Abstract
The equine sarcoid is the most common dermatologic neoplasm reported in horses. Bovine papillomavirus (BPV) types 1 and 2 are associated with sarcoids, in which the expression of the major transforming oncoprotein (E5) is often recorded. The transformation activity of the virus is due to the binding of the E5 to the platelet-derived growth factor β receptor (PDGFβ-r). In the present study, we show by Western blot in 4 sarcoid samples and 3 normal equine skin samples that the PDGFβ-r is more phosphorylated in sarcoid tissue than in normal skin (P < .001). Furthermore, the physical interaction between the activated receptor and the 85-kDa regulatory subunit (p85) of phosphatidylinositol-3-kinase (PI3K) is shown by coimmunoprecipitation. The PI3K-AKT-cyclin D3 molecular pathway downstream to the activation of the PDGFβ-r is shown to be expressed, and the amount of the investigated molecules is higher than normal (P < .001), suggesting an activation of these effectors in sarcoids. Further, we demonstrate that phospho-JNK and phospho-JUN are more expressed in sarcoids than in normal skin. Our results provide new insights into the pathogenesis of equine sarcoids and support the validity of this in-vivo model to further characterize the molecular pathways underlying BPV E5-induced carcinogenesis.
Introduction
Sarcoids are the most common fibroblastic skin tumor affecting horses, mules, and donkeys. They are histologically characterized by disorganized dermal proliferation of spindle-shaped fibroblasts that form whorls and by epidermal hyperplasia, hyperkeratosis, and rete peg formation. 33,40 They are locally invasive and often occur at sites of a previous injury or scar, the most common sites being the skin of the head, the ventral abdomen, the legs, and the paragenital region. 38
In the early 1950s, bovine papillomavirus (BPV) was proposed as the causative agent of sarcoids on the evidence that the experimental inoculation of horses with bovine warts induced tumors. 34 Since then many research groups from different nations have reported the close association between BPV and sarcoids. 11 In the USA, both BPV-1 and BPV-2 types have been detected in sarcoid tumors in equal frequency, whereas the BPV-1 type is the predominant type in Europe. 1,4,10,11 The genome of BPV contains early and late coding regions. The early coding genes regulate viral replication, the maintenance of the episomal state, 42 and the activation of cell proliferation. The late coding genes encode structural proteins, such as L1, which has been recently demonstrated to be in complex, in some cases, with BPV-1 DNA. 6 This suggests that sarcoids may also be productive lesions. In addition, it has been well known that the BPV genes E5, E6, and E7 are each oncogenes. 39
E5 is a transmembrane protein with 44 amino acids that dimerizes via 2 cysteine residues (Cys 37 and Cys 39) that are located in the predicted extracellular domain of E5. 7 It is the major oncoprotein of BPV and is expressed in the natural host in the deep layers of the epithelium, 2,46 being localized largely to the membranes of the endoplasmic reticulum and Golgi apparatus of the host cells. 7,35
Because of its relatively small size and structure, it is unlikely that E5 induces transformation by an enzymatic function. E5 interacts with the 16-K subunit c protein, a component of the vacuolar H+-ATPase pump. 13,18,20,22 The pump is critical for the function of cellular compartments that process growth factors, and the binding of the E5 protein may result in alteration of this processing.
The main mechanism underlying BPV carcinogenesis lies in the binding (both in vitro and in vivo) of the E5 to the platelet derived growth factor β receptor (PDGFβ-r). 5,16,17 The binding of dimeric ligand, PDGF-BB, to the extracellular domain of the receptor induces dimerization and receptor activation, resulting in autophosphorylation of key tyrosine residues. Once phosphorylated, these tyrosine residues become docking sites for important SH-2 domain-containing substrates, 23 whose activation induces signaling pathways that lead to cell proliferation, morphologic, changes, and motility.
Downstream effectors of the E5-PDGFβ-r binding were unknown until a recent report showed the expression of various substrates due to sustained activation of the PDGFβ-r in mortal human fibroblasts. 37 The present study investigates the expression of some of the molecules related to the PI3K-AkT pathway, one of several downstream effector pathways of E5-PDGFβ-r. This pathway is a key signaling transduction cascade controlling the regulation of cell growth, proliferation, and cell survival, which constitute critical steps towards tumor formation and malignant cell dissemination. 47 Activation of PI3K and phosphatidylinositol generation are required for AKT, which is commonly dysregulated in a variety of human tumors. AKT regulates many of the key effector molecules involved in apoptosis, anoikis, and cell cycle progression. 9 In addition, the activation of the PI3K/AKT pathway induces expression of D-type cyclins, which acts as a sensor of the external stimuli elicited by transmembrane receptors, allowing cells to progress through the G1 phase of the cell cycle. 41
It has also been shown that the E5-PDGFβ-r binding may activate 2 transforming signals: c-JUN NH2-terminal kinase (JNK) and c-JUN. JNK has been implicated in oncogenic transformation. 14 A major outcome of JNK activation is the phosphorylation of the transcription factor c-JUN and augmentation of AP-1 transcriptional activity. 14 Furthermore, it has also been established that JNK activation is regulated by the AKT signaling pathway. 27
There are few studies relating to the mechanisms of cell transformation in equine sarcoids. In a previous study on a subset of sarcoids, the presence of BPV DNA, the expression of oncoproteins E5 and E7, and the expression of the PDGFβ-r were demonstrated. 4 We investigated the activation of PDGFβ-r (phospho-PDGFβ-r) and the expression of some of the downstream signaling intermediates required for growth proliferation, namely, the 85-kDa regulatory subunit (p85) of phosphatidylinositol-3-kinase (PI3K), AKT, phosphoAKT (p-AKT), cyclin D3, phospho-JNK (p-JNK), and phospho-JUN (p-JUN) in E5 positive sarcoid samples.
Materials and Methods
Sample collection
Equine sarcoid samples were obtained from 3 horses (1 verrucous and 2 nodular types) and 1 donkey (nodular type). A normal skin sample was obtained 10 cm away from the sarcoid of one of the horses (T2), and 2 more skin samples were included from sarcoid-free horses. The animals were between 5 and 14 years old. Tissue samples were divided into 2 parts: 1 part was fixed in formalin for routine histologic diagnosis, and 1 part of the fresh material was perfused thoroughly with cold 0.9% NaCl and frozen at −80°C. All tumor samples were histologically classified as sarcoids and were found to harbor BPV-1 DNA and express oncoprotein E5 (data not shown).
Sodium dodecyl sulfate polyacrylamide gel electrophoresis/Western blotting of normal skin and tumor samples
Molecular analysis was performed on all 4 tumor samples and on the 3 normal skin samples. They were homogenized in 7.5 ml JS buffer (5 ml 1 M hepes pH 7.5, 3 ml 5 M NaCl, 1 ml glycerol, 10 ml 10% Triton, 0.15 ml 1 M MgCl2, 5 ml 0,1 M ethylene glycol tetraacetic acid) added with 2 ml 0.1 M sodium pyrophosphate, 0.2 ml aprotinin at 5 mg/ml, 0.2 ml 0.1 M phenylmethylsulfonyl fluoride (PMSF), 0.2 ml 500 mM sodium orthovanadate (Na2VO3), and 0.25 ml 2 M NaF. Homogenates were divided into small aliquots and stored at −80°C until use. The quantity of proteins was determined by use of a protein assay kit (Bio-Rad protein assay). Equal amounts of proteins were boiled for 5 minutes in sodium dodecyl sulfate (SDS) sample buffer, loaded on bis/acrylamide gel with different percentage, and electrophoresed. After electrophoresis, the samples were blotted from the gel onto PVDF membranes using a Trans-Blot apparatus (Bio-Rad) according to the manufacturer's instructions. The membranes were blocked with 5% nonfat dry milk in Tris-buffered saline (TBS) at room temperature, washed with TBS-0.1% Tween, and incubated with the following primary antibodies: 1) goat anti-pPDGFβ-r (BD Pharmingen) at 1 : 500, 2) rabbit anti-PI3K (p85) (Upstate Biotechnology) at 1 : 2,000, 3) rabbit anti-AKT (Cell Signaling Technology) at 1 : 1,000, 4) rabbit anti-pAKT (pSer 473) (Cell Signaling Technology) at 1 : 1,000, 5) mouse anti-cyclin D3 at 1 : 500 (BD Pharmingen), 6) rabbit anti-p-JNK at 1 : 2,000 (BD Pharmingen), and 7) rabbit anti-p-JUN at 1 : 2,000 (Upstate Biotechnology).
After appropriate washing steps, peroxidase-conjugated anti-rabbit IgG anti-mouse IgG or anti-goat IgG (Santa Cruz Biotechnology, Inc.) were applied for 2 hours at a 1 : 1,000 dilution. After washing, bound antibody was visualized on ECL film (Amersham Pharmacia Biotech). The blots were stripped and reprobed against mouse anti-actin antibody (Calbiochem) at 1 : 5,000 to confirm equal loading of proteins in each lane. Protein expression levels were quantitatively estimated by densitometry using a Discover Pharmacia scanner equipped with a Sun Spark Classic densitometric workstation. The protein concentrations were normalized to the actin level and expressed as the densitometric ratio.
Sodium dodecyl sulfate polyacrylamide gel electrophoresis/Western blotting of cell lines
Young HSF4012 (NHDF4012) human foreskin fibroblasts (HDFs) and an HDF strain stably expressing wild-type BPV E5 (HDFwtE5), which were previously shown to express the investigated pathway, were kindly provided by Dr. Lisa Petti (Center for Immunology and Microbial Disease, Albany Medical College, New York, NY, USA).
To prepare HDF extracts, cells were washed twice in phosphate-buffered saline and then lysed in radioimmunoprecipitation assay (RIPA) buffer supplemented with the following inhibitors: 20 mM 3-(N-morpholino)propanesulfonic acid (MOPS), 150 mM NaCl, 1% nonylphenoxylpolyethoxylethanol (Nonidet P-40), 0.1 M ethylenediaminetetraacetic acid (EDTA), 1% sodium deoxycholate, 1 M PMSF, 500 mM Na2VO3, 0.1% SDS. The quantity of proteins was determined by use of a protein assay kit (Bio-Rad protein assay). Equal amounts of proteins were boiled for 5 minutes in SDS sample buffer, loaded on bis/acrylamide gel with different percentage, and electrophoresed. After electrophoresis, the samples were blotted from the gel onto PVDF membranes using a Trans-Blot apparatus (Bio-Rad) according to the manufacturer's instructions. The membranes were blocked with 5% nonfat dry milk in TBS buffer at room temperature, washed with TBS-0.1% Tween, and incubated with the following primary antibodies: 1) goat anti-pPDGFβ-r (BD Pharmingen) at 1 : 500, 2) rabbit anti-PI3K (p85) (Upstate Biotechnology) at 1 : 2,000, 3) rabbit anti-AKT (Cell Signaling Technology) at 1 : 1,000, 4) rabbit anti-pAKT (pSer 473) (Cell Signaling Technology) at 1 : 1,000, 5) mouse anti-cyclin D3 at 1 : 500 (BD Pharmingen), 6) rabbit anti-p-JNK at 1 : 2,000 (BD Pharmingen), 7) rabbit anti-p-JUN at 1 : 2,000 (Upstate Biotechnology).
After appropriate washings steps, peroxidase-conjugated anti-rabbit IgG anti-mouse IgG or anti-goat IgG (Santa Cruz Biotechnology, Inc.) were applied for 2 hours at a 1 : 1,000 dilution. After washing, bound antibody was visualized on ECL film (Amersham Pharmacia Biotech). The blots were stripped and reprobed against mouse anti-actin antibody (Calbiochem) at 1 : 5,000 to confirm equal loading of proteins in each lane. Protein expression levels were quantitatively estimated by densitometry using a Discover Pharmacia scanner equipped with a sun spark classic densitometric workstation. The protein concentrations were normalized to the actin level and expressed as the densitometric ratio.
Coimmunoprecipitation and sodium dodecyl sulfate polyacrylamide gel electrophoresis/Western blotting
Proteins from the tumor samples and the normal skin were recovered as described above. The quantity of proteins was determined by use of a protein assay kit (Bio-Rad protein assay). A-Sepharose beads (Pharmacia) (0.08 g/ml) were swollen for 1 hour in TBS (10 mM Tris-HCl, pH 7.4, 165 mM NaCl). A-Sepharose beads (0.5 ml) was added to 0.5 ml of the homogenate, and the mixture was rotated overnight at 4°C for preclearing. Anti-pPDGFβ receptor (2.5 µl) was added to 0.5 ml of A-Sepharose beads, and the mixture was rotated overnight at 4°C. The beads were washed 5 times with TBS or RIPA buffer (20 mM Mops, 150 mM NaCl, 1% Nonidet P-40, 0,1 M EDTA, 1% sodium deoxycholate, 1 M PMSF, 500 mM Na2VO3, 0.1% SDS) and were added to 0.5 ml of the precleared homogenized. The mixture was rotated overnight at 4°C for immunoprecipitation. The beads were washed 5 times with TBS or RIPA buffer and then resuspended in Laemmli sample buffer (SDS, Tris-HCl pH 6.8, glycerol, Bromophenol blu, β-mercaptoethanol) and boiled 5 minutes. The beads were pelleted, and the supernatant was decanted in a new tube. The immunoprecipitates were loaded on 7.5% bis/acrylammide gel and electrophoresed. The proteins were blotted from the gel onto nitrocellulose membranes. The membranes were blocked with 5% nonfat dry milk in TBS buffer at room temperature, washed with TBS-0.1% Tween, and incubated with the rabbit anti-PI3K (p85) (Upstate Biotechnology) at 1 : 2,000. After appropriate washings steps, peroxidase-conjugated anti-rabbit IgG (Santa Cruz Biotechnology, Inc.) was applied for 2 hours at a 1 : 1,000 dilution. After washing, bound antibody was visualized on ECL film (Amersham Pharmacia Biotech).
Statistical analysis
Statistical analyses were performed using independent samples t-tests, and P < .001 was considered to indicate a significant difference. All statistical analyses were performed using SPSS 13 software.
Results
PDGFβ-r activation and its physical interaction with p85-PI3K in sarcoids and normal skin
The 4 sarcoids and the 3 normal skin samples were examined to determine the phosphorylation status of the PDGFβ-r. An increase of the pPDGFβ-r expression levels in sarcoid samples compared with normal (P < .001) was recorded by densitometric analysis (Fig. 1). The expression levels of p85-PI3K in sarcoids were increased compared with normal tissue, and the p85-PI3K/actin densitometric ratios showed that p85-PI3K expression levels were increased in sarcoids compared with normal tissue (Fig. 2a).
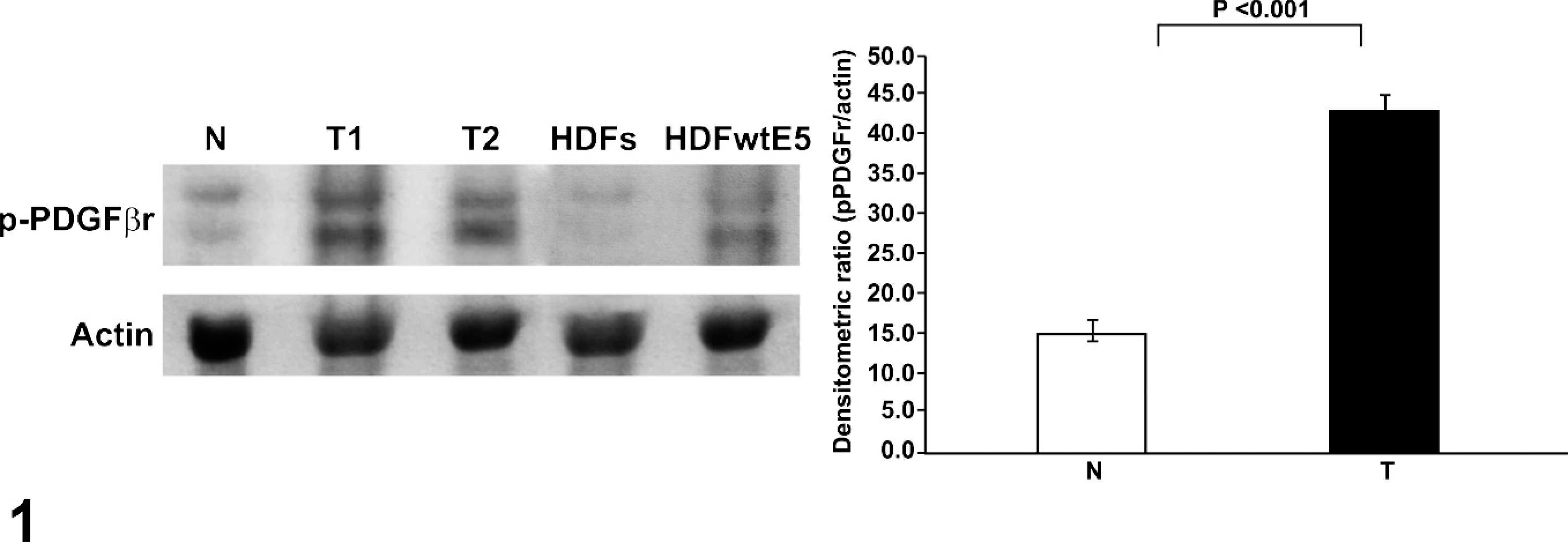
Equine sarcoids, normal skin, human diploid fibroblasts (HDFs), and HDFs expressing wild-type bovine papillomavirus E5 (HDFwtE5). Western blot analysis for activated platelet-derived growth factor β receptor (PDGFβ-r) in sarcoids. Homogenized samples from 2 representative sarcoids (T) and normal equine skin (N) and protein extracts from HDFs and HDFwtE5 were electrophoresed on sodium dodecyl sulfate polyacrylamide gel electrophoresis and analyzed by Western blot with goat IgG anti-pPDGFβ-r at 1 : 500. The blots were stripped and reprobed against mouse IgG anti-actin 1 : 5,000 (lower blot). A comparison of average p-PDGFβ-r expression levels in equine sarcoids (T) and normal equine skin (N). Data represent the mean ± SD.
To determine whether the pPDGFβ receptor was in a molecular complex with the p85-PI3K protein, the ability of pPDGFβ receptor to coprecipitate with the p85-PI3K was assessed. Protein lysates from the tumor specimens, as well as the normal equine skins, were immunoprecipitated with anti-pPDGFβ receptor antibody. The immunoprecipitates were probed for the presence of the p85-PI3K by Western blotting with the respective antibody. p85-PI3K was detected in 1 of 4 tumors and in the normal pPDGFβ receptor-immunoprecipitates (Fig. 2b), suggesting that p85-PI3K and the pPDGF β receptor are present in a stable complex in BPV-1–positive naturally occurring equine sarcoids.
PI3K-AKT signaling pathway in sarcoids and normal skin
We then aimed to see the expression of signaling intermediates downstream of PI3K. Specifically, we ascertained the expression of AKT, p-AKT, and cyclin D3 by immunoblot. In all of the tumor samples, the anti-AKT antibody recognized a band of the right molecular weight in both normal and neoplastic tissue. To further confirm the activation of AKT, we then performed immunoblots with an anti-p-AKT antibody. An increase of the amount of p-AKT levels in sarcoid samples was recorded (Fig. 2c) compared with normal skin samples. The blots were stripped and reprobed with an anti-actin antibody to be sure of equal loading in all lanes. The densitometric ratio of p-AKT/actin showed that p-AKT expression levels were significantly higher in samples of equine sarcoids than in normal tissue (P < .001).
Cyclin D3 expression was also examined by immunoblots, and densitometric analysis showed an increase of cyclin D3 in sarcoids versus normal (P < .001), thus suggesting that cyclin D3 is expressed in cancer more than in normal tissue (Fig. 2d).
Finally, expression levels of p-JNK and p-JUN, major substrates for JNK, were assessed using phospho-specific antibodies. Both p-JNK and p-JUN levels were increased in cancer samples when compared with normal. Densitometric ratios of p-JNK/actin and p-JUN/actin showed that p-JNK and p-JUN expression levels were higher in sarcoids than in normal tissue (P < .001) (Fig. 3a, b).
PDGFβ-r activation and PI3K-AKT signaling pathway in cell lines
HDFs and HDFwtE5 cell lines were investigated by immunoblots to further evaluate the expression of the investigated molecules. The pPDGFβ-r was expressed at higher levels in HDFwtE5 compared with HDFs (Fig. 1). Similarly, the levels of pAKT, Cyclin D3 (Fig. 2), p-JUN, and p-JNK (Fig. 3) were also found to be higher in HDF wtE5 cell lines compared with HDF cell lines.
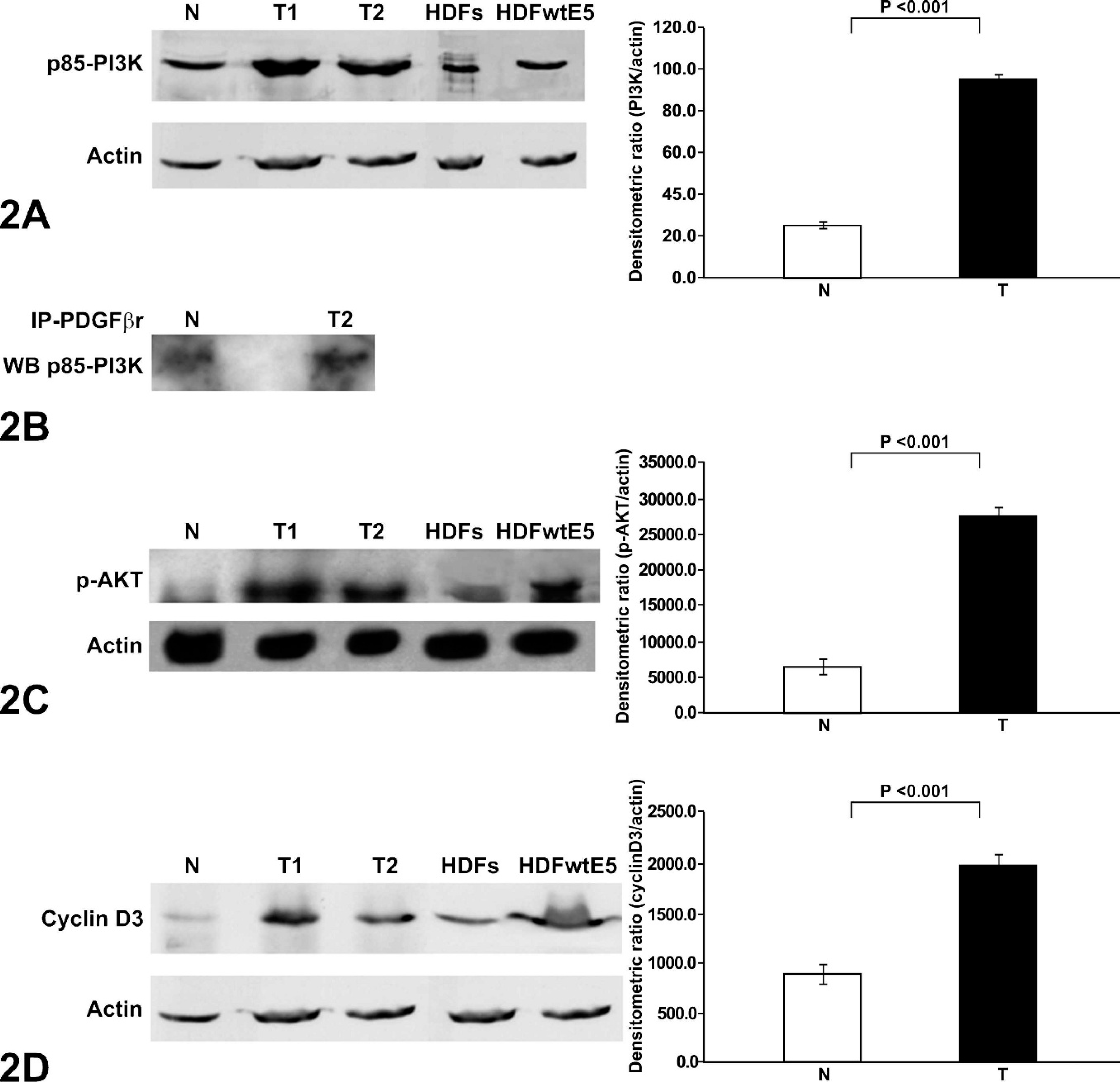
Equine sarcoids, normal skin human diploid fibroblasts (HDFs), and HDFs expressing wild-type BPV E5 (HDFwtE5). p-PDGFβ-r and PI3K coprecipitate in sarcoids overexpressing PI3K, p-AKT, and cyclin D3. Homogenized samples from 2 representative sarcoids (T) and normal equine skin (N) and protein extracts from HDFs and HDFwtE5 were electrophorized on sodium dodecyl sulfate polyacrylamide gel electrophoresis and analyzed by Western blot with rabbit IgG anti-PI3K at 1 : 2,000 (p85) (panel A upper blots); Coimmunoprecipitation of the PI3K with the anti-p-PDGFβ-r (panel B); rabbit IgG anti-pAKT (pSer 473) 1 : 1,000 (panel C, upper blot), mouse IgG anti-cyclin D3 1 : 500 (panel D, upper blot). The blots were stripped and reprobed against mouse IgG anti-actin 1 : 5,000 (lower blots). A comparison of average PI3K, p-AKT and cyclin D3 expression levels in equine sarcoids (T) and normal equine skin (N). Data represent the mean ± SD.
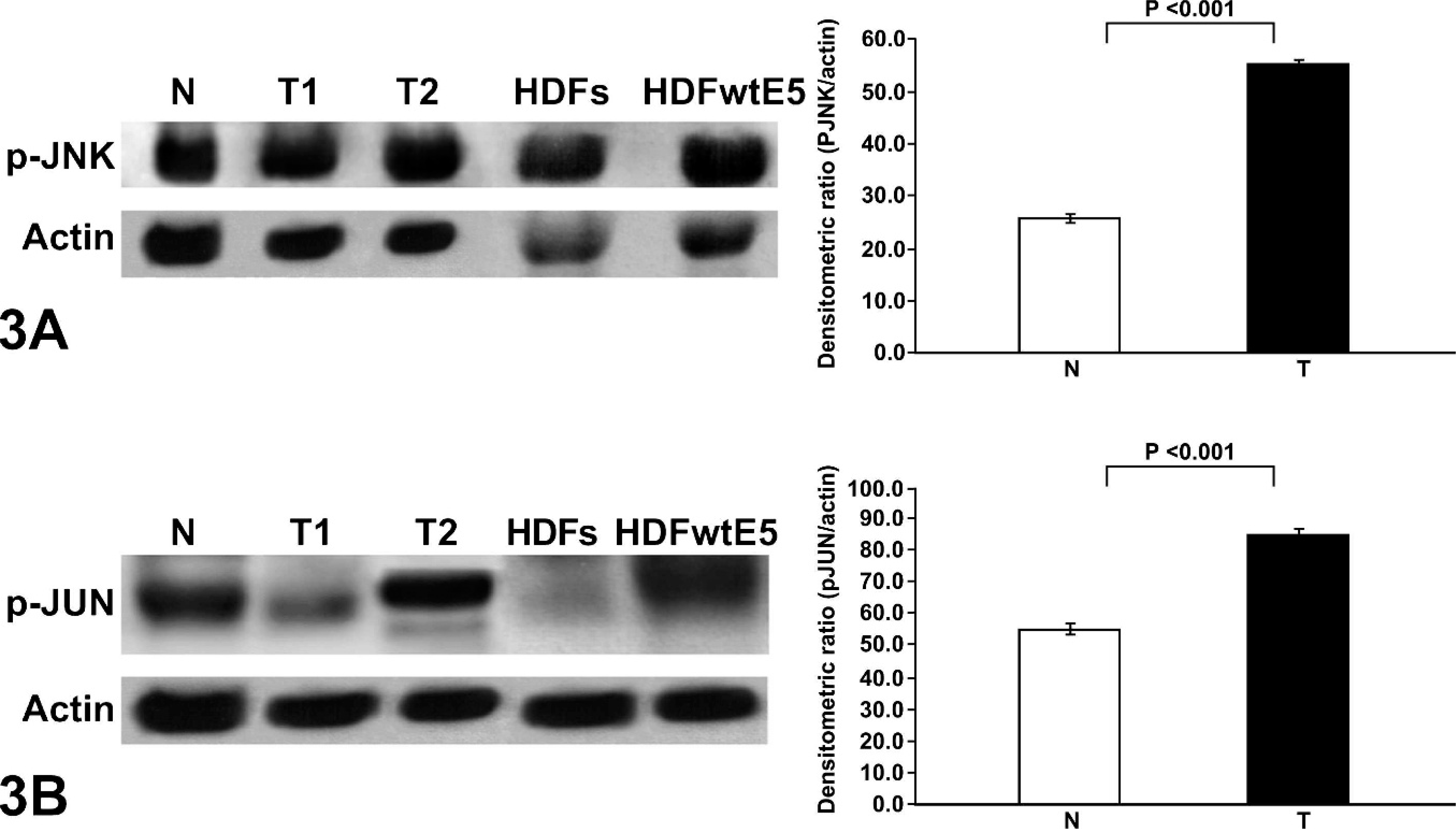
Equine sarcoids, normal skin, human diploid fibroblasts (HDFs), and HDFs expressing wild-type BPV E5 (HDFwtE5). Western blot analysis showing p-JUN and p-JNK hyperphosphorylation in sarcoids versus normal. Homogenized samples from 2 representative sarcoids (T) and normal equine skin (N) and protein extracts from HDFs and HDFwtE5 were electrophorized on sodium dodecyl sulfate polyacrylamide gel electrophoresis and analyzed by Western blot with rabbit IgG anti-p-JNK 1 : 2,000 (panel A upper blot) and rabbit IgG anti-p-JUN 1 : 2,000 (panel B upper blot). The blots were stripped and reprobed against mouse IgG anti-actin 1 : 5,000 (lower blot). A comparison of average p-JNK and p-JUN expression levels in equine sarcoids (T) and normal equine skin (N). Data represent the mean ± SD.
Discussion
In a recent previous study, we showed that in a subset of BPV-associated sarcoids the major BPV E5 oncoprotein is expressed along with the PDGFβ-r. 4 The present study demonstrates that in sarcoids 1) the PDGFβ-r is activated and interacts with the PI3K, 2) the PI3K-AKT signaling pathway is activated, and 3) p-Jun and p-JNK are overexpressed.
Equine sarcoids are biologically attractive tumors, because it is the only known case of cross-species infection by BPV-1 or BPV-2 in horses and other equids. 8 The oncogenic activity of BPV is mostly importantly due to its E5 oncoprotein. E5 induces cell transformation of cultured rodent fibroblast and keratinocytes by binding to and activating the PDGFβ-r, a type I transmembrane receptor tyrosine kinase, whereas it does not directly activate either the α subunit of the PDGF-or the epidermal growth factor receptor. 16,31,36 Each E5 dimer binds to 2 molecules of the PDGFβ-r forming a stable complex with it and resulting in tyrosine phosphorylation of each receptor molecule and activation of cellular SH2 domain-containing signaling proteins. 17,29,30 This mechanism takes place in vitro, as well as in naturally occurring bovine bladder epithelial cancer. 5
Here we demonstrate that in a subset of E5-positive sarcoids expressing the PDGFβ-r, the receptor itself is hyperphosphorilated and thus activated. We speculate that the activated receptor may be in complex with E5, inducing receptor dimerization and trans-phosphorylation, as generally demonstrated for E5 in artificial systems. 29,30
The PI3K-AKTt is one of the signal transduction pathways activated by the phosphorylated PDGFβ-r to transform cells. 37 Class IA PI3Ks are composed of heterodimers, including the 85-kDa regulatory subunit (p85) of phosphoinositide 3-kinase. The p85 binds and integrates signals from various cellular proteins, including transmembrane tyrosine kinase–linked receptors and intracellular proteins, such as protein kinase C, SHP1, Ras, and Src. 24 Genetic screens in model organisms have shown that upon activation by receptor tyrosine kinases, PI3K generates inositol phospholipids that trigger AKT activation. For full complete activation, phosphorylation in the hydrophobic C terminal domain (serine 473) is necessary. This phosphorylation affects multiple cellular processes driving tumor progression, such as proliferation, growth, and motility. 46 This pathway has been shown to be disregulated in different human malignancies. Furthermore, it is well known that the pathway can be stimulated by different viruses, including PVs and related PV-like particles. 21 Different in vitro studies have shown that human papillomavirus (HPV) E5 and BPV E5 activate the PI3K/AKT pathway through the activation of a tyrosine kinase receptor. 28,37 However, there is also evidence that some BPV E5 mutants may activate PI3K independently of PDGFβ-r activation. 44 Our data strongly support the hypothesis that phosphorylated (activated) PDGFβ-r form a stable complex with the PI3K leading to the activation of the pathway in naturally occurring BPV-associated equine sarcoids. Furthermore, this study adds another line of evidence supporting the role of the activated PDGFβ-r in the pathogenesis of a naturally occurring BPV-associated neoplasia.
Cyclin D3 is known to be overexpressed in different types of cancers, and recent studies provide evidence that the cyclin D3 is likely to be a target for PI3K/AKT signaling. 15,19,43,50 In addition, HPV-transforming proteins E6 and E7 are able to induce high levels of cyclin D3. 50 We show that sarcoids expressing E5 and the PI3K/AKT have higher levels of cyclin D3 compared with normal, thus suggesting that this kind of equine tumor is likely associated with abnormal cell cycle control mechanisms.
p-Jun, a major substrate for JNK, and p-JNK are implicated in oncogenic transformation. 45,48 A role for the JNK pathway in tumorigenesis is supported by the high levels of JNK activity found in several cancer cell lines. 25 A major outcome of JNK activation is the phosphorylation of the transcription factor c-Jun, thereby inducing augmentation of the AP-1 transcriptional activity, which can be associated with transformation. 49 Published studies have linked JNK activation to the AKT signaling pathway, 26,27 and activated JNK seems to play an important role in cancer development. 49 Furthermore, JNK is constitutively phosphorylated in keratinocytes immortalized with HPV-16 E6/E7 oncogenes. 12 Our findings suggest a possible role for activated JNK and p-JUN in sarcoids based on a recent report showing their phosphorylation upon BPV-E5 interaction via p-PDGFβ-r. 37
The important finding emerging from this study is a new scenario in sarcoid molecular pathology. Even though further studies are needed to gain new insights into the molecular mechanisms underlying this common equine neoplasia, to the best of our knowledge, this is the first report demonstrating the involvement of the PI3K-AKT molecular pathway in an in vivo model of BPV-associated neoplasia.
Currently, none of the presently available treatments for equine sarcoids is 100% effective. 11 A recent attempt to therapeutically vaccinate sarcoid-bearing donkeys with chimeric virus-like particles did not achieve statistically significant results. 3 More than 400 recent patents cover pharmaceutical targeting of PI3K. Several drugs targeting the PI3K pathway have entered clinical trials (phase I) for different human solid tumors. Notably, many of the PI3K-targeted drugs being developed for cancer therapy have been found to be very selective, with low levels of toxicity. 32 In this regard, our results may provide the basis for further developing and testing therapeutic drugs against sarcoids.
Footnotes
Acknowledgements
We are indebted to Dr. Lisa M. Petti (Center for Immunology and Microbial Disease, Albany Medical College, New York, NY, USA) for her kind gift of the HDF cell culture. Drs. Laura Rinaldi Vincenzo Musella (Department of Pathology and Animal Health, Division of Parasitology) and Valeria Marina Monetti (Department of Agricultural Engineering, Faculty of Agriculture, University of Naples Federico II, Naples, Italy) are gratefully acknowledged for their help in statistical analysis. This work was supported by Ministero dell'Istruzione, Università e Ricerca scientifica (MIUR) – PRIN n° prot. 2006078044 - 2006/2008 and Legge Regionale N° 5 (2006) from Regione Campania.
