Abstract
RAD51 is a key enzyme of homologous recombination and repair of DNA double-strand breaks. RAD51 mRNA expression levels are significantly increased in laser-microdissected mammary simple carcinomas and their lymph node metastases when compared to adenomas or nonneoplastic mammary gland of the same dog. Here, RAD51 protein expression was analyzed by immunohistochemistry in paraffin-embedded mammary carcinomas and their lymph node metastases of 40 dogs, adenomas of 48 dogs, and nonneoplastic mammary gland of 88 dogs. Number of cells with nuclear RAD51 expression was significantly (P ≤ .05) increased in carcinomas when compared to adenomas and metastases. In contrast, no significant differences in the number of RAD51-expressing cells were detected when metastases were compared with adenomas and nonneoplastic gland. RAD51 expression in carcinomas was correlated with expression in metastases but not with histologic grade. In conclusion, the increased number of RAD51-expressing cells in carcinomas might indicate genomic instability in these cells. Nevertheless, the increased RAD51 mRNA expression in metastases could not be confirmed by immunohistochemistry.
RAD51 is a protein that interacts directly or indirectly with a large number of proteins involved in DNA repair and cell cycle control. 22 RAD51 protein functions in DNA repair by mediating homologous pairing and strand exchange reactions. 5 Its expression is regulated mainly by BRCA1 and BRCA2, important proteins involved in DNA damage signaling. 26 In general, it is assumed that RAD51 can compensate for some aspects of BRCA2 gene defects and that RAD51 expression levels may be an important modifier of the BRCA2 defective genotype. 7 Besides the interactions of RAD51 with key players in human breast tumorigenesis in normal mammary gland, there is additional evidence to support a direct role for RAD51 in the development of human breast cancer. 15 Both RAD51 overexpression and decreased expression are associated with a poor prognosis in human breast cancer, and RAD51 expression levels have therefore been proposed as a clinically relevant tumor marker. 16,17,25 Nevertheless, direct carcinogenic effects due to RAD51 genomic variation have not been found in comprehensive studies on human breast cancer patients. 4,15,24 The function of RAD51 protein in the carcinogenesis of canine mammary tumors is unclear, but a reduced and aberrant expression of its inducer BRCA1 has been shown in canine mammary tumors. 20
We recently observed an up-to-fourfold increase in RAD51 mRNA expression levels in laser-microdissected tissue samples of 50% of canine mammary carcinomas and 80% of their lymph node metastases when compared to nonneoplastic mammary gland of the same dog. 12 In contrast, none of the canine mammary adenomas analyzed had elevated RAD51 mRNA expression, but 40% had reduced expression when compared to nonneoplastic mammary gland of the same dog. To corroborate the significance of these findings on the posttranslational level, RAD51 protein expression was analyzed immunohistochemically in canine mammary adenomas, carcinomas, and lymph node metastases that were recently used for mRNA quantification recently and additional tissue samples. 12
Formalin-fixed and paraffin-embedded tissue samples of 48 dogs with simple mammary adenomas and of 40 dogs with simple mammary carcinomas metastatic to the lymph node, as well as the nonneoplastic (normal and hyperplastic) mammary gland of all 88 dogs, were selected from the archive of the Department of Veterinary Pathology. Paraffin sections (2 μm) were stained with hematoxylin and eosin, and tumors were classified according to the World Health Organization and the grading system by Elston and Ellis. 9,19 Histologic grading of the simple carcinomas (according to Elston and Ellis) found 20 of 48 to be grade 2 and 28 of 48 to be grade 3.
RAD51 expression was immunohistochemically detected using the avidin-biotin complex method. 10 In brief, anti-RAD51 specific antibody (polyclonal antibody, diluted 1:800; catalog No. sc-34566, Calbiochem, San Diego, CA) was diluted in Tris-buffered saline (50 mM, pH 7.6) and incubated at 4°C overnight after a blocking step with 50% goat serum in Tris-buffered saline for 30 minutes at room temperature. Detection of RAD51 required antigen retrieval by preincubation with pronase (10 μg/ml) for 30 minutes. As secondary antibody, goat antirabbit immunoglobulin G (pab, 1:200; catalog No. BA1000, Vector, Peterborough, UK) was used. Diaminobenzidine tetrahydrochloride (catalog No. D8001, Sigma Aldrich, St. Louis, MO) was used as chromogen, and slides were counterstained with hematoxylin (Merck GmbH, Darmstadt, Germany). As positive control, formalin-fixed and paraffin-embedded MCF7 cells were used. For negative controls, sections were incubated with an irrelevant rabbit serum at the dilution used. Nuclear RAD51 immunolabeling was evaluated in 10 random 400× magnification fields, as has been shown recently. 20,21 Numbers of RAD51-expressing and RAD51-nonexpressing mammary epithelial cells were counted in all fields, and percentage of positive cells per total cell number was calculated. Statistical analyses included the Kolgomorov–Smirnov test and analysis of variance. Data were found to be normally distributed, and analysis of variance was applied. Differences between tissue groups were considered statistically significant for P ≤ .05. Kendall–Tau b test was used for calculation of correlation analysis. All analyses were performed using SPSS 16.0. Nuclear RAD51 expression was found in 21.4% ± 10.1 (156 of 732 nuclei) of nonneoplastic mammary gland epithelial cells (Figs. 1A, 2). In adenomas, nuclear RAD51 expression was observed in 27.9% ± 12.9 (225 of 805 nuclei) of the tumor cells (Fig. 1B). RAD51 expression was heterogeneous within the tumor. Nuclear RAD51 expression was found in 35.9% ± 8.5 (218 of 783 nuclei) of carcinoma cells (Fig.1C). Again, RAD51 expression was heterogeneous within the tumors. In sum, 26.7% ± 14.2 (143 of 534 nuclei) of the metastastic carcinomas cells in the lymph nodes had nuclear RAD51 expression (Fig. 1D). Cytoplasmic RAD51 expression was not found in any tissue analyzed. Nuclear RAD51 expression was also found in few single fibroblasts, epidermal keratinocytes, and apocrine glands and in few single lymphocytes in lymph nodes. Statistical analysis of RAD51 expression revealed a significant increase in average percentage of RAD51-positive cells when nonneoplastic gland was compared with adenomas or carcinomas (Fig. 2). Furthermore, RAD51 was expressed in a significantly higher percentage of positive nuclei when carcinomas were compared with adenomas or metastases. No significant differences were found between adenomas and metastases or between nonneoplastic gland and metastases. RAD51 expression in primary tumors correlated significantly (P < .001) with RAD51 expression in metastases. RAD51 expression and histologic grade of the primary tumor were not correlated.
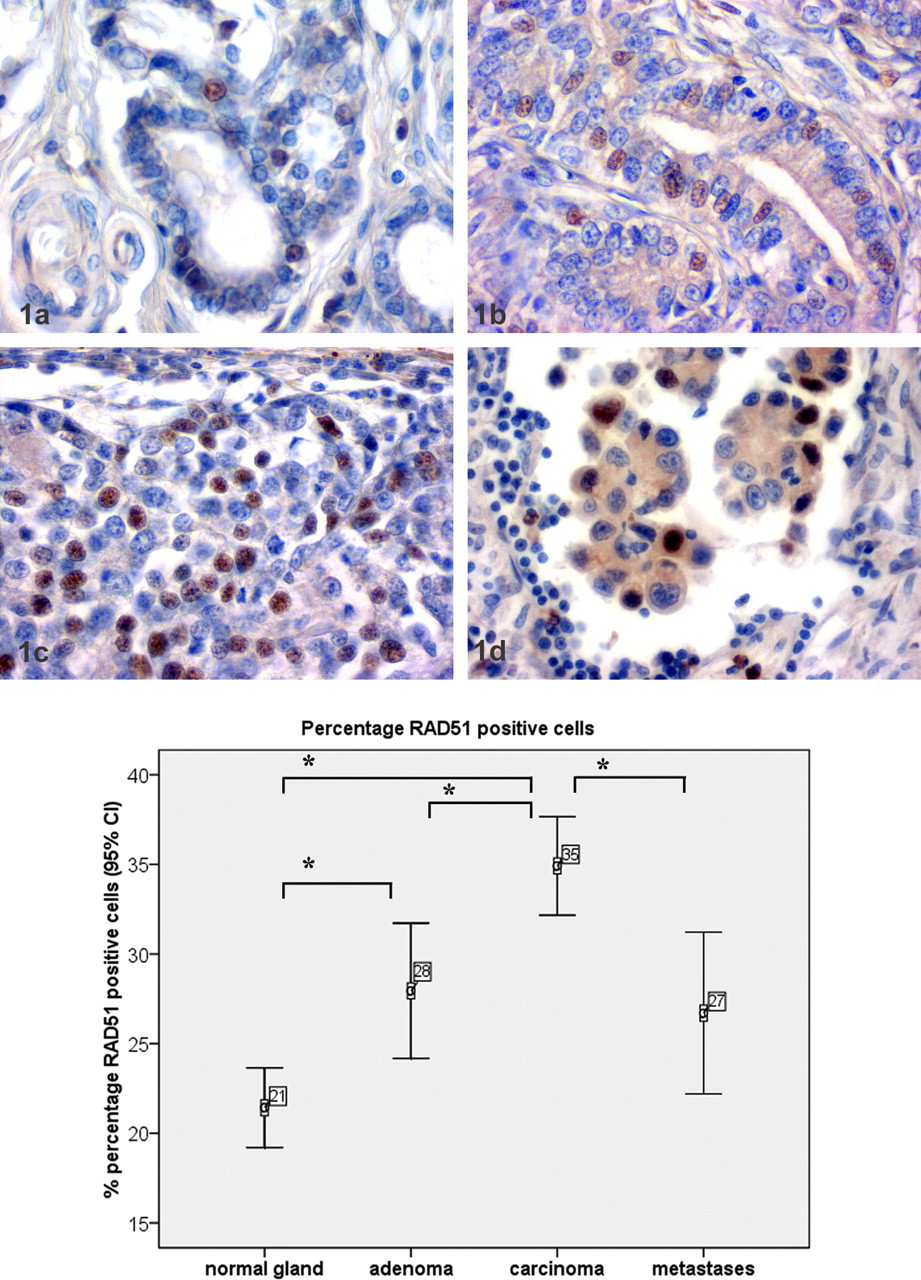
The results presented in this study have two main results. First, the number of RAD51-positive cells is increased in benign and malignant primary canine mammary tumors when compared to nonneoplastic gland, and the number of RAD51-positive cells is higher in malignant tumors when compared with benign tumors. Furthermore, RAD51 expression in primary tumors correlated significantly with RAD51 expression in the lymph node metastases. RAD51 overexpression can stimulate but also reduce homologous recombination and can potentially lead to chromosome rearrangements in tumor progression. 3,14,27,28 Nevertheless, inoculation of immunodeficient mice with neoplastic cells overexpressing RAD51 did not result in an increased tumor burden, whereas RAD51-negative cells without RAD51-mediated homologue recombination had twofold-increased tumorigenesis. 6 The application of this experimental in vitro and in vivo data on spontaneous tumors is difficult. In human breast cancer, both increased and decreased levels of RAD51 expression have been found. For example, decreased RAD51 expression was found in mostly sporadic and some BRCA1-associated breast cancer samples, whereas in another study, histological grading of sporadic invasive ductal breast cancer was found to correlate significantly with overexpression of wild-type RAD51. 17,29
The second major finding of this study is that the significantly increased mRNA levels in laser-microdissected lymph node metastases could not be confirmed by immunohistochemistry. 12 The reasons for this discrepancy between mRNA and protein expression in lymph node metastases are unclear. So far, DNA damage is still the main inducer of increased RAD51 transcription, but p53 and flavopiridol also suppress RAD51 transcription. 1,2,11,13 It is therefore assumable that a posttranscriptional regulation of RAD51 in lymph node metastases might be involved, although a similar regulation has not been described so far in any species. The ataxia telangiectasia mutated gene and the cellular Abelson murine leukemia viral oncogene homolog 1 are necessary for correct posttranslational assembly of the RAD51 complex. 8 Loss of ataxia telangiectasia mutated gene and increased cellular Abelson murine leukemia expression are involved in dysregulated RAD51 expression and carcinogenesis of human breast cancer, and a similar mechanism might be true for canine mammary tumors. 18,23 Furthermore, the selective process during metastatic progression or the specific microenvironment of the lymph node may play a role in the different RAD51 protein expression in primary tumors and their lymph node metastases.
In conclusion, expression of the RAD51 protein is significantly increased in primary carcinomas but not in adenomas and lymph node metastases. The significantly increased RAD51 mRNA expression level in microdissected tumor samples of lymph node metastases could not be confirmed by immunohistochemistry, suggesting defective protein translation after malignant progression. The results therefore argue that increased DNA repair response in benign and malignant mammary tumors might be indicative of genomic instability in this tumor type.
Footnotes
Acknowledgements
We thank Monika Schärig for excellent technical assistance.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
