Abstract
A high prevalence of urinary bladder transitional-cell carcinoma (TCC) has been noted in captive fishing cats (Prionailurus viverrinus). Of the 91 adult deaths between 1995 and 2004, 12 (13%) were attributed to TCC. To help elucidate mechanisms of carcinogenesis, archival sections of urinary bladder from 14 fishing cats were examined histologically and immunohistochemically for p53, cyclooxygenase (COX)-1, and COX-2 expression. Ten cats had TCC, and 4 were unaffected. The average age at death was 10.8 years in affected individuals and 10.5 years in unaffected individuals. There was no sex predilection. Fishing cat TCCs were characterized histologically as papillary and infiltrating (n = 6), nonpapillary and infiltrating (n = 3), or carcinoma in situ (n = 1). Glandular and squamous metaplasia, necrosis, and lymphatic invasion were prominent histologic features. Two individuals had documented metastasis. p53 nuclear immunolabeling was detected in 4/10 (40%) TCCs. In two cases, immunolabeling was limited to less than 10% of the neoplastic cellular population and was comparable to staining of normal fishing cat bladder. Therefore, p53 gene mutation did not appear to be an essential component of TCC carcinogenesis in examined fishing cats. COX-1 immunohistochemistry was negative in all cases. All TCCs had some degree of COX-2 cytoplasmic immunolabeling, which was exclusively within the invasive portions of the neoplasms. Papillary portions were uniformly negative. COX-2 overexpression was a prominent feature in the majority of the examined fishing cat TCCs, suggesting that COX-2-mediated mechanisms of carcinogenesis are important in this species and that COX-inhibiting drugs may be of therapeutic benefit.
Keywords
Fishing cats (Prionailurus viverrinus) are small, primarily piscivorous felids native to Southeast Asia and Indonesia, and are currently listed in Appendix II of the Convention on International Trade in Endangered Species (CITES), 17 which provides the species with protection against overutilization incompatible with long-term survival (http://www.cites.org). Over recent years, an abnormally high prevalence of urinary bladder transitional-cell carcinoma (TCC) has been noted in captive individuals of this species. Of the 91 worldwide, captive adult (>1 year) deaths between 1995 and 2004, 12 (13%) were attributed to TCC. 6, 21 In contrast, urinary TCC is a rare tumor in domestic cats, with a prevalence of less than 1%. 5, 19, 22 Among domestic species, TCC occurs most commonly in the dog. 12 In cattle, a high prevalence of TCC associated with prolonged bracken fern (Pteridium sp.) ingestion is noted in certain geographic regions. 3 In humans, TCC is the second most common cancer of elderly (>60 years) men. 10
TCC is a tumor of older domestic animals and presents with clinical signs attributable to lower-urinary-tract disease, including hematuria, dysuria, stranguria, and urinary obstruction. The clinical aspects of TCC in 4 fishing cats have recently been described. 21 In that study, the average age at death was 9.5 years, no sex predilection was identified, and the major clinical sign in all cases was persistent hematuria unresponsive to medical therapy. In domestic cats and dogs, the trigone/neck region of the bladder is most often affected, and multiple tumors have been noted. Local invasion of the bladder wall is a prominent feature, and metastases to regional lymph nodes and/or lungs are noted in 25 to 50% of domestic canine and feline cases. 12
The etiopathogenesis of TCC is not completely understood; however, several important risk factors have been associated with the development of disease in humans and dogs. Canine TCC has been linked to topical flea/insecticide application, environmental herbicide exposure, and obesity. A breed predisposition in Scottish Terriers and a female sex predilection have also been noted. 16 In humans, established risk factors for the development of TCC include cigarette smoking, industrial exposure to arylamines, certain parasitic bladder infections (i.e., Schistosoma haematobium), long-term analgesic usage, and long-term exposure to cyclophosphamide. 10
In addition to these risk factors, investigations into human-bladder carcinogenesis have identified several specific genetic alterations associated with the development/progression of TCC. 4, 18 Elucidation of the molecular pathways that underlie bladder carcinogenesis has been essential for understanding pathogenesis, monitoring disease progression, and developing effective treatments in humans. p53 and retinoblastoma (Rb) gene mutation and overexpression of epidermal growth factor receptor (EGFR) and the cyclooxygenase (COX)-2 enzyme are among the genetic alterations noted. 1, 4, 15, 20 In human TCC, p53 mutations are frequent in high-grade tumors and have been associated with invasiveness. 20, 23 Rb gene alterations occur in an estimated 30% of human TCCs and are associated with higher recurrence and lower survival rates. 4, 18 EGFR overexpression is a feature of high-grade, poorly differentiated human TCCs. 4 In addition to human TCC, COX-2 overexpression has also been noted in rat, bovine, canine, and domestic feline TCC. 2, 3, 14, 15
To help elucidate possible mechanisms of bladder carcinogenesis in fishing cats, archival sections of urinary bladder were examined histologically and by immunohistochemistry for expression of p53, Rb, EGFR, COX-1, and COX-2.
Materials and Methods
Paraffin-embedded tissue or unstained 5 μm sections of previously diagnosed urinary bladder TCCs from 10 fishing cats housed in 6 different zoological facilities and normal urinary bladder from 4 fishing cats housed in 3 different zoological facilities, along with available clinical histories and/or necropsy reports, were obtained for retrospective histopathologic and immunohistochemical analyses.
Paraffin-embedded samples were sectioned at 5 μm, and all slides were stained with hematoxylin and eosin (HE) for routine light microscopy. A diagnosis of TCC was confirmed, and TCCs were classified according to the World Health Organization (WHO) International Classification of Tumors of the Urinary System of Domestic Animals. 12
Immunolabeling was performed by using a streptavidin-biotin-peroxidase complex method. Primary antibodies used were rabbit polyclonal anti-p53 (p53-CM1; Novocastra Laboratories, Newcastle upon Tyne, UK) diluted 1 ° 200 in 0.1 M phosphate-buffered saline solution (PBS); rabbit polyclonal anti-COX-2 (PG27B; Oxford Biomedical Research, Oxford, MI) diluted 1 ° 200 in PBS; mouse monoclonal anti-COX-1 (160110; Cayman Chemical, Ann Arbor, MI) diluted 1 ° 100 in PBS; rabbit polyclonal anti-Rb gene product (sc-50; Santa Cruz Biotechnology, Inc., Santa Cruz, CA) diluted 1 ° 200 in PBS; and ready-to-use rabbit polyclonal anti-EGFR (AR335-5R; BioGenex, San Ramon, CA).
Domestic feline oral squamous-cell carcinoma and normal oral mucosa were used as COX-1, Rb, EGFR, and p53 positive-control tissues based upon manufacturer recommendation and/or previous studies. 14 Domestic feline urinary bladder TCC was used as the COX-2 positive-control tissue based upon previous studies. 2, 3, 14
For COX-1, COX-2, Rb, and EGFR, after deparaffinization and rehydration, endogenous peroxidases were blocked by incubation in 0.3% hydrogen peroxide in methanol for 30 minutes. After a 5-minute rinse in PBS, slides were steamed for 30 minutes in citrate buffer at pH 6 for antigen retrieval. Slides were rinsed for 5 minutes in PBS and then incubated in 10% normal goat serum (31872; Pierce Biotechnology, Rockford, IL) at room temperature for 20 minutes. After a 15-minute rinse in PBS, Avidin D and biotin solutions (SP-2001; Vector Laboratories, Burlingame, CA) were applied sequentially for 15 minutes each. Slides were rinsed for 15 minutes in PBS before application of primary antibodies. Negative control slides were incubated with 10% normal goat serum. All slides were incubated overnight at 4°C. After a 15-minute rinse in PBS, secondary antibodies were applied for 30 minutes at room temperature (COX-1–biotinylated goat anti-mouse immunoglobulin (Ig) G H+L, BA-9200, Vector Laboratories; COX-2, Rb, EGFR–biotinylated goat anti-rabbit IgG H+L, BA-1000, Vector Laboratories). Slides were then rinsed for 15 minutes in PBS before the application of a peroxidase blocking reagent (S2001; DakoCytomation, Carpinteria, CA) for 5 minutes. After a 5-minute rinse in PBS, slides were incubated with horseradish-peroxidase-streptavidin conjugate (43-4323; Zymed Laboratories Inc., San Francisco, CA) diluted 1°500 in PBS for 30 minutes at room temperature. Slides were rinsed for 15 minutes in PBS before the chromagen, 3,3′-diaminobenzidine tetrachloride (D4168; Sigma-Aldrich, St. Louis, MO) was applied. Slides were counterstained with Mayer's hematoxylin (MHS-32; Sigma-Aldrich).
For p53, specific modifications were made to the protocol described for COX-1, COX-2, Rb, and EGFR. Antigen retrieval with steaming in citrate buffer was done before incubation in 0.3% hydrogen peroxide in methanol. An additional antigen retrieval step that involved a 10-minute incubation in 0.05% saponin solution (S7900-25G; Sigma-Aldrich) was performed before the application of normal goat serum. Lastly, the incubation time for the peroxidase blocking solution was increased from 5 to 10 minutes.
Immunoreactivity was assessed and assigned a grade based upon the percentage of positive neoplastic cells. Grades were determined from evaluating five 10× fields and were assigned as follows: 0, negative; 1, <10% of neoplastic cells staining positively; 2, 10–50%; 3, >50%.
Results
Demographic, pathologic, and immunohistochemical results are summarized in Table 1.
Demographic, pathologic, and immunohistochemical characterization of fishing cat urinary bladder TCC cases.
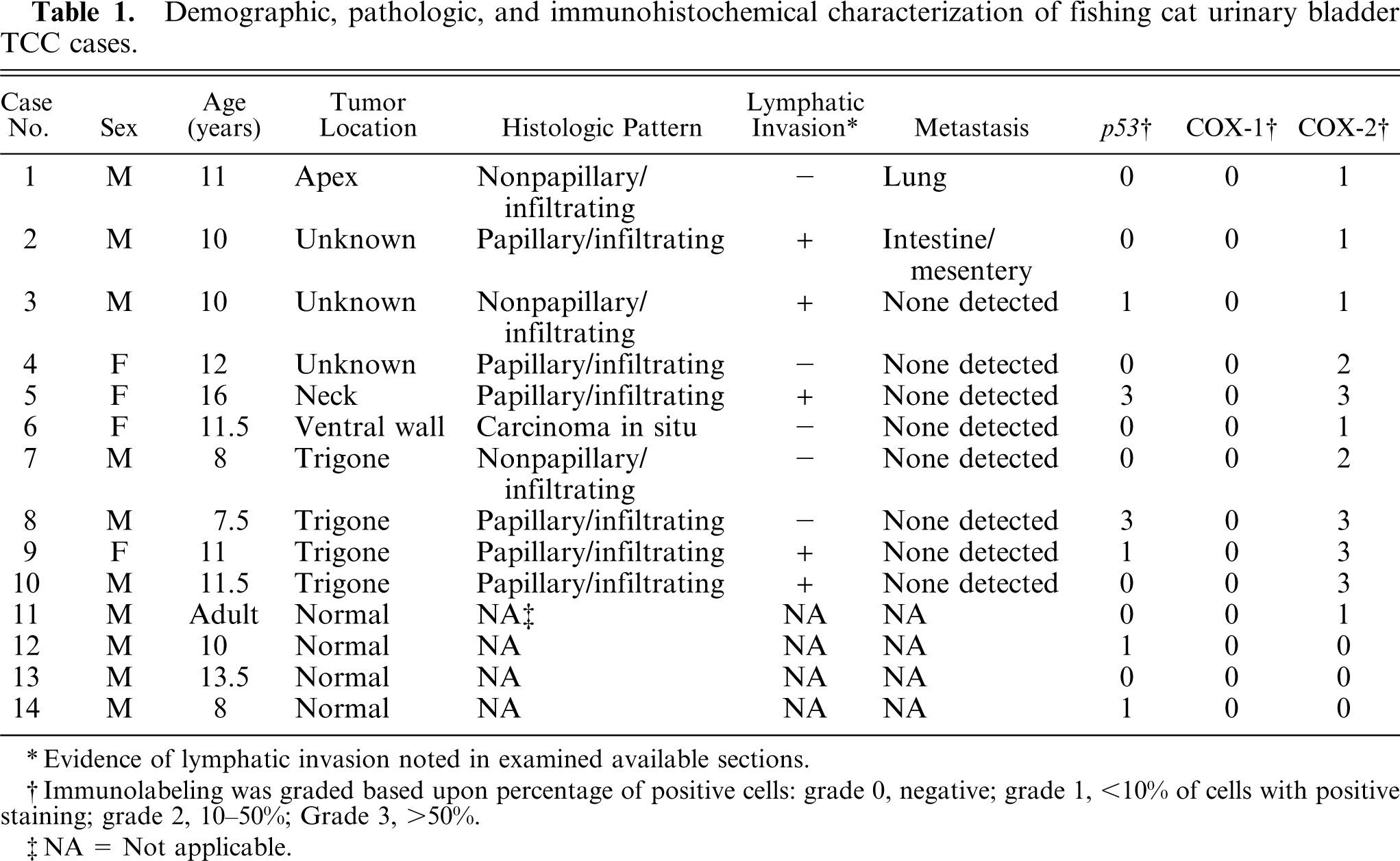
∗ Evidence of lymphatic invasion noted in examined available sections.
†Immunolabeling was graded based upon percentage of positive cells: grade 0, negative; grade 1, <10% of cells with positive staining; grade 2, 10–50%; Grade 3, >50%.
‡NA = Not applicable.
All fishing cats were captive-born individuals residing in U.S. zoological facilities. Unaffected and affected individuals were housed together at the various facilities. Six of the affected animals were male, and 4 were female. The average age at death was 10.8 years (7.5–16 years). All unaffected animals were males, with an average age at death of 10.5 years (8–13.5 years). All affected animals were euthanized as a result of urinary TCC. Unaffected animals died or were euthanized because of a variety of causes, including mandibular osteosarcoma, adrenal cortical carcinoma, osteoarthritis, and septic peritonitis.
Grossly, neoplasms were characterized as discrete, firm, tan to white, nodular to papillary masses, or indistinct, irregular, regional mural thickenings often with mucosal ulceration. Neoplasms were most commonly identified in the trigone/neck region of the bladder. TCC was confirmed histologically in all cases. Urinary bladders of the unaffected animals were histologically normal.
Based upon the WHO classification system, 12 examined fishing cat TCCs were categorized as papillary and infiltrating in 6 of 10 cases (60%) (Fig. 1), nonpapillary and infiltrating in 3 of 10 (30%), and carcinoma in situ in 1 of 10 cases (10%). Invasive neoplasms markedly expanded the bladder wall, effaced smooth-muscle fibers, and often extended to the serosal surface. Three distinct patterns of neoplastic cellular arrangement were noted. Papillary components of the neoplasms consisted of arborizing luminal projections of crowded, disorganized, neoplastic epithelial cells supported by slender central stalks of fibrovascular stroma (Fig. 2). Arrangement in infiltrative portions of the neoplasms ranged from densely packed thick cords and haphazard nests of neoplastic cells supported by small amounts of fibrovascular stroma (Fig. 3) to irregular islands and branching trabeculae, with or without glandular metaplasia that were separated by moderate to large accumulations of dense collagenous stroma (Fig. 4). Both patterns were commonly present within infiltrative portions of the same neoplasm. In 6 of 10 cases (60%), large portions of the neoplasms were necrotic. Multifocal squamous metaplasia was noted in 3 of 10 cases (30%). Lymphatic vessels that contained intravascular rafts of neoplastic cells were detected in examined sections from 5 of 10 cases (50%). In case No. 6, the bladder mucosa was regionally thickened. Epithelial cells in this region were crowded and disorganized, with multifocal loss of nuclear polarity, consistent with carcinoma in situ. Distant metastasis was documented in case Nos. 1 and 2, with spread to the lungs and intestines/mesentery, respectively.
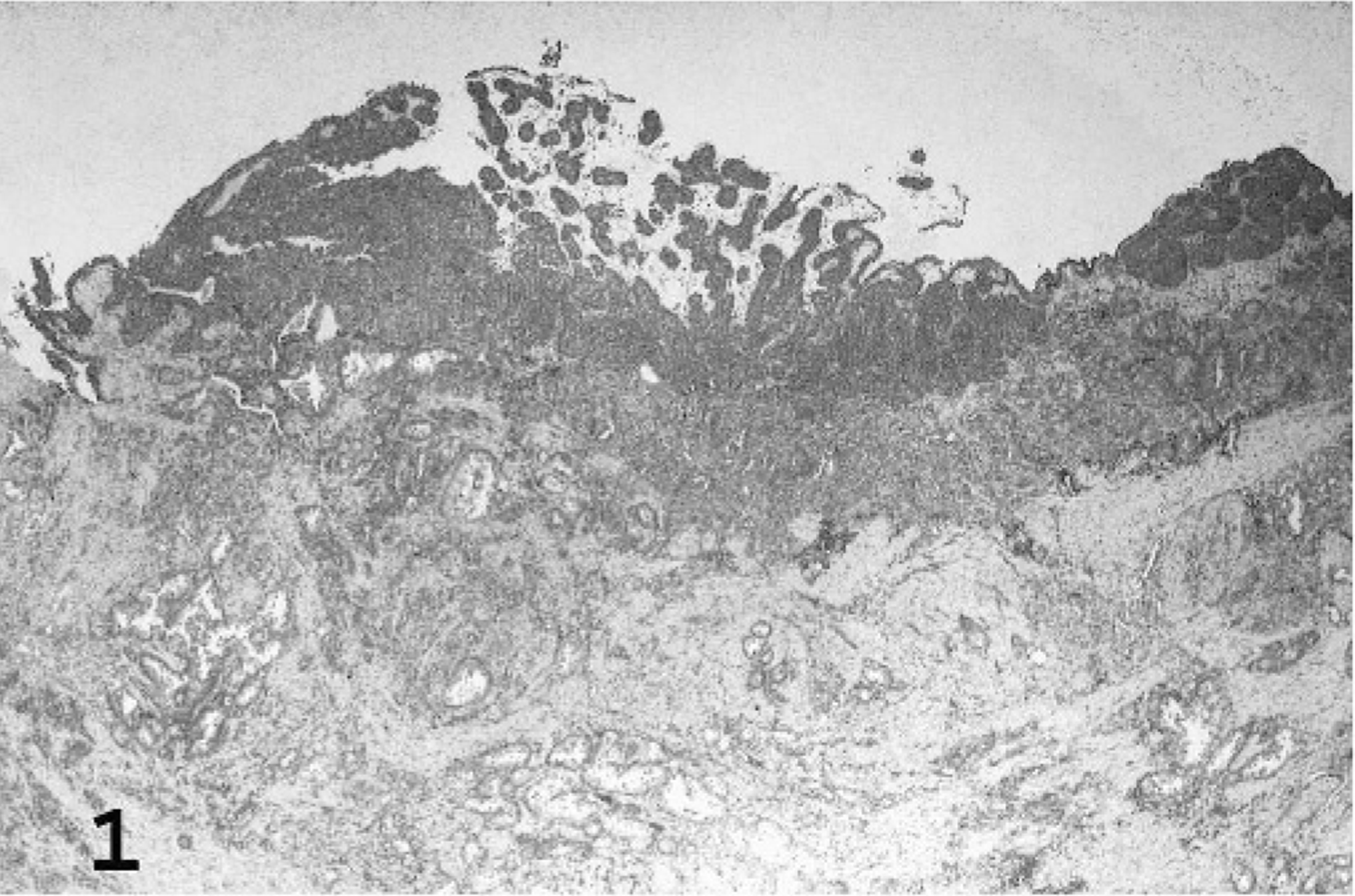
Urinary bladder; fishing cat No. 5. Papillary and infiltrating TCC expanded the bladder wall with replacement of smooth muscle. HE.
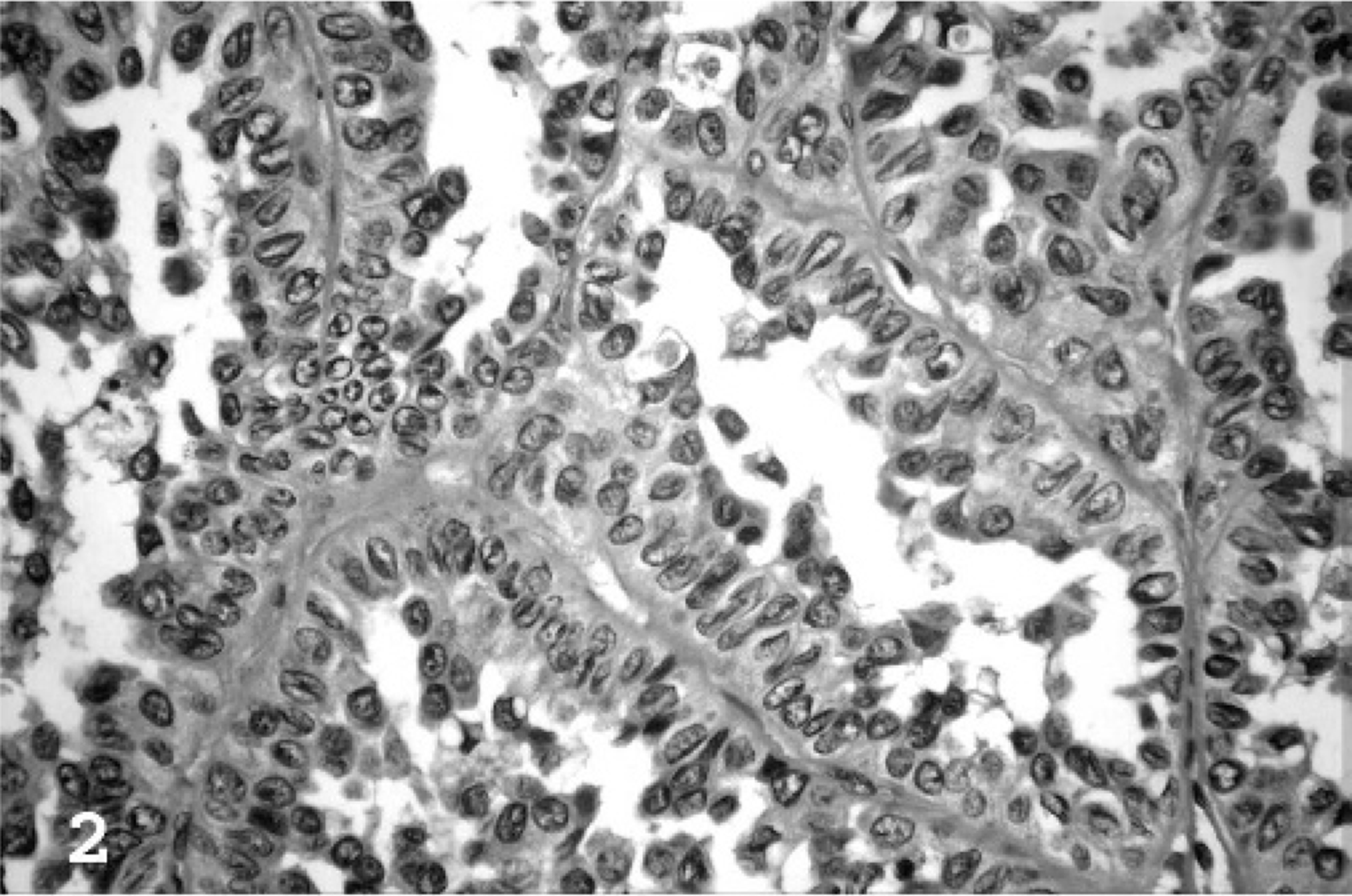
Urinary bladder; fishing cat No. 2. In papillary portions of the neoplasms, cells formed arborizing projections of crowded, disorganized neoplastic epithelial cells supported by slender central stalks of fibrovascular stroma. HE.
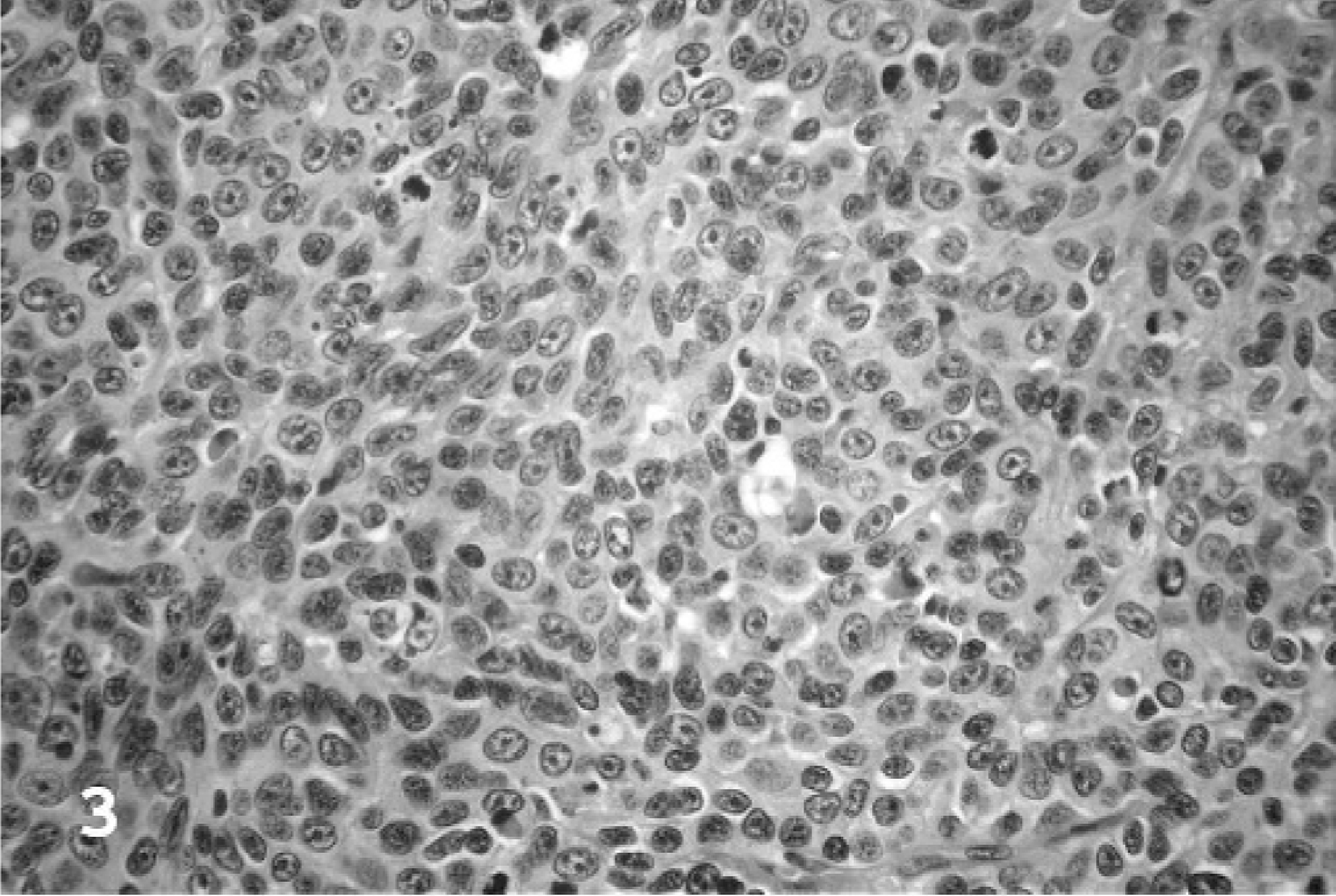
Urinary bladder; fishing cat No. 3. Neoplastic cells in infiltrative portions of the neoplasms were commonly arranged in densely packed thick cords and haphazard nests supported by small amounts of fibrovascular stroma. HE.
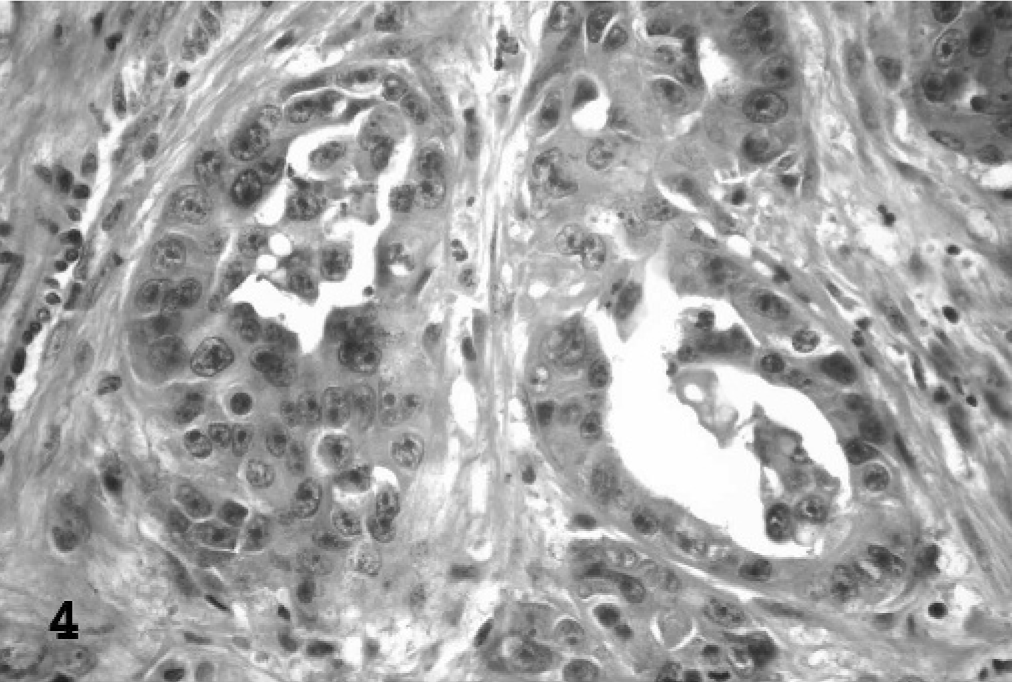
Urinary bladder; fishing cat No. 9. In infiltrative portions of the neoplasms, a second pattern consisting of irregular islands and branching trabeculae of neoplastic cells separated by moderate to large accumulations of dense collagenous stroma was noted. Many of the islands/trabeculae contained central areas of glandular metaplasia. HE.
Positive p53 immunolabeling was detected in 4 of 10 TCCs (40%). Positive staining was distributed throughout the nucleus of labeled cells and was distinctly granular and of high intensity in all cases (Fig. 5). Grade 1 staining noted in 2 TCC cases was similar to the level of immunolabeling noted in 2 normal fishing cat bladders.
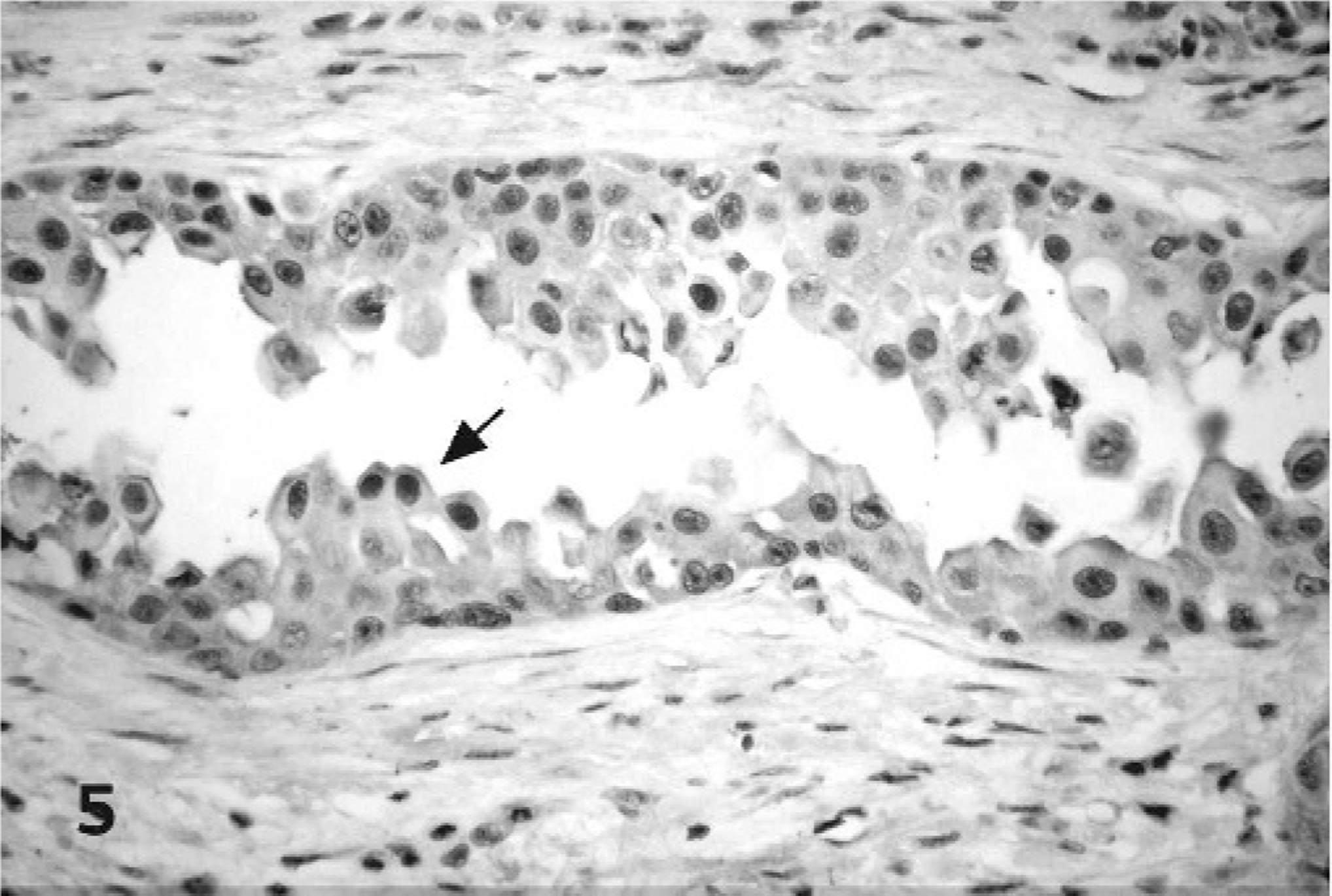
Urinary bladder; fishing cat No. 5. p53 immunolabeling was seen in the nucleus of scattered neoplastic cells (arrow). Streptavidin-biotin-peroxidase, Mayer's hematoxylin counterstain.
COX-1 immunohistochemistry was negative in all 10 TCCs and all 4 normal urinary bladders. In contrast, all 10 TCCs had some degree of positive immunolabeling for COX-2, with all labeled cells located within infiltrative portions of the neoplasms. Papillary portions were uniformly negative. Staining was perinuclear to diffuse cytoplasmic, finely granular, and of moderate to high intensity (Fig. 6). In grade 1 cases, positive cells were most often individual or small clusters of cells along the deep infiltrative margins of the neoplasms. One normal bladder had rare COX-2 positive individual epithelial cells.
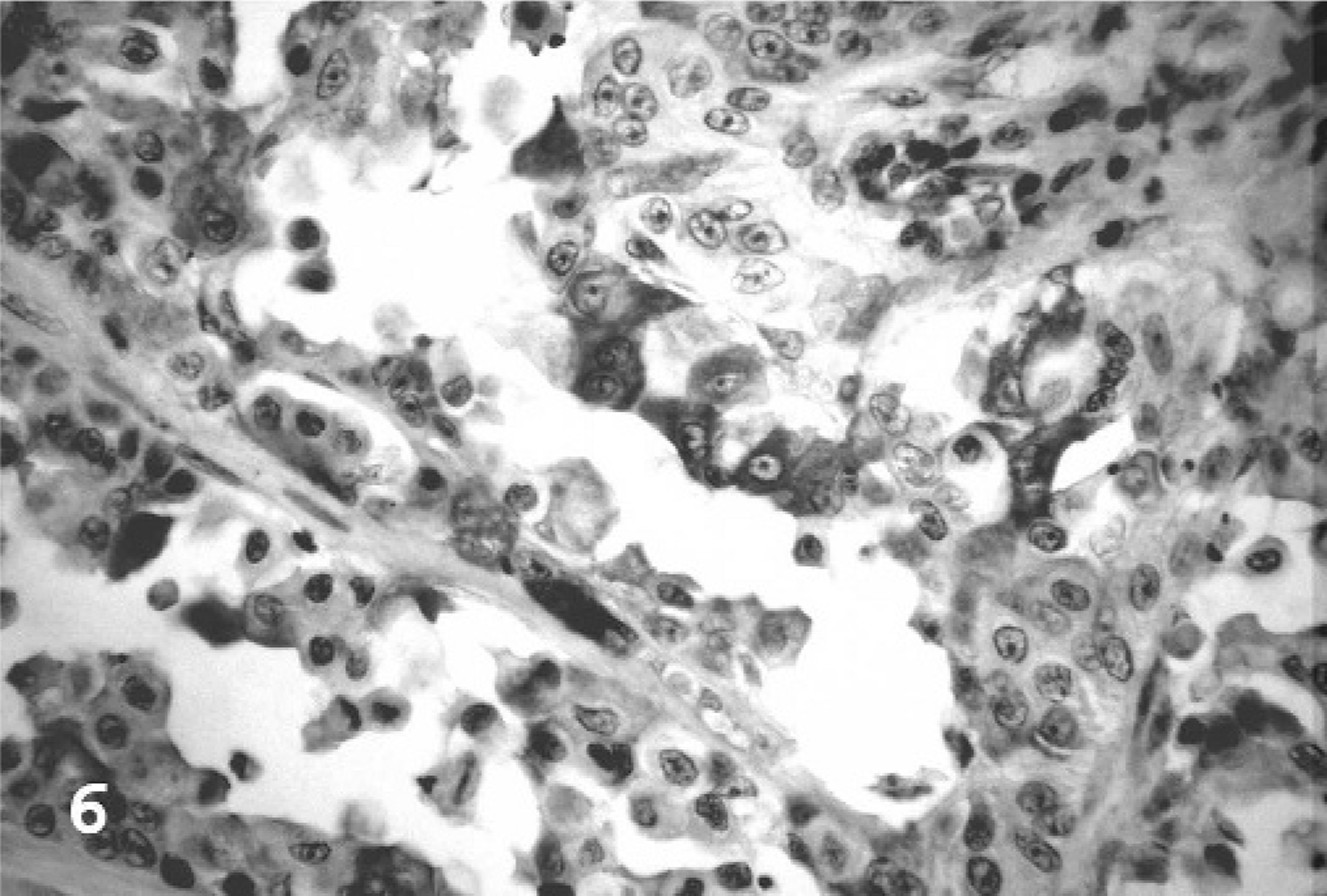
Urinary bladder; fishing cat No. 5. COX-2 immunolabeling was seen as moderate to high intensity, perinuclear to diffuse cytoplasmic staining of neoplastic cells. Streptavidin-biotin-peroxidase, Mayer's hematoxylin counterstain.
No immunolabeling for Rb was detected in any of the domestic feline control tissues or normal fishing cat bladders examined. Application of EGFR antibody to both domestic feline control tissues and normal fishing cat bladder resulted in consistent, nonspecific, diffuse cytoplasmic and nuclear background staining. Thus, immunohistochemistry with these antibodies was not pursued on the fishing cat TCC cases.
The genealogy of the fishing cat TCC cases is depicted in Fig. 7. Eight of the 10 cases can be traced back to a single, unaffected breeding pair that consisted of a wild-caught female and a captive-born male with wild-caught parents.
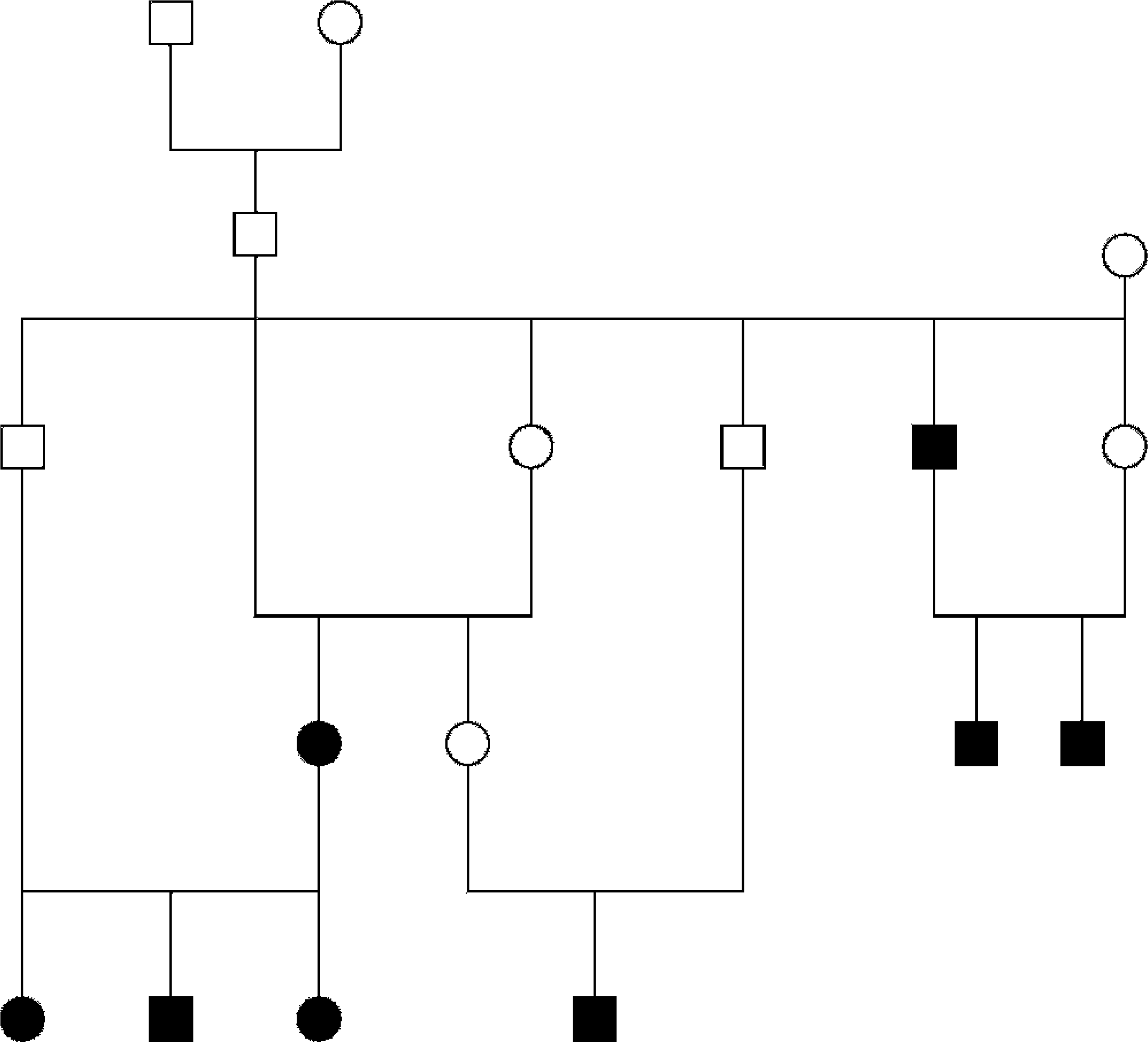
Genealogy of 8 fishing cat TCC cases. Eight of the 10 confirmed TCC cases could be traced back to a single breeding pair. Squares represent males; circles represent females. Shaded figures represent TCC cases.
Discussion
Histologically, all examined fishing cat TCCs, except the lone case of carcinoma in situ, had extensive mural invasion that often extended through to the serosal surface of the bladder. A subset of these tumors also had luminal papillary components. Papillary and infiltrating TCC is the most common morphologic variant in domestic species. In domestic animals, histologic evidence of mural invasion is a risk factor for metastasis. Carcinoma in situ is the least common variant of TCC in domestic species and likely represents early neoplastic transformation of the bladder epithelium. 12
The high degree of relatedness within the fishing cat American Zoo and Aquarium Association Species Survival Plan captive population (mean F value = 0.186) 21 combined with the increased prevalence of bladder TCC therein suggest genetics may play a significant role in fishing cat TCC carcinogenesis. The relatively high level of inbreeding present within the captive fishing cat population is because of the low number of original founder animals. The resultant population dynamics with repeated line breeding could have facilitated the perpetuation of a TCC-related genetic defect(s). Eight of the confirmed cases described in this report were descended from a single unaffected breeding pair. The remaining 2 cases were litter mates. The potential genetic basis for TCC in this species spurred the investigation of genetic alterations known to occur in TCCs of other species, including mutation of the p53 and Rb genes and overexpression of EGFR and COX-2.
Wild-type p53 has a very short half-life, with the normal expression generally undetectable by immunohistochemistry. Mutated p53 has a prolonged half-life and thus, p53 immunolabeling is considered generally indicative of mutation. 4 In the fishing cats, only 2 of 10 TCCs (20%) had significant positive immunoreactivity for p53. In 2 additional cases, grade 1 staining was detected. This low level of staining was comparable with that noted in sections of 2 histologically normal fishing cat bladders. It was unknown whether staining in the normal bladders was indicative of early accumulation of significant p53 mutations or, alternatively, representative of normal wild-type p53 expression. Mutations of p53 are detected in 10 to 20% of all human bladder tumors, however, the percentage markedly increases with the exclusion of low-grade, papillary phenotypes in which p53 mutations are rare. In human TCC, p53 mutations most often occur late in the course of the disease and are associated with muscle invasion and a high-grade, rapidly progressing phenotype. 18, 20, 23 The majority of the examined fishing cat TCCs had prominent invasive components. Despite the apparent high-grade phenotype of the examined fishing cat TCCs, p53 gene mutation does not appear to be an essential component of TCC pathogenesis in this species.
COX-2 overexpression was noted in all examined cases of fishing cat TCC, and was a prominent feature in 6 of 10 TCCs (60%), suggesting that COX-2-mediated mechanisms of carcinogenesis are important in this species. COX-2 overexpression has been detected in several types of epithelial neoplasia in both humans and animals, suggesting a mechanistic link between COX-2 expression and the development of neoplasms. 2, 3, 9, 11, 14, 15 Genetic studies that involved transgenic and knockout mice have further illustrated a cause-effect relation between COX-2 overexpression and carcinogenesis in certain types of epithelial neoplasms. 7 COX-2 overexpression is the result of the deregulation of transcriptional and posttranscriptional control. Various growth factors, oncogenes, cytokines, and tumor promoters have the ability to stimulate COX-2 transcription via protein kinase C and Ras-mediated signaling. The resulting COX-2 overexpression leads to increased prostaglandin synthesis, which, in turn, affects numerous processes implicated in carcinogenesis, including increased cellular proliferation, inhibition of apoptosis, stromal invasion, decreased immune surveillance, and angiogenesis. 7, 8 In addition to these prostaglandin-mediated effects, the inherent oxidative properties of COX-2 can result in the direct formation of a variety of mutagenic substances (i.e., byproducts of arachidonic acid metabolism and/or converted xenobiotics). 7, 8
COX-2 immunoreactivity was limited to the infiltrative portions of the neoplasms. In the 4 cases with grade 1 COX-2 immunolabeling, the positive staining was notably most prominent in cells along the deep infiltrative margins of the neoplasms. This differential immunolabeling based on the morphologic pattern of the neoplasms suggested COX-2 expression might play a role in neoplastic cellular invasion in fishing cat TCC. In humans, COX-2 has been associated with activation of matrix metalloproteinases, enzymes responsible for the digestion of basement membrane collagen and facilitation of neoplastic cellular migration and metastasis. 3, 7
The role of COX-2 overexpression in carcinogenesis makes this enzyme an important target for chemopreventative therapeutics. Nonsteroidal anti-inflammatory drugs (NSAID) function by inhibiting cyclooxygenase enzymes and have been used in both humans and animals to treat/prevent a variety of neoplastic diseases. Selective COX-2 inhibitors are of particular interest because of their tendency to result in less adverse side effects, such as upper-gastrointestinal mucosal injury. In human, canine, and laboratory rodent cases of TCC, NSAID therapy resulted in partial clinical remission, with a reduction in tumor size 13 . In addition, in laboratory rats, certain selective COX-2 inhibitors have been shown experimentally to prevent formation of bladder neoplasms. 7, 8 When considering the prominent differential immunolabeling noted in the examined fishing cat TCCs, NSAID therapy in this species may prove useful in preventing/halting muscle invasion and metastasis. The lack of immunolabeling within papillary portions of the examined TCCs, however, suggests that NSAID therapy may not be as effective in resolving disease or clinical signs related to these components of the neoplasms. Two of the fishing cats (case Nos. 6 and 8) included in this study were treated with Piroxicam (Feldene; Pfizer, Inc., U.S. Animal Health Operations, New York, NY), a potent oxicam-derivative NSAID, and exhibited partial clinical remission manifested by the resolution of clinical signs (hematuria) for a period of weeks to months before euthanasia. 21 Though these cases provide encouraging anecdotal evidence, additional prospective studies are needed to determine the efficacy and the proper treatment regimen for effective NSAID therapy in cases of fishing cat TCC.
Mutation of the Rb gene and overexpression of EGFR are other important genetic alterations recognized in human TCC. 4, 18 Unfortunately, commercially available Rb antibodies used in this study failed to stain any of the control domestic feline tissues or normal fishing cat bladder used. Lack of antibody cross-reactivity with feline Rb may have accounted for the absence of immunolabeling in examined tissues. In the case of EGFR, insurmountable procedural difficulties that resulted in consistent, nonspecific, diffuse, cytoplasmic, and nuclear background staining, despite multiple attempted modifications to the immunohistochemistry antigen retrieval and blocking protocols, hindered meaningful interpretation of immunolabeling and precluded evaluation of EGFR expression in the fishing cat TCC cases.
Urinary bladder TCC is an important disease that impacts the individual health as well as the overall population survival of captive fishing cats. The relatedness of affected individuals and the abnormally high prevalence of TCC in this limited population suggest that genetic factors play a role in carcinogenesis. In addition, the overexpression of COX-2, noted particularly in invasive portions of the neoplasms, implicated this enzyme in fishing cat TCC invasion. Therapeutic targeting of COX-2 with NSAIDs may assist in preventing or halting the progression of invasive TCC-related disease.
Future studies that investigate the possible role of nutrition in fishing cat TCC carcinogenesis would be beneficial. Most captive fishing cat diets contain a relative paucity of fish (W. Swanson, personal communication). The omega-3 fatty acids and natural antioxidants, e.g., vitamin E, abundant in fish, may be useful in combating COX-2's direct oxidative damage and its contribution to carcinogenesis.
Footnotes
Acknowledgements
We thank Stacy Schultz, Jane Chladny, and the University of Illinois histology laboratory for technical assistance and slide preparation and the Chicago Zoological Society's Brookfield Zoo for providing laboratory space. Drs. Michael Kinsel and Lisa Farina provided critical review of the manuscript. We also thank Dr. William Swanson, Dr. Meg Sutherland-Smith, and Linda Curtis for providing the Fishing Cat International Studbook and helping to identify fishing cat TCC cases. Finally, we thank the veterinarians and staff of the Audubon Zoo; Capron Park Zoo; Chicago Zoological Society's Brookfield Zoo; Cincinnati Zoo and Botanical Gardens; Department of Veterinary Population Medicine, College of Veterinary Medicine, University of Minnesota; Memphis Zoo; Minnesota Zoo; Northwest ZooPath; San Diego Zoo and Wild Animal Park; and Zoo/Exotic Pathology Service for their contributions of case materials.
