Abstract
This article describes the histopathology of grossly normal mesenteric lymph nodes (MLNs) of New Zealand farmed red deer (Cervus elaphus). Eighty MLNs were sourced from 10 deer from 5 North Island herds and 5 South Island herds classified as low risk and high risk of Mycobacterium avium subspecies paratuberculosis (MAP) infection, respectively. Fixed sections were stained with hematoxylin and eosin; Ziehl-Neelsen; and, selectively, periodic acid–Schiff, Perl’s, and Sudan black. Positive Ziehl-Neelsen stain, follicular hyperplasia, capsular eosinophil infiltration, focal granulomas, foci of macrophages containing lipopigment, parasitic granulomas, and calcified foci are described and severity graded where appropriate. Animal age, sex, and herd of origin are variably associated with the presence of one or more features. Trabecular fibrosis and dilated edema-filled sinusoids are described. These observations allow differentiation between likely nonpathologic histologic features in deer MLNs and features possibly attributable to infection with a pathogen such as MAP.
The New Zealand farmed deer industry is the largest producer of venison worldwide, with more than 700,000 deer commercially slaughtered in 2006. Every deer carcass is assessed by an accredited meat inspector, and lymph nodes (LNs), including the mesenteric lymph nodes (MLNs), are visually inspected, palpated, and/or incised for lesions resembling tuberculosis (Tb) (Mycobacterium bovis). Nodes identified as being suspicious for Tb are sampled and assessed, primarily by histologic examination. 9,10
Histopathologic features of grossly abnormal deer LNs infected with M bovis or the closely related bacterium Mycobacterium avium subspecies paratuberculosis (MAP) have been well described. 4,10 However, there has not been a published description of the range and prevalence of histopathologic features in grossly normal deer MLNs or the typical cellular architecture of those nodes. Such knowledge may be useful to histopathologists to help differentiate between nonpathologic features in grossly abnormal MLNs and those likely to be related to infection with M bovis or MAP.
This article describes the cellular architecture of grossly normal MLNs of red deer from 2 populations with differing risk of MAP infection. The range and prevalence of selected histopathologic features in MLNs, associations between each feature and (1) each remaining MLN feature and (2) each animal risk factor, and additional histologic features of interest are also determined.
Selection of Deer Herds for Sampling
Herds were selected from those in a 2005 epidemiologic study of Johne’s disease (JD) in New Zealand farmed deer herds. Five North Island herds were selected as being low risk for MAP infection based on an absence of clinical JD or lesions reported from deer slaughter premises and a negative culture of 6 pools of 10 fecal samples from adult hinds for MAP. It was confirmed in 2007 that no deer on those farms had exhibited clinical signs of JD and/or grossly abnormal MLNs at slaughter in the intervening 2 years. Five South Island herds were selected as being high risk for MAP infection based on a positive pooled fecal culture in 2005. In addition, one or more deer had been observed with clinical JD, and/or one or more deer had a false-positive reaction to an intradermal test for M bovis in the period to early 2007. All herds had a Tb status of C7 to C10, indicating no evidence of M bovis infection in the previous 7 to 10 years.
Selection of MLNs
MLNs were selected from deer slaughtered in a deer slaughter premise between January 1, 2007, and December 31, 2007. Eighty MLNs, 40 from low-risk herds and 40 from high-risk herds, were randomly selected, including 2 to 15 animals per herd of origin. Age and sex strata were hinds and stags, young (12 to 24 months old) and adult (> 24 months old). Only normal MLNs were selected, with each containing no grossly visible pathology along its entire length after incision and having a maximum circumference of 60 mm.
A 15-mm-wide section was taken from each MLN at 25%, 50%, and 75% of its length, anterior to posterior, and individually stored in 10% buffered formalin. Fixed tissue samples were dehydrated, cleared, and embedded in paraffin. Sections were stained with hematoxylin and eosin (HE) and Ziehl-Neelsen (ZN). Additional stains, including periodic acid–Schiff (PAS), Perl’s, and Sudan black, were applied to sections from 6 animals to aid identification of a granular material present within macrophages. One histopathologist (R.G.C.) examined all sections without knowledge of animal age, sex, or herd of origin. Sections were examined under low (2.5 × 10), mid (10 × 10), and high (10 × 40) dry magnification with a Leitz Wetzlar microscope and, as necessary, oil immersion (10 × 100).
Sections were photographed with an Olympus BX51 microscope with a mounted Olympus DP70 camera and an Olympus U-RFL-T light source. Ultraviolet examination employed a mercury burner at wavelengths 365/366, 404.7, 435, 546.1, and 577/579.1 nm. For ultraviolet light, an Olympus WU filter was used: excitation, 380–385 nm; emission > 420 nm.
Data Collection and Management
Animal age, sex, and regional location of the herd of origin were obtained. Histopathologic features were categorized, graded, and coded for later analysis (Table 1 ). The prevalence of each feature was summarized into a frequency table. Statistical analyses were performed to examine the relationships between each feature and animal age, sex, and herd-level MAP risk; MLN incision site; and remaining MLN features.
Description and Subjectively Assessed Grades of Common Histopathologic Features Observed in Grossly Normal Mesenteric Lymph Nodes of Red Deer

Cellular Architecture
The MLN cellular architecture was typical of that seen in other ruminants. The MLN was surrounded by an outer cortex, with trabeculae extending into the parenchyma. A cross section of an afferent lymph vessel was visible, which opened into the subcapsular sinus. The outer cortex of the node contained secondary follicles separated by diffuse lymphatic tissue. Medullary cords were separated by a network of sinuses and connective tissue trabeculae.
Features of Interest
In 92% of lymph nodes, a variable amount of fine, granular, irregularly sized material was observed within macrophages in the cortex and medullary cords. This material was often observed with a pale brown appearance on HE, black on a ZN stain, and rarely contained a red granule, as illustrated in Figure 1 . This material was also Sudan black positive, PAS positive, and positive to a variable extent on Perl’s stain, indicating the presence of iron (Fig. 2 ). Some foci also contained a crystalline-like material (Fig. 1), which may be due to ingested soil, or possibly fat crystals, such as cholesterol.
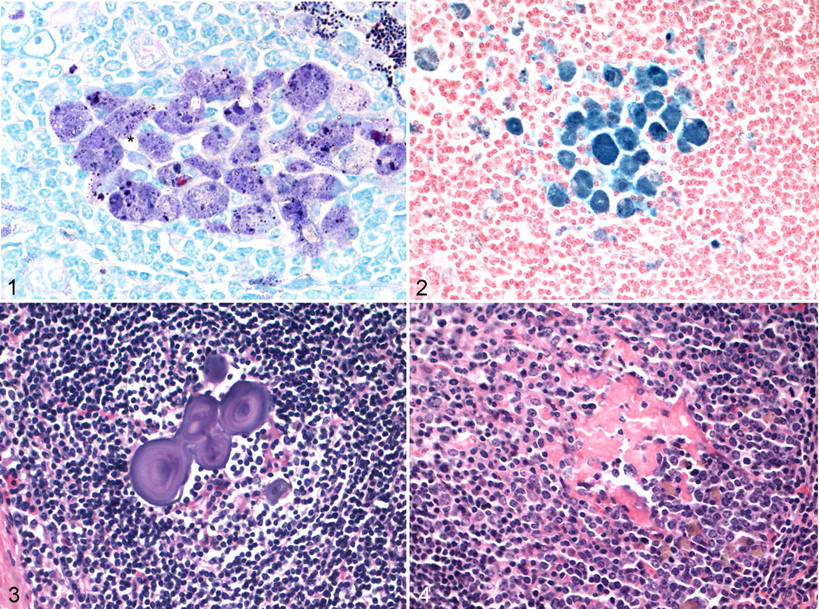
On ultraviolet light examination of unstained sections, there were scattered focal areas of a golden yellow color, which related to focal concentrations of macrophages seen on HE and other special stained slides. The ultraviolet light and special stain results are indicative of a lipopigment. 7 Although there are no published reports describing the lipopigment, it has been observed in deer MLNs (R.G.C., personal observation). Lipopigments, also referred to as lipofuscins, include ceroid, age pigments, and pigments found in the inherited storage disease ceroid lipofuscinosis. 7 The pigment was more likely to be observed in adult deer than yearling deer and so may be an age-related change. However, this does not mean that it is an age pigment. The iron present within the lipopigment was variable, suggesting a variable source, such as hemosiderin (eg, from hemolysis or copper deficiency) or iron from soil ingestion. An unidentified pigment that increases with age has been reported in cattle LNs, particularly in MLNs. That pigment was positive to Schmorl’s reaction and under ultraviolet light showed yellow autofluorescence, similar to the pigment observed in the present study. However, it was negative to PAS, ZN, Perl’s Prussian blue, and fat stain Sudan IV–oil red O staining procedures. These staining reactions were different to those observed in the present study, suggesting a different pigment.
Calcified foci, often with a laminated appearance, were present within germinal centers (Fig. 3). These generally had no inflammatory response but were occasionally associated with a multinucleated giant cell. Trabecular fibrosis was present in one or 2 sections in 3 animals, and dilated edema-filled sinusoids were seen in all sections from 2 animals. Dilated sinusoids have been reported in cattle, and likely causes include local obstruction or congenital defects in efferent lymphatics. 8 Hemosiderin-like material was present within the medullary sinusoids of a number of animals, being particularly marked in one case. Less common histopathologic features observed were small focal granulomas containing coccidian zoites and follicular germinal centers with perivascular fibrinoid material (Fig. 4 ). Fibrinoid foci and vascular hyalinization within follicular germinal centers have been considered a postreactive change in cattle; for humans, vascular hyalinization has been shown to increase with age. 8
Prevalence of Features in MLNs
The prevalence of each histopathologic feature in normal MLNs may be used as a baseline reference by histopathologists when examining grossly abnormal MLNs for possible infection with pathogens such as MAP (Table 2 ). Infiltration by eosinophils into the nodal capsule was observed in over 60% of MLNs. Possible explanations include migration of nematode larva, usually liver fluke (Fasciola hepatica) in other species and/or lungworm (Dictyocaulus spp), 6 or aberrant coccidia.
Prevalence of Each Feature in Grossly Normal Deer Mesenteric Lymph Nodes (n = 80) a
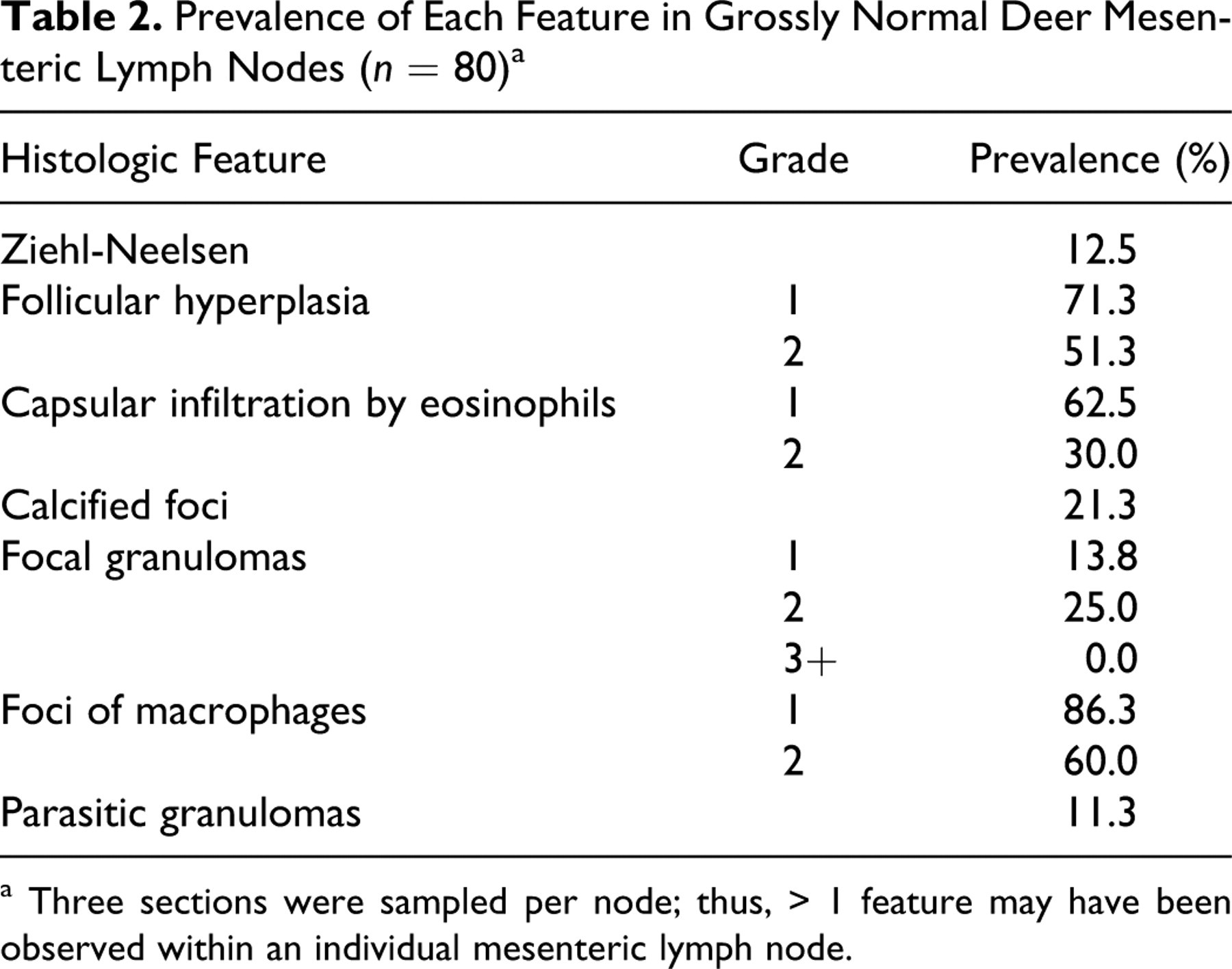
a Three sections were sampled per node; thus, > 1 feature may have been observed within an individual mesenteric lymph node.
Follicular hyperplasia was commonly observed, involving proliferation of the normal cell population within the lymphoid follicle’s germinal center with consequent formation of secondary lymphoid follicles with germinal centers of variable size. Follicular hyperplasia is typically seen in MLNs as a result of constant antigenic stimulation from challenge of the intestinal mucosa by dietary antigens and commensal and pathogenic microbes and their products.
Calcified foci within the germinal centers of MLNs were also observed. Similar mineralized bodies of varying size and shape, including laminated corpora amylacea, are most often located in germinal centers and are occasionally observed in bovine LNs. In cattle, they have been interpreted as post-reactive changes and the larger ones are considered to be an outcome of parasitic infection. However, calcified foci have also been observed in grossly normal and abnormal MLNs of known MAP-infected deer (R.G.C., personal observation).
Association Between Features and Risk Factors
Only 1 focal granuloma (2.5%) was observed in 40 MLN sections sourced from low-risk MAP herds, whereas 30 MLNs with focal granulomas (75.0%) were sourced from 3 of 5 MAP herds. This histopathologic feature has been described in grossly normal and abnormal MAP-infected MLNs of deer. 2,3,5 Granulomas have also been shown to be more common in MLNs than in the adjacent intestinal mucosa, especially in the mild stages of disease. 2 This study found that focal granulomas were more likely to be observed in grossly normal MLNs from herds at high risk of MAP, indicating that this feature may be useful as a predictor of subclinical MAP infection. However, this feature can develop in deer MLNs in response to infection with other pathogens, such as M bovis. 9,10 The likelihood of M bovis infection in study herds was low because selection criteria included a negative Tb test result and a herd-level status for 7 to 10 or more years.
Both mild and moderate follicular hyperplasias were less likely to be observed in MLNs sourced from adult deer. This association may be explained by possibly higher antigenic stimulation of the MLNs occurring relatively early in life, owing to constant challenge by new dietary antigens, microbes, and other stimuli.
Moderate follicular hyperplasia, a moderate to marked capsular infiltration by eosinophils, and foci of macrophages with lipopigment in the cortex were more likely to be clustered at 25% of the MLN length. However, the distribution of focal granulomas, presumably due to MAP infection, was not significantly associated with incision location on the MLN. This finding has relevance to selection of the site of the LNs for investigation into MAP infection. Carrigan and Seaman found that the most severe gross pathology due to MAP infection in sheep occurred in the last 3 to 4 m of the small intestine. 1 This lesion distribution was presumed to be associated with the varying presence of lymphoid tissue within the intestinal wall. In contrast, in cases of advanced disease due to MAP infection in deer, grossly visible abnormalities are most commonly located in the first half of the MLN.
These results confirm that grossly normal MLNs of farmed red deer in New Zealand contain a variety of histopathologic features. A number of these features, such as mild to moderate follicular hyperplasia, occur in reaction to antigenic stimulation, particularly early in life, and can be considered common and relatively nonpathologic. However, focal granulomas may suggest a mycobacterial infection justifying further diagnostic tests, such as culture and polymerase chain reaction, regardless of the gross appearance of the node.
Footnotes
Acknowledgements
We would like to sincerely thank the management, staff, and AsureQuality meat assessors at Venison Packers, Feilding, and Alliance Sockburn, Christchurch, New Zealand. Mr Dan Lynch is acknowledged and thanked for his tireless work to ensure that the project ran smoothly.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The primary author was supported by Johne’s Management Limited and the Deer Industry’s Johne’s Research Group1 (JRG1) in association with DEEResearch and the Foundation for Research, Science and Technology via a subcontract with AgResearch. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
