Abstract
The BRCA/RAD51 complex of tumor suppressor genes plays a major role in the DNA damage response. In this explorative study, BRCA1, BRCA2, and RAD51 mRNA expression was quantified in highly defined laser microdissected tissue samples of simple adenomas, adenocarcinomas of the mammary gland, and their lymph node metastases by real-time quantitative reverse transcription polymerase chain reaction. Expression levels in the tumors were normalized to the geometric mean of 3 housekeeping genes and quantified relative to normal mammary epithelium of the same dog. In adenomas, mRNA expression was reduced for BRCA1 (6/10 dogs, 60%), BRCA2 (4/10 dogs, 40%), and RAD51 (4/10, 40%). In adenocarcinomas BRCA1 expression varied with increased expression in 3 of 10 (30%) dogs and no differences in 7 of 10 (70%) dogs when compared with normal mammary gland. BRCA2 and RAD51 were overexpressed in 5 of 10 (50%) and 6 of 10 (60%) of adenocarcinomas, respectively. An overexpression of RAD51 and BRCA2 was found in 8 of 10 (80%) and 5 of 10 (50%) of the lymph node metastases, respectively. Direct comparison of primary tumors and metastases revealed increased mRNA expression of BRCA1 (2/10 dogs, 20%), BRCA2 (2/10 dogs, 20%), and RAD51 (3/10 dogs, 30%) in lymph node metastases. Taken together, the results suggest that RAD51 is upregulated in the majority of lymph node metastases of canine mammary tumors. Further experimental studies are needed to clarify whether these changes in gene expression are a direct carcinogenetic stimulus or a protective response due to genetic instability during tumor progression.
Introduction
Mammary tumors are among the most prevalent tumors in dogs. A fatal outcome of the disease is virtually always due to metastasis of the primary tumor to distant organs. The exact molecular pathogenesis of mammary tumor development and metastasis is unknown. Several studies have been reported on the expression of mRNA and protein levels of such genes as p53, hormone receptors, ErbB2, and BRCA1 and their association with carcinogenesis and prognosis of canine mammary tumors. 4, 11, 17 Unfortunately, these studies lead to several contradictory results, such as on the expression of c-erb and E-cadherin. 8, 9, 15, 20
In human breast cancer, overexpression of BRCA2 and RAD51 is associated with a poor prognosis, whereas BRCA1 expression is often decreased during progression of sporadic breast cancer. 1, 7, 19 The expression levels of these genes have therefore been proposed as clinically relevant tumor markers. 1, 7, 19 Homologous genes of the human BRCA1, BRCA2, and RAD51 have been identified in dogs. 12, 18 However, the role of BRCA and RAD51 gene expression in canine mammary tumors is unclear. The only available immunohistochemical study on this multiprotein complex in dogs revealed underexpression of nuclear BRCA1, while cytoplasmic BRCA1 was overexpressed in some of the tumors. 11 So far, the regulation of BRCA2 and RAD51 gene expression has not been investigated in normal canine mammary gland epithelium or mammary tumors.
BRCA1 and BRCA2 play important roles in the preservation of genomic stability, thereby functioning as “caretakers” of the genome. 23 They are essential for DNA damage signaling and DNA repair. These tasks are mainly accomplished by the interaction and activation of DNA repair proteins. Especially the interaction of both proteins with Rad51, a DNA-repair protein, is thought to be of prime relevance. 16 Nevertheless, only BRCA2 directly interacts with Rad51, while the biologic relevance of the indirect BRCA1/RAD51 interaction is unclear. 2, 16
In this explorative study, we characterized the differential mRNA expression of BRCA1, BRCA2, and RAD51 in laser-microdissected tissue samples of normal mammary gland epithelium, simple canine adenomas, and adenocarcinomas. Using laser microdissection, we obtained highly defined tissue samples without contamination by stromal and non-neoplastic epithelial cells. For studies on mRNA expression levels in canine mammary tumors, this is especially of great importance due to the unavoidable variance in proportion of neoplastic and non-neoplastic cells in whole mount tissue samples. Furthermore, we compared BRCA1, BRCA2, and RAD51 expression between simple adenocarcinomas and their lymph node metastases to elucidate their role in the metastatic progression of canine mammary tumors. We show decreased BRCA2 and RAD51 mRNA expression levels in adenomas and increased expression in lymph node metastases when compared with non-neoplastic mammary epithelium. Moreover, the similar regulation of BRCA2 and RAD51 is indicative of a common role of these genes in canine mammary tumor carcinogenesis.
Materials and Methods
Dogs and tissue processing
From a total of 130 canine patients with mammary tumors, all animals with simple mammary adenocarcinomas metastatic to the regional lymph node at the time of tumor resection were selected (n = 10). Furthermore, 6 patients with histologically well-differentiated simple adenomas of the mammary gland were selected (n = 10). All dogs had a comprehensive clinical examination, including complete blood count and blood biochemistry. None of the dogs had radiologic evidence of pulmonary metastases or abnormalities in the peripheral blood. Tissue specimens were sliced at 5-mm thickness, and every other slice was immersion fixed in neutral-buffered 4% formaldehyde or snap frozen in liquid nitrogen at −80°C within 15 minutes after resection and stored until further use.
Formaldehyde-fixed tissues were routinely embedded in paraffin. Sections of 2-µm thickness were mounted on adhesive glass slides and stained with HE. Histologic evaluation of the tumors was performed independently by 2 board-certified pathologists following the criteria of the World Health Organization classification of canine mammary tumors. 10
Laser-capture microdissection and reverse transcription
To obtain highly specific microdissected tissue samples without contamination by stromal and inflammatory cells or by non-neoplastic cells or neoplastic cells, respectively, 5 consecutive sections of 6- to 8-µm thickness from the frozen tissue samples were mounted on glass slides covered with a polyethylene naphthalate membrane (PALM Microlaser Technologies, Bernried, Germany) for laser microdissection. After fixation for 2 minutes in 95% ethanol at −20°C, sections were stained with HE in diethylpyrocarbonate-treated water. Subsequently, sections were dehydrated in ascending graded ethanol (30, 50, 70, and 99%) and air dried at room temperature. To obtain sufficient amounts of mRNA 25 × 106 µm2 were excised from tissue sections of adenomas, adenocarcinomas, and lymph node metastases. Non-neoplastic epithelial cells were obtained from tissue sections of adjacent mammary complexes. Cells were laser pressure catapulted into the caps of 0.5-ml Eppendorf tubes containing 30 µl of lysis buffer (NucleoSpin RNA XS; Macherey & Nagel, Düren, Germany). Total RNA was extracted and purified using a commercial kit (NucleoSpin RNA XS). Total RNA was reverse transcribed using the iScript cDNA synthesis kit (Biorad, Germany).
Quantitative real-time polymerase chain reaction
Primer sequences for BRCA1, BRCA2, RAD51, and the housekeeping genes hypoxanthine-phosphoribosyl transferase, ATP-synthase subunit 5B, and ribosomal protein L32 are shown in Table 1. Real-time quantitative reverse transcription polymerase chain reaction and data analyses were performed using the MX 3000P Quantitative PCR System and MX Pro software (Stratagene, La Jolla, CA, USA). The reactions were carried out in 96-well polypropylene plates covered with optical caps (Stratagene). The plates contained triplicates of each cDNA sample and no-template controls with water instead of cDNA templates. During initial optimization runs, the exact primer concentrations and polymerase chain reaction time and temperature conditions were determined. Polymerase chain reaction products were sequenced to evaluate specificity of the primer pairs. The 15-µl reaction mix contained 5 µl cDNA and 12.5 µl Brilliant SYBR Green QPCR Master Mix (Stratagene) with 300 nM of each primer. Cycling conditions were 10 minutes at 95°C, followed by 40 cycles of 30 seconds at 95°C, 1 minute at 58°C, and 30 seconds at 72°C. The cDNA of all samples were amplified on the same plate for every primer pair to ensure equal amplification conditions. Specificity of amplification products was confirmed by melting curve analyses. For each sample, results were documented as cycle threshold (threshold set to 100 relative fluorescence units) values of background-subtracted QPCR fluorescence kinetics using the MX Pro Stratagene analysis software; applying the adaptive baseline, amplification based threshold; and moving average algorithm enhancement.
Sequences of primer used for qPCR.

Quantification of target gene expression
Relative expression of the target gene in neoplastic and non-neoplastic tumor cells was determined using a comparative computed tomographic method with multiple housekeepers, as previously described. 5, 21 The housekeeper genes used were selected from a panel of reference genes according to the GeNorm algorithm and represent the best reference genes for canine mammary gland. 3 Data are presented as fold change in gene expression level in the sample of interest normalized to the housekeepers and relative to the sample control. Briefly, normalization factor was calculated using the geometric mean of 2ΔCT of the 3 reference genes and the gene of interest in the sample of interest and sample control. Finally the fold change of gene of interest expression was calculated with the ΔCTGOI multiplied by the normalization factor. Cut-off values for the fold change were set at >2.0 for increase and fold change values <0.5 for decrease in gene expression. 1 Specifically, gene expression levels of all adenomas, adenocarcinomas, and lymph node metastases (sample of interest) was normalized to reference gene expression and compared with non-neoplastic gland epithelium (sample control) of the same dog. Furthermore, gene expression levels in lymph node metastases (sample of interest) were compared with their respective primary tumor (sample control).
Results
Histopathology
Simple adenomas were well differentiated and partly encapsulated with mild expansive growth but no infiltration of the surrounding connective tissue (Fig. 1). The mildly pleomorphic neoplastic cells were arranged in plump tubules lined by 2 to 3 layers of moderately well-differentiated epithelial cells. Cellular pleomorphism was only mild, and mitotic figures were less than 2 mitotic figures per 400× magnification. Infiltrative growth or angioinvasion was absent, and regional lymph nodes were free of metastases. All adenocarcinomas were poorly differentiated with marked infiltrative growth into the surrounding tissues and invasion of lymph and blood vessels (Fig. 2). Neoplastic cells showed marked cellular pleomorphism, anisokaryosis, and a high mitotic index, with >3 mitoses per 400× magnification. Furthermore, all adenocarcinomas displayed multifocal necrosis of a varying degree and mild to moderate scirrhous reaction. Lymph node metastases were found in regional lymph nodes (Fig. 3). Here, metastatic tumor cells were highly pleomorphic with tubular to solid arrangement and replaced and compressed the surrounding lymphatic tissue. Mitotic index was high with >4 mitoses per 400× magnification. Non-neoplastic mammary epithelium was characterized by isomorphic epithelial cells that were arranged in 1-layered tubules that formed well-demarcated glandular lobules (Fig. 4). Dysplasia, angioinvasion, and infiltration of the adjacent tissue were absent.
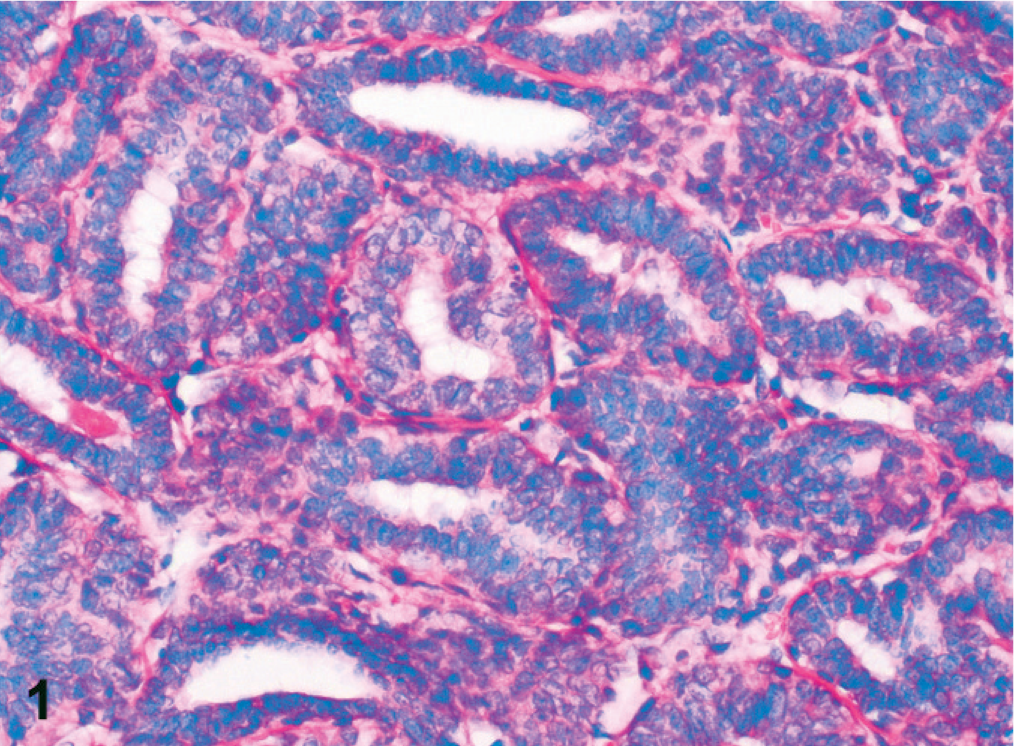
Mammary gland, dog No. 2. Simple adenoma composed of mildly pleomorphic neoplastic cells arranged in plump tubules. HE staining. Bar = 50 µm.
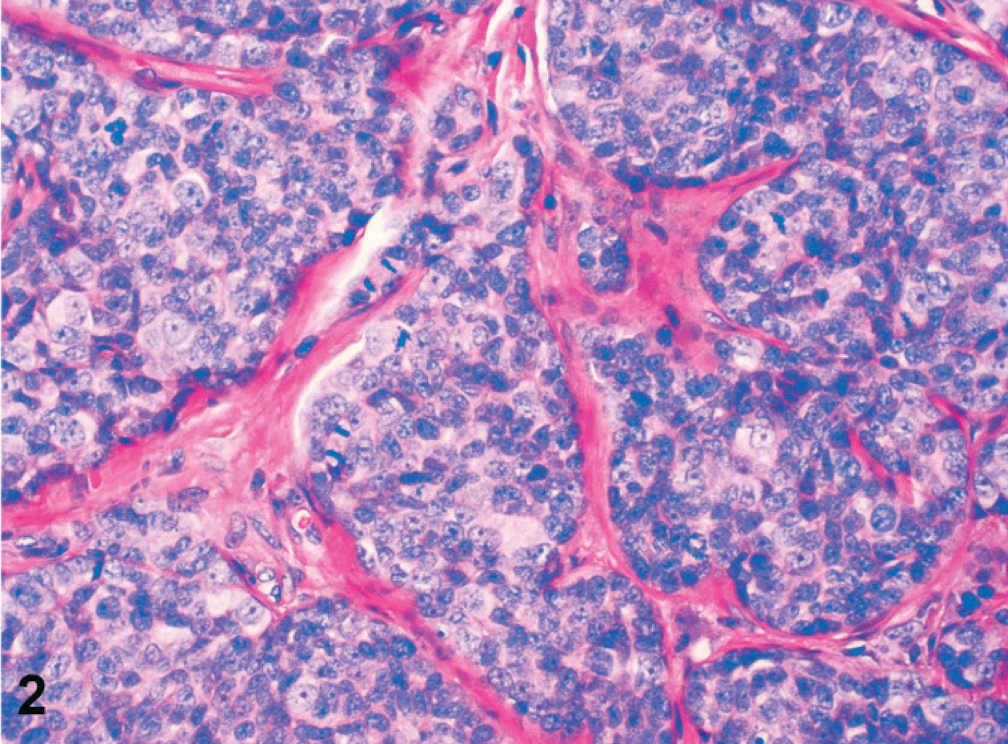
Mammary gland, dog No. 16. Simple adenocarcinoma with marked infiltration of the surrounding connective tissue. Neoplastic cells are arranged in nests and solid sheets. Cellular pleomorphism, anisokaryosis, and high mitotic index were typical features of all adenocarcinomas. HE staining. Bar = 50 µm.
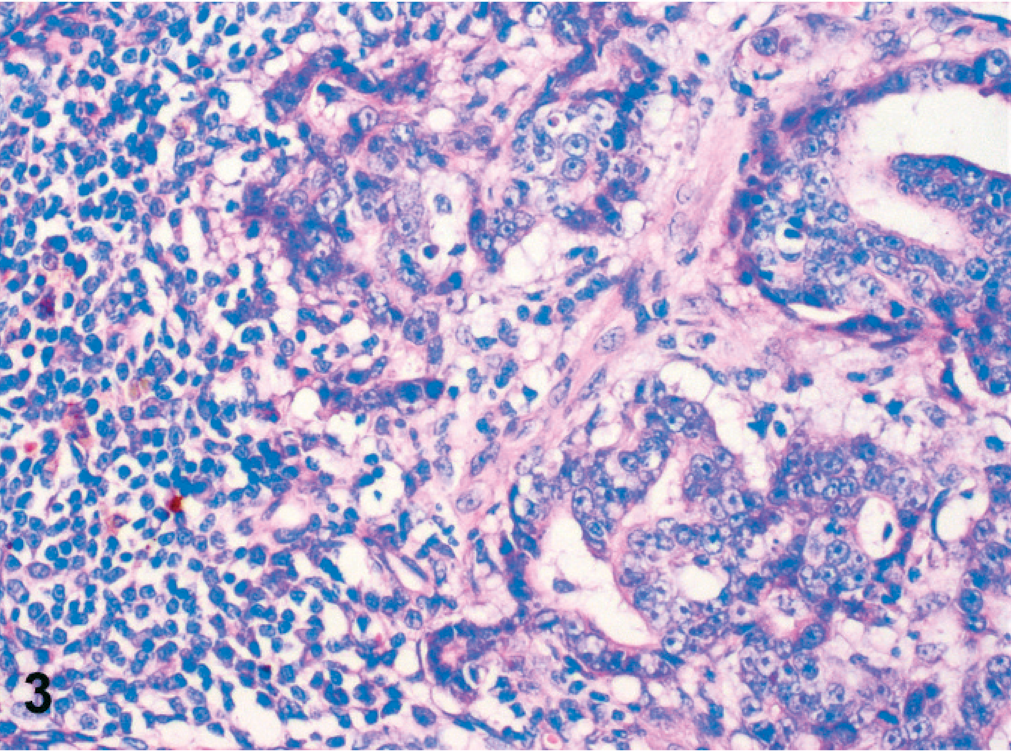
Inguinal lymph node, dog No. 16. Lymph node metastasis of a mammary gland adenocarcinoma composed of nests and primitive tubules and that replaces the preexisting lymphatic tissue. Tumor cells are highly pleomorphic and have a high mitotic index. HE staining. Bar = 50 µm.
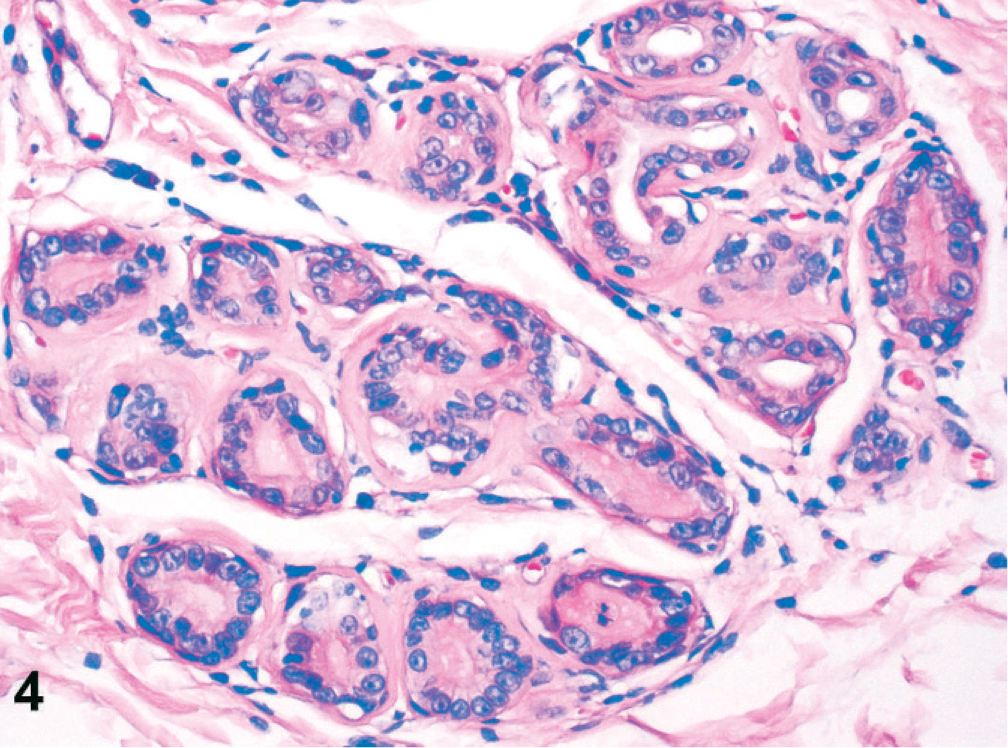
Mammary gland, dog No. 16. Non-neoplastic mammary gland epithelium characterized by well-differentiated tubular arrangement of epithelial cells without cellular pleomorphism and infiltration of the adjacent tissue. HE staining. Bar = 50 µm.
Expression levels of BRCA1, BRCA2, and RAD51 in adenomas versus normal gland
To test whether the cellular expression levels of BRCA1, BRCA2, and RAD51 differ in adenomas when compared with non-neoplastic mammary epithelium of the same dog, their relative mRNA copy numbers per mRNA copy numbers of 3 housekeeping genes were determined using real-time quantitative reverse transcription polymerase chain reaction after laser microdissection of pure tumor or non-neoplastic tissue areas. The relative copy numbers of BRCA1 in adenomas of 6 of 10 dogs was reduced to <0.5-fold when compared with normal gland epithelium (Fig. 5). Similarly, expression levels of BRCA2 and RAD51 were also reduced in adenomas of 4 of 10 dogs (Figs. 6, 7). Of note, BRCA1 expression was increased in 1 of 10 adenomas.
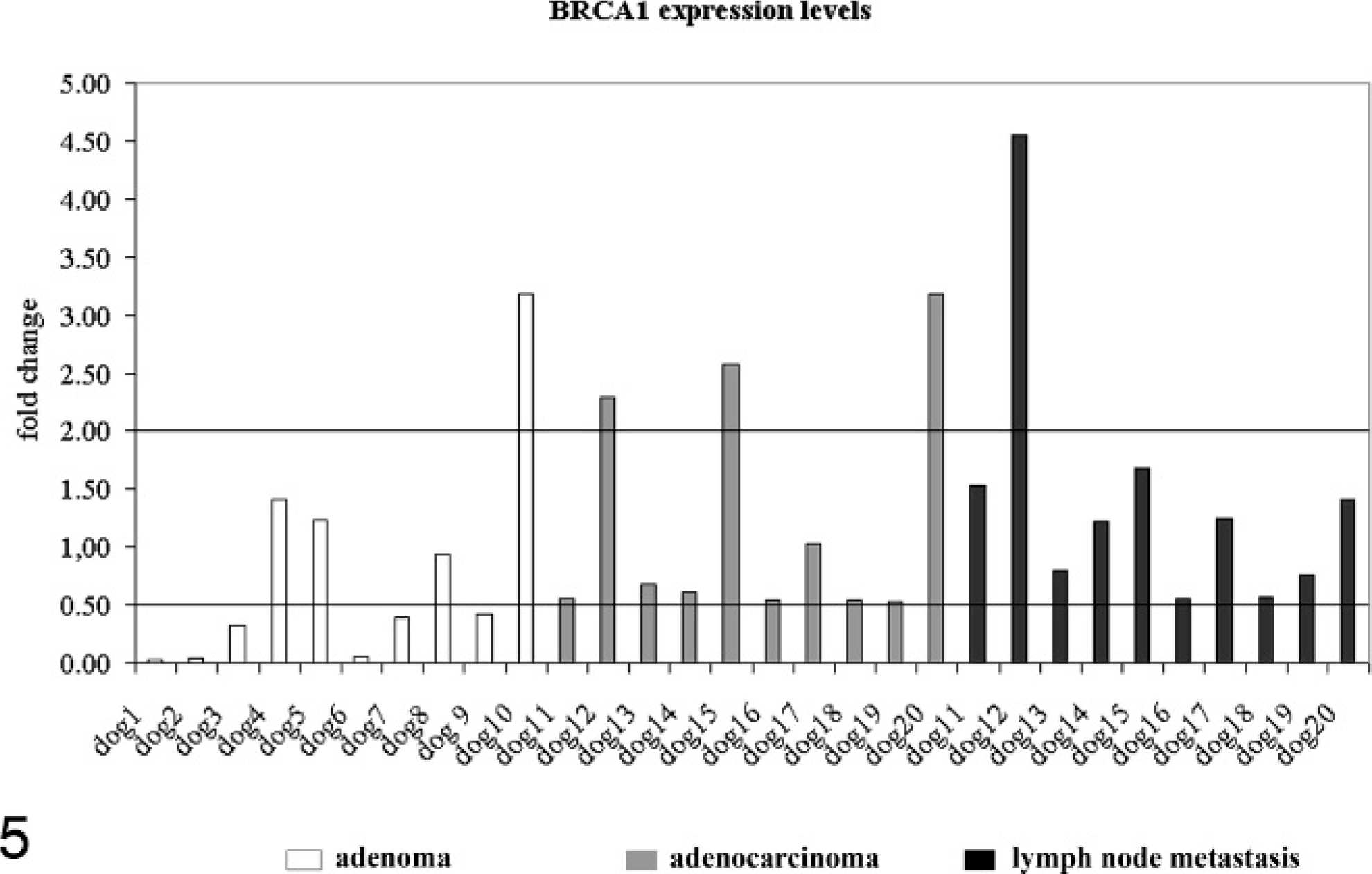
BRCA1 expression in mammary adenomas (dogs Nos. 1–10, white columns), adenocarcinomas (dogs Nos. 10–20, gray columns), and their lymph node metastases (dogs Nos. 10–20, black columns) compared with non-neoplastic mammary epithelium of the same dog. Values are represented as fold change relative to normal gland. Values ≥2.0 were considered overexpression, and values ≤0.5 were considered reduced gene expression.
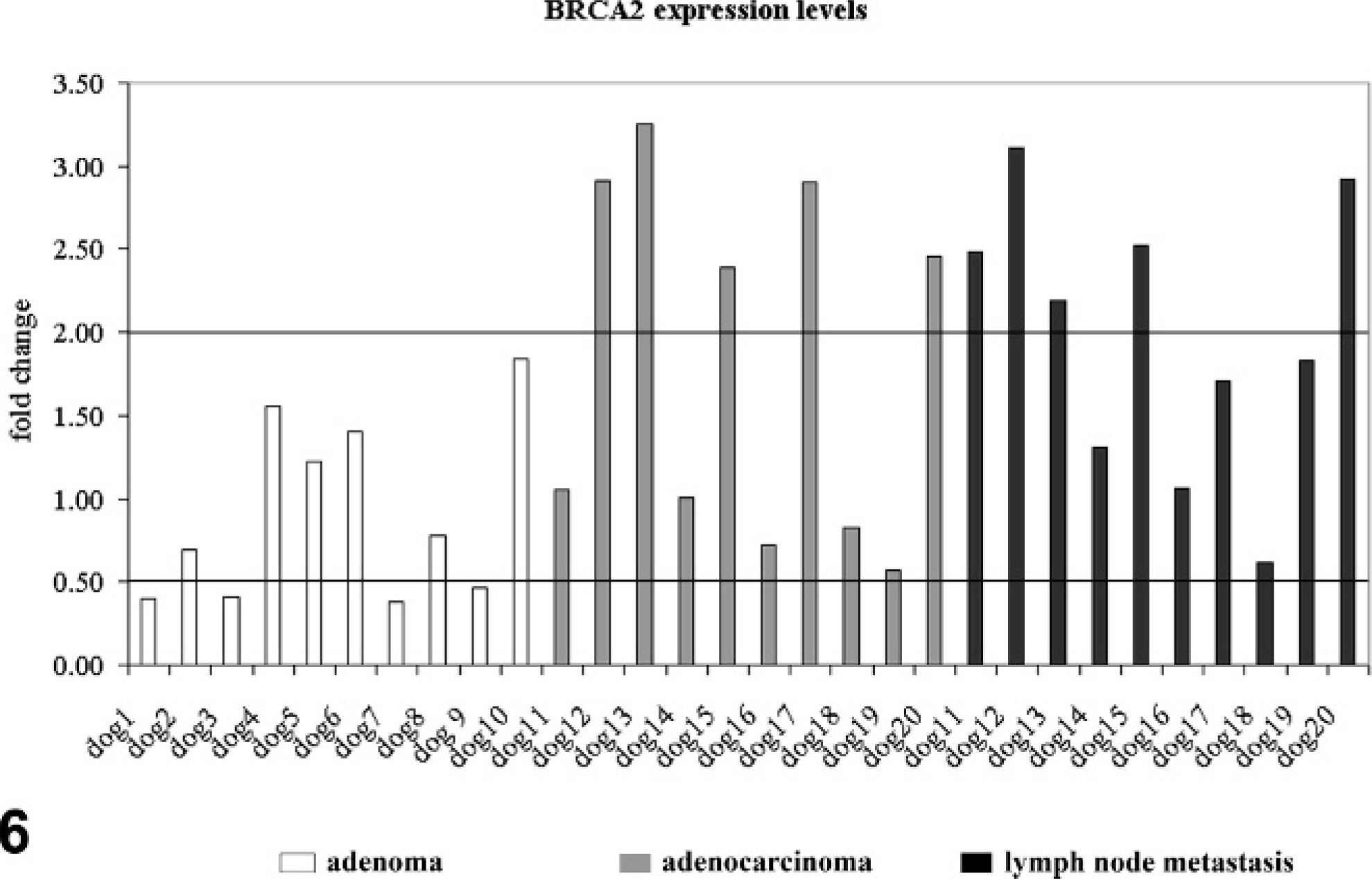
BRCA2 expression in mammary adenomas (dogs Nos. 1–10, white columns), adenocarcinomas (dogs Nos. 10–20, gray columns), and their lymph node metastases (dogs Nos. 10–20, black columns) compared with non-neoplastic mammary epithelium of the same dog. Values are presented as fold change relative to the primary tumors. Values ≥2.0 were overexpression, and values ≤0.5 were considered reduced gene expression.
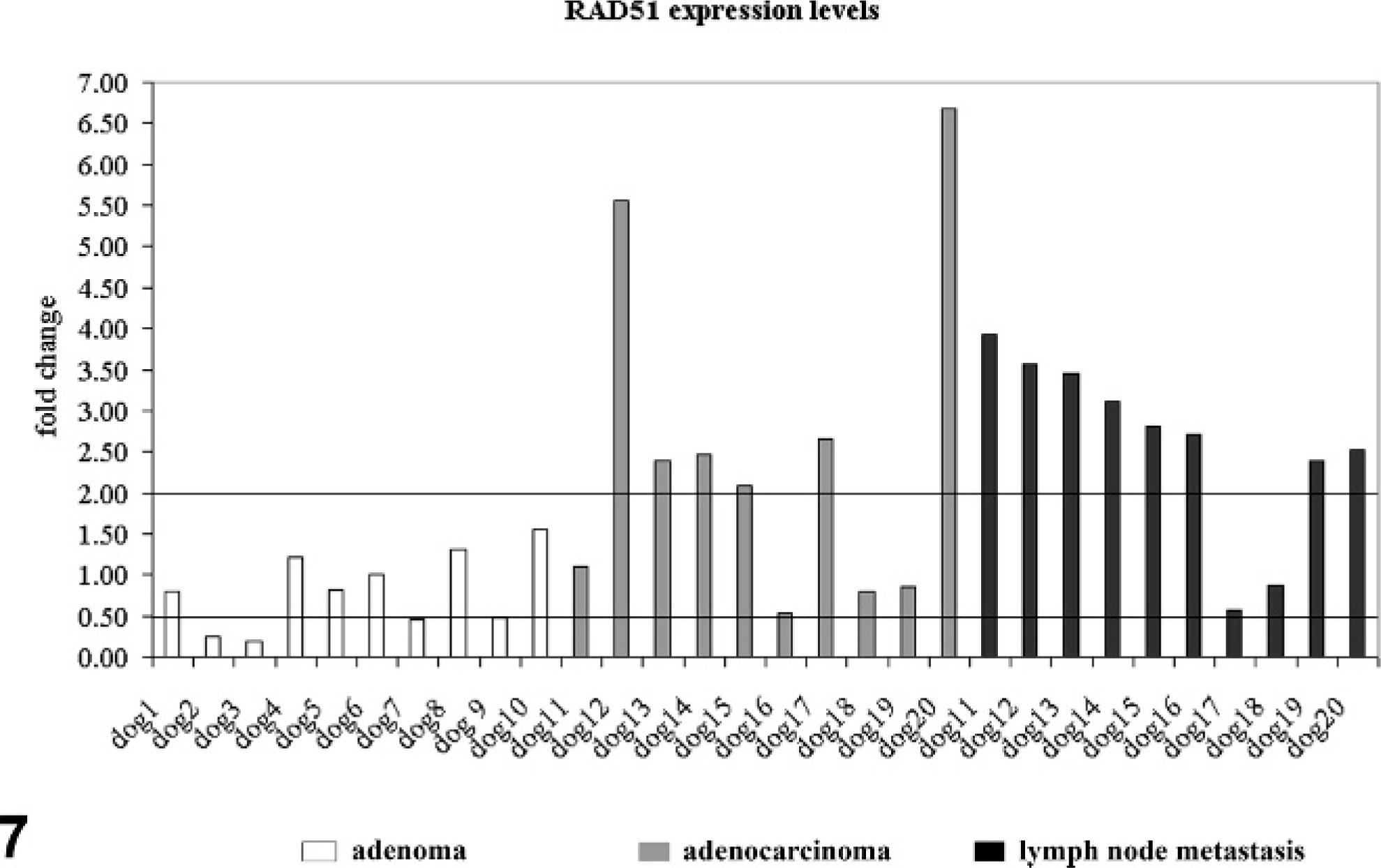
RAD51 expression in mammary adenomas (dogs Nos. 1–10, white columns), adenocarcinomas (dogs Nos. 10–20, gray columns), and their lymph node metastases (dogs Nos. 10–20, black columns) compared with non-neoplastic mammary epithelium of the same dog. Values are presented as fold change relative to the primary tumors. Values ≥2 were overexpression, and values ≤0.5 underexpression.
Expression levels of BRCA1, BRCA2, and RAD51 in adenocarcinomas versus normal gland
In adenocarcinomas, no consistent differential BRCA1 expression was observed in the 10 adenocarcinomas. BRCA1 was overexpressed in 3 of 10 dogs, and 7 of 10 dogs had expression levels comparable to non-neoplastic epithelium (Fig. 5). Overexpression of BRCA2 was observed in 5 of 10 dogs (Fig. 6). RAD51 was overexpressed in 6 of 10 dogs (Fig. 7). Interestingly, BRCA2 overexpression was always (5/10 dogs) associated with RAD51 overexpression.
Expression levels of BRCA1, BRCA2, and RAD51 in lymph node metastases versus normal gland
BRCA1 was overexpressed in microdissected lymph node metastases of 1 of 10 dogs (Fig. 5). In contrast, BRCA2 was upregulated in 5 of 10 dogs (Fig. 6). A more consistent upregulation of gene expression was observed for RAD51 (Fig. 7). Overexpression of RAD51 was observed in 8 of 10 lymph node metastases when compared with non-neoplastic epithelium (Figs. 5–7). Similarly to the primary adenocarcinomas, BRCA2 overexpression was always (5/10 dogs) associated with RAD51 overexpression.
Expression levels of BRCA1, BRCA2, and RAD51 in lymph node metastases versus primary tumors
To test whether the expression levels of BRCA1, BRCA2, and RAD51 were differentially regulated between adenocarcinomas and their lymph node metastases, relative mRNA copy numbers of the 3 genes per mRNA copy numbers of the 3 housekeeping genes were determined (Fig. 8). Microdissected tissue samples of the lymph node metastases were defined as sample of interest and tissue samples of the primary tumor as sample control. Calculation of fold change in gene expression was then conducted as described for the comparison of neoplastic versus normal gland epithelium. Increased BRCA1 and BRCA2 expression was observed in 2 of 10 dogs. RAD51 expression was increased 3 of 10 dogs, and 1 of 10 dogs had decreased RAD51 expression in lymph node metastases when compared with its primary tumor.
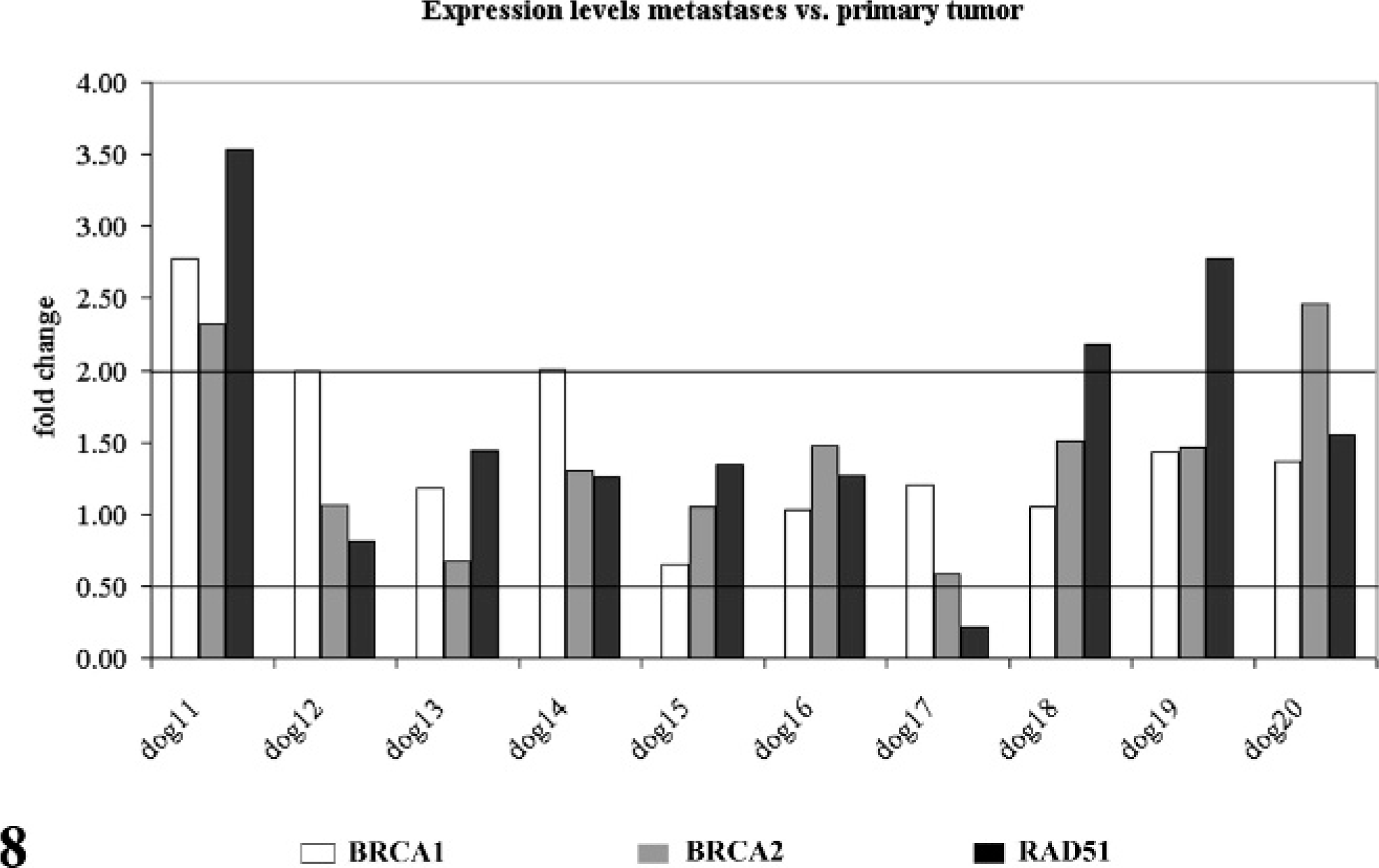
BRCA1 (white columns), BRCA2 (gray columns), and RAD51 (black columns) expression in lymph node metastases compared with their primary tumor masses (dogs Nos. 10–20). Values are presented as fold change relative to the primary tumors. Values ≥2 were overexpression, and values ≤0.5 underexpression.
Discussion
The role of the BRCA-RAD51 complex in the carcinogenesis and metastasis of canine mammary tumors is unclear at this point. So far, only a differential protein expression of BRCA1 in canine mammary tumors has been shown. 11 The results of this explorative study on highly defined microdissected tissue samples identified reduced expression of BRCA1 in 60% of simple adenomas. In contrast, 80% of the adenocarcinomas had no differential expression of BRCA1 (fold change < 0.5 or fold change > 2.0). Reduced BRCA1 mRNA levels in human breast cancer have been associated with nonsense mutations and lead to an increased resistance to apoptosis. No information about similar genetic changes is available for dogs, but the results show that in adenomas but not adenocarcinomas, a similar mechanism might be feasible. Similarly to the primary tumors, BRCA1 was not differentially expressed in 90% of the lymph node metastases when compared with normal gland. Altogether, it thus seems that in canine mammary tumors, BRCA1 expression is not unequivocally associated with histologic criteria of malignancy and therefore differs from the human malignancy.
For BRCA2, upregulation of gene expression was observed for 50% of the lymph node metastases. This was in contrast to the adenomas, in which 40% of the tumors had reduced expression. Nevertheless, the use of this gene as a tumor marker to differentiate between adenomas and potentially metastasizing adenocarcinomas is doubtful, because no clear cut expression differences were observed between adenomas and the primary adenocarcinomas. In human breast cancer, BRCA2 expression might also exert positive effects on proliferation. 14, 22, 23 This would be doubtless in contrast to the assumption that overexpression of the tumor suppressor BRCA2 should inhibit proliferation. 1 Hence, it has been suggested that overexpression might be the result of a negative regulatory loop in which BRCA2 is induced by cellular proliferation and then inhibits proliferation. 13 In this model, overexpression of BRCA2 is a protective response that decreases cancer progression. Nevertheless, the reduced BRCA2 expression levels in 4 of 10 adenomas indicate that proliferation alone does not upregulate BRCA2 expression in canine mammary tumors. Although not aggressive in behavior, adenomas consist of dysregulated and proliferating tumor cells and therefore should have BRCA2 overexpression too. Besides proliferation, increased genetic instability in rapidly proliferating metastatic tumor cells might also be a possible stimulus for BRCA2 expression. This is supported by the findings that RAD51 is overexpressed in 80% of lymph node metastases. Similarly, 50% of the primary tumors also had RAD51 overexpression. The parallel overexpression of BRCA2 and RAD51 in 5 of 10 lymph node metastases points at a direct interaction of both proteins like in human mammary epithelium. Enhanced expression of Rad51 protein in tumor cells is associated with high DNA repair capacity and elevated recombination rates in human cells, a mechanism that can also be expected in canine cells. 6 So far, there is no evidence that these differences in gene expression are due to alterations in the Rad51 coding sequence in spontaneous human breast cancer or canine mammary tumors. 7 Differential gene expression of RAD51 is therefore a physiologic response to changed genetic stability induced by the BRCA/RAD51-complex. Nevertheless, more experimental data are needed to substantiate this hypothesis.
In summary, the results of this explorative study indicate that RAD51 is overexpressed in most lymph node metastases of canine mammary tumors when compared with non-neoplastic mammary tissues, and RAD51 overexpression therefore might be associated with malignancy of canine mammary tumors. In further experimental studies with larger tumor numbers, it will be fascinating to characterize the regulatory mechanisms that induce BRCA2 and RAD51 overexpression lymph node metastases of canine mammary gland tumors. Nevertheless, heterogeneous expression in adenomas and primary adenocarcinomas excludes expression levels of the 3 gene products as unequivocal prognostic factors for future metastasis of canine mammary tumors in routine diagnostics.
Footnotes
Acknowledgements
We thank Monika Schärig for her excellent technical assistance.
