Abstract
The largest outbreak of highly pathogenic avian
The United States Department of Agriculture (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed the first report of H5N2 highly pathogenic avian
A H5N8 HPAIV, similar to the H5N8 HPAIV previously identified in Asia, was identified in captive wild birds in the western United States in December 2014. 8 This H5N8 virus was suspected to have spread from Asia to North America along wild bird migratory pathways (http://goo.gl/ldBWn4) and was subsequently identified in captive birds, backyard poultry, or commercial chicken and turkey flocks in Washington, Idaho, Oregon, and California (http://goo.gl/CxV2GB). The mixing of this H5N8 virus with an AIV of North American lineage generated a new reassortant H5N2 HPAIV that contains the highly pathogenic Eurasian H5 (clade 2.3.4.4) and the low pathogenic North American N2 (http://goo.gl/ldBWn4).8,11 This H5N2 reassortant HPAIV is composed of 5 gene segments (PB2, PA, HA, M, NS) related to the Eurasian H5N8 HPAIV and 3 gene segments (PB1, NP, NA) related to North American lineage AIVs. 11
AIV is in the family
AIVs can be classified as low pathogenic AIV (LPAIV) or HPAIV. The World Organization for Animal Health (OIE) has established molecular criteria for the identification of HPAIV based on HA protein sequences (http://goo.gl/qssVJG). In poultry, the presence of multiple basic amino acids located at the endoproteolytic cleavage site for HA is the molecular marker of HPAIV.5,6,12 Of the 16 HA subtypes, to date, only H5 and H7 viruses have been associated with naturally occurring HPAI in poultry (chickens and turkeys). Even so, the vast majority of H5 and H7 AIVs are LPAIV, and only a small proportion of H5 and H7 AIVs are HPAIV.2,15
Clinical signs of HPAIV include torticollis, ruffled feathers, loss of appetite, depression, respiratory signs (nasal discharge, coughing, sneezing), decreased egg production, misshapen eggs, and high mortality. 3 Gross and microscopic lesions can vary as a result of susceptibility and duration of infection prior to death. 14 Common gross findings include sinusitis, subcutaneous hemorrhages, swelling of the head, cyanosis of the combs and wattles, multifocal subcutaneous hock hemorrhage and petechiae, and fibrinous peritonitis.3,13 Microscopic lesions vary in severity and location but commonly consist of necrosis, hemorrhage, and/or inflammation within multiple visceral organs, especially the heart, brain, pancreas, and skin. 14
We describe 2 cases of H5N2 infection in layers in Iowa and describe gross and microscopic lesions and viral antigen distribution in infected birds along with other laboratory findings. The first farm was located in Kossuth County in northwest Iowa (A/Chicken/Iowa/21981/2015 [ISUVDL2015021981]), and the second farm was located ~225 km (140 miles) away in Madison County in central Iowa (A/Chicken/Iowa/14399-4/2015 [ISUVDL2015022716]). Both farms reported a rapid increase in mortality that resulted in the submission of 5 and 10 dead birds, respectively, to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, Iowa).
External examination consistently revealed marked, diffuse edema and multifocal-to-extensive hemorrhage of the comb, as well as accumulation of mucus in the oropharynx (Fig. 1A) and proximal trachea. Less consistently, epicardial petechiae and ecchymoses (Fig. 1B), locally extensive-to-diffuse necrotizing pancreatitis (Fig. 1C), and multifocal-to-locally extensive splenic hemorrhage and necrosis (Fig. 1D) were observed.
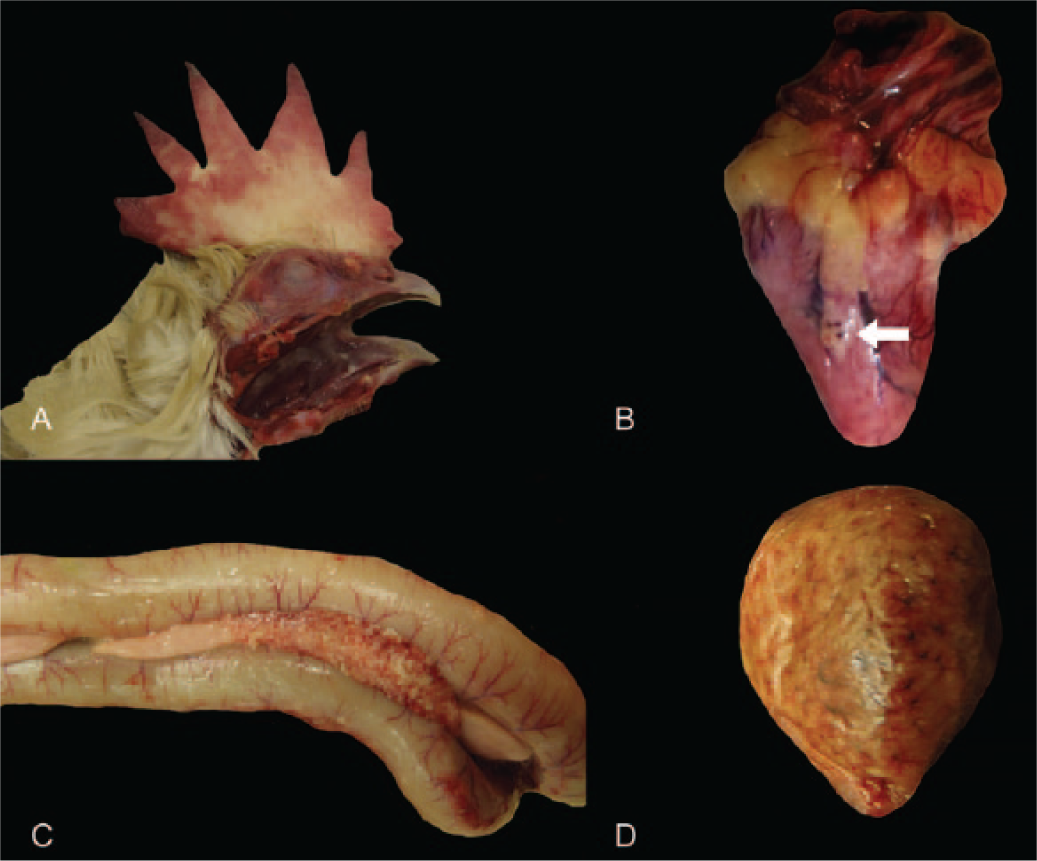
Common macroscopic lesions in chickens naturally infected with highly pathogenic avian
Microscopic changes were variable, with none-to-many lesions observed in each bird; no individual bird had all of the following described lesions. Mild multifocal lymphohistiocytic meningoencephalitis, with perivascular cuffing and mild gliosis, as well as rare degenerate neurons in the immediately adjacent neuropil were observed (Fig. 2A). There were a few foci of myocardial necrosis or lymphohistiocytic and necrotizing myocarditis (Fig. 2B). Also noted were multifocal-to-locally extensive necrotizing pancreatitis (Fig. 2C) and splenitis (Fig. 2D), perivasculitis and edema in the comb (Fig. 2E), necrotizing bronchitis, and lymphohistiocytic interstitial pneumonia (Fig. 2F) and sparse accumulations of lymphocytes in the tracheal propria-submucosa. These histologic lesions are similar to those described in chickens experimentally inoculated with a H5N2 HPAIV isolated from commercial poultry in central Mexico. 13 Neurotropism and epitheliotropism as seen in these cases has been documented in different species of birds infected with HPAIV under experimental conditions. 9 Tubular necrosis and interstitial nephritis, which has been described in birds infected with HPAIV, were not observed in the tissue sections evaluated from these cases. 13 The extent of renal lesions in cases of HPAIV can vary from absent 1 to severe 7 and may vary according to the stage of disease.
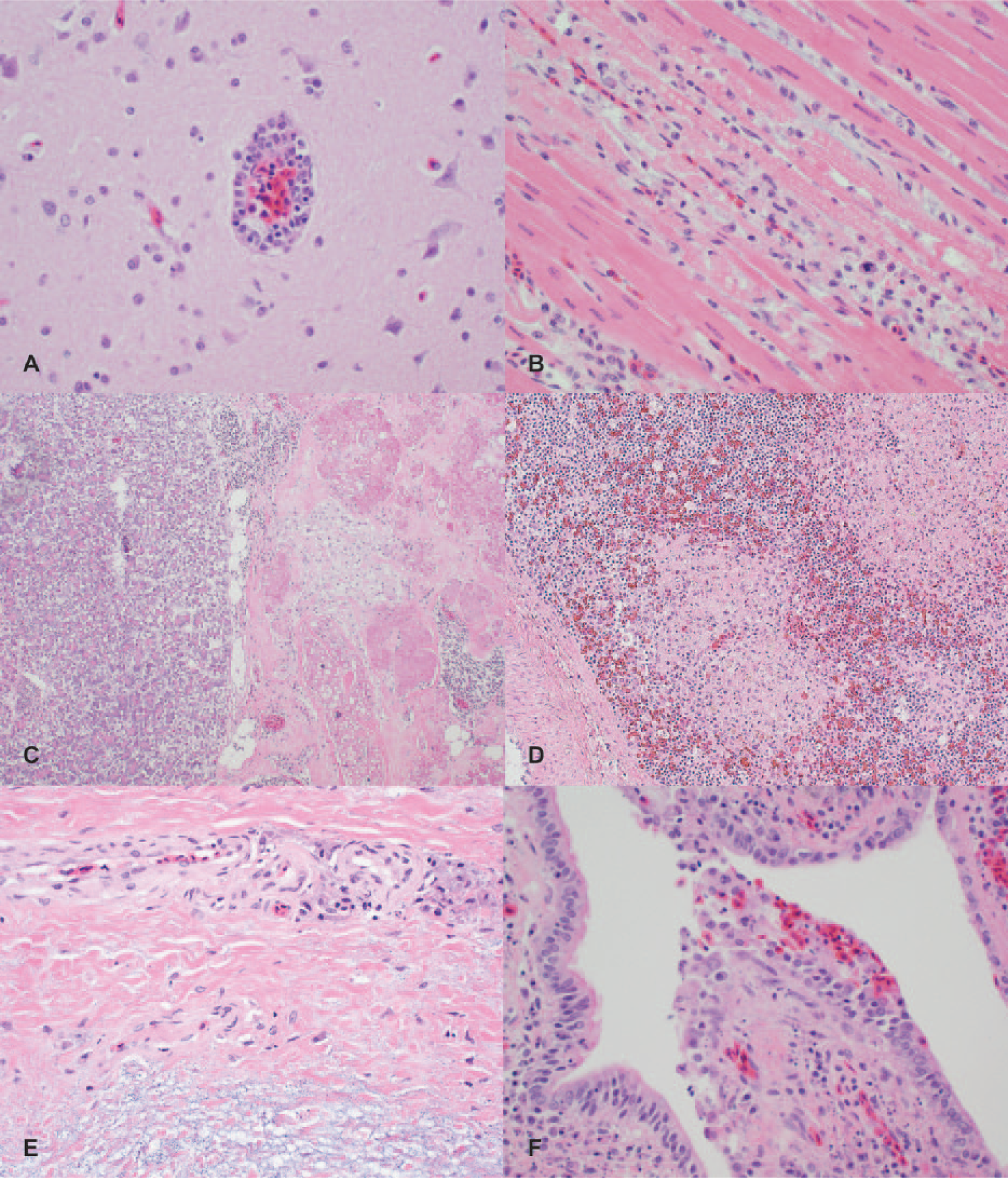
Selected microscopic lesions in chickens naturally infected with highly pathogenic avian
Based on clinical signs, gross anatomic lesions, and history of an ongoing outbreak of HPAI in Iowa, oropharyngeal and trachea swabs
a
were collected individually and placed in a tube containing 5 mL of brain-heart infusion broth
b
for influenza PCR testing. Testing at ISU-VDL and USDA National Veterinary Services Laboratories (Ames, Iowa) for AIV confirmed the presence of H5N2
On confirmation of AIV, comb, brain, lung, heart, spleen, kidney, liver, pancreas, and small and large intestines collected from these cases were further examined by immunohistochemistry for
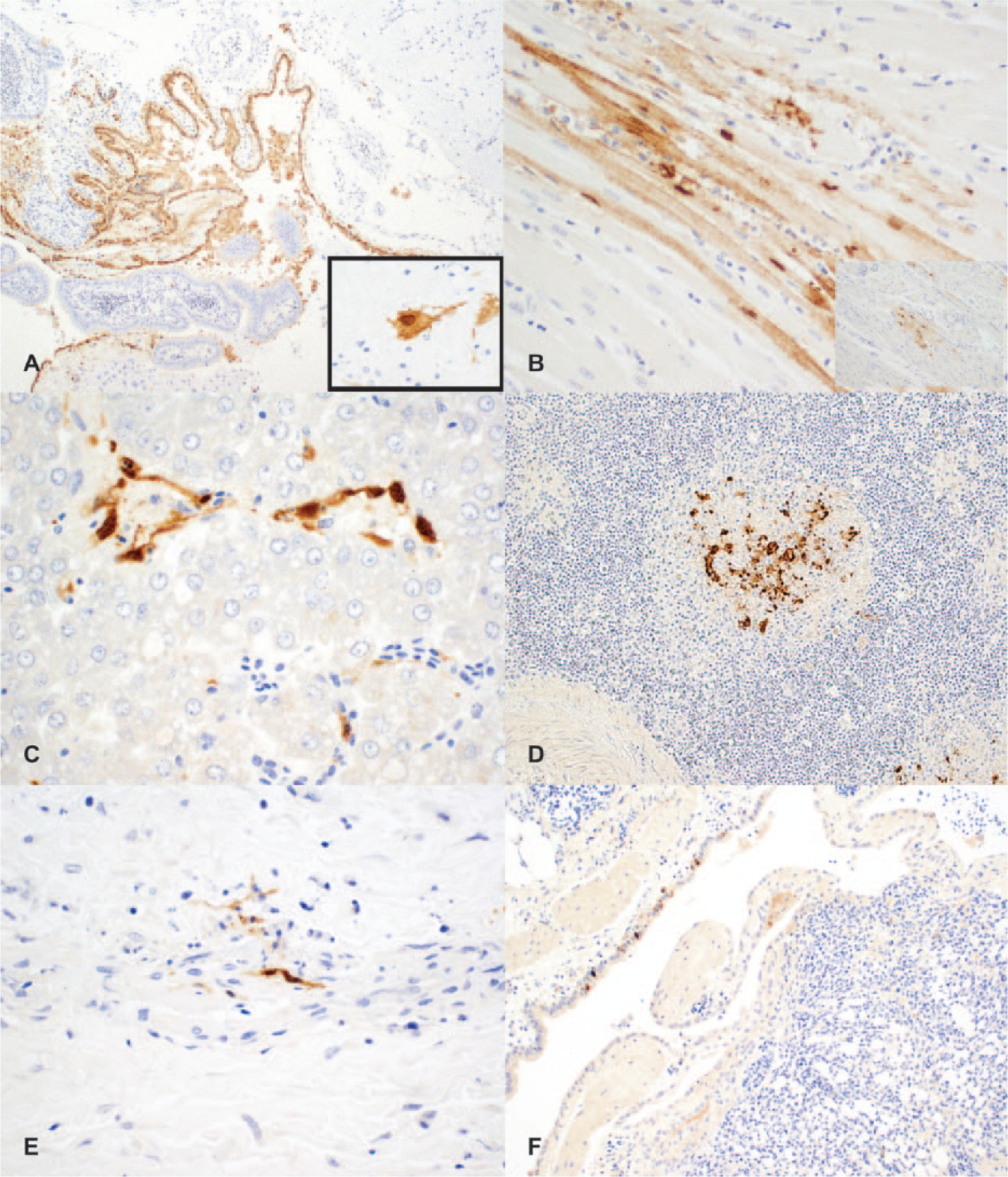
Chickens naturally infected with highly pathogenic avian
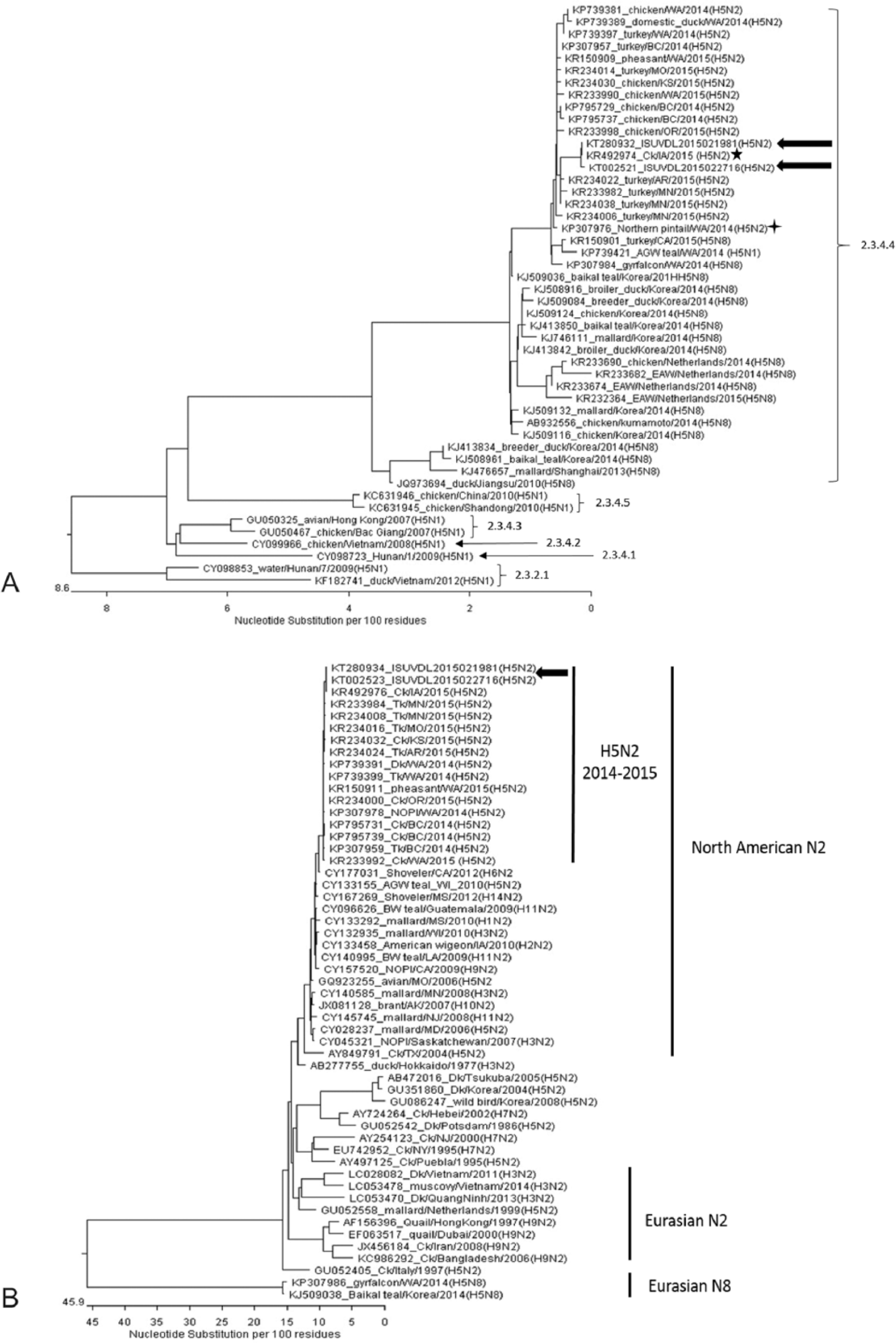
Further molecular characterization of H5N2 HPAIV identified in each case (A/Chicken/Iowa/21981/2015 [ISUVDL2015021981] and A/Chicken/Iowa/14399-4/2015 [ISUVDL2015022716]) was performed by sequencing DNA directly from clinical specimens for all 8 genomic DNA segments using a next-generation sequencing technique. d Sequences of the viruses were compared with those of H5 HPAIVs available in GenBank. Overall sequence analyses demonstrated that these 2 H5N2 HPAIVs are a reassortant between H5 AIV of Eurasian lineage and N2 AIV of North American lineage as reported by other investigators.4,8,11
BLAST (http://goo.gl/nOVQhQ) searches revealed that all genome segments of both viruses (GenBank accessions KT280929–KT280936 [A/Chicken/Iowa/21981/2015; ISUVDL2015021981] and KT002518–KT002525 [A/Chicken/Iowa/14399-4/2015; ISUVDL2015022716]) share 99.4–99.8% and 99.4–100% nucleotide identity with the U.S. H5N2 index case (KP307973–KP307980 [A/Northern pintail/Washington/40964/2014]) 8 and Iowa layer H5N2 index case (KR492971–KR492978 [A/chicken/Iowa/04-20/2015]), 4 respectively. As illustrated in Figure 4A, the HA gene segment of both viruses are closely related to Asian-origin HPAIV H5 clade 2.3.4.4 11 reported in both Asia and North America. Although the 2 viruses shared 99.9% identity with each other in their HA gene segments, the BLAST search revealed that the HA gene segment of the 2 viruses shared 99.4–99.5% and 99.9–100% nucleotide identity with the U.S. H5N2 index case and the Iowa layer H5N2 index case (Oceola County, Iowa) respectively, suggesting that the virus may have been introduced to the 2 index farms by area spreading through an unidentified mechanism or source. The NA gene segment of both Iowa HPAIVs was close to that of North American lineage H5N2 (Fig. 4B), which has been identified in migratory water fowl over U.S. flyways and showed 100% nucleotide identity with that of the Iowa layer H5N2 index case. In addition to the NA gene segment, the polymerase PA gene segment, polymerase PB2 gene segment, and matrix protein gene segment also showed 100% nucleotide identity with those of the Iowa layer H5N2 index case.
Footnotes
Authors’ contributions
PHE Arruda and BL Arruda contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. GW Stevenson, ML Killian, ER Burrough contributed to acquisition, analysis, and interpretation of data. PC Gauger and KJ Yoon contributed to conception of the study, and contributed to acquisition, analysis, and interpretation of data. KM Harmon and J Zhang contributed to acquisition and analysis of data. DR Magstadt and P Piñeyro contributed to conception of the study, and contributed to acquisition of data. DM Madson, RJ Derscheid, VL Cooper, RG Main, and Y Sato contributed to acquisition of data. KJ Schwartz contributed to conception and design of the study, and contributed to interpretation of data. PG Halbur contributed to interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Sterile polyester tipped applicators, Puritan Medical Products, Guilford, ME.
b.
Remel BHI broth (brain heart infusion broth), Thermo Scientific, Pittsburgh, PA.
c.
Anti-Influenzavirus A nucleoprotein monoclonal antibody, Iowa State University Hybridoma Facility, Ames, IA.
d.
Ion Torrent platform, Life Technologies, Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
