital carcinomas is significantly more common that primary neoplastic transformation of endometrial epithelium.
Similar to the findings in this study, Ki67 positive cells are confined to the parabasal layer in normal cervical epithelium of women, and there is an increase in positively labeled cells throughout all layers of epithelium in cervical intraepithelial neoplasia. 24 In women, proliferation indices are often higher in high-grade intraepithelial lesions compared with low-grade intraepithelial lesions. 36 In this study, however, a statistically significant increase in Ki67 index in HGIL compared with LGIL lesions was only noted in the prepuce. Low sample size may account for lack of significant differences in the majority of tissues examined in this study. It is also possible that some lesions graded as LGIL were higher grade lesions. The higher Ki67 index in normal genital epithelium of male compared with female sea lions may indicate that there is higher cell renewal in the penis and prepuce, which are more exposed to the external environment, than the cervix and vagina.
Although wild-type p53 has a short half-life, p53 mutation increases the half-life, thus allowing detection via immunohistochemistry. 45 The p53 immunohistochemical staining in this study was highly variable, with complete absence of immunostaining in some tumors and intraepithelial lesions. Given the notorious difficulty in antigen retrieval using p53 antibodies, false negative results are possible since sea lion samples having variable duration of fixation were utilized in this investigation. 45 Mutation of p53 is common, however, in many types of neoplasia and the positive expression in some intraepithelial lesions suggests that p53 mutation could play a role in tumor progression. 47 Of particular interest was the finding of positive p53 immunostaining in nonaffected genital epithelium adjacent to intraepithelial lesions. A study in women with cervical cancer found high epithelial expression of p53 and increased proliferation in unaffected margins of hysterectomy samples, suggesting that molecular alterations may occur prior to histologically detectable neoplastic change. 42 Oncogenic gammaherpesvirus and papillomavirus proteins can also affect p53 function and increased expression of p53 has been observed in Epstein-Barr virus–associated nasopharyngeal carcinomas. 39,40,44 Future evaluation of the potential oncogenic properties of sea lion genital viruses should include examination of viral proteins known to interact with p53.
Carcinogenesis is most often multifactorial and can involve initiators that cause molecular damage, co-carcinogens that act synergistically or enhance the activity of initiators, promoters that lead to increased proliferation of damaged cells, and genetic susceptibility. 7 This study identifies several additional factors that may play a role in sea lion genital carcinogenesis. Further research will be needed to completely elucidate how all of these factors may interact in tumor development.
Footnotes
Acknowledgements
We thank Dr. Dennis Wilson and Dr. Chuck Mohr for advice and critical review of the manuscript. The histology laboratory at the VMTH, University of California, Davis is acknowledged for their assistance in obtaining slides for this study. We especially thank the staff and volunteers of The Marine Mammal Center and Tracey Goldstein, Denise Greig, and Alisa Hall for advice on statistical analysis. Funding for this project was provided by the West Coast Center for Oceans and Human Health through the NOAA Oceans and Human Health Initiative. A NIEHS training grant (ES007055) provided support for KMC.
