Abstract
Cyclooxygenase-2 (COX-2), the rate-limiting enzyme in the biosynthesis of prostaglandins, plays an important role in inflammation and tumorigenesis. COX-2 primary structure has been characterized in many species and its expression demonstrated in a variety of cancers in humans and dogs, including mammary cancer. In contrast, there is currently little information on the structure of feline COX-2. Also, information on COX-2 expression in feline mammary cancer is limited and conflicting. The objectives of this study were therefore to characterize the molecular structure of feline COX-2 and to evaluate by immunohistochemistry its expression in mammary carcinomas. Our results show that the predicted coding region of feline COX-2 encodes a 604-amino acid protein, which is identical in length to several COX-2 homologs. Feline COX-2 amino acid sequence is highly similar to other mammalian COX-2 homologs. Immunohistochemical analysis of 40 mammary carcinomas showed that the majority of tumors studied (35/40; 87%) expressed COX-2 at a level varying from low (20/40; 50%) to intermediate (13/40; 32%) and high (2/40; 5%). These results provide the first molecular characterization of feline COX-2 and demonstrate that COX-2 is expressed in the majority of feline mammary carcinomas.
Introduction
Cyclooxygenase (COX), also known as prostaglandin endoperoxide synthase, is a key enzyme in the biosynthetic pathway leading to the conversion of arachidonic acid into prostaglandins. COX catalyzes the transformation of arachidonic acid into prostaglandin G2 and subsequently to prostaglandin H2. Two structurally different forms of COX have been identified and characterized. 27 COX-1 is constitutively expressed and mainly associated with the production of prostaglandins involved in important physiological processes such as the cytoprotection of the gastric mucosa and renal blood flow. 27 In contrast, the expression of COX-2 is inducible, and the enzyme is responsible for the production of prostaglandins implicated in ovulation, inflammation, and cancer. 27 COX-2–derived prostaglandins are thought to promote cancer progression through their actions on tumor-associated angiogenesis, apoptosis resistance, the immune system, and tumor cell invasiveness. 5 Upregulated expression of COX-2 has been documented in many cancers in humans. 28 Moreover, experimental evidence derived from several studies, particularly on human colorectal and breast cancer, also has provided strong evidence for the association of COX-2 and tumorigenesis. 15, 25 Inhibition of COX-2 as a strategy for cancer prevention has proved to reduce the risk of some cancers, although unexpected cardiovascular toxic effects have raised serious questions about their use as chemotherapeutic agents in humans. 2, 5, 15
Many canine cancers have also been reported to overexpress COX-2. 7, 12, 16, 20 In contrast, COX-2 expression has been demonstrated in only a limited number of feline neoplasms. Beam et al. analyzed by immunohistochemistry various feline cancers and found significant COX-2 expression in transitional cell carcinomas. 1 Expression of COX-2 also has been reported in feline oral squamous cell carcinomas, with a recent study suggesting that cats with naturally occurring oral squamous cell carcinoma could represent a good animal model to study anticancer activity of COX inhibitors. 1, 6, 9 The COX-2 status of feline mammary carcinomas is less clear, with 2 studies presenting conflicting results. 1, 17 Indeed, one group reported the absence of COX-2 expression in 9 feline mammary carcinomas, while Millanta et al. found COX-2 in 96% of the feline mammary tumors they evaluated. 1, 17 Lymphomas and vaccine-associated sarcomas as well as pulmonary and intestinal adenocarcinomas have been reported to be COX-2 negative in cats. 1
COX-2 mRNA encodes a 70-KDa protein that is approximately 75% homologous to the COX-1 protein. 26 COX-2 can be induced by various stimuli such as lipopolysaccharides and inflammatory cytokines such as interleukin-1, tumor necrosis factor, or transforming growth factor alpha. 27 The molecular structure of COX-2 has been characterized in a wide variety of animal species. 3, 4, 13, 14 However, the molecular structure of feline COX-2 has not yet been reported. The main objectives of the present study were therefore to characterize at the molecular level the structure of feline COX-2 and, in view of the conflicting results in the literature, to analyze by immunohistochemistry the expression of COX-2 in mammary carcinomas, a common and deadly form of cancer in cats.
Materials and Methods
Cloning of feline COX-2
The full-length feline COX-2 cDNA was isolated by a combination of reverse transcription polymerase chain reaction (RT-PCR) and 5′- and 3′-rapid amplification of cDNA ends. RT-PCR was performed using sense and antisense primers designed from a highly conserved region identified after sequence alignments of human and mouse COX-2 homologues and total RNA extracted from feline pulmonary fibroblasts (ATCC, Manassas, VA).
Immunohistochemistry
Forty feline mammary carcinomas submitted to the Département de Pathologie et Microbiologie of the Faculté de Médecine Vétérinaire (Université de Montréal) were evaluated. Immunohistochemical staining was performed using the Vectastain ABC kit (Vector Laboratories, Burlington, Ontario, Canada). Formalin-fixed tissues were paraffin-embedded, and 3-μm-thick sections were prepared and deparaffined through graded alcohol series. Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in methanol for 30 minutes. After rinsing in phosphate buffered solution (PBS) for 15 minutes, sections were incubated with diluted normal goat serum for 20 minutes at room temperature. The anti-COX-2 antibody used was MF243, a polyclonal antibody that was raised in rabbits against ovine placental COX-2 and whose selectivity for COX-2 in cats has previously been characterized. 6 The antibody (1 : 7,500 dilution) was applied, and sections were incubated overnight at 4°C. Control sections were incubated with PBS or with nonimmune rabbit serum. After rinsing in PBS for 10 minutes, a biotinylated goat anti-rabbit antibody (1 : 222 dilution) was applied, and sections were incubated for 45 minutes at room temperature. Sections were washed in PBS for 10 minutes and incubated with the avidin DH-biotinylated horseradish peroxidase H reagents for 45 minutes at room temperature. After PBS wash for 10 minutes, the reaction was revealed using diaminobenzidine tetrahydrochloride as the chromogen. Sections were counterstained with Gill's hematoxylin stain, mounted, and immunoreactivity was evaluated. The percentage of COX-2 positive tumor cells was graded as 0 = <5%, 1 = 5–25%, 2 = 26–50%, 3 = 51–75%, and 4 = >75%, and the intensity of COX-2 immunoreactivity was graded as − = no, + = weak, ++ = moderate, and +++ = strong staining. Each COX-2 score represented the product of percentage of positive tumor cells and intensity, as previously described. 7 Scores from 1 to 4 were considered as low COX-2 expression, from 5 to 7 as intermediate COX-2 expression, and greater than 8 as high COX-2 expression.
Immunoblot analysis
Proteins were resolved by one-dimensional sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred to polyvinyldene-fluoride membranes. Blocking of membranes was done using 5% nonfat dry milk in 0.1% TTBS (0.1% Tween-20, 10 mM Tris-buffered saline, pH 7.5) for 1 hour at room temperature, then washed twice for 2 minutes at room temperature with 0.1% TTBS. The membrane was incubated with MF243 (1 : 6,000 dilution) for 2 hours at room temperature, then incubated with a horseradish-peroxidase-labeled donkey anti-rabbit secondary antibody (1 : 15,000 dilution) for 1 hour at room temperature. It was washed, and the bound secondary antibody was detected using the enhanced chemiluminescence system (ECL plus, Amersham Biosciences Corp., Baie d'Urfé, Québec, Canada).
Results
Molecular characterization of feline COX-2
Results showed that the feline COX-2 cDNA is composed of a 5′-UTR of 129 bp, an ORF of 1815 bp (including the stop codon), and a 3′-UTR of 646 bp. The nucleotide sequence was submitted to GenBank with accession number EF036473. The predicted complete coding region of feline COX-2 encodes a 604-amino acid protein, which is identical in length to several COX-2 homologs, but is 1 amino acid longer than ovine COX-2. Comparative analyses revealed that the feline COX-2 amino acid sequence is highly similar to its homologs, being identical at 96% with canine, 93% with equine, 92% with mink, 91% with porcine and rabbit, 89% with bovine, human and mouse, 88% with rat and ovine, and 82% with chicken COX-2 (Fig. 1). Moreover, structural domains putatively involved in COX-2 function are conserved in the feline protein, including an epidermal growth factor (EGF) domain, a membrane-binding domain (MBD), and a short C-terminal domain corresponding to an endoplasmic retention (ER) signal (Fig. 1).

Predicted amino acid sequence of feline COX-2 and comparison with other mammalian homologs. The deduced amino acid sequence of feline (fel) COX-2 is aligned with the human (hum), canine (can), porcine (pig), rabbit (rab), equine (equ), bovine (bov), ovine (ovi), murine (mou), mink (min), rat (rat), and chicken (chi) homologs. Identical residues are indicated by a printed period, and the signal peptide cleavage site is indicated with an arrowhead. Putative N-glycosylation sites are marked with an asterisk; proximal and distal heme coordination residues are overlined; the tyrosine associated with the cyclooxygenase active site is underlined, and the aspirin-acetylation site is indicated by a number sign. Boxed regions include an EGF domain, an MBD, and a short C-terminal domain thought to represent an ER signal. Numbers on the right refer to the last amino acid on that line, and the percentage in parentheses indicates the degree of identity when compared with feline COX-2.
Expression of COX-2 in feline mammary carcinomas
Forty feline mammary carcinomas were immunostained for COX-2 expression using a polyclonal antibody that has previously been shown to recognize a protein of the right molecular weight in cats. 6 Immunohistochemical results showed that the majority of mammary tumors studied (35/40; 87%) expressed COX-2, with the intensity of COX-2 immunoreactivity varying from weak to moderate (Fig. 2) and strong (Fig. 3). Of the 35 COX-2 positive mammary carcinomas, 20 displayed a low expression (mean COX-2 score = 2.5 ± 1.1; 50%), 13 had an intermediate level of COX-2 expression (mean COX-2 score = 5.6 ± 0.5; 32%), while 2 showed a high COX-2 expression (mean COX-2 score = 8.5 ± 0.5; 5%) and 5 tumors were negative. COX- 2 expression appeared mainly as a diffuse cytoplasmic staining in neoplastic epithelial cells (Fig. 2), with some cells also displaying a perinuclear localization (Fig. 3). Frozen tissues from 1 mammary carcinoma were available for immunoblot analysis of COX-2 protein. This tumor was one of the 20 that displayed a low COX-2 expression by immunohistochemistry (score of 2), and, as expected, the immunoblot showed a weak signal of the right molecular weight in protein extracts from this carcinoma (Fig. 4).
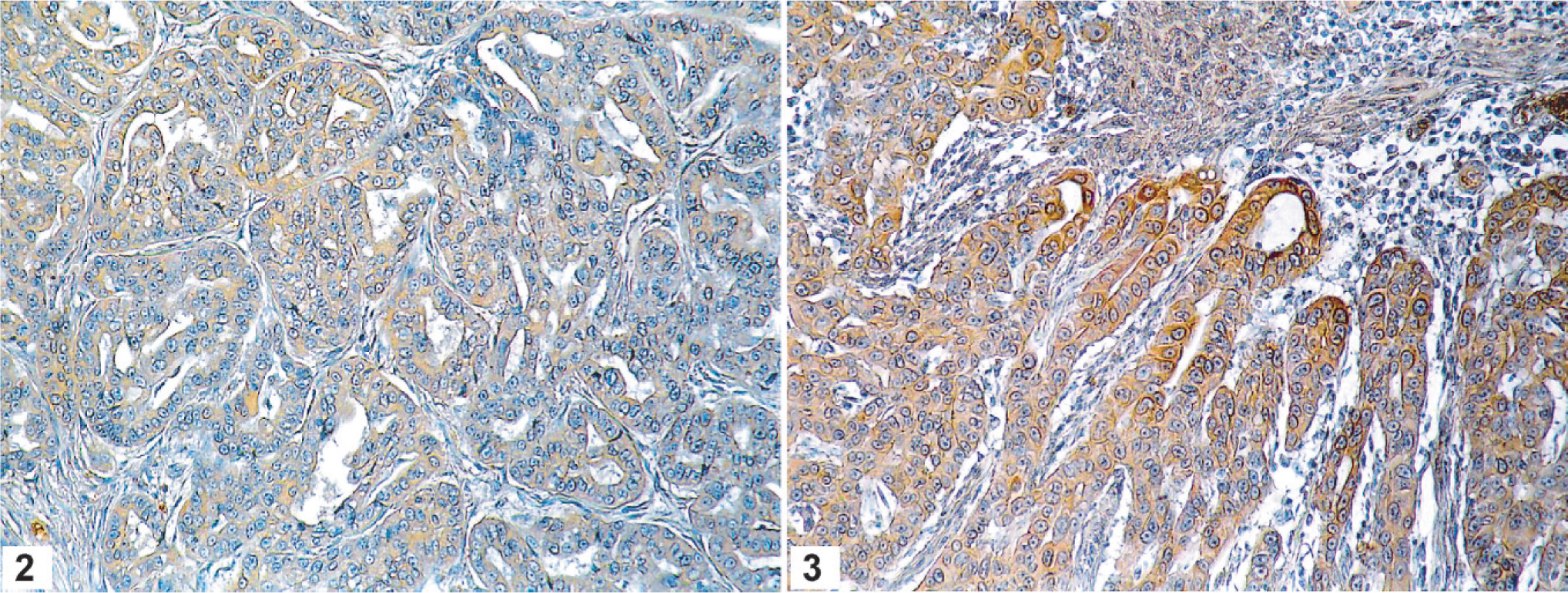
COX-2 expression in feline mammary carcinomas. Tumors with neoplastic epithelial cells displaying a moderate (Fig. 2) to strong (Fig. 3) intensity of COX-2 immunoreactivity are shown. COX-2 expression appeared mainly as a diffuse cytoplasmic staining in neoplastic cells, with some cells also showing a perinuclear localization. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain.
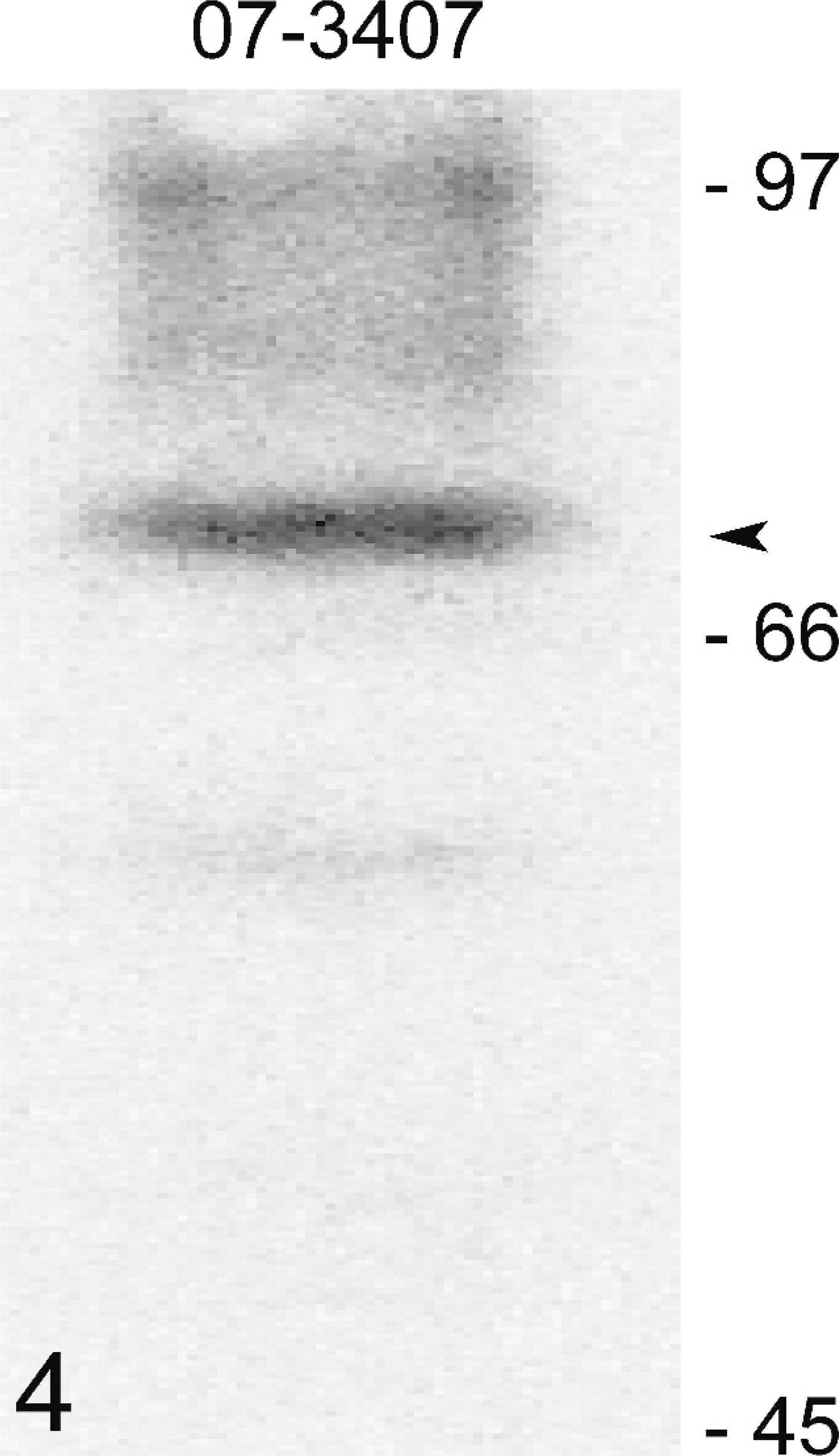
COX-2 protein expression in a feline mammary carcinoma. A protein extract was prepared from a mammary carcinoma and analyzed by one-dimensional SDS-PAGE and immunoblotting techniques using a COX-2 selective antibody. A signal of the right molecular weight is present (arrowhead).
Discussion
This is the first study reporting the molecular characterization of feline COX-2. Our results indicate that feline COX-2 is very similar to its homolog in other species, with all the essential structural domains being conserved in the feline protein. 23 Indeed, the EGF domain important for the dimerization of the protein, the membrane-binding domain, and the signal for retention in the endoplasmic reticulum are all conserved in the feline enzyme. Similar to other mammalian COX-2, feline COX-2 contains 4 N-glycosylation sites, heme coordination residues, a COX active-site tyrosine, and a serine at position 516 that corresponds to the aspirin-acetylation site. Feline COX-2 is a 604-amino acid protein, which is identical in length to several COX-2 homologs but 1 amino acid longer than ovine COX-2. 29 Moreover, feline COX-2 shares a high level of identity with other mammalian COX-2, with the highest identity being with canine COX-2 (96%). 4 This close proximity between feline and canine COX-2 is in keeping with the recently published information on the cat genome, where the highest mean percent identity of the reciprocal best alignments between cat sequences and other mammalian genomes was with the dog genome. 21 In many species, COX-2 mRNA and protein can be induced by agonists such as phorbol 12-myristate 13-acetate, lipopoly saccharides, and inflammatory cytokines. 26, 27 As expected, an induction of COX-2 was observed in cultures of feline fibroblasts stimulated with these agonists (data not shown).
Our results also demonstrate that a large proportion (87%) of feline mammary carcinomas express COX-2. This conclusion is based on the evaluation of COX-2 by immunohistochemistry with confirmation of the specificity of the immunostaining using immunoblot analysis, as COX-2 detection by immunohistochemistry has been shown to produce false-positives in some mammalian cell lines. 11 The high percentage of feline mammary carcinomas expressing COX-2 in the present study is comparable with that reported by Millanta et al., who found immunoreactivity for COX-2 in 96% of the feline mammary carcinomas they evaluated. 17 In their study, 81% of the cases demonstrated strong COX-2 immunoreactivity, while our study found a lower percentage of cases (37%) with intermediate to high COX-2 expression. Possible explanations for this difference in the proportion of tumors with high COX-2 staining intensity include differences in the antibodies and the scoring system that were used. Like those of Millanta et al., our results are in sharp contrast to the absence of COX-2 expression in feline mammary carcinomas previously reported by Beam et al. 1 Again, difference in the antibody is a likely explanation for these contradictory results.
COX-2 expression has previously been reported in canine mammary adenocarcinomas. 7, 10, 17, 18, 22 We 7 and others 10, 18 found that 56 to 62% of canine mammary adenocarcinomas express COX-2, while 2 studies reported higher percentages (100%). 17, 22 Results of the present study indicate that COX-2 expression is also prevalent in feline mammary cancer, suggesting that COX-2 could play some role in feline mammary carcinogenesis, as previously postulated for canine and human mammary cancers. 7, 10, 15, 17, 18, 22, 24 In humans, overexpression of COX-2 has been well documented in breast cancer, with a large variation in prevalence between studies (4.5 to 85%), possibly caused by differences in antibodies and scoring systems used. 24 Some studies have found a correlation between COX-2 expression in breast cancer and increased tumor size, high grade, development of distant metastases, and reduced survival in women. 24 In canine and feline mammary carcinomas, increased levels of COX-2 have been correlated with a poorer prognosis. 17
In women, epidemiologic studies have revealed a reduction of up to 28% in the relative risk of breast cancer with regular use of nonsteroidal anti-inflammatory drugs (NSAIDs) for 10 years or more. 8 This risk reduction by NSAIDs is the result of the inhibition of prostaglandin synthesis, which acts at several levels of the oncogenic process, including stimulation of cell proliferation, suppression of the immune surveillance, reduced apoptosis, increased cell survival, proangiogenesis, and increased activity of matrix metalloproteinase-2 favoring an invasive behavior of the cells. 24 In cats, piroxicam, a nonselective COX inhibitor, has recently been proposed as a potential beneficial treatment for oral squamous cell carcinomas where an overexpression of COX-2 can be demonstrated, representing approximately 20% of the cases. 6 Treatment options for feline mammary carcinomas are very limited. Aggressive surgical excision remains the most common therapeutic intervention, and development of additional therapeutic strategies could help to improve survival. Indeed, adjunctive chemotherapy using doxorubicin has recently been shown to improve the long-term survival of cats with mammary carcinomas. 19 It is possible that other chemotherapeutics agents, such as COX-2 inhibitors, could help as adjunct therapy in the treatment options for feline mammary cancer.
In summary, the present study provides the first molecular characterization of the feline COX-2 enzyme and reveals that it is highly homologous to other mammalian homologs. It also confirms that a high percentage of feline mammary carcinomas express COX-2. Further studies will be essential to elucidate the role played by COX-2 and prostaglandins in pathologic conditions affecting the feline species, including the various forms of cancer.
Footnotes
Acknowledgements
We thank Dr. Stacia Kargman, Merck Frosst Centre for Therapeutic Research, Pointe-Claire-Dorval, Québec, for kindly providing antibody MF243 and Danielle Rannou for technical assistance. This work was supported by a grant from the Winn Feline Foundation (M. D.). GenBank accession number EF036473.
