Abstract
The following case report is, to the best of our knowledge, the first documentation of a squamous cell carcinoma in a full-blooded American bison (
An ulcerated lesion was first noticed on the left flank during the summer of 1997 of an 8-year-old, full-blooded American bison cow, at the site of a brand. The original owner stated that it was a “hard [deep] brand that took months to heal.” The lesion did not resolve as expected during the following winter, and in the summer of 1998, it developed into an ulcer, which attained a size of 12–15 cm in diameter. In the fall, there was marked growth of the lesion, and it became exophytic. By December, it achieved a diameter of approximately 30 cm, and on 3 December 1998, a biopsy was performed by a veterinarian. The histopathologic report was prepared by the Colorado Veterinary Diagnostic Laboratory of the Colorado State University College of Veterinary Medicine and Biomedical Sciences. The tumor consisted of sheets of large cells with highly pleomorphic nuclei, large prominent nucleoli, and an abundant amount of cytoplasm. The cells formed clumps and cords, and some squamous differentiation was present. Mitoses were reported to be numerous, and since the cells extended to the edge of section, the chances for recurrence were reported to be high. The diagnosis was a poorly differentiated squamous cell carcinoma. The animal's condition deteriorated, and a decision was made to euthanatize the bison in February 1999, when she could no longer stand. At that time, the primary flank tumor was 30 by 40 cm and had a depth of 12 cm (Fig. 1). A postmortem examination was performed, and sections of skin, liver, lung, brain, prefemoral lymph node, kidney, spleen, and gut were obtained for microscopic examination. Blood samples were also obtained to determine purebred status.

Presentation of the lesion on the flank of the euthanatized bison, measuring approximately 30 by 40 cm.
Postmortem examination by the attending veterinarian, Wayne R. Davis, DVM (Craig, CO), on 8 February 1999 revealed the following: a female bison, pregnant, exhibited a large mass on the left hip where she had been previously branded. Necropsy revealed numerous tumor masses in the liver and a large tumor mass in the abdomen where the aorta bifurcates into the internal and external iliac arteries. The abdominal serosa was inflamed, and petechial hemorrhages were present on the epicardium. There was also serous atrophy of fat in many body structures.
Specimens submitted for histopathologic examination included primary skin lesion, lung, liver, prefemoral lymph node, skeletal muscle, kidney, stomach, spleen, brain, and adrenal gland.
Histologic examination of a skin specimen from the necropsy revealed a piece of soft tissue covered by both normal and dysplastic stratified squamous epithelium, with abrupt transition from one to the other. The dysplastic epithelium was invasive into the underlying fibrous connective tissues and formed numerous small, but somewhat variably sized and shaped, tumor islands. Most tumor tissue was poorly differentiated, with little tendency to keratinize, although some prekeratinized cells and tiny keratin pearls were present. Tumor cells were uniformly hyperchromatic, and 250× magnification revealed 8–10 mitotic figures per field, some of which were atypical. Nuclei were often pleomorphic, and some were enlarged and vesicular, with chromatin clumping. Intercellular bridging was easily recognized, and there was an accompanying marked inflammatory infiltrate composed mostly of neutrophils (Fig. 2). The final diagnosis was a moderate to poorly differentiated squamous cell carcinoma of the skin.
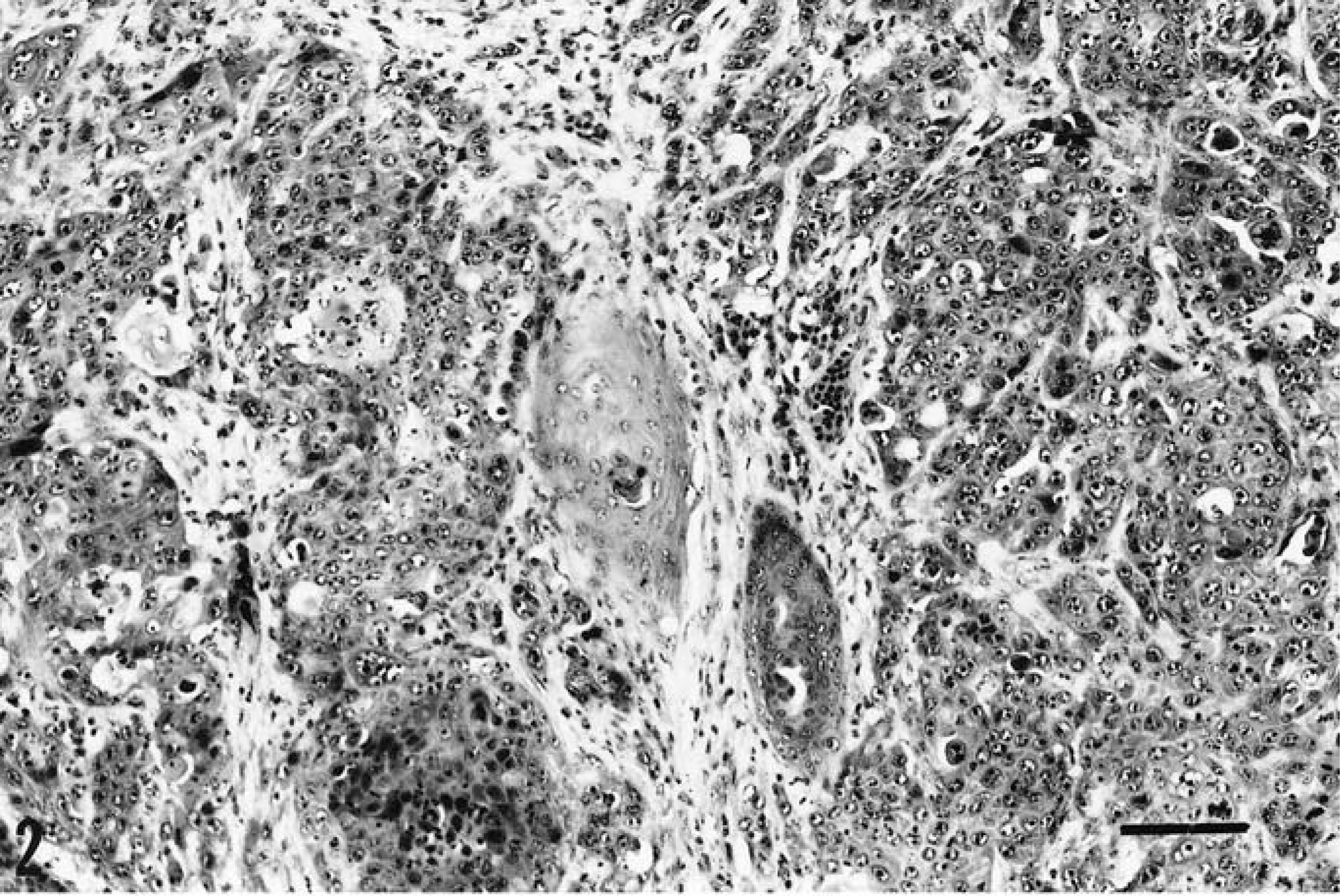
Islands of malignant invasive squamous epithelium at the primary skin site. HE. Bar = 100 µm.
Metastatic tumor that was identical to the primary was found in multiple foci within lung tissue (Fig. 3) and in the liver (Fig. 4). Tumor was also found deep within skeletal muscle at the primary site and in the prefemoral lymph node. Again, all distant tumor foci were identical to the primary squamous cell carcinoma.
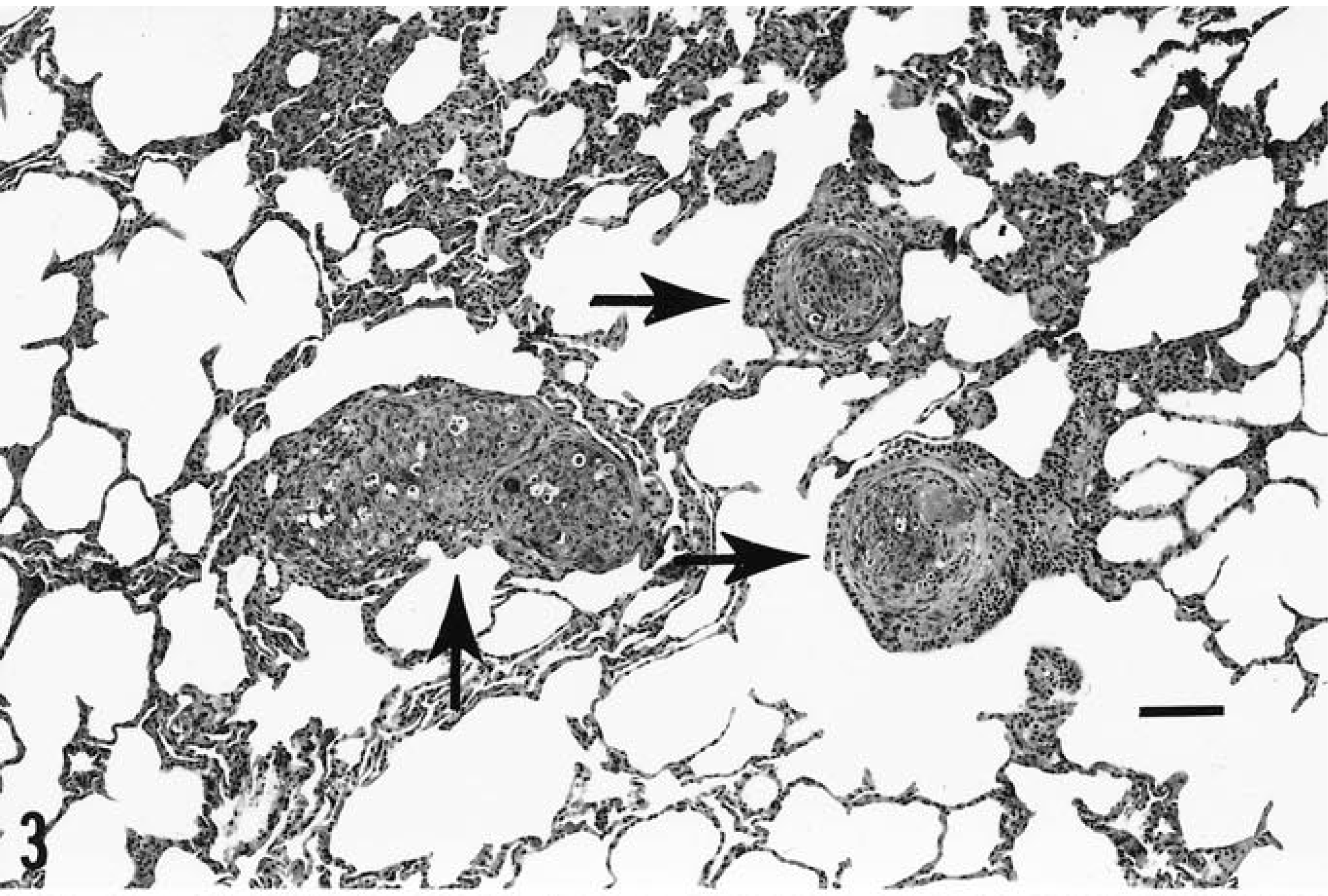
Three islands (arrows) of metastatic squamous cell carcinoma within lung tissue. HE. Bar = 100 µm.
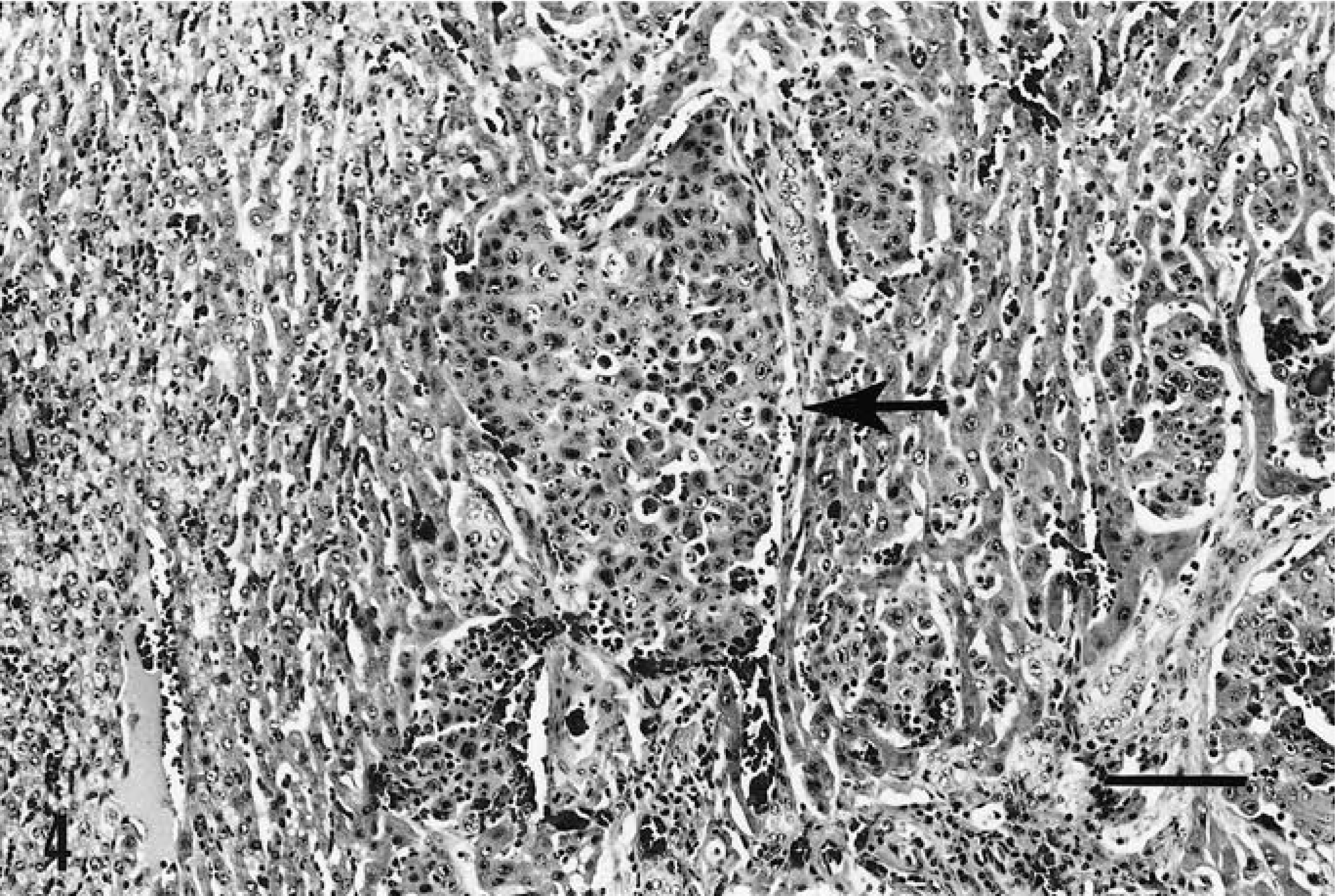
Metastatic squamous cell carcinoma within liver tissue (arrow). HE. Bar = 100 µm.
Cytokeratin staining utilizing the avidin–biotin complex method 1 , with reagents obtained from Vector Laboratories Inc. (Burlingame, CA), utilizing mouse monoclonal antibody as the primary antiserum, was positive, further verifying that the tumor was indeed a squamous cell carcinoma.
Blood samples obtained immediately prior to the bison's being euthanatized were sent for laboratory analysis (Stormont Laboratories Inc., Woodland, CA), which included standard blood-typing 2 and microsatellite markers for DNA polymorphism 3 to determine parentage and species specificity. The results of testing revealed that this animal was a full-blooded American bison and eligible to enter the bison registry.
Our literature search did not reveal any references to cancer in bison. The literature does, however, contain articles on cancer in “Buffalo,”
4–6
but this must be clarified. The term “Buffalo” has been applied generically to those animals included in the families Bubalus and Bovidae.
7
The former includes the African (or Cape) buffalo and the Asiatic water buffalo. The latter includes the American buffalo.
8
The family Bubalus is considered the only true buffalo, and they do not crossbreed with the American buffalo, who belong to the genus
Of possible significance is the fact that this cancer developed at the site of previous trauma in the form of heat branding. Although it is difficult to establish a direct cause-and-effect relationship, this association has been noted in cattle. 9
At this time, we are unable to make a statement regarding the incidence of cancer in American bison. We are currently attempting to ascertain whether or not other purebred bison have been so affected and can be documented.
Footnotes
Acknowledgements
We thank Barbara E. Powers DVM, PhD, of the College of Veterinary Medicine and Biomedical Sciences (Colorado State University), for preparing the histopathologic report; Howard Gittelman, DVM, New City, NY; and Sandy Oelschlegel, of the Tufts University School of Veterinary Medicine, North Grafton, MA.
