Abstract
We investigated the immunohistochemical expression of progesterone receptors (PRs) in the prostate and bulbourethral glands of thirty-two 10–14-mo-old Charolais bulls following treatment with a low dosage of estrogens. Animals were divided into 2 groups: 16 animals (group T) were treated for 71 d with a therapeutic dose of trenbolone acetate and estradiol by subcutaneous implant, 16 animals (group C) received no treatment. Urine samples were collected both at the beginning of the trial and 9 times during the study. A semiquantitative analysis of immunohistochemistry (IHC) was performed by counting the number of positive cells in 10 randomly selected high-power fields (hpf). Both groups showed no significant histologic lesions. IHC examination showed positive cells in the epithelium of both glands, with different patterns of distribution between groups. In group C, IHC-positive cells per hpf varied from 0 to 40 in the prostate and from 0 to 32 in the bulbourethral gland. In group T, positive cells varied from 0 to 85 per hpf in the prostate and from 0 to 75 in the bulbourethral gland. The treated group showed significantly higher median numbers of positively stained cells in both organs than the controls ( p < 0.001). Chemical analysis of the urine samples confirmed that the experimental treatment mimics continuous, low-dose administration of anabolic steroids. IHC quantification showed good sensitivity with a high predictive power to correctly classify treated animals and could be used as a preliminary screening test in bulls.
Introduction
Estrogens are steroid hormones involved in the development of the female reproductive structures. They play an important role in other physiologic processes, such as mineral, fat, sugar, and protein metabolism, intestinal motility, blood coagulation, cholesterol metabolism, and in sodium and water conservation by the kidney. 17 Moreover, they improve protein anabolism and mineral retention of important tissues (Meyer HHD, et al. Ruminant physiology: digestion, metabolism, growth and reproduction. Proc 8th Int Symp Ruminant Physiol. Stuttgart: Ferdinand Enke Verlag, 1995:475–482). 21
In 2008, the European Union (EU) banned the use of estrogens in food-producing animals because of the potential risk for humans to develop cancer through the consumption of meat from treated animals. 9 However, considering the systemic effects of these hormones in the body, in some parts of Europe, some producers and veterinarians continue to use estrogens as illicit growth promoters in calves to stimulate and boost meat production.14,16,22 In 1996, a Council Directive required EU member states to implement a national surveillance system to monitor the illegal use of growth promoters to stop the abuse of these substances. 5
According to current legislation, only analytical screening and confirmatory methods can be applied to detect the abuse of anabolic substances by employing chemical analysis of specific target compounds. However, the chemical determination of steroid residues in matrices of biological origin may be hampered either by the rapid metabolism of these molecules or by the administration of new or unknown substances.2,15,23
Detection of the illicit use of anabolic substances is largely based on the direct detection of drug residues in various matrices. 6 The advent of new designer drugs, together with the continuing illegal use of hormones, highlights a need to develop new techniques to detect growth promoter residues in livestock. 11 For example, Italian legislation recommends serum as the matrix for official analysis to detect the presence of estradiol. Histology and immunohistochemistry (IHC) could be used as indirect screening methods. 8 Histology can identify the morphologic and functional changes in tissues caused by anabolic drugs, with relatively low cost and short analytical time. 11 Several authors 12 demonstrated that one of the biological effects induced by estradiol in the body is the upregulation of progesterone receptors (PRs). Overexpression and earlier detection of a gene or its encoded protein in tissue are potentially powerful measures of a specific molecule-related induction. 19 The expression of PRs in epithelium has been detected immunohistochemically in accessory sex glands of male mice as a specific consequence of estrogen treatment. 20 As reported in previous studies, PRs in prepuberal male calves could be used as specific estrogen-related biomarkers. PRs are not normally expressed in the accessory sex glands of male calves and can be increased by exposure to exogenous estrogens.8,19 We evaluated the usefulness and accuracy of semiquantitative IHC as a screening test to detect overexpression of PRs in low-dose steroid–treated bulls.
Materials and methods
Animals and experimental program
Thirty-two 10–14-mo-old Charolais bulls were divided into 2 homogeneous groups. Each animal was individually housed with a feeder and automatic waterer, under natural light and dark cycle conditions. Feed and hay consumption were recorded; water was available ad libitum, and the amount was not recorded. Treatment started at least 15 d after the animals were housed. A veterinarian evaluated the animals clinically twice a week. On day 16, 1 group of 16 animals (group T) was treated with trenbolone acetate (TBA) and estradiol (E2) at the therapeutic dose of 200 mg of TBA (20 mg/pellet) and 40 mg of E2 (4 mg/pellet), by subcutaneous implant a behind the ear. The other group of 16 animals (group C) received no treatment and was considered the control group. The implant, which consists of 10 pellets, was administered only once because the pellet allows a delayed and controlled release of the active principles, as specified in the product leaflet. After 71 d of treatment, animals in both groups were slaughtered in a certified slaughterhouse.
Our study was conducted according to the European Parliament Council Directives.4,10 The procedure was screened and approved by the Animal Experimentation Ethic Committee of the University of Bologna on January 31, 2011.
Histopathology and immunohistochemistry
Samples of the prostate and bulbourethral glands were collected at the slaughterhouse from each animal. The samples were fixed in 10% neutral-buffered formalin for 48 h, routinely processed for histology, and 4-μm sections were stained with hematoxylin and eosin. The specimens were examined for metaplasia of the glandular and ductal epithelium, features related to estrogen treatment.
IHC was performed on 3-μm sections using an automated immunostainer. b Antigen retrieval was performed using a commercial prediluted solution c (pH 8.4) at 95°C for 64 min. The slides were incubated with an anti–PR monoclonal antibody d applied at 1:50 dilution for 1 h at room temperature. An indirect biotin-free system e was used as the detection system. A positive control consisting of an estrogen-treated veal calf prostate and a negative control consisting of untreated veal calf prostate were included in each run.
A semiquantitative analysis of IHC was performed. The following 3 distribution patterns of glandular tissue positivity were considered: focal (rare and randomly distributed positive cells), multifocal (multiple and randomly distributed foci of positive cells), and disseminated (several foci of positive cells distributed throughout the section). The count of positive cells, identified as a dark-brown deposit in nuclei of epithelial cells, was made in 10 randomly selected high-power fields (hpf; 400×; 2.37 mm2).
Chemical analysis
Urine samples were collected the day before the beginning of the trial (T0), and at T7, T21, T28, T35, T42, T49, T56, T63, and T68. The collected samples were frozen and stored at −20°C. Urine samples were clarified by centrifugation (5,000 × g for 5 min). Each sample was analyzed in duplicate (5 mL/aliquot). Fifty microliters of internal standards, containing D3-17β-TBA and D4-17β-E2, were added to each aliquot to obtain a final concentration of the 2 hormones/aliquot of 3 µg/L. The pH was adjusted to 5.0 by adding 15 mL of 0.2 M acetate buffer and, when needed, a few drops of 6N hydrochloric acid. Hydrolysis was carried out for 2 h at 55°C using 50 µL of β-glucuronidase and 50 µL of sulfatase from Helix pomatia.
Samples were cooled at room temperature, and the urine extract was purified using a HLB solid-phase extraction (SPE) cartridge f (200 mg, 6 mL) previously activated with 5 mL of methanol and conditioned with 5 mL of water. The cartridge was washed with 5 mL of water and 5 mL of methanol:water (50:50, v/v). The components were eluted with 4 mL of ethyl acetate:methanol (50:50, v/v). The eluted solution was filtered through a NH2 SPE cartridge g (100 mg, 1 mL) previously conditioned with 1 mL of methanol; the purified extracts were collected directly during the loading. The final solution was evaporated at 45°C, and the dry residue was reconstituted with 0.5 mL of water:acetonitrile (70:30, v/v) and analyzed by liquid chromatography–tandem mass spectrometry (LC-MS/MS).
Analysis of steroid residues was achieved using a high-performance LC system h interfaced to a triple quadrupole mass spectrometer i with a turbo ion spray source. j Purified extracts were analyzed by reversed-phase LC using a C18 column k (100 × 2.1 mm, 2.6 µm, 100 Å), under different ionization and elution conditions: positive electrospray (ESI+) and a mobile phase consisting of 0.02% (v/v) ammonium hydroxide in water and acetonitrile:methanol (70:30, v/v) for trenbolone; negative electrospray (ESI–) and 0.02% (v/v) ammonium hydroxide in water and 0.02% (v/v) ammonium hydroxide in acetonitrile for estradiol.
The urinary concentrations of exogenous steroids (17α-TBA, 17β-TBA, 17β-E2) were calculated using a matrix (bovine urine)-matched calibration curve. The matrix-matched calibration curve for 17α-E2, an endogenous steroid, was prepared by using urine samples previously treated with charcoal to remove physiologically present endogenous compounds. The method used for steroid quantification was validated according to EU Commission Decision 3 and is characterized by the parameters summarized in Table 1.
Performance characteristics of the analytical method used for the quantification of steroids.*
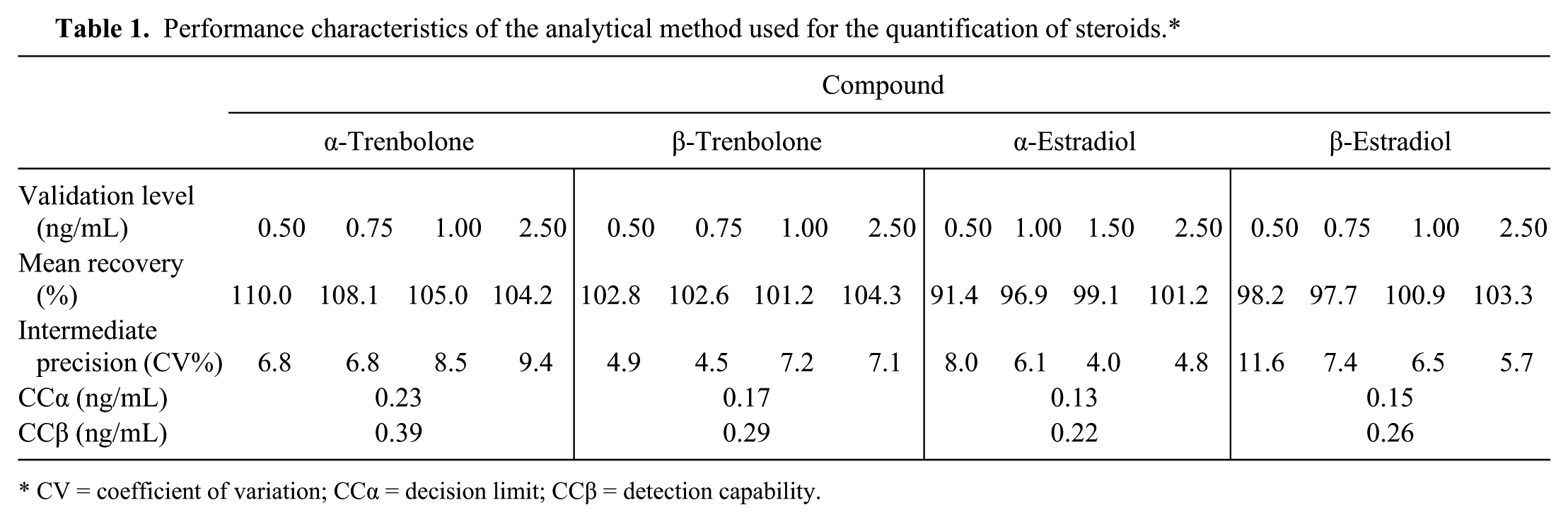
CV = coefficient of variation; CCα = decision limit; CCβ = detection capability.
Statistical methods
Immunohistochemical analysis
The quantification of PRs was summarized by using the median value of each animal considering the substantial intragroup variability of the PR-positive cells. The nonparametric Mann–Whitney 2-sample statistic was used to compare the distributions of the median values between the 2 groups, after evaluating the equality of variances by Levene robust test statistic. An alternative approach used to analyze the data was based on a new dichotomous variable: for each sample, the fields with at least 1 positive cell were converted as “1” or as “0” otherwise. A generalized linear mixed model was adopted, including the group and the sample as the fixed and random effect, respectively. The predicted probabilities of being classified as a treated or control animal were reported with the 95% confidence interval. All statistical analyses were separately performed both for the prostate and bulbourethral glands using commercial software. l
Chemical analysis
Steroid concentrations in the urine samples collected on T68 were reported as the mean ± standard error of the mean. The difference in the measured concentration values between control and treated groups was assessed using Student unpaired t-tests, considering p < 0.05 as statistically significant.
Results
Histopathology and immunohistochemistry
Histologic evaluation of the target organs showed no significant difference between the 2 groups of animals. Lesions were not evident in most of the prostates (12/16 in C and 12/16 in T) and bulbourethral glands (13/16 in C and 13/16 in T). The histologic changes highlighted in the prostate and bulbourethral glands were mild, namely multifocal lymphocytic infiltrates and multifocal glandular ectasia. These histologic findings were interpreted as age-related lesions not associated with the treatment.
Positive cells were present in both glands as focal (Fig. 1) to multifocal patterns in control animals, and multifocal to disseminated (Fig. 2) in treated bulls. In the control group, the number of positive cells per HPF ranged from 0 to 40 in the prostate and from 0 to 32 in the bulbourethral gland. In the treated group, the values ranged from 0 to 85 in the prostate and from 0 to 75 in the bulbourethral gland (Figs. 3, 4)
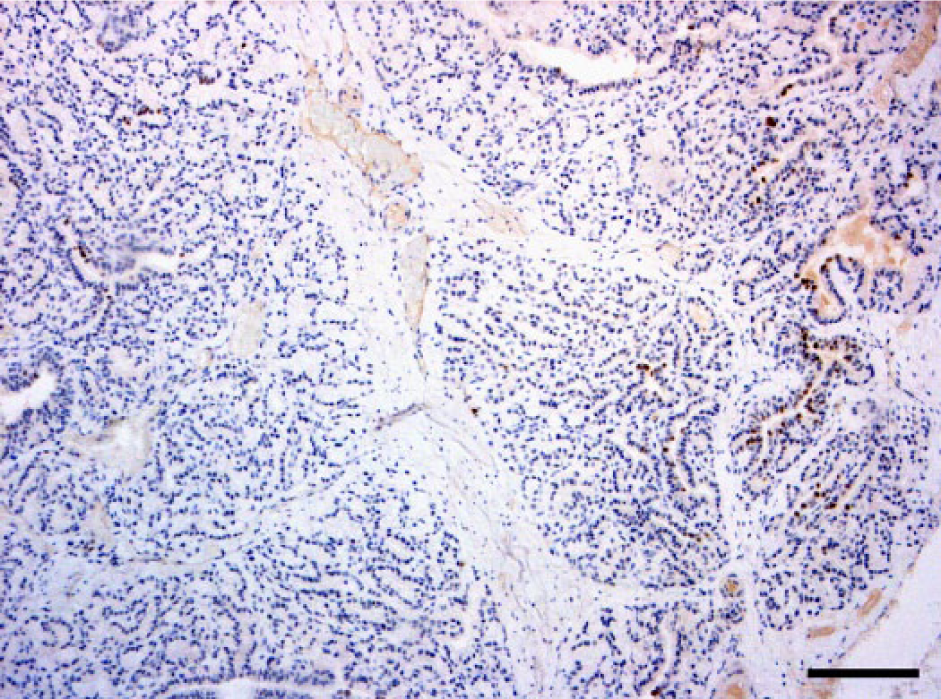
Focal dark-brown nuclear immunopositivity of progesterone receptors in the ductal and glandular epithelial cells of a control bull prostate. Immunohistochemistry. 10×. Bar = 500 µm.
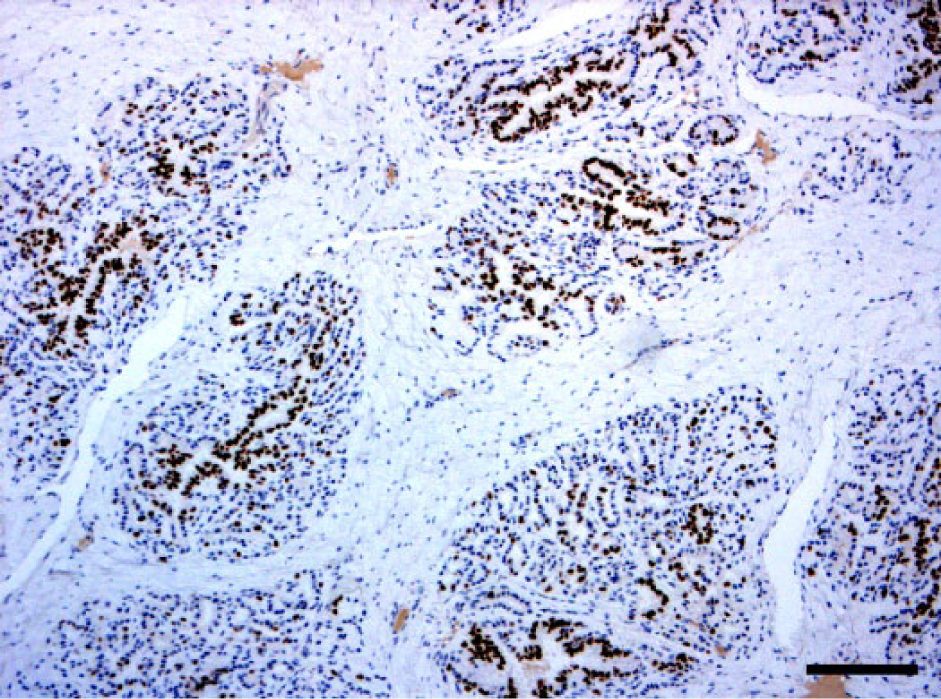
Disseminated foci of dark-brown nuclear immunopositivity of progesterone receptors in the ductal and glandular epithelial cells of a treated bull prostate. Immunohistochemistry. 10×. Bar = 500 µm.
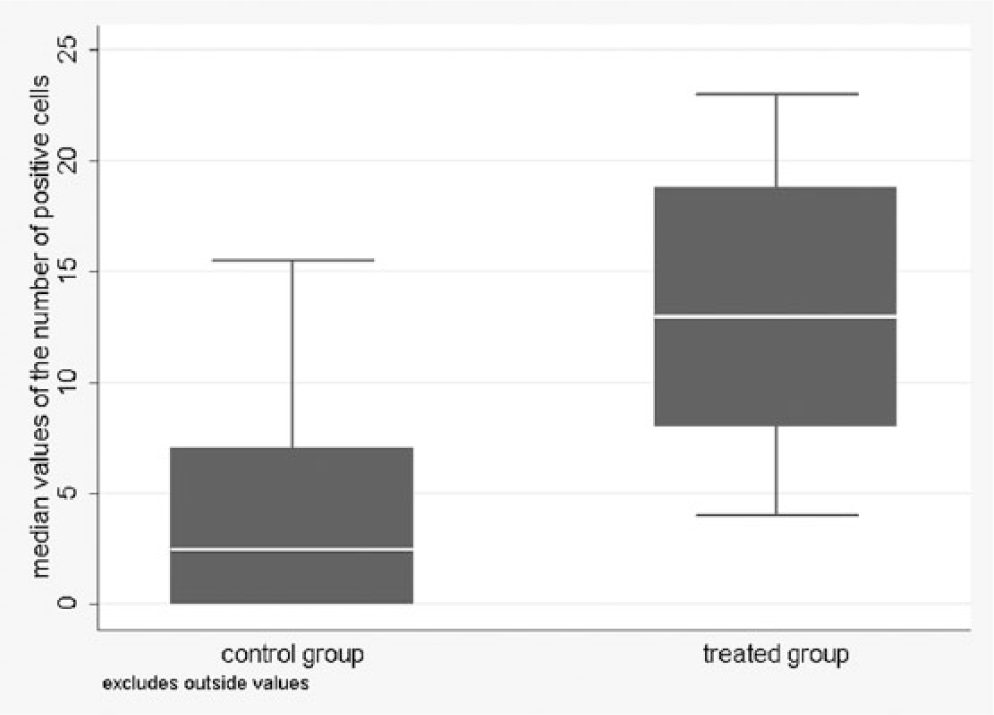
Distribution of median values of samples of the prostate of the control and treated animals.
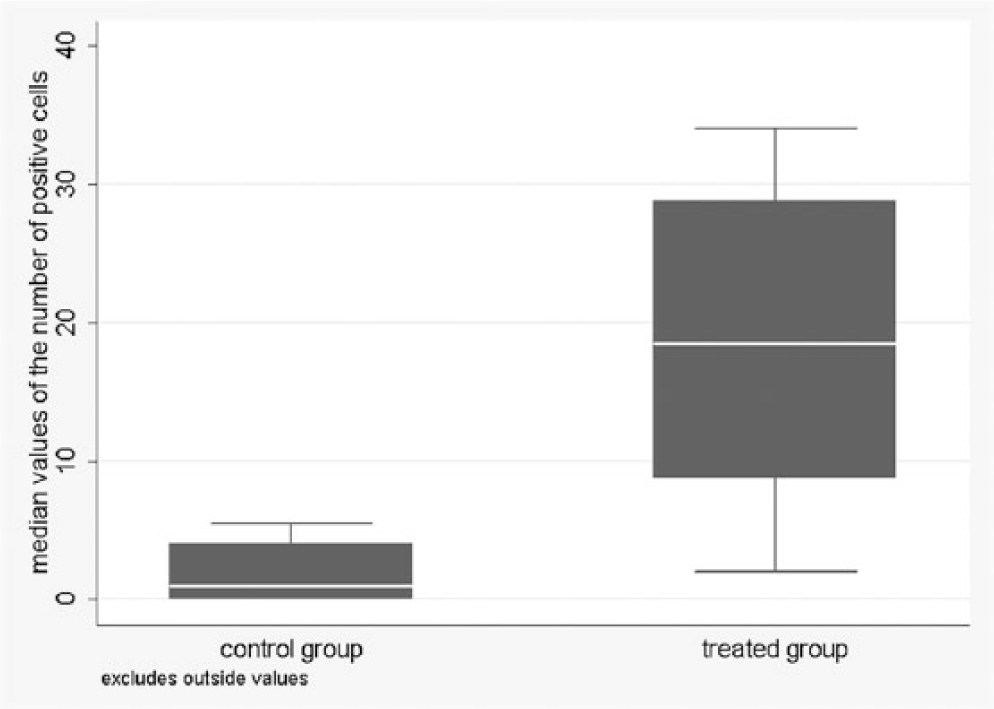
Distribution of median values of samples of the bulbourethral gland of the control and treated animals.
Treated animals had significantly higher median values of stained cells than controls for both organs ( p < 0.001). Moreover, in bulbourethral glands, a significant difference was observed in the variability of the median values between the 2 groups. Analysis of the presence or absence of at least 1 positive cell per field confirmed the significant association with the group for both matrices (Table 2).The predicted probability to observe at least 1 positive cell had high values and wide confidence intervals in the control group: 0.65 (95% CI: 0.05–0.98) for the prostate and 0.59 (95% CI: 0.02–0.98) for the bulbourethral gland.
Results of the generalized linear mixed model applied after having transformed the fields of each sample into “0” and “1,” depending on the presence of positive cells.*

The table reports coefficients and predicted probabilities, with 95% confidence intervals (in parentheses), for the variable group, stratified by type of samples.
Chemical analysis
The urinary excretion profile of 17α-TBA, 17β-TBA, 17α-E2, and 17β-E2 was monitored during the trial for a total of 320 analyzed urine samples (data not shown) from both treated and untreated animals. No drug residues of the exogenous compounds were detectable in the urine samples from the control group. The 17α-E2 was consistently detectable in both groups, but untreated animals showed an average concentration lower than that found in treated animals on the corresponding sampling day. In treated animals, 17α-TBA and 17β-TBA were present in urine since the beginning of the trial. The average urinary concentrations of 17α-E2 and 17β-E2, measured on the last sampling day (t = day 68), was significantly different between the 2 groups ( p < 0.01; Table 3).
Urinary concentrations of 17α-TBA, 17β-TBA, 17α-E2, and 17β-E2 after 68 d of treatment.
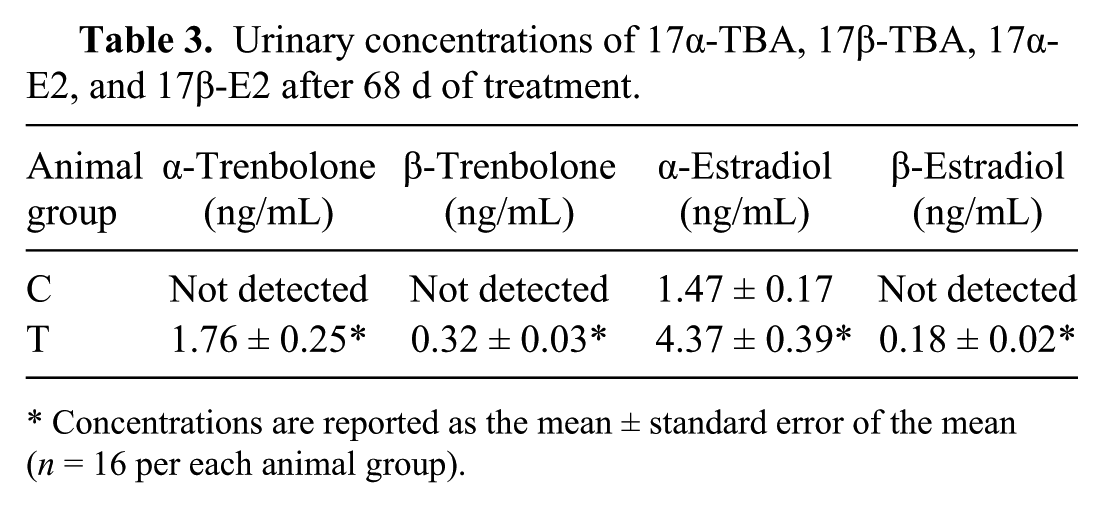
Concentrations are reported as the mean ± standard error of the mean (n = 16 per each animal group).
Discussion
The goal of our study was to investigate the usefulness and accuracy of IHC as an indirect screening method to detect treatment with a low dosage of trenbolone acetate and estradiol in 10–14-mo-old bulls. Results from our urine chemical analysis confirm that the experimental treatment adopted in this study reproduced a low-dose anabolic steroid continuous administration, a condition that presumably mimics the illegal steroid supplementation in bulls. Results also demonstrate that, under these conditions, the detection of 17β-E2 in urine is challenging.
As seen in mouse models, estrogenic treatments induce dose-dependent metaplastic changes in accessory sex glands of male calves.1,8,13 Previous studies have shown that the overexposure of prepubertal male calves to exogenous estrogens induced not only epithelial hyperplasia and metaplastic changes of the prostate and the bulbourethral gland but also widespread PR expression in both organs.7,18 The same authors showed that PRs are not normally expressed in the accessory sex glands of male veal calves but can be induced by brief prepubertal estrogen exposure. In fact, in male veal calves, 17β-E2 induces transcriptional regulation of PR complementary DNA in the prostate and bulbourethral glands in a dose-dependent manner. Therefore, IHC detection of PR could be used as a specific estrogen-related biomarker in the detection of the illegal treatment. 8 Moreover, histopathology, in contrast with official tests, could identify illegal estrogen treatment, even 15 d after the last drug administration. 1 Consequently, histology and IHC have been proposed as the official screening method in the National Residue Monitoring Plan. Both tests are reliable and inexpensive methods to detect illegal steroid treatment in food-producing animals, especially in male calves. However, if the effects of PR expression on estrogen-treated calves’ male sex glands are well known, limited data on bulls are available.
A primary finding of our study was a significant increase of PR expression in those animals treated with low doses of 17β-E2, confirming PR transcriptional upregulation induced by estrogens in the prostate and bulbourethral glands. PR immunopositivity found in both prostate and bulbourethral glands of control animals confirmed that PRs can be normally expressed in the accessory sex glands of bulls. In a previous study, 11 PR gene and protein expression were evaluated in bulls treated with a subcutaneous implant a for 89 d. Treated animals exhibited diffuse PR positivity in both accessory glands, whereas control animals showed weak multifocal positivity, mainly at the periphery of the glands. Although quantitative evaluation of the IHC results was not performed in that study, significant up-regulation of the PR messenger RNA was highlighted in both glands of treated animals (age range: 13–22 mo). The more diffuse staining of PR expression observed by these authors 11 could result from the longer period of drug administration compared with our study (89 vs. 70 d). Histology did not reveal any changes related to the treatment in either study. The PR overexpression is dose-dependent and may not necessarily be linked to histologic lesions, especially in animals treated with low-dosage cocktails. 11 Based on the similar pattern observed in both control and treated groups, as well as the high variability in the number of PR-positive cells, an attempt to quantify the IHC results was conducted by counting positive cells in 10 randomly selected hpf. Statistical analysis demonstrated that the applied method has a high predictive power to correctly identify treated animals as a result of their significantly higher median values of PR expression than those of the controls. The high variability and wide confidence intervals observed in both glands of control animals indicate a lack of predictive power to correctly classify them. In other words, this method showed good sensitivity, but low specificity. Considering the control group, values of the bulbourethral gland had less variability than the prostate, leading to better distinction between treated and untreated animals. The bulbourethral gland could be considered a more reliable organ than prostate to differentiate the 2 groups. Age and breed as well as plant steroids and concurrent drug use may affect PR basal expression in sexual glands, even though no definitive evidence is available to date. In our survey, because the control group was homogeneous for the aforementioned variables, individual biological variability in PR expression must be considered.
Our results demonstrate that IHC might be a useful and cost-effective preliminary screening test in bulls to address subsequent confirmatory chemical analysis, based on chromatography coupled to mass spectrometry. More accurate quantification methods are mandatory in cases of multifocal positivity to better discriminate treated and untreated animals.
Footnotes
Authors’ contributions
C Zanardello and M Vascellari contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. K Capello, R Stella, and G Biancotto contributed to design of the study, and contributed to analysis and interpretation of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Revalor-XS, Merck Animal Health, Madison, NJ.
b.
BenchMark ULTRA instrument, Ventana Medical Systems, Tucson, AZ.
c.
ULTRA cell conditioning solution (ULTRA CC1), Ventana Medical Systems, Tucson, AZ.
d.
Progesterone receptor Ab-2 (clone hPRa 2, MS-192-P), Thermo Fisher Scientific, Fremont, CA.
e.
UltraView universal DAB detection kit (052 697 806 001 code), Ventana Medical Systems, Tucson, AZ.
f.
HLB SPE cartridge, Waters, Milford, MA.
g.
NH2 SPE cartridge, Phenomenex, Torrance, CA.
h.
Prominence HPLC system, Shimadzu, Kyoto, Japan.
i.
API 4000 LC-MS/MS system, SCIEX, Framingham, MA.
j.
TurboIon spray, SCIEX, Framingham, MA.
k.
Accucore C18 column, Superchrom, Milan, Italy.
l.
STATA v.12, StataCorp, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by Italian Ministry of Health (Funded Research 2008).
