Abstract
Highly pathogenic avian influenza (HPAI) is a major viral disease of poultry characterized by acute onset, systemic infection, and rapid death. In January 2015, H5N2 HPAI was identified by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) and gene sequencing as the cause of rapid death in 40 of 390 ring-necked pheasants (approximately 10% mortality), raised in a game bird farm in Washington State. We report clinicopathologic findings and viral antigen distribution in pheasants that died during the outbreak. Affected birds were depressed with reluctance to move, ruffled feathers, and drooping heads. Congestion of the cerebellar meningeal blood vessels was the only consistent gross pathologic finding. Meningoencephalitis with vasculitis and necrosis in the spleen and heart were the major microscopic lesions in the birds. Viral antigen was consistently detected in the brain, heart, and ovary with variable presence in other organs.
Avian influenza virus (AIV), a member of the genus Influenza virus A of the family Orthomyxoviridae, infects a wide range of major and minor gallinaceous species. Based on the difference in virulence, AIV is generally classified into 2 pathogenicity groups: low pathogenic avian influenza (LPAI) and highly pathogenic avian influenza (HPAI). 5 LPAI typically causes localized respiratory infections, resulting in a mild disease and very low mortality, whereas HPAI is known to cause systemic infections, resulting in a severe disease and very high mortality in susceptible bird species. Two well-known surface antigens of AIV that play a significant role in pathogenesis and immune response are hemagglutinin (HA) and neuraminidase (NA). To date, 16 HA (H1 to H16) and 9 NA (N1 to N9) subtypes of AIV have been identified in birds. 1 Notably, only H5 and H7 subtypes with multiple basic amino acids at the cleavage site have been shown to possess HPAI characteristics. Pathologic findings of HPAI have been extensively documented in major gallinaceous species such as chickens and turkeys. 8 However, pathology studies in minor gallinaceous species such as pheasants are very limited, especially under natural conditions. The goal of this report is to document gross and histologic lesions and antigen distribution in ring-necked pheasants that died during recent outbreaks of HPAI in the state of Washington.
On January 25, 2015, on a game bird farm located in the northern Washington state, 40 of 390 ring-necked pheasants (approximately 10%) and 12 of 12 turkeys (100%) died overnight. The farm also had 3000 mallard ducks, 75 chukars, 7 geese, 70 partridges, and 20 chickens; however, no mortalities were observed in these birds. The surviving affected pheasants were reluctant to move, lay on their back with flapping of wings, and were depressed with ruffled feathers and drooping heads (Fig. 1). HPAI was suspected as the cause of death because of ongoing outbreaks in the region at that time. Thirteen dead pheasants and 1 dead turkey were submitted to the Washington Animal Disease Diagnostic Laboratory (WADDL) for AIV testing and necropsy. Choanal/tracheal samples were collected from all submitted birds in the laminar flow hood using Dacron polyester-tipped swabs, and the swabs were stored in brain heart infusion (BHI) medium. RNA was isolated from these samples (Ambion AM1836 MagMAX-96 Viral RNA Isolation Kit; Life Technologies, Austin TX) according to the manufacturer’s instructions. Initial screening for AIV was performed by a quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) specific for the AIV matrix gene, and RNA samples that tested positive for the AIV matrix gene were subjected to additional qRT-PCR subtyping assays specific for hemagglutinin types H5 and H7 following National Veterinary Services Laboratory (NVSL) SOP-AV-0068 protocol. 10 –12 Thirteen birds (12 pheasants and 1 turkey) were positive for AIV matrix and H5 genes, and none of these birds were positive for the H7 gene. Samples that tested positive for both matrix and H5 genes were further tested by the USDA-Animal and Plant Health Inspection Service (APHIS), NVSL in Ames, Iowa. Virus isolation and sequencing of HA cleavage site confirmed that the pheasants were infected with Eurasian H5N2 HPAI.
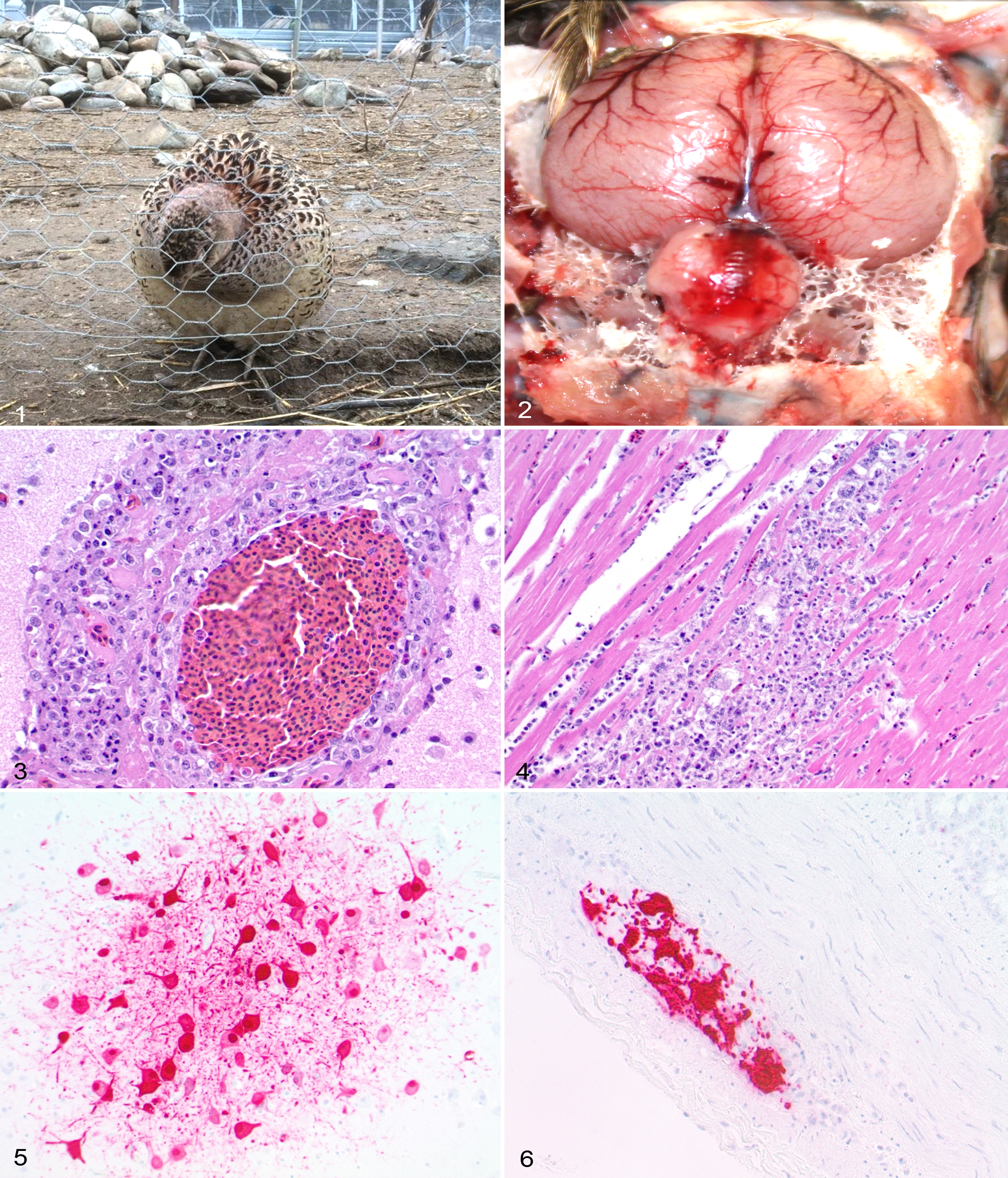
A complete necropsy of 3 pheasants (2 females and 1 male) was performed in the laminar flow hood. Moderate to severe congestion of meningeal blood vessels, especially in the cerebellum, was observed in all 3 birds (Fig. 2). The spleen of 1 bird was mildly enlarged and mottled red and white. No other significant gross lesions were noted. Tissues were fixed in buffered formalin, and histologic sections were routinely prepared for hematoxylin and eosin staining.
Histologically, lesions were most common in the brain (3/3), spleen (3/3), heart (3/3), pancreas (2/3), and liver (2/3). Multifocally, the meningeal blood vessels in the cerebellum were moderately to severely congested. Several sections of the brain showed multifocal meningoencephalitis characterized by expansion of perivascular (Virchow-Robin) spaces by 2 to 10 layers of inflammatory cells consisting of moderate numbers of macrophages and lymphocytes, small numbers of heterophils, and occasional plasma cells, admixed with a small to moderate amount of fibrin and necrotic cellular debris (Fig. 3). Vasculitis characterized by infiltration of the wall of blood vessels by small numbers of the above-noted inflammatory cells was also observed with occasional endothelial cells containing pyknotic nuclei (necrosis). The vasculitis was most prominent in the meninges, with inflammation following the blood vessels into the subjacent Virchow-Robin’s spaces. Rare single-cell neuronal necrosis with minimal inflammation was observed in the cerebrum. The spleen had multifocal, mild to severe necrosis with mild inflammation composed of macrophages, lymphocytes, and heterophils. Multifocal necrosis with variably severe inflammation was also observed in the heart (Fig. 4), pancreas, and liver. The adrenal glands of 1 bird had multifocal, moderate necrosis with accumulation of moderate numbers of macrophages, lymphocytes, and heterophils. Multifocal, mild to moderate renal tubular necrosis and ovarian necrosis with inflammation composed of macrophages and lymphocytes were observed in 1 bird. No significant histological lesions were observed in the lungs, esophagus, trachea, testicle, proventriculus, ventriculus, intestines, skin, eye, and thyroid.
All tissues examined by routine histopathology were also examined by immunohistochemistry (IHC) to detect AIV antigen. IHC testing was carried out using an automated immunostaining system (Benchmark; Ventana Medical Systems, Tucson, AZ). The primary antibody was mouse monoclonal antibody to influenza A virus nucleoprotein (cat. C65331 M; Meridian Life Science, Memphis, TN). Visualization of influenza A monoclonal antibody binding was done following the manufacturer’s instructions for the alkaline phosphatase–based detection system (UltraView Red, cat. 760-501; Ventana Medical Systems). The negative antibody control consisted of irrelevant, isotype-matched monoclonal antibody to Babesia bovis 23/28.57 applied at the same concentration to a serial section of the same test tissue. 7 Positive tissue control consisted of archived avian lung tissue, shown positive by rRT-PCR, immunostained with influenza A monoclonal antibody.
Antigen was consistently detected multifocally in the brain (3/3), heart (3/3), spleen (2/3), pancreas (2/3), and ovary (2/2). Occasional positive immunoreactivity was observed in intestines, esophagus, and skin of 1 or 2 birds. In the brain, immunoreactivity within the nucleus and cytoplasm was observed in neurons in the cerebrum and cerebellum, as well as within the nucleus and cytoplasm of glial cells, macrophages, and endothelial cells (Fig. 5). Immunoreactivity was observed in areas with or without microscopic lesions. Multifocally, cardiac myocytes showed strong immunoreactivity both in cytoplasm and nucleus. Immunoreactivity was restricted to areas of necrosis in the spleen and pancreas. In ovary and intestines, the immunoreactivity was predominantly detected in the neurons of the ovarian plexus and myenteric plexus (Fig. 6), respectively, with occasional immunoreactivity in the theca externa of the ovarian follicles. Immunoreactivity was occasionally observed in the esophagus (submucosal gland epithelium) and skin (all layers of epidermis and follicular epithelium).
This report describes the gross and microscopic lesions and antigen distribution of natural H5N2 HPAI infections in pheasants from the 2014 to 2015 outbreak in Washington State. In November 2014, H5N2 HPAI infection had been first confirmed in several poultry farms in British Columbia, Canada, and the virus was soon found in wild birds in Washington State. 3 Subsequently, the birds in other states of the western and central United States have been shown to be infected with the Eurasian H5N2 HPAI virus.
Although clinical signs were nonspecific, pathological findings and molecular test results confirmed H5N2 HPAI as the cause of sudden death in ring-necked pheasants in this outbreak. Congestion of the cerebellar meningeal blood vessels was the consistent gross change observed in these pheasants. Histologically, meningoencephalitis with vasculitis was a common feature. Necrosis with or without inflammation was observed in the spleen, liver, pancreas, heart, and adrenal glands. Virus antigen, detected by IHC, was confirmed in the brain, spleen, heart, and pancreas. Although there were no significant lesions, viral antigen was also detected in the small intestines, ovary, esophagus, and skin.
Fifteen HA subtypes of AIV (H1 to H15) have experimentally been shown to replicate in Chinese ring-necked pheasants without showing any disease-related clinical signs. 2 Experimental infection of H5N1 HPAI induced necrosis in the nasal cavity, heart, brain, spleen, and adrenal glands in ring-necked pheasants but not pancreatic necrosis. 9 In these pheasants, viral antigen was most commonly found in the adrenal glands, followed by heart and brain. 9 Although pheasants were susceptible to experimental infection with H5N2 HPAI (A/Chicken/Penn/1370/83), most did not show any clinical signs. 13 Reports describing natural HPAI infections in pheasants are scarce. During a 1999 outbreak in Italy, H7N1 HPAI-infected pheasants exhibited tremors and incoordination and consistently had necrotizing pancreatitis. 6
In the farm of the present report, the game birds were at risk of developing AIV infection from exposure to migrating waterfowl, as they were raised in mesh-covered outdoor runs. In addition, during summer months, the farm had used water from a nearby river as a source of water for birds, further increasing the risk of AIV transmission since AIV can remain infectious in water for months under suitable conditions. 4 Samples from the birds were tested as part of AIV surveillance prior to their release, and LPAI infections (H2N5 and H4N6) were confirmed based on RT-PCR, virus isolation, and sequencing in 2005, 2010, and 2014. During this outbreak, HPAI H5N2 was also confirmed as the cause of death in 1 turkey, which also exhibited cerebellar meningeal vessel congestion. Tissues from this bird were not examined histologically. No mortality was observed in chukar partridges, which were raised on a wire mesh floor, and the farm personnel did not walk into these pens on regular basis. Following the diagnosis of HPAI, the game bird farm was completely depopulated and appropriately disinfected. Recommendations such as pretreatment of water with chemicals were provided to the farm to increase biosecurity. Recent testing after the repopulation confirmed the absence of AIV on the farm.
In summary, this study documents the clinicopathological findings and viral antigen distribution of natural HPAI H5N2 infection in ring-necked pheasants. Consistently, congestion of the cerebellar meningeal blood vessels was observed in affected birds. All the birds exhibited meningoencephalitis with vasculitis. Variable necrosis was observed in the spleen, liver, pancreas, heart, and adrenal glands. Viral antigen was commonly found in the brain, spleen, heart, pancreas, and ovary. Exposure to the migrating waterfowl was suspected as the source of infection for the outbreak.
Footnotes
Acknowledgements
We thank the owners of the farm for providing the image of a sick bird for use in this report and WADDL histology staff for preparing slides.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
