Abstract
Susceptibility of Canada geese (Branta canadensis) to highly pathogenic avian influenza (HPAI) virus (H5N1) infection was studied by inoculating 10 naïve (antibody-negative) animals (5 adults and 5 juveniles) with A/chicken/Vietnam/14/05 (H5N1) virus. In the adults, 1 of 5 became infected, and 4 of 5 remained normal; in the juvenile group, 5 of 5 became infected. The pathology observed in the affected animals was similar to that reported in natural occurrences. Peripheral and parasympathetic nervous systems were examined and found infected, as well as cerebrospinal fluid-contacting neurons. In some locations with significant virus infection in cells, the expected inflammatory reaction was absent or very mild. Immunohistochemistry was used to locate influenza A virus nucleoprotein in brain, spinal cord, respiratory and digestive systems, pancreas, heart, and peripheral and parasympathetic nervous systems. Further studies are needed to explain age-related differences in susceptibility.
Highly pathogenic avian influenza (HPAI) viruses of subtypes H5 and H7 characteristically induce fatal systemic infection in terrestrial poultry. It has been shown that HPAI virus replicates in endothelial cells throughout the vascular system during initial infection in chickens. 1 Penetration of the blood-brain barrier by virus is followed by infection of neurons and glial cells, 9 and concurrently virus is able to infect a wide variety of parenchymal cells in other organs. 6
Waterfowl have long been considered the natural hosts for influenza A viruses and usually are inapparently affected. Since 2002, HPAI viruses have been increasingly associated with deaths in a variety of aquatic birds and other wild bird species. 10 Canada goose (Branta canadensis) mortality due to H5N1 HPAI virus natural infection was reported from the 2002 Hong Kong outbreak 3 and from the European continent in 2006. 11 Migratory birds are suggested to have played a role in the long-range spread of H5N1 HPAI virus from Asia into Europe and subsequently Africa. 4 Domestic geese (Anser anser domesticus), bar-headed geese (Anser indicus), and Cackling geese (Branta hutchinsii) have been shown to be susceptible to H5N1 HPAI virus infection under experimental conditions. 2,8
In North America, a major migratory species is the Canada goose. Little was known regarding susceptibility and/or carrier status of Canada geese until the recent study by Pasick et al. 7 Histopathology results from that study are reported here.
Ten naïve geese (no detectable antibody to AI) were exposed to A/chicken/Vietnam/14/05 (H5N1) HPAI virus via the nasal cavity, oral cavity, and ocular route. The group was composed of 5 adults and 5 juveniles. A timed necropsy was performed at 3 days post inoculation (dpi) (1 adult and 1 juvenile). Thereafter, necropsies were performed when clinical signs of disease became severe at 4 dpi (1 juvenile) and 5 dpi (1 adult, 3 juveniles) (Table 1). The 3 remaining naïve adults survived the initial exposure and were euthanatized at 20 dpi.
Histopathology lesions in naïve Canada geese infected with H5N1 highly pathogenic avian influenza virus.
∗ DPI = days post inoculation; n = number of animals necropsied at this day post inoculation.
† Lesions directly associated with the presence of viral antigen.
‡ = no lesion; + = mild lesion; ++ = intermediate lesion; +++ = severe lesion.
Tissue samples collected at necropsy (Table 1) were fixed in 10% neutral phosphate-buffered formalin, routinely processed, and stained with HE for histopathologic examination. For immunohistochemistry, paraffin sections were stained using a horseradish peroxidase–labeled polymer, Envision + system (anti-mouse) (Dykomation, Carpenteria, CA, USA). The sections were quenched for 10 minutes in aqueous 3% hydrogen peroxide then pretreated with proteolytic enzyme for 15 minutes. The primary antibody was a mouse monoclonal antibody specific for influenza A nucleoprotein (Clone 1331, BIODESIGN International, Saco, ME, USA) and was used at a 1 : 5,000 dilution for 1 hour. The sections were then counterstained with Gill's hematoxylin.
Histologic lesions in the 5 juveniles and 1 adult that died or were necropsied at 3, 4, and 5 dpi are summarized in Table 1. The adults examined at 3 dpi (1 animal) and 20 dpi (3 animals) did not have any lesions. At 3 dpi, lesions were well developed in 1 juvenile, especially in the central nervous system. In the cerebrum and brain stem, there was focal neuronal pycnosis and mild edema, usually near or surrounding a capillary (Fig. 1). Purkinje cell necrosis and gliosis was prominent in the cerebellum. Ependymal cells lining the ventricles of the brain and spinal canal (Fig. 2) were surrounded by a mild glial reaction, and focal gliosis was present in some gray matter horns of the spinal cord. Immunohistochemically there was specific immunolabeling for influenza A virus nucleoprotein in neurons, axons, and cerebrospinal fluid–contacting neurons surrounding ependymal cells (Fig. 2). The ependymal cells were not stained by immunohistochemistry for nucleoprotein, suggesting that they were not infected at this stage. However, the foot processes of the neurons clearly project into the central canal, and their cytoplasm is positive by immunohistochemistry for influenza virus. Lesions in the upper digestive system were not prominent, but there was a subtle, mild inflammatory reaction, usually oriented around capillaries in the submucosa of the esophagus and proventriculus. In the trachea, there was occasional mild inflammatory reaction composed of heterophils, lymphoplasmacytic cells, and macrophages in the submucosa. This reaction was much more prominent in the marrow cavities of the tracheal cartilage (Fig. 3), and immunohistochemistry was positive for nucleoprotein in many macrophages and osteoclasts (Fig. 3). In the lung, there was mild interstitial perivascular extravasation of fluid, and a few endothelial cells in scattered capillaries were positive for AI virus nucleoprotein. Pulmonary inflammatory reaction was minor. Severe focal necrosis of pancreatic acinar exocrine cells was prominent in all animals (Fig. 4). In the sections of duodenum and ceca, the ganglion cells of the myenteric plexus were pycnotic and appeared to be undergoing early stages of necrosis (Fig. 5). There was no evidence of an associated inflammatory reaction. However, positive staining for AI virus nucleoprotein was very prominent (Fig. 5). In the myocardium of some animals, there were small focal areas of myocardial muscle fiber disruption. Myocardial muscle fibers in these focal areas were positive by immunohistochemistry for AI virus nucleoprotein. In the sciatic nerves, there were occasional focal aggregates of lymphocytes around small capillaries, and immunohistochemistry confirmed the presence of AI virus nucleoprotein in the capillary endothelium in these nerves (Fig. 6).
Brain, cerebrum; Canada goose, 3 days post inoculation. Pycnotic neurons and neuropil vacuolation near a capillary. HE stain. Bar = 20 µm. Inset: Serial section with specific immunolabeling for avian influenza virus nucleoprotein showing positive staining in neurons and endothelium in a capillary.
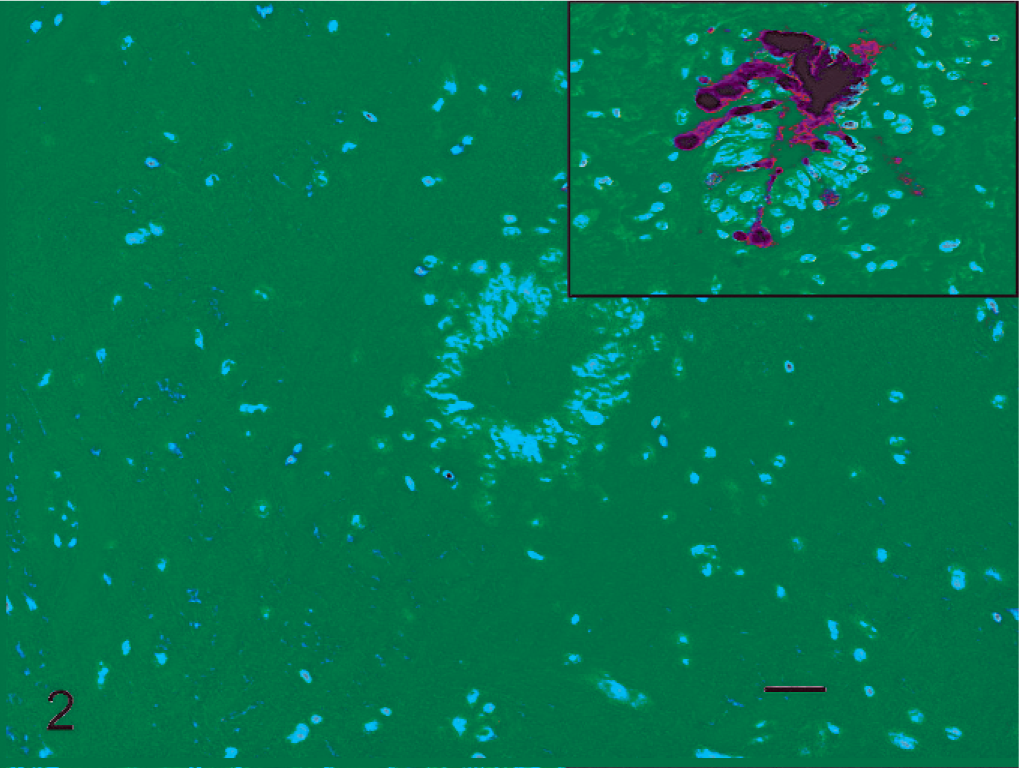
Cervical spinal cord; Canada goose, 3 days post inoculation. Mild pycnosis of neurons surrounding ependymal cells of the spinal canal. HE stain. Bar = 20 µm. Inset: Serial section with immunolabelling for avian influenza virus nucleoprotein showing cerebrospinal fluid–contacting neurons with cytoplasmic extensions into the spinal canal lumen.
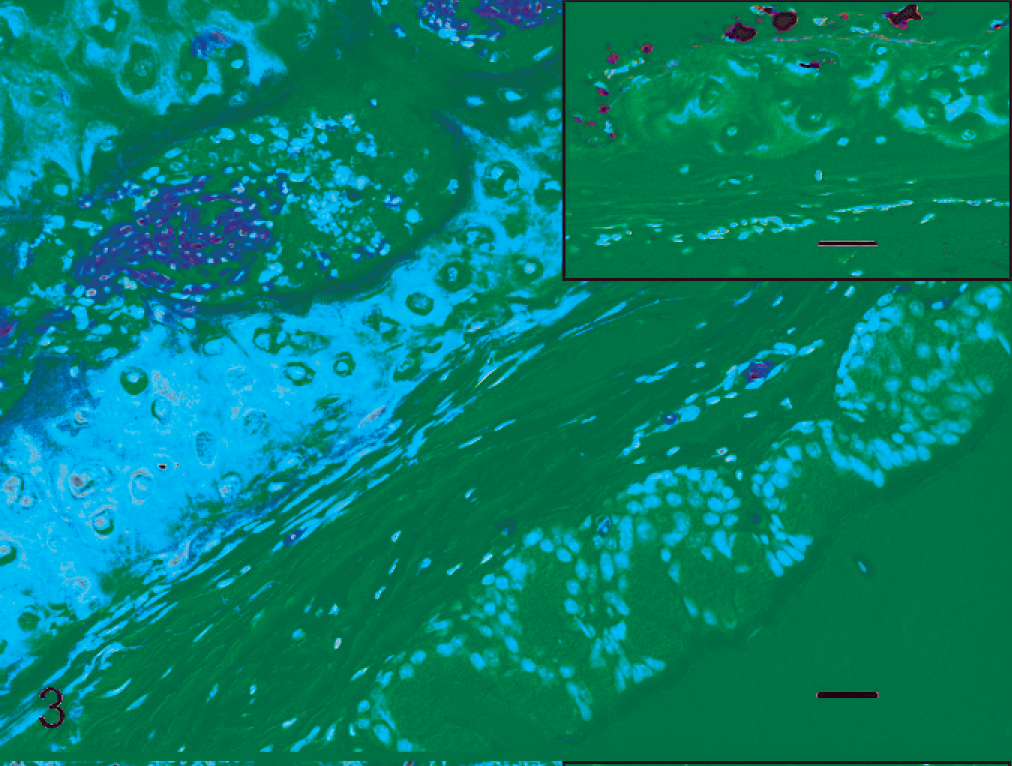
Trachea; Canada goose, 3 days post inoculation. Mild inflammatory reaction in the marrow cavity of the tracheal cartilage. HE stain. Bar = 20 µm. Inset: Serial section of tracheal cartilage with immunolabeling for avian influenza virus nucleoprotein in cells lining the marrow cavity.

Pancreas; Canada goose, 3 days post inoculation. Area of necrosis of pancreatic excretory cells. HE stain. Bar = 20 µm. Inset: Serial section of pancreas with specific immunolabeling for avian influenza virus nucleoprotein in nuclei and cytoplasm of acinar cells.

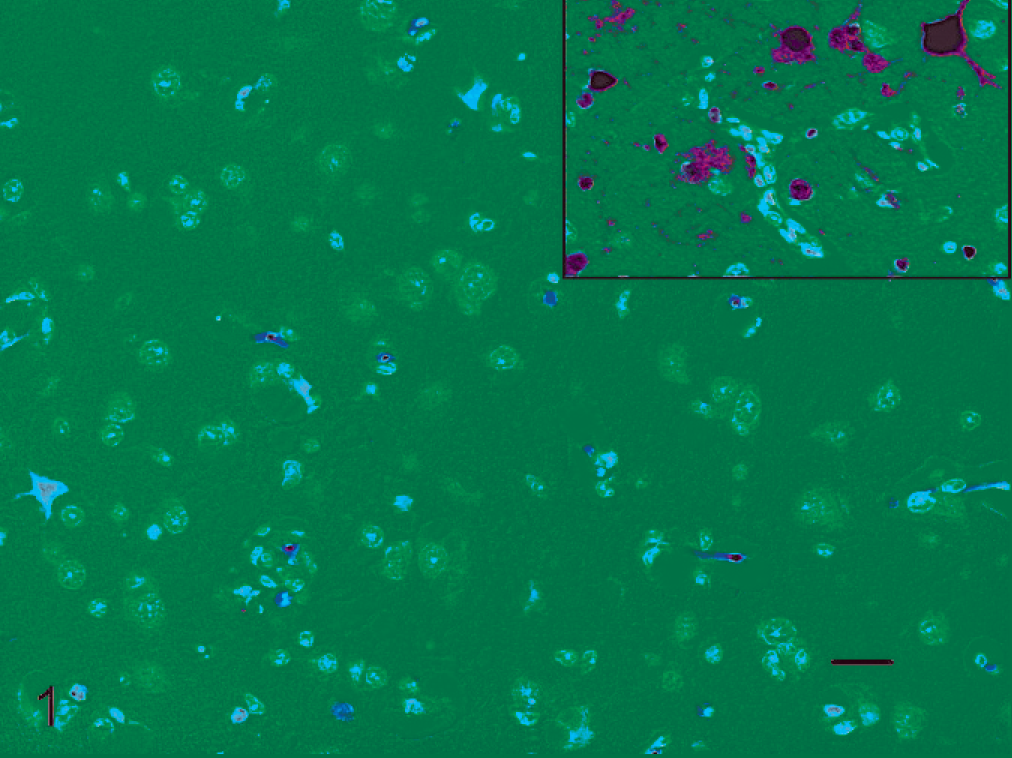
Myenteric plexus, cecal intestine; Canada goose, 3 days post inoculation. Pycnotic neurons within the nerve bundle, no inflammatory response. HE stain. Bar = 20 µm. Inset: Serial section of myenteric plexus with specific immunolabeling for avian influenza virus nucleoprotein in neurons within the plexus.
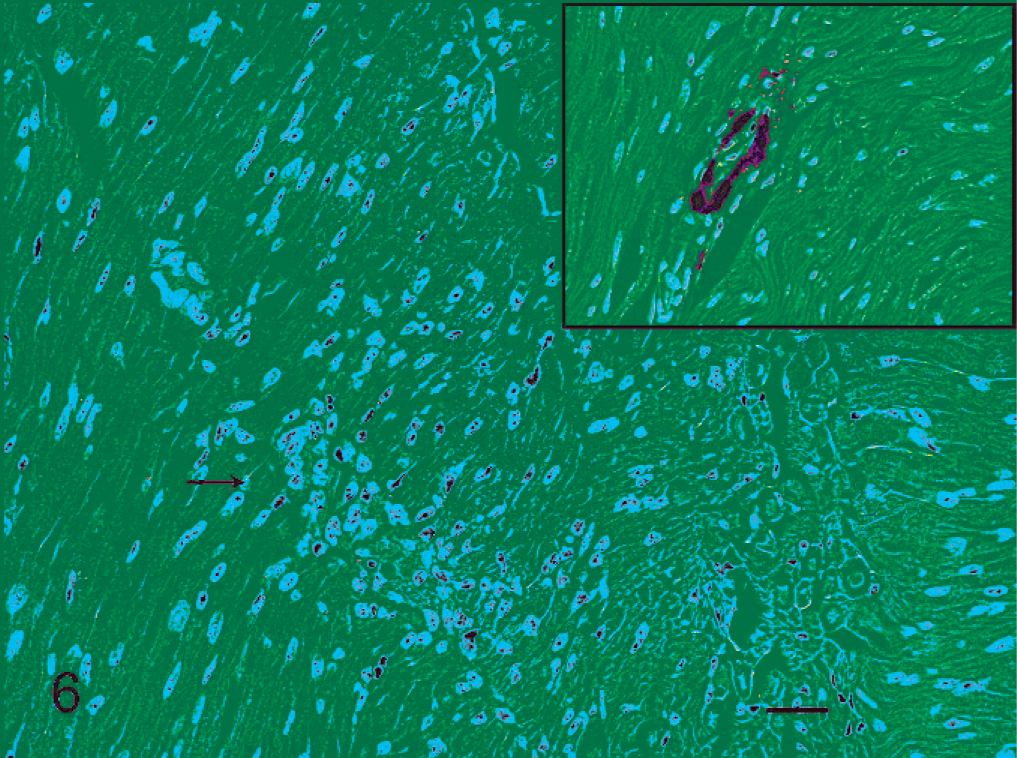
Sciatic nerve; Canada goose, 3 days post inoculation. Inflammatory reaction near a capillary (arrow). Inset: Capillary within same sciatic nerve with specific immunolabeling for avian influenza virus nucleoprotein within the capillary endothelium.
The histopathology of acute H5N1 HPAI virus infection in naïve juvenile Canada geese has not been previously reported. This report correlates tissue damage with the presence of H5N1 HPAI virus antigen by using immunohistochemistry and is part of an effort to better understand the pathogenesis of AI virus infection in waterfowl.
Lesions in the acutely affected animals were most severe in the central nervous system and also involved the parasympathetic nervous system. A remarkable feature of the infection in the nervous system, including parasympathetic ganglia, was the minimal inflammatory response at 3 to 5 dpi. Significant viral damage also occurred in many other organs, including the heart, trachea, and pancreas. These well-developed lesions in organs other than the central nervous system, occurring in the same period as those in the central nervous system, suggest a means of virus spread other than through a peripheral nerve route. The widespread infection of cerebral and midbrain neurons and the presence of viral antigen in cerebral fluid–contacting neurons may indicate a hematogenous route of spread through the blood-brain barrier (choroid plexus) and/or the capillaries of the circulatory system. 9
Studies in several species in the order Anseriformes using H5N1 HPAI virus have shown there are species variations in susceptibility and ability to excrete virus. 5,11 We have shown in this study that young of the Canada geese are highly susceptible to challenge (dying within 5 days). The adults in this case seem to have some inherent ability to resist infection. In the 1 adult that was affected by 5 dpi, the lesions were the same as those in the juveniles. The reason for lower susceptibility in adults remains to be determined. It is possible that exposure to other AI virus subtypes may confer some ability to resist H5N1 HPAI virus infection. In addition, studies will be required to confirm the initial site of invasion by AI virus and its tropism for certain cell types in organ systems. By 3 dpi there is substantial damage in most organ systems. Therefore, pathogenesis studies in earlier stages in the infection are required. Naïve juvenile Canada geese have been shown to be useful for this type of study.
Footnotes
Acknowledgements
Expert technical assistance was provided by E. Moffat, B. Collignon, A. Bukhari, and L. Burton.
